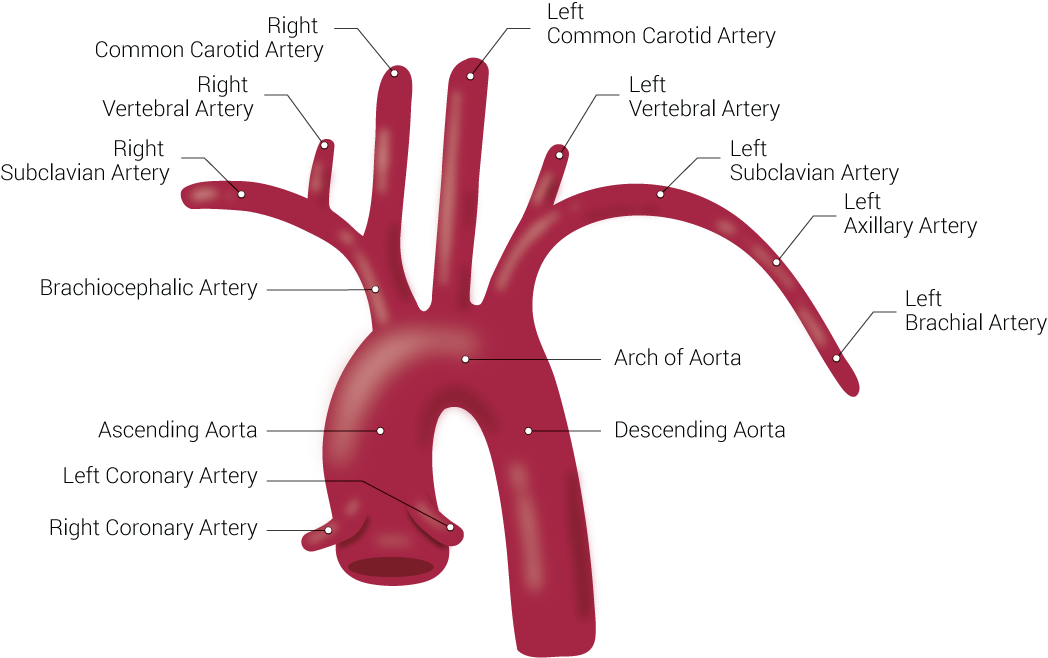
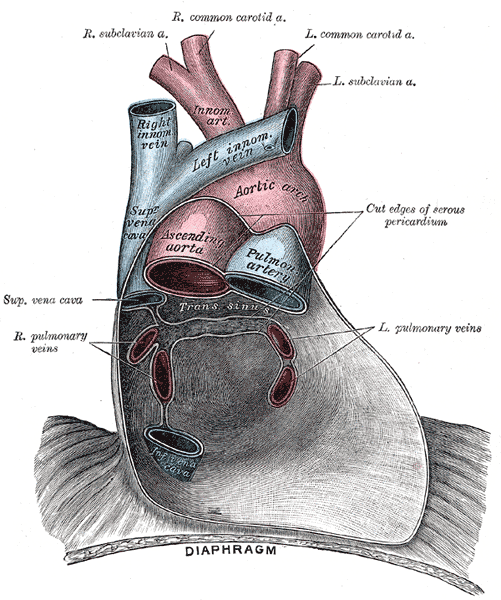
[1]
Collins JA, Munoz JV, Patel TR, Loukas M, Tubbs RS. The anatomy of the aging aorta. Clinical anatomy (New York, N.Y.). 2014 Apr:27(3):463-6. doi: 10.1002/ca.22384. Epub 2014 Feb 12
[PubMed PMID: 24523152]
[2]
Komutrattananont P, Mahakkanukrauh P, Das S. Morphology of the human aorta and age-related changes: anatomical facts. Anatomy & cell biology. 2019 Jun:52(2):109-114. doi: 10.5115/acb.2019.52.2.109. Epub 2019 Jun 30
[PubMed PMID: 31338225]
[3]
Loukas M, Bilinsky E, Bilinsky S, Blaak C, Tubbs RS, Anderson RH. The anatomy of the aortic root. Clinical anatomy (New York, N.Y.). 2014 Jul:27(5):748-56. doi: 10.1002/ca.22295. Epub 2013 Sep 2
[PubMed PMID: 24000000]
[4]
Dagenais F. Anatomy of the thoracic aorta and of its branches. Thoracic surgery clinics. 2011 May:21(2):219-27, viii. doi: 10.1016/j.thorsurg.2010.12.004. Epub
[PubMed PMID: 21477772]
[5]
FELLER I, WOODBURNE RT. Surgical anatomy of the abdominal aorta. Annals of surgery. 1961 Dec:154(6)Suppl(Suppl 6):239-52
[PubMed PMID: 13892214]
[6]
Peidro J, Boufi M, Loundou AD, Hartung O, Dona B, Vernet F, Bensoussan D, Alimi YS. Aortic Anatomy and Complications of the Proximal Sealing Zone after Endovascular Treatment of the Thoracic Aorta. Annals of vascular surgery. 2018 Apr:48():141-150. doi: 10.1016/j.avsg.2017.09.029. Epub 2017 Dec 8
[PubMed PMID: 29225128]
[7]
Santos PVD, Barbosa ABM, Targino VA, Silva NA, Silva YCM, Barbosa F, Oliveira ASB, Assis TO. ANATOMICAL VARIATIONS OF THE CELIAC TRUNK: A SYSTEMATIC REVIEW. Arquivos brasileiros de cirurgia digestiva : ABCD = Brazilian archives of digestive surgery. 2018 Dec 6:31(4):e1403. doi: 10.1590/0102-672020180001e1403. Epub 2018 Dec 6
[PubMed PMID: 30539978]
Level 1 (high-level) evidence
[8]
Negoi I, Beuran M, Hostiuc S, Negoi RI, Inoue Y. Surgical Anatomy of the Superior Mesenteric Vessels Related to Colon and Pancreatic Surgery: A Systematic Review and Meta-Analysis. Scientific reports. 2018 Mar 8:8(1):4184. doi: 10.1038/s41598-018-22641-x. Epub 2018 Mar 8
[PubMed PMID: 29520096]
Level 1 (high-level) evidence
[9]
Kau T, Sinzig M, Gasser J, Lesnik G, Rabitsch E, Celedin S, Eicher W, Illiasch H, Hausegger KA. Aortic development and anomalies. Seminars in interventional radiology. 2007 Jun:24(2):141-52. doi: 10.1055/s-2007-980040. Epub
[PubMed PMID: 21326792]
[10]
Murono K, Kawai K, Kazama S, Ishihara S, Yamaguchi H, Sunami E, Kitayama J, Watanabe T. Anatomy of the inferior mesenteric artery evaluated using 3-dimensional CT angiography. Diseases of the colon and rectum. 2015 Feb:58(2):214-9. doi: 10.1097/DCR.0000000000000285. Epub
[PubMed PMID: 25585080]
[11]
Schleich JM. Images in cardiology. Development of the human heart: days 15-21. Heart (British Cardiac Society). 2002 May:87(5):487
[PubMed PMID: 11997429]
[12]
Sano M, Unno N, Sasaki T, Baba S, Sugisawa R, Tanaka H, Inuzuka K, Yamamoto N, Sato K, Konno H. Topologic distributions of vasa vasorum and lymphatic vasa vasorum in the aortic adventitia--Implications for the prevalence of aortic diseases. Atherosclerosis. 2016 Apr:247():127-34. doi: 10.1016/j.atherosclerosis.2016.02.007. Epub 2016 Feb 12
[PubMed PMID: 26897260]
[13]
Beveridge TS, Johnson M, Power A, Power NE, Allman BL. Anatomy of the nerves and ganglia of the aortic plexus in males. Journal of anatomy. 2015 Jan:226(1):93-103. doi: 10.1111/joa.12251. Epub 2014 Nov 9
[PubMed PMID: 25382240]
[14]
Weinberg PM. Aortic arch anomalies. Journal of cardiovascular magnetic resonance : official journal of the Society for Cardiovascular Magnetic Resonance. 2006:8(4):633-43
[PubMed PMID: 16869315]
[15]
Fox EB, Latham GJ, Ross FJ, Joffe D. Perioperative and Anesthetic Management of Coarctation of the Aorta. Seminars in cardiothoracic and vascular anesthesia. 2019 Jun:23(2):212-224. doi: 10.1177/1089253218821953. Epub 2019 Jan 7
[PubMed PMID: 30614372]
[16]
Tchervenkov CI, Jacobs JP, Sharma K, Ungerleider RM. Interrupted aortic arch: surgical decision making. Seminars in thoracic and cardiovascular surgery. Pediatric cardiac surgery annual. 2005:():92-102
[PubMed PMID: 15818364]
[17]
Holloway BJ, Rosewarne D, Jones RG. Imaging of thoracic aortic disease. The British journal of radiology. 2011 Dec:84 Spec No 3(Spec Iss 3):S338-54. doi: 10.1259/bjr/30655825. Epub
[PubMed PMID: 22723539]
[18]
DE BAKEY ME, COOLEY DA, CREECH O Jr. Surgical considerations of dissecting aneurysm of the aorta. Annals of surgery. 1955 Oct:142(4):586-610; discussion, 611-2
[PubMed PMID: 13259422]
[19]
Carpenter SW, Kodolitsch YV, Debus ES, Wipper S, Tsilimparis N, Larena-Avellaneda A, Diener H, Kölbel T. Acute aortic syndromes: definition, prognosis and treatment options. The Journal of cardiovascular surgery. 2014 Apr:55(2 Suppl 1):133-44
[PubMed PMID: 24796906]
[20]
Dijkema EJ, Leiner T, Grotenhuis HB. Diagnosis, imaging and clinical management of aortic coarctation. Heart (British Cardiac Society). 2017 Aug:103(15):1148-1155. doi: 10.1136/heartjnl-2017-311173. Epub 2017 Apr 4
[PubMed PMID: 28377475]
[21]
González-Salvado V, Bazal P, Alonso-González R. Aortic Coarctation With Extensive Collateral Circulation. Circulation. Cardiovascular imaging. 2018 Aug:11(8):e007918. doi: 10.1161/CIRCIMAGING.118.007918. Epub
[PubMed PMID: 30354495]
[22]
Wahl L, Tubbs RS. A review of the clinical anatomy of hypertension. Clinical anatomy (New York, N.Y.). 2019 Jul:32(5):678-681. doi: 10.1002/ca.23369. Epub 2019 Apr 3
[PubMed PMID: 30873636]
[23]
Bossone E, Pluchinotta FR, Andreas M, Blanc P, Citro R, Limongelli G, Della Corte A, Parikh A, Frigiola A, Lerakis S, Ehrlich M, Aboyans V. Aortitis. Vascular pharmacology. 2016 May:80():1-10. doi: 10.1016/j.vph.2015.11.084. Epub 2015 Dec 22
[PubMed PMID: 26721213]
[25]
Wooten C, Hayat M, du Plessis M, Cesmebasi A, Koesterer M, Daly KP, Matusz P, Tubbs RS, Loukas M. Anatomical significance in aortoiliac occlusive disease. Clinical anatomy (New York, N.Y.). 2014 Nov:27(8):1264-74. doi: 10.1002/ca.22444. Epub 2014 Jul 25
[PubMed PMID: 25065617]