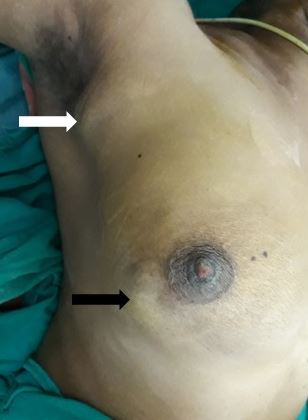
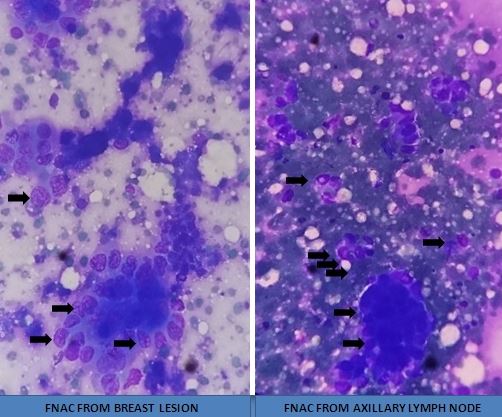
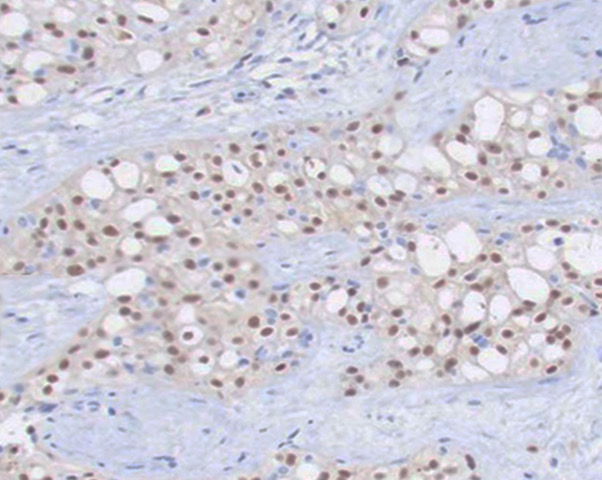
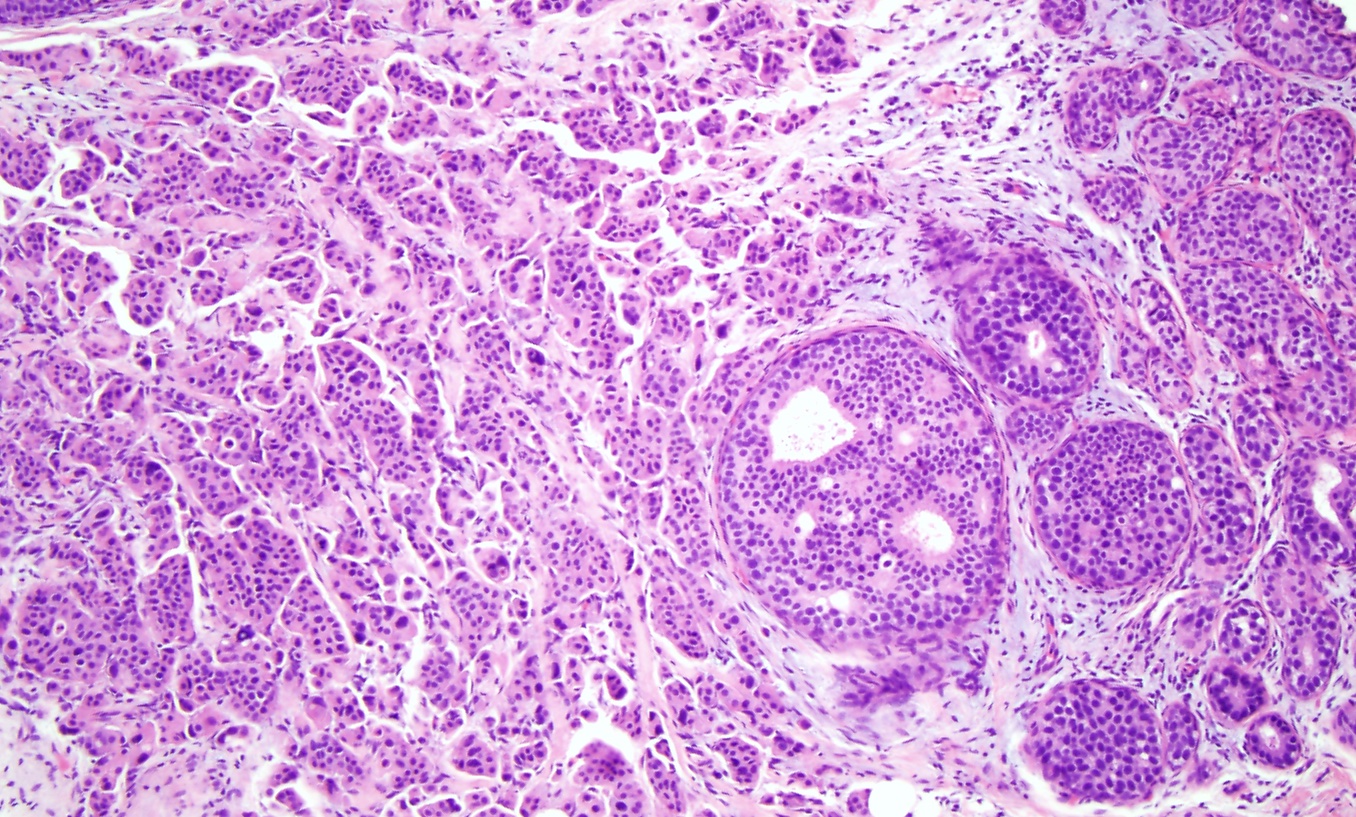
[1]
Watkins EJ. Overview of breast cancer. JAAPA : official journal of the American Academy of Physician Assistants. 2019 Oct:32(10):13-17. doi: 10.1097/01.JAA.0000580524.95733.3d. Epub
[PubMed PMID: 31513033]
Level 3 (low-level) evidence
[2]
Alex A, Bhandary E, McGuire KP. Anatomy and Physiology of the Breast during Pregnancy and Lactation. Advances in experimental medicine and biology. 2020:1252():3-7. doi: 10.1007/978-3-030-41596-9_1. Epub
[PubMed PMID: 32816256]
Level 3 (low-level) evidence
[3]
Kashyap D, Pal D, Sharma R, Garg VK, Goel N, Koundal D, Zaguia A, Koundal S, Belay A. Global Increase in Breast Cancer Incidence: Risk Factors and Preventive Measures. BioMed research international. 2022:2022():9605439. doi: 10.1155/2022/9605439. Epub 2022 Apr 18
[PubMed PMID: 35480139]
[4]
PDQ Screening and Prevention Editorial Board. Breast Cancer Screening (PDQ®): Health Professional Version. PDQ Cancer Information Summaries. 2002:():
[PubMed PMID: 26389344]
[5]
Doren A, Vecchiola A, Aguirre B, Villaseca P. Gynecological-endocrinological aspects in women carriers of BRCA1/2 gene mutations. Climacteric : the journal of the International Menopause Society. 2018 Dec:21(6):529-535. doi: 10.1080/13697137.2018.1514006. Epub 2018 Oct 8
[PubMed PMID: 30295091]
Level 2 (mid-level) evidence
[6]
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA: a cancer journal for clinicians. 2021 May:71(3):209-249. doi: 10.3322/caac.21660. Epub 2021 Feb 4
[PubMed PMID: 33538338]
[7]
Parada H Jr, Sun X, Tse CK, Olshan AF, Troester MA. Lifestyle Patterns and Survival Following Breast Cancer in the Carolina Breast Cancer Study. Epidemiology (Cambridge, Mass.). 2019 Jan:30(1):83-92. doi: 10.1097/EDE.0000000000000933. Epub
[PubMed PMID: 30299404]
[8]
White AJ, Bradshaw PT, Hamra GB. Air pollution and Breast Cancer: A Review. Current epidemiology reports. 2018 Jun:5(2):92-100. doi: 10.1007/s40471-018-0143-2. Epub 2018 Mar 27
[PubMed PMID: 30271702]
[9]
Gucalp A, Traina TA, Eisner JR, Parker JS, Selitsky SR, Park BH, Elias AD, Baskin-Bey ES, Cardoso F. Male breast cancer: a disease distinct from female breast cancer. Breast cancer research and treatment. 2019 Jan:173(1):37-48. doi: 10.1007/s10549-018-4921-9. Epub 2018 Sep 28
[PubMed PMID: 30267249]
[10]
Azamjah N, Soltan-Zadeh Y, Zayeri F. Global Trend of Breast Cancer Mortality Rate: A 25-Year Study. Asian Pacific journal of cancer prevention : APJCP. 2019 Jul 1:20(7):2015-2020. doi: 10.31557/APJCP.2019.20.7.2015. Epub 2019 Jul 1
[PubMed PMID: 31350959]
[11]
Collaborative Group on Hormonal Factors in Breast Cancer. Familial breast cancer: collaborative reanalysis of individual data from 52 epidemiological studies including 58,209 women with breast cancer and 101,986 women without the disease. Lancet (London, England). 2001 Oct 27:358(9291):1389-99
[PubMed PMID: 11705483]
Level 2 (mid-level) evidence
[12]
Roulot A, Héquet D, Guinebretière JM, Vincent-Salomon A, Lerebours F, Dubot C, Rouzier R. Tumoral heterogeneity of breast cancer. Annales de biologie clinique. 2016 Dec 1:74(6):653-660
[PubMed PMID: 27848916]
[13]
Prat A, Pineda E, Adamo B, Galván P, Fernández A, Gaba L, Díez M, Viladot M, Arance A, Muñoz M. Clinical implications of the intrinsic molecular subtypes of breast cancer. Breast (Edinburgh, Scotland). 2015 Nov:24 Suppl 2():S26-35. doi: 10.1016/j.breast.2015.07.008. Epub 2015 Aug 5
[PubMed PMID: 26253814]
[14]
Perou CM, Sørlie T, Eisen MB, van de Rijn M, Jeffrey SS, Rees CA, Pollack JR, Ross DT, Johnsen H, Akslen LA, Fluge O, Pergamenschikov A, Williams C, Zhu SX, Lønning PE, Børresen-Dale AL, Brown PO, Botstein D. Molecular portraits of human breast tumours. Nature. 2000 Aug 17:406(6797):747-52
[PubMed PMID: 10963602]
[15]
McCart Reed AE, Kalinowski L, Simpson PT, Lakhani SR. Invasive lobular carcinoma of the breast: the increasing importance of this special subtype. Breast cancer research : BCR. 2021 Jan 7:23(1):6. doi: 10.1186/s13058-020-01384-6. Epub 2021 Jan 7
[PubMed PMID: 33413533]
[16]
Roux P, Knight S, Cohen M, Classe JM, Mazouni C, Chauvet MP, Reyal F, Colombo PE, Jouve E, Chopin N, Daraï E, Coutant C, Lambaudie E, Houvenaeghel G. Tubular and mucinous breast cancer: results of a cohort of 917 patients. Tumori. 2019 Feb:105(1):55-62. doi: 10.1177/0300891618811282. Epub 2018 Dec 20
[PubMed PMID: 30900967]
[17]
Cserni G. Histological type and typing of breast carcinomas and the WHO classification changes over time. Pathologica. 2020 Mar:112(1):25-41. doi: 10.32074/1591-951X-1-20. Epub
[PubMed PMID: 32202537]
[18]
. Practice Bulletin Number 179: Breast Cancer Risk Assessment and Screening in Average-Risk Women. Obstetrics and gynecology. 2017 Jul:130(1):e1-e16. doi: 10.1097/AOG.0000000000002158. Epub
[PubMed PMID: 28644335]
[19]
Baines CJ. Physical examination of the breasts in screening for breast cancer. Journal of gerontology. 1992 Nov:47 Spec No():63-7
[PubMed PMID: 1430885]
[20]
Menta A, Fouad TM, Lucci A, Le-Petross H, Stauder MC, Woodward WA, Ueno NT, Lim B. Inflammatory Breast Cancer: What to Know About This Unique, Aggressive Breast Cancer. The Surgical clinics of North America. 2018 Aug:98(4):787-800. doi: 10.1016/j.suc.2018.03.009. Epub 2018 May 24
[PubMed PMID: 30005774]
[23]
Mann RM, Kuhl CK, Moy L. Contrast-enhanced MRI for breast cancer screening. Journal of magnetic resonance imaging : JMRI. 2019 Aug:50(2):377-390. doi: 10.1002/jmri.26654. Epub 2019 Jan 18
[PubMed PMID: 30659696]
[24]
Nielsen S, Narayan AK. Breast Cancer Screening Modalities, Recommendations, and Novel Imaging Techniques. The Surgical clinics of North America. 2023 Feb:103(1):63-82. doi: 10.1016/j.suc.2022.08.004. Epub 2022 Oct 17
[PubMed PMID: 36410354]
[26]
Radovic N, Ivanac G, Divjak E, Biondic I, Bulum A, Brkljacic B. Evaluation of Breast Cancer Morphology Using Diffusion-Weighted and Dynamic Contrast-Enhanced MRI: Intermethod and Interobserver Agreement. Journal of magnetic resonance imaging : JMRI. 2019 May:49(5):1381-1390. doi: 10.1002/jmri.26332. Epub 2018 Oct 16
[PubMed PMID: 30325549]
[27]
Pediconi F, Marzocca F, Cavallo Marincola B, Napoli A. MRI-guided treatment in the breast. Journal of magnetic resonance imaging : JMRI. 2018 Dec:48(6):1479-1488. doi: 10.1002/jmri.26282. Epub 2018 Oct 14
[PubMed PMID: 30318672]
[28]
Watanabe Y, Anan K. The decision to perform or omit sentinel lymph node biopsy during mastectomy for ductal carcinoma in situ should be tailored in accordance with preoperative findings. Breast cancer (Tokyo, Japan). 2019 Mar:26(2):261-262. doi: 10.1007/s12282-018-0917-x. Epub 2018 Oct 16
[PubMed PMID: 30328007]
[29]
Verma P, Sharma R, Sharma N, Gulati A, Parashar A, Kaundal A. Fine-Needle Aspiration Cytology versus Core-Needle Biopsy for Breast Lesions: A Dilemma of Superiority between the Two. Acta cytologica. 2021:65(5):411-416. doi: 10.1159/000517005. Epub 2021 Jun 30
[PubMed PMID: 34192704]
[30]
Gradishar WJ, Moran MS, Abraham J, Aft R, Agnese D, Allison KH, Anderson B, Burstein HJ, Chew H, Dang C, Elias AD, Giordano SH, Goetz MP, Goldstein LJ, Hurvitz SA, Isakoff SJ, Jankowitz RC, Javid SH, Krishnamurthy J, Leitch M, Lyons J, Mortimer J, Patel SA, Pierce LJ, Rosenberger LH, Rugo HS, Sitapati A, Smith KL, Smith ML, Soliman H, Stringer-Reasor EM, Telli ML, Ward JH, Wisinski KB, Young JS, Burns J, Kumar R. Breast Cancer, Version 3.2022, NCCN Clinical Practice Guidelines in Oncology. Journal of the National Comprehensive Cancer Network : JNCCN. 2022 Jun:20(6):691-722. doi: 10.6004/jnccn.2022.0030. Epub
[PubMed PMID: 35714673]
Level 1 (high-level) evidence
[31]
Kerr AJ, Dodwell D, McGale P, Holt F, Duane F, Mannu G, Darby SC, Taylor CW. Adjuvant and neoadjuvant breast cancer treatments: A systematic review of their effects on mortality. Cancer treatment reviews. 2022 Apr:105():102375. doi: 10.1016/j.ctrv.2022.102375. Epub 2022 Mar 4
[PubMed PMID: 35367784]
Level 1 (high-level) evidence
[32]
Shien T, Iwata H. Adjuvant and neoadjuvant therapy for breast cancer. Japanese journal of clinical oncology. 2020 Mar 9:50(3):225-229. doi: 10.1093/jjco/hyz213. Epub
[PubMed PMID: 32147701]
[33]
Waks AG, Winer EP. Breast Cancer Treatment: A Review. JAMA. 2019 Jan 22:321(3):288-300. doi: 10.1001/jama.2018.19323. Epub
[PubMed PMID: 30667505]
[38]
Wapnir IL, Dignam JJ, Fisher B, Mamounas EP, Anderson SJ, Julian TB, Land SR, Margolese RG, Swain SM, Costantino JP, Wolmark N. Long-term outcomes of invasive ipsilateral breast tumor recurrences after lumpectomy in NSABP B-17 and B-24 randomized clinical trials for DCIS. Journal of the National Cancer Institute. 2011 Mar 16:103(6):478-88. doi: 10.1093/jnci/djr027. Epub 2011 Mar 11
[PubMed PMID: 21398619]
Level 1 (high-level) evidence
[39]
Litière S, Werutsky G, Fentiman IS, Rutgers E, Christiaens MR, Van Limbergen E, Baaijens MH, Bogaerts J, Bartelink H. Breast conserving therapy versus mastectomy for stage I-II breast cancer: 20 year follow-up of the EORTC 10801 phase 3 randomised trial. The Lancet. Oncology. 2012 Apr:13(4):412-9. doi: 10.1016/S1470-2045(12)70042-6. Epub 2012 Feb 27
[PubMed PMID: 22373563]
Level 1 (high-level) evidence
[40]
Wang X, Xu L, Yin Z, Wang D, Wang Q, Xu K, Zhao J, Zhao L, Yuan Z, Wang P. Locoregional recurrence-associated factors and risk-adapted postmastectomy radiotherapy for breast cancer staged in cT1-2N0-1 after neoadjuvant chemotherapy. Cancer management and research. 2018:10():4105-4112. doi: 10.2147/CMAR.S173628. Epub 2018 Oct 2
[PubMed PMID: 30323666]
[41]
Tang L, Matsushita H, Jingu K. Controversial issues in radiotherapy after breast-conserving surgery for early breast cancer in older patients: a systematic review. Journal of radiation research. 2018 Nov 1:59(6):789-793. doi: 10.1093/jrr/rry071. Epub
[PubMed PMID: 30321392]
Level 1 (high-level) evidence
[42]
Kirby AM. Updated ASTRO guidelines on accelerated partial breast irradiation (APBI): to whom can we offer APBI outside a clinical trial? The British journal of radiology. 2018 May:91(1085):20170565. doi: 10.1259/bjr.20170565. Epub 2018 Mar 23
[PubMed PMID: 29513031]
[43]
Vicini FA, Cecchini RS, White JR, Arthur DW, Julian TB, Rabinovitch RA, Kuske RR, Ganz PA, Parda DS, Scheier MF, Winter KA, Paik S, Kuerer HM, Vallow LA, Pierce LJ, Mamounas EP, McCormick B, Costantino JP, Bear HD, Germain I, Gustafson G, Grossheim L, Petersen IA, Hudes RS, Curran WJ Jr, Bryant JL, Wolmark N. Long-term primary results of accelerated partial breast irradiation after breast-conserving surgery for early-stage breast cancer: a randomised, phase 3, equivalence trial. Lancet (London, England). 2019 Dec 14:394(10215):2155-2164. doi: 10.1016/S0140-6736(19)32514-0. Epub 2019 Dec 5
[PubMed PMID: 31813636]
Level 1 (high-level) evidence
[44]
Bartelink H, Horiot JC, Poortmans PM, Struikmans H, Van den Bogaert W, Fourquet A, Jager JJ, Hoogenraad WJ, Oei SB, Wárlám-Rodenhuis CC, Pierart M, Collette L. Impact of a higher radiation dose on local control and survival in breast-conserving therapy of early breast cancer: 10-year results of the randomized boost versus no boost EORTC 22881-10882 trial. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2007 Aug 1:25(22):3259-65
[PubMed PMID: 17577015]
Level 1 (high-level) evidence
[45]
Overgaard M, Nielsen HM, Tramm T, Højris I, Grantzau TL, Alsner J, Offersen BV, Overgaard J, DBCG Radiotherapy Group. Postmastectomy radiotherapy in high-risk breast cancer patients given adjuvant systemic therapy. A 30-year long-term report from the Danish breast cancer cooperative group DBCG 82bc trial. Radiotherapy and oncology : journal of the European Society for Therapeutic Radiology and Oncology. 2022 May:170():4-13. doi: 10.1016/j.radonc.2022.03.008. Epub 2022 Mar 11
[PubMed PMID: 35288227]
[46]
Donker M, van Tienhoven G, Straver ME, Meijnen P, van de Velde CJ, Mansel RE, Cataliotti L, Westenberg AH, Klinkenbijl JH, Orzalesi L, Bouma WH, van der Mijle HC, Nieuwenhuijzen GA, Veltkamp SC, Slaets L, Duez NJ, de Graaf PW, van Dalen T, Marinelli A, Rijna H, Snoj M, Bundred NJ, Merkus JW, Belkacemi Y, Petignat P, Schinagl DA, Coens C, Messina CG, Bogaerts J, Rutgers EJ. Radiotherapy or surgery of the axilla after a positive sentinel node in breast cancer (EORTC 10981-22023 AMAROS): a randomised, multicentre, open-label, phase 3 non-inferiority trial. The Lancet. Oncology. 2014 Nov:15(12):1303-10. doi: 10.1016/S1470-2045(14)70460-7. Epub 2014 Oct 15
[PubMed PMID: 25439688]
Level 1 (high-level) evidence
[47]
Whelan TJ, Olivotto IA, Parulekar WR, Ackerman I, Chua BH, Nabid A, Vallis KA, White JR, Rousseau P, Fortin A, Pierce LJ, Manchul L, Chafe S, Nolan MC, Craighead P, Bowen J, McCready DR, Pritchard KI, Gelmon K, Murray Y, Chapman JA, Chen BE, Levine MN, MA.20 Study Investigators. Regional Nodal Irradiation in Early-Stage Breast Cancer. The New England journal of medicine. 2015 Jul 23:373(4):307-16. doi: 10.1056/NEJMoa1415340. Epub
[PubMed PMID: 26200977]
[48]
Choi KH, Ahn SJ, Jeong JU, Yu M, Kim JH, Jeong BK, Lee JH, Kim SH, Lee JH. Postoperative radiotherapy with intensity-modulated radiation therapy versus 3-dimensional conformal radiotherapy in early breast cancer: A randomized clinical trial of KROG 15-03. Radiotherapy and oncology : journal of the European Society for Therapeutic Radiology and Oncology. 2021 Jan:154():179-186. doi: 10.1016/j.radonc.2020.09.043. Epub 2020 Sep 24
[PubMed PMID: 32980384]
Level 1 (high-level) evidence
[49]
Pignol JP, Truong P, Rakovitch E, Sattler MG, Whelan TJ, Olivotto IA. Ten years results of the Canadian breast intensity modulated radiation therapy (IMRT) randomized controlled trial. Radiotherapy and oncology : journal of the European Society for Therapeutic Radiology and Oncology. 2016 Dec:121(3):414-419. doi: 10.1016/j.radonc.2016.08.021. Epub 2016 Sep 13
[PubMed PMID: 27637858]
Level 1 (high-level) evidence
[50]
Darby SC, Ewertz M, McGale P, Bennet AM, Blom-Goldman U, Brønnum D, Correa C, Cutter D, Gagliardi G, Gigante B, Jensen MB, Nisbet A, Peto R, Rahimi K, Taylor C, Hall P. Risk of ischemic heart disease in women after radiotherapy for breast cancer. The New England journal of medicine. 2013 Mar 14:368(11):987-98. doi: 10.1056/NEJMoa1209825. Epub
[PubMed PMID: 23484825]
[51]
Epler GR, Kelly EM. Systematic review of postradiotherapy bronchiolitis obliterans organizing pneumonia in women with breast cancer. The oncologist. 2014 Dec:19(12):1216-26. doi: 10.1634/theoncologist.2014-0041. Epub 2014 Oct 31
[PubMed PMID: 25361622]
Level 1 (high-level) evidence
[52]
Taghian AG, Assaad SI, Niemierko A, Floyd SR, Powell SN. Is a reduction in radiation lung volume and dose necessary with paclitaxel chemotherapy for node-positive breast cancer? International journal of radiation oncology, biology, physics. 2005 Jun 1:62(2):386-91
[PubMed PMID: 15890579]
[53]
Williams NR, Williams S, Kanapathy M, Naderi N, Vavourakis V, Mosahebi A. Radiation-induced fibrosis in breast cancer: A protocol for an observational cross-sectional pilot study for personalised risk estimation and objective assessment. International journal of surgery protocols. 2019:14():9-13. doi: 10.1016/j.isjp.2019.02.002. Epub 2019 Feb 13
[PubMed PMID: 31851743]
Level 2 (mid-level) evidence
[54]
Collette S, Collette L, Budiharto T, Horiot JC, Poortmans PM, Struikmans H, Van den Bogaert W, Fourquet A, Jager JJ, Hoogenraad W, Mueller RP, Kurtz J, Morgan DA, Dubois JB, Salamon E, Mirimanoff R, Bolla M, Van der Hulst M, Wárlám-Rodenhuis CC, Bartelink H, EORTC Radiation Oncology Group. Predictors of the risk of fibrosis at 10 years after breast conserving therapy for early breast cancer: a study based on the EORTC Trial 22881-10882 'boost versus no boost'. European journal of cancer (Oxford, England : 1990). 2008 Nov:44(17):2587-99. doi: 10.1016/j.ejca.2008.07.032. Epub 2008 Aug 29
[PubMed PMID: 18757193]
[55]
Jacobson G, Bhatia S, Smith BJ, Button AM, Bodeker K, Buatti J. Randomized trial of pentoxifylline and vitamin E vs standard follow-up after breast irradiation to prevent breast fibrosis, evaluated by tissue compliance meter. International journal of radiation oncology, biology, physics. 2013 Mar 1:85(3):604-8. doi: 10.1016/j.ijrobp.2012.06.042. Epub 2012 Jul 28
[PubMed PMID: 22846413]
Level 1 (high-level) evidence
[56]
Gross JP, Whelan TJ, Parulekar WR, Chen BE, Rademaker AW, Helenowski IB, Donnelly ED, Strauss JB. Development and Validation of a Nomogram to Predict Lymphedema After Axillary Surgery and Radiation Therapy in Women With Breast Cancer From the NCIC CTG MA.20 Randomized Trial. International journal of radiation oncology, biology, physics. 2019 Sep 1:105(1):165-173. doi: 10.1016/j.ijrobp.2019.05.002. Epub 2019 May 11
[PubMed PMID: 31085285]
Level 1 (high-level) evidence
[57]
DiSipio T, Rye S, Newman B, Hayes S. Incidence of unilateral arm lymphoedema after breast cancer: a systematic review and meta-analysis. The Lancet. Oncology. 2013 May:14(6):500-15. doi: 10.1016/S1470-2045(13)70076-7. Epub 2013 Mar 27
[PubMed PMID: 23540561]
Level 1 (high-level) evidence
[58]
Bartels SAL, Donker M, Poncet C, Sauvé N, Straver ME, van de Velde CJH, Mansel RE, Blanken C, Orzalesi L, Klinkenbijl JHG, van der Mijle HCJ, Nieuwenhuijzen GAP, Veltkamp SC, van Dalen T, Marinelli A, Rijna H, Snoj M, Bundred NJ, Merkus JWS, Belkacemi Y, Petignat P, Schinagl DAX, Coens C, van Tienhoven G, van Duijnhoven F, Rutgers EJT. Radiotherapy or Surgery of the Axilla After a Positive Sentinel Node in Breast Cancer: 10-Year Results of the Randomized Controlled EORTC 10981-22023 AMAROS Trial. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2023 Apr 20:41(12):2159-2165. doi: 10.1200/JCO.22.01565. Epub 2022 Nov 16
[PubMed PMID: 36383926]
Level 1 (high-level) evidence
[59]
Pierce SM, Recht A, Lingos TI, Abner A, Vicini F, Silver B, Herzog A, Harris JR. Long-term radiation complications following conservative surgery (CS) and radiation therapy (RT) in patients with early stage breast cancer. International journal of radiation oncology, biology, physics. 1992:23(5):915-23
[PubMed PMID: 1639653]
[60]
Meric F, Buchholz TA, Mirza NQ, Vlastos G, Ames FC, Ross MI, Pollock RE, Singletary SE, Feig BW, Kuerer HM, Newman LA, Perkins GH, Strom EA, McNeese MD, Hortobagyi GN, Hunt KK. Long-term complications associated with breast-conservation surgery and radiotherapy. Annals of surgical oncology. 2002 Jul:9(6):543-9
[PubMed PMID: 12095969]
[61]
Grantzau T, Overgaard J. Risk of second non-breast cancer after radiotherapy for breast cancer: a systematic review and meta-analysis of 762,468 patients. Radiotherapy and oncology : journal of the European Society for Therapeutic Radiology and Oncology. 2015 Jan:114(1):56-65. doi: 10.1016/j.radonc.2014.10.004. Epub 2014 Nov 7
[PubMed PMID: 25454172]
Level 1 (high-level) evidence
[62]
Ye JC, Yan W, Christos P, Nori D, Chao KS, Ravi A. Second cancer, breast cancer, and cardiac mortality in stage T1aN0 breast cancer patients with or without external beam radiation therapy: a national registry study. Clinical breast cancer. 2015 Feb:15(1):54-9. doi: 10.1016/j.clbc.2014.07.003. Epub 2014 Aug 15
[PubMed PMID: 25223278]
[63]
Dracham CB, Shankar A, Madan R. Radiation induced secondary malignancies: a review article. Radiation oncology journal. 2018 Jun:36(2):85-94. doi: 10.3857/roj.2018.00290. Epub 2018 Jun 29
[PubMed PMID: 29983028]
[64]
Burguin A, Diorio C, Durocher F. Breast Cancer Treatments: Updates and New Challenges. Journal of personalized medicine. 2021 Aug 19:11(8):. doi: 10.3390/jpm11080808. Epub 2021 Aug 19
[PubMed PMID: 34442452]
[66]
Syed YY. Oncotype DX Breast Recurrence Score(®): A Review of its Use in Early-Stage Breast Cancer. Molecular diagnosis & therapy. 2020 Oct:24(5):621-632. doi: 10.1007/s40291-020-00482-7. Epub
[PubMed PMID: 32613290]
[67]
Andre F, Ismaila N, Allison KH, Barlow WE, Collyar DE, Damodaran S, Henry NL, Jhaveri K, Kalinsky K, Kuderer NM, Litvak A, Mayer EL, Pusztai L, Raab R, Wolff AC, Stearns V. Biomarkers for Adjuvant Endocrine and Chemotherapy in Early-Stage Breast Cancer: ASCO Guideline Update. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2022 Jun 1:40(16):1816-1837. doi: 10.1200/JCO.22.00069. Epub 2022 Apr 19
[PubMed PMID: 35439025]
[69]
Korde LA, Somerfield MR, Carey LA, Crews JR, Denduluri N, Hwang ES, Khan SA, Loibl S, Morris EA, Perez A, Regan MM, Spears PA, Sudheendra PK, Symmans WF, Yung RL, Harvey BE, Hershman DL. Neoadjuvant Chemotherapy, Endocrine Therapy, and Targeted Therapy for Breast Cancer: ASCO Guideline. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2021 May 1:39(13):1485-1505. doi: 10.1200/JCO.20.03399. Epub 2021 Jan 28
[PubMed PMID: 33507815]
[70]
Wu YT, Xu Z, Zhang K, Wu JS, Li X, Arshad B, Li YC, Wang ZL, Li HY, Wu KN, Kong LQ. Efficacy and cardiac safety of the concurrent use of trastuzumab and anthracycline-based neoadjuvant chemotherapy for HER2-positive breast cancer: a systematic review and meta-analysis. Therapeutics and clinical risk management. 2018:14():1789-1797. doi: 10.2147/TCRM.S176214. Epub 2018 Sep 26
[PubMed PMID: 30310287]
Level 1 (high-level) evidence
[71]
Dieci MV, Vernaci G, Guarneri V. Escalation and de-escalation in HER2 positive early breast cancer. Current opinion in oncology. 2019 Jan:31(1):35-42. doi: 10.1097/CCO.0000000000000492. Epub
[PubMed PMID: 30325338]
Level 3 (low-level) evidence
[72]
Ye F, Dewanjee S, Li Y, Jha NK, Chen ZS, Kumar A, Vishakha, Behl T, Jha SK, Tang H. Advancements in clinical aspects of targeted therapy and immunotherapy in breast cancer. Molecular cancer. 2023 Jul 6:22(1):105. doi: 10.1186/s12943-023-01805-y. Epub 2023 Jul 6
[PubMed PMID: 37415164]
[73]
Husinka L, Koerner PH, Miller RT, Trombatt W. Review of cyclin-dependent kinase 4/6 inhibitors in the treatment of advanced or metastatic breast cancer. Journal of drug assessment. 2020 Dec 18:10(1):27-34. doi: 10.1080/21556660.2020.1857103. Epub 2020 Dec 18
[PubMed PMID: 33414982]
[74]
Debien V, De Caluwé A, Wang X, Piccart-Gebhart M, Tuohy VK, Romano E, Buisseret L. Immunotherapy in breast cancer: an overview of current strategies and perspectives. NPJ breast cancer. 2023 Feb 13:9(1):7. doi: 10.1038/s41523-023-00508-3. Epub 2023 Feb 13
[PubMed PMID: 36781869]
Level 3 (low-level) evidence
[75]
Loibl S, Poortmans P, Morrow M, Denkert C, Curigliano G. Breast cancer. Lancet (London, England). 2021 May 8:397(10286):1750-1769. doi: 10.1016/S0140-6736(20)32381-3. Epub 2021 Apr 1
[PubMed PMID: 33812473]
[76]
Al-Hilli Z, Wilkerson A. Breast Surgery: Management of Postoperative Complications Following Operations for Breast Cancer. The Surgical clinics of North America. 2021 Oct:101(5):845-863. doi: 10.1016/j.suc.2021.06.014. Epub 2021 Aug 7
[PubMed PMID: 34537147]