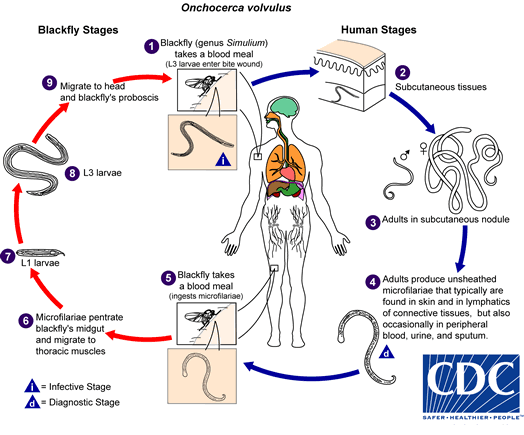
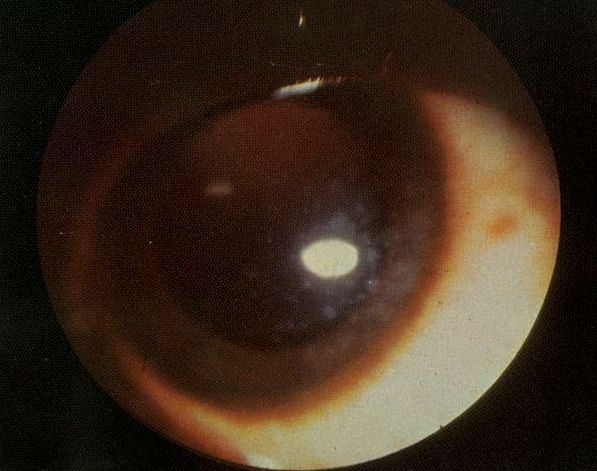
[1]
Herricks JR,Hotez PJ,Wanga V,Coffeng LE,Haagsma JA,Basáñez MG,Buckle G,Budke CM,Carabin H,Fèvre EM,Fürst T,Halasa YA,King CH,Murdoch ME,Ramaiah KD,Shepard DS,Stolk WA,Undurraga EA,Stanaway JD,Naghavi M,Murray CJL, The global burden of disease study 2013: What does it mean for the NTDs? PLoS neglected tropical diseases. 2017 Aug;
[PubMed PMID: 28771480]
[2]
Post RJ,Onyenwe E,Somiari SA,Mafuyai HB,Crainey JL,Ubachukwu PO, A guide to the Simulium damnosum complex (Diptera: Simuliidae) in Nigeria, with a cytotaxonomic key for the identification of the sibling species. Annals of tropical medicine and parasitology. 2011 Jun;
[PubMed PMID: 21871165]
[3]
Duke BO, The population dynamics of Onchocerca volvulus in the human host. Tropical medicine and parasitology : official organ of Deutsche Tropenmedizinische Gesellschaft and of Deutsche Gesellschaft fur Technische Zusammenarbeit (GTZ). 1993 Jun;
[PubMed PMID: 8367667]
[4]
Schulz-Key H, Observations on the reproductive biology of Onchocerca volvulus. Acta Leidensia. 1990;
[PubMed PMID: 2378210]
[5]
Udall DN, Recent updates on onchocerciasis: diagnosis and treatment. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2007 Jan 1;
[PubMed PMID: 17143815]
[6]
Mackenzie CD,Huntington MK,Wanji S,Lovato RV,Eversole RR,Geary TG, The association of adult Onchocerca volvulus with lymphatic vessels. The Journal of parasitology. 2010 Feb;
[PubMed PMID: 19803543]
[7]
Taylor MJ,Awadzi K,Basáñez MG,Biritwum N,Boakye D,Boatin B,Bockarie M,Churcher TS,Debrah A,Edwards G,Hoerauf A,Mand S,Matthews G,Osei-Atweneboana M,Prichard RK,Wanji S,Adjei O, Onchocerciasis Control: Vision for the Future from a Ghanian perspective. Parasites
[PubMed PMID: 19154624]
Level 3 (low-level) evidence
[8]
Komlan K,Vossberg PS,Gantin RG,Solim T,Korbmacher F,Banla M,Padjoudoum K,Karabou P,Köhler C,Soboslay PT, Onchocerca volvulus infection and serological prevalence, ocular onchocerciasis and parasite transmission in northern and central Togo after decades of Simulium damnosum s.l. vector control and mass drug administration of ivermectin. PLoS neglected tropical diseases. 2018 Mar;
[PubMed PMID: 29494606]
[9]
Budden FH, Route of entry of Onchocerca volvulus microfilariae into the eye. Transactions of the Royal Society of Tropical Medicine and Hygiene. 1976;
[PubMed PMID: 982529]
[10]
Basak SK,Hazra TK,Bhattacharya D, Persistent corneal edema secondary to presumed dead adult filarial worm in the anterior chamber. Indian journal of ophthalmology. 2007 Jan-Feb;
[PubMed PMID: 17189893]
[11]
Egbert PR,Jacobson DW,Fiadoyor S,Dadzie P,Ellingson KD, Onchocerciasis: a potential risk factor for glaucoma. The British journal of ophthalmology. 2005 Jul;
[PubMed PMID: 15965151]
[12]
Berger RB,Blackwell NM,Lass JH,Diaconu E,Pearlman E, IL-4 and IL-13 regulation of ICAM-1 expression and eosinophil recruitment in Onchocerca volvulus keratitis. Investigative ophthalmology
[PubMed PMID: 12202520]
[13]
Keiser PB,Reynolds SM,Awadzi K,Ottesen EA,Taylor MJ,Nutman TB, Bacterial endosymbionts of Onchocerca volvulus in the pathogenesis of posttreatment reactions. The Journal of infectious diseases. 2002 Mar 15;
[PubMed PMID: 11920298]
[14]
Higazi TB,Filiano A,Katholi CR,Dadzie Y,Remme JH,Unnasch TR, Wolbachia endosymbiont levels in severe and mild strains of Onchocerca volvulus. Molecular and biochemical parasitology. 2005 May;
[PubMed PMID: 15811532]
[15]
Hall LR,Pearlman E, Pathogenesis of onchocercal keratitis (River blindness). Clinical microbiology reviews. 1999 Jul;
[PubMed PMID: 10398675]
[16]
Semba RD,Murphy RP,Newland HS,Awadzi K,Greene BM,Taylor HR, Longitudinal study of lesions of the posterior segment in onchocerciasis. Ophthalmology. 1990 Oct;
[PubMed PMID: 2243684]
[17]
Banla M,Tchalim S,Karabou PK,Gantin RG,Agba AI,Kére-Banla A,Helling-Giese G,Heuschkel C,Schulz-Key H,Soboslay PT, Sustainable control of onchocerciasis: ocular pathology in onchocerciasis patients treated annually with ivermectin for 23 years: a cohort study. PloS one. 2014;
[PubMed PMID: 24887413]
[18]
Braun G,McKechnie NM,Connor V,Gilbert CE,Engelbrecht F,Whitworth JA,Taylor DW, Immunological crossreactivity between a cloned antigen of Onchocerca volvulus and a component of the retinal pigment epithelium. The Journal of experimental medicine. 1991 Jul 1;
[PubMed PMID: 2056276]
[19]
Chandrashekar R,Ogunrinade AF,Alvarez RM,Kale OO,Weil GJ, Circulating immune complex-associated parasite antigens in human onchocerciasis. The Journal of infectious diseases. 1990 Nov;
[PubMed PMID: 2230240]
[20]
Bird AC,el-Sheikh H,Anderson J,Fuglsang H, Changes in visual function and in the posterior segment of the eye during treatment of onchocerciasis with diethylcarbamazine citrate. The British journal of ophthalmology. 1980 Mar;
[PubMed PMID: 7387952]
[21]
Paul EV,Zimmerman LE, Some observations on the ocular pathology of onchocerciasis. Human pathology. 1970 Dec;
[PubMed PMID: 5535426]
[22]
RODGER FC, The pathogenesis and pathology of ocular on-chocerciasis. Part IV. The pathology. American journal of ophthalmology. 1960 Mar;
[PubMed PMID: 14438157]
[23]
Lupi O,Downing C,Lee M,Pino L,Bravo F,Giglio P,Sethi A,Klaus S,Sangueza OP,Fuller C,Mendoza N,Ladizinski B,Woc-Colburn L,Tyring SK, Mucocutaneous manifestations of helminth infections: Nematodes. Journal of the American Academy of Dermatology. 2015 Dec;
[PubMed PMID: 26568337]
[24]
Ali MM,Baraka OZ,AbdelRahman SI,Sulaiman SM,Williams JF,Homeida MM,Mackenzie CD, Immune responses directed against microfilariae correlate with severity of clinical onchodermatitis and treatment history. The Journal of infectious diseases. 2003 Feb 15;
[PubMed PMID: 12599094]
[25]
Tamarozzi F,Halliday A,Gentil K,Hoerauf A,Pearlman E,Taylor MJ, Onchocerciasis: the role of Wolbachia bacterial endosymbionts in parasite biology, disease pathogenesis, and treatment. Clinical microbiology reviews. 2011 Jul;
[PubMed PMID: 21734243]
[26]
Semba RD,Donnelly JJ,Young E,Green WR,Scott AL,Taylor HR, Experimental ocular onchocerciasis in cynomolgus monkeys. IV. Chorioretinitis elicited by Onchocerca volvulus microfilariae. Investigative ophthalmology
[PubMed PMID: 2016131]
[28]
Showler AJ,Nutman TB, Imported onchocerciasis in migrants and travelers. Current opinion in infectious diseases. 2018 Oct;
[PubMed PMID: 30113326]
Level 3 (low-level) evidence
[29]
Unnasch TR,Golden A,Cama V,Cantey PT, Diagnostics for onchocerciasis in the era of elimination. International health. 2018 Mar 1;
[PubMed PMID: 29471336]
[30]
Anderson J,Fuglsang H, Further studies on the treatment of ocular onchocerciasis with diethylcarbamazine and suramin. The British journal of ophthalmology. 1978 Jul;
[PubMed PMID: 678497]
[31]
Basáñez MG,Pion SD,Boakes E,Filipe JA,Churcher TS,Boussinesq M, Effect of single-dose ivermectin on Onchocerca volvulus: a systematic review and meta-analysis. The Lancet. Infectious diseases. 2008 May;
[PubMed PMID: 18471776]
Level 1 (high-level) evidence
[32]
Opoku NO,Bakajika DK,Kanza EM,Howard H,Mambandu GL,Nyathirombo A,Nigo MM,Kasonia K,Masembe SL,Mumbere M,Kataliko K,Larbelee JP,Kpawor M,Bolay KM,Bolay F,Asare S,Attah SK,Olipoh G,Vaillant M,Halleux CM,Kuesel AC, Single dose moxidectin versus ivermectin for Onchocerca volvulus infection in Ghana, Liberia, and the Democratic Republic of the Congo: a randomised, controlled, double-blind phase 3 trial. Lancet (London, England). 2018 Oct 6;
[PubMed PMID: 29361335]
Level 1 (high-level) evidence
[33]
Hoerauf A,Mand S,Volkmann L,Büttner M,Marfo-Debrekyei Y,Taylor M,Adjei O,Büttner DW, Doxycycline in the treatment of human onchocerciasis: Kinetics of Wolbachia endobacteria reduction and of inhibition of embryogenesis in female Onchocerca worms. Microbes and infection. 2003 Apr;
[PubMed PMID: 12706439]
[34]
Evangelisto A,Werth V,Schumacher HR, What is that nodule? A diagnostic approach to evaluating subcutaneous and cutaneous nodules. Journal of clinical rheumatology : practical reports on rheumatic
[PubMed PMID: 17023809]
[35]
Sauerbrey M, The Onchocerciasis Elimination Program for the Americas (OEPA). Annals of tropical medicine and parasitology. 2008 Sep;
[PubMed PMID: 18718151]
[36]
Fischer PU,King CL,Jacobson JA,Weil GJ, Potential Value of Triple Drug Therapy with Ivermectin, Diethylcarbamazine, and Albendazole (IDA) to Accelerate Elimination of Lymphatic Filariasis and Onchocerciasis in Africa. PLoS neglected tropical diseases. 2017 Jan;
[PubMed PMID: 28056015]
[37]
Vinkeles Melchers NVS,Coffeng LE,Boussinesq M,Pedrique B,Pion SDS,Tekle AH,Zouré HGM,Wanji S,Remme JH,Stolk WA, Projected number of people with onchocerciasis-loiasis co-infection in Africa, 1995 to 2025. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2019 Jul 13;
[PubMed PMID: 31304961]
[38]
Wanji S,Kengne-Ouafo JA,Esum ME,Chounna PW,Tendongfor N,Adzemye BF,Eyong JE,Jato I,Datchoua-Poutcheu FR,Kah E,Enyong P,Taylor DW, Situation analysis of parasitological and entomological indices of onchocerciasis transmission in three drainage basins of the rain forest of South West Cameroon after a decade of ivermectin treatment. Parasites
[PubMed PMID: 25886166]
[39]
Kelly-Hope L,Paulo R,Thomas B,Brito M,Unnasch TR,Molyneux D, Loa loa vectors Chrysops spp.: perspectives on research, distribution, bionomics, and implications for elimination of lymphatic filariasis and onchocerciasis. Parasites
[PubMed PMID: 28381279]
Level 3 (low-level) evidence
[40]
Gardon J,Gardon-Wendel N,Demanga-Ngangue,Kamgno J,Chippaux JP,Boussinesq M, Serious reactions after mass treatment of onchocerciasis with ivermectin in an area endemic for Loa loa infection. Lancet (London, England). 1997 Jul 5;
[PubMed PMID: 9217715]
[41]
Francis H,Awadzi K,Ottesen EA, The Mazzotti reaction following treatment of onchocerciasis with diethylcarbamazine: clinical severity as a function of infection intensity. The American journal of tropical medicine and hygiene. 1985 May;
[PubMed PMID: 4003668]
[42]
Awadzi K,Opoku NO,Attah SK,Lazdins-Helds JK,Kuesel AC, Diagnosis of O. volvulus infection via skin exposure to diethylcarbamazine: clinical evaluation of a transdermal delivery technology-based patch. Parasites
[PubMed PMID: 26452723]
[43]
Stingl P,Pierce PF,Connor DH,Gibson DW,Straessle T,Ross MA,Ribas JL, Does dexamethasone suppress the Mazzotti reaction in patients with onchocerciasis? Acta tropica. 1988 Mar;
[PubMed PMID: 2896449]
[44]
Chandler RE, Serious Neurological Adverse Events after Ivermectin-Do They Occur beyond the Indication of Onchocerciasis? The American journal of tropical medicine and hygiene. 2018 Feb;
[PubMed PMID: 29210346]
[45]
Newland HS,White AT,Greene BM,Murphy RP,Taylor HR, Ocular manifestations of onchocerciasis in a rain forest area of west Africa. The British journal of ophthalmology. 1991 Mar;
[PubMed PMID: 2012784]
[46]
Cooper PJ,Proaño R,Beltran C,Anselmi M,Guderian RH, Onchocerciasis in Ecuador: ocular findings in Onchocerca volvulus infected individuals. The British journal of ophthalmology. 1995 Feb;
[PubMed PMID: 7696237]
[47]
Akama T,Freund YR,Berry PW,Carter DS,Easom EE,Jarnagin K,Lunde CS,Plattner JJ,Rock F,Stefanakis R,Fischer C,Bulman CA,Lim KC,Suzuki BM,Tricoche N,Mansour A,DiCosty U,McCall S,Carson B,McCall JW,McKerrow J,Hübner MP,Specht S,Hoerauf A,Lustigman S,Sakanari JA,Jacobs RT, Macrofilaricidal Benzimidazole-Benzoxaborole Hybrids as an Approach to the Treatment of River Blindness: Part 1. Amide Linked Analogs. ACS infectious diseases. 2020 Feb 14;
[PubMed PMID: 31876154]