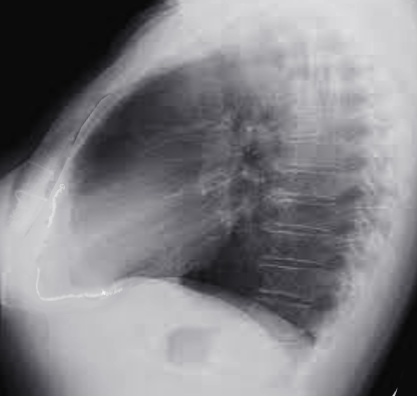
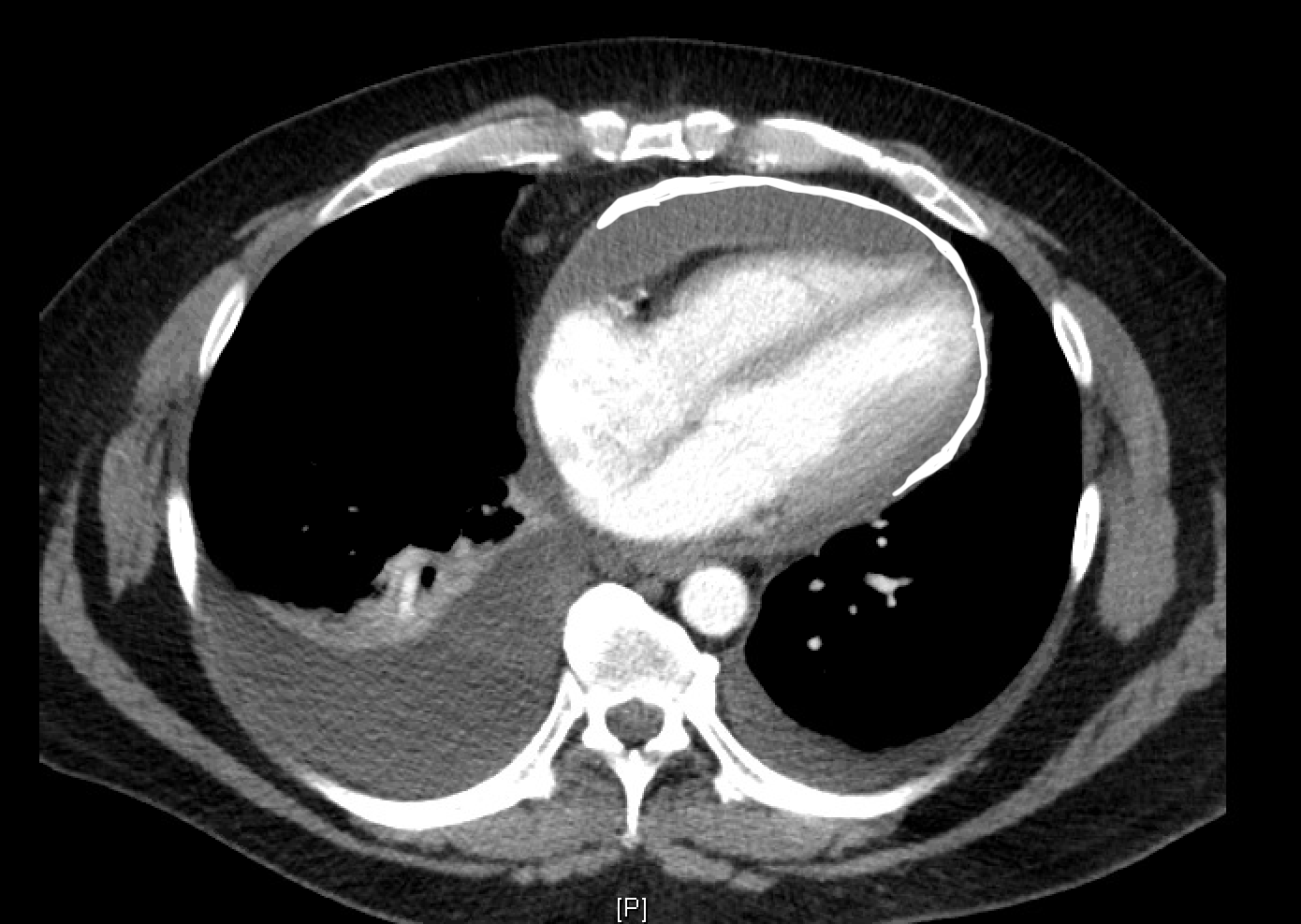
[1]
Watkins MW,LeWinter MM, Physiologic role of the normal pericardium. Annual review of medicine. 1993;
[PubMed PMID: 8476238]
[2]
Nguyen T,Phillips C,Movahed A, Incidental findings of pericardial calcification. World journal of clinical cases. 2014 Sep 16;
[PubMed PMID: 25232549]
Level 3 (low-level) evidence
[3]
Umer A,Khalid N,Chhabra L,Spodick DH, Role of Pericardiectomy in Postcardiac Transplant Constrictive Pericarditis. The Annals of thoracic surgery. 2015 Dec;
[PubMed PMID: 26652564]
[4]
Umer A,Khalid N,Chhabra L,Memon S,Spodick DH, Constrictive pericarditis complicating cardiac transplantation. Journal of cardiothoracic surgery. 2015 Aug 25;
[PubMed PMID: 26302865]
[5]
Cameron J,Oesterle SN,Baldwin JC,Hancock EW, The etiologic spectrum of constrictive pericarditis. American heart journal. 1987 Feb;
[PubMed PMID: 3812191]
[6]
Ling LH,Oh JK,Breen JF,Schaff HV,Danielson GK,Mahoney DW,Seward JB,Tajik AJ, Calcific constrictive pericarditis: is it still with us? Annals of internal medicine. 2000 Mar 21;
[PubMed PMID: 10733443]
[7]
Miranda WR,Oh JK, Constrictive Pericarditis: A Practical Clinical Approach. Progress in cardiovascular diseases. 2017 Jan - Feb;
[PubMed PMID: 28062267]
[9]
Imazio M,Brucato A,Maestroni S,Cumetti D,Belli R,Trinchero R,Adler Y, Risk of constrictive pericarditis after acute pericarditis. Circulation. 2011 Sep 13;
[PubMed PMID: 21844077]
[10]
Strang JI,Kakaza HH,Gibson DG,Girling DJ,Nunn AJ,Fox W, Controlled trial of prednisolone as adjuvant in treatment of tuberculous constrictive pericarditis in Transkei. Lancet (London, England). 1987 Dec 19;
[PubMed PMID: 2891992]
[11]
Ling LH,Oh JK,Schaff HV,Danielson GK,Mahoney DW,Seward JB,Tajik AJ, Constrictive pericarditis in the modern era: evolving clinical spectrum and impact on outcome after pericardiectomy. Circulation. 1999 Sep 28;
[PubMed PMID: 10500037]
[13]
Diaz-Arocutipa C,Saucedo-Chinchay J,Imazio M,Argulian E, Natriuretic peptides to differentiate constrictive pericarditis and restrictive cardiomyopathy: A systematic review and meta-analysis. Clinical cardiology. 2021 Dec 30
[PubMed PMID: 34967020]
Level 1 (high-level) evidence
[14]
Bull RK,Edwards PD,Dixon AK, CT dimensions of the normal pericardium. The British journal of radiology. 1998 Sep;
[PubMed PMID: 10195005]
[15]
Talreja DR,Edwards WD,Danielson GK,Schaff HV,Tajik AJ,Tazelaar HD,Breen JF,Oh JK, Constrictive pericarditis in 26 patients with histologically normal pericardial thickness. Circulation. 2003 Oct 14;
[PubMed PMID: 14517161]
[16]
Wang ZJ,Reddy GP,Gotway MB,Yeh BM,Hetts SW,Higgins CB, CT and MR imaging of pericardial disease. Radiographics : a review publication of the Radiological Society of North America, Inc. 2003 Oct
[PubMed PMID: 14557510]
[17]
Burns N,Shriki JE,Farvid AM,El-Sherief KE,Cunningham MJ,Colletti PM,Shinbane J, Calcific constrictive pericarditis demonstrated on 99mTc-MDP bone scintigraphy. Journal of radiology case reports. 2009;
[PubMed PMID: 22470658]
Level 3 (low-level) evidence
[18]
Al-Mallah MH,Almasoudi F,Ebid M,Ahmed AM,Jamiel A, Multimodality Imaging of Pericardial Diseases. Current treatment options in cardiovascular medicine. 2017 Oct 12;
[PubMed PMID: 29027095]
[19]
Talreja DR,Nishimura RA,Oh JK,Holmes DR, Constrictive pericarditis in the modern era: novel criteria for diagnosis in the cardiac catheterization laboratory. Journal of the American College of Cardiology. 2008 Jan 22;
[PubMed PMID: 18206742]
[20]
Haley JH,Tajik AJ,Danielson GK,Schaff HV,Mulvagh SL,Oh JK, Transient constrictive pericarditis: causes and natural history. Journal of the American College of Cardiology. 2004 Jan 21;
[PubMed PMID: 14736448]
[21]
Chang SA, Oh JK. Constrictive Pericarditis: A Medical or Surgical Disease? Journal of cardiovascular imaging. 2019 Jul:27(3):178-186. doi: 10.4250/jcvi.2019.27.e28. Epub 2019 Apr 30
[PubMed PMID: 31161752]
[22]
Inglessis I I,Dec GW, Constrictive Pericarditis. Current treatment options in cardiovascular medicine. 1999 Jun
[PubMed PMID: 11096470]
[23]
Depboylu BC,Mootoosamy P,Vistarini N,Testuz A,El-Hamamsy I,Cikirikcioglu M, Surgical Treatment of Constrictive Pericarditis. Texas Heart Institute journal. 2017 Apr
[PubMed PMID: 28461794]
[24]
Bertog SC,Thambidorai SK,Parakh K,Schoenhagen P,Ozduran V,Houghtaling PL,Lytle BW,Blackstone EH,Lauer MS,Klein AL, Constrictive pericarditis: etiology and cause-specific survival after pericardiectomy. Journal of the American College of Cardiology. 2004 Apr 21;
[PubMed PMID: 15093882]
[25]
Goland S,Caspi A,Malnick SD, Idiopathic chronic pericardial effusion. The New England journal of medicine. 2000 May 11;
[PubMed PMID: 10809614]