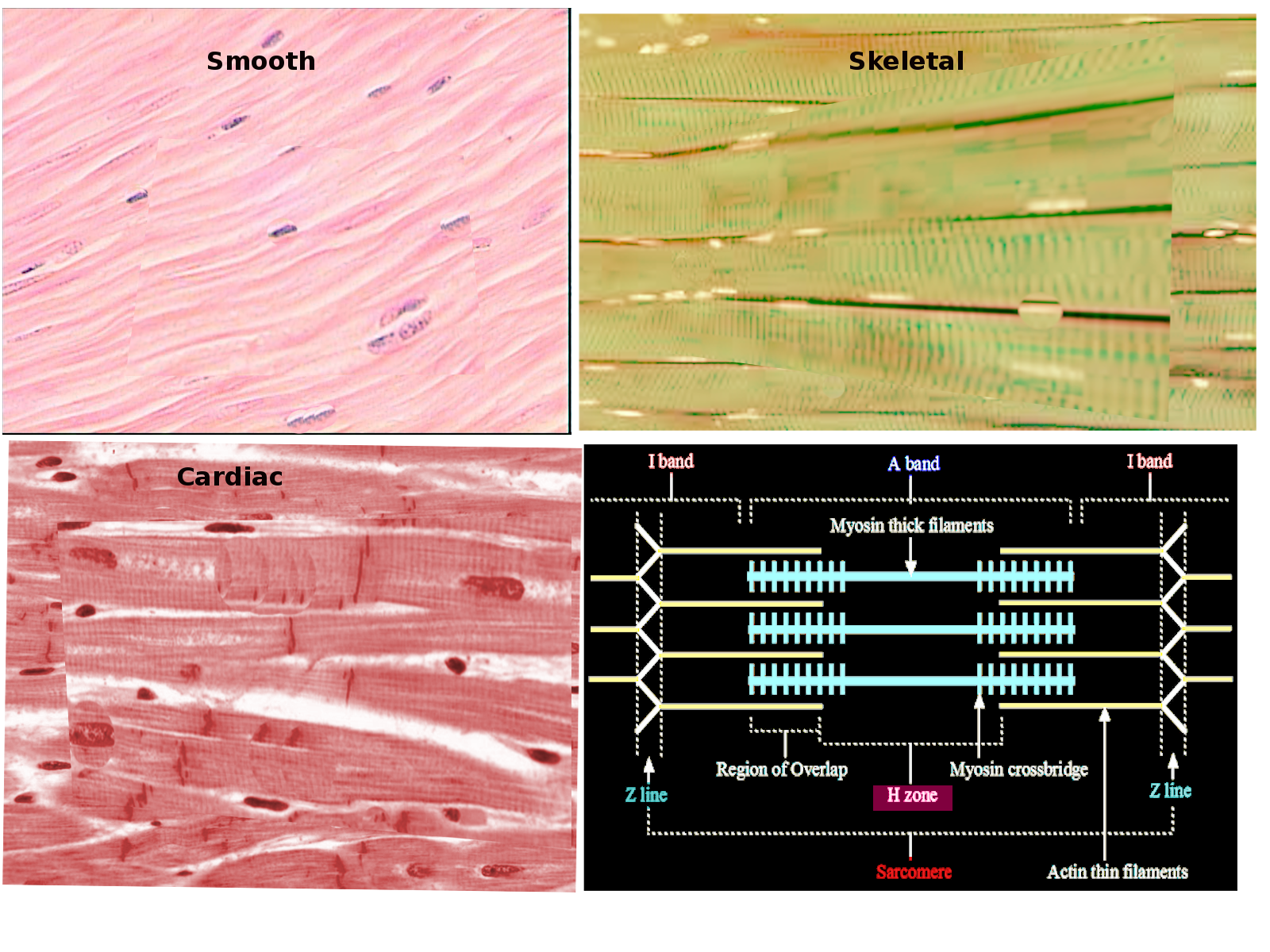
[1]
Shadrin IY, Khodabukus A, Bursac N. Striated muscle function, regeneration, and repair. Cellular and molecular life sciences : CMLS. 2016 Nov:73(22):4175-4202
[PubMed PMID: 27271751]
[2]
Brozovich FV, Nicholson CJ, Degen CV, Gao YZ, Aggarwal M, Morgan KG. Mechanisms of Vascular Smooth Muscle Contraction and the Basis for Pharmacologic Treatment of Smooth Muscle Disorders. Pharmacological reviews. 2016 Apr:68(2):476-532. doi: 10.1124/pr.115.010652. Epub
[PubMed PMID: 27037223]
[3]
Jafri MS. Models of excitation-contraction coupling in cardiac ventricular myocytes. Methods in molecular biology (Clifton, N.J.). 2012:910():309-35. doi: 10.1007/978-1-61779-965-5_14. Epub
[PubMed PMID: 22821602]
[4]
McLoon LK, Vicente A, Fitzpatrick KR, Lindström M, Pedrosa Domellöf F. Composition, Architecture, and Functional Implications of the Connective Tissue Network of the Extraocular Muscles. Investigative ophthalmology & visual science. 2018 Jan 1:59(1):322-329. doi: 10.1167/iovs.17-23003. Epub
[PubMed PMID: 29346490]
[5]
Henderson CA, Gomez CG, Novak SM, Mi-Mi L, Gregorio CC. Overview of the Muscle Cytoskeleton. Comprehensive Physiology. 2017 Jun 18:7(3):891-944. doi: 10.1002/cphy.c160033. Epub 2017 Jun 18
[PubMed PMID: 28640448]
Level 3 (low-level) evidence
[6]
Sieck GC, Ferreira LF, Reid MB, Mantilla CB. Mechanical properties of respiratory muscles. Comprehensive Physiology. 2013 Oct:3(4):1553-67. doi: 10.1002/cphy.c130003. Epub
[PubMed PMID: 24265238]
[7]
Birnkrant DJ, Bushby K, Bann CM, Apkon SD, Blackwell A, Brumbaugh D, Case LE, Clemens PR, Hadjiyannakis S, Pandya S, Street N, Tomezsko J, Wagner KR, Ward LM, Weber DR, DMD Care Considerations Working Group. Diagnosis and management of Duchenne muscular dystrophy, part 1: diagnosis, and neuromuscular, rehabilitation, endocrine, and gastrointestinal and nutritional management. The Lancet. Neurology. 2018 Mar:17(3):251-267. doi: 10.1016/S1474-4422(18)30024-3. Epub 2018 Feb 3
[PubMed PMID: 29395989]
Level 3 (low-level) evidence
[8]
Banks GB, Combs AC, Odom GL, Bloch RJ, Chamberlain JS. Muscle structure influences utrophin expression in mdx mice. PLoS genetics. 2014 Jun:10(6):e1004431. doi: 10.1371/journal.pgen.1004431. Epub 2014 Jun 12
[PubMed PMID: 24922526]
[9]
Goldstein SA, Newby LK, Cyr DD, Neely M, Lüscher TF, Brown EB, White HD, Ohman EM, Roe MT, Hamm CW. Relationship Between Peak Troponin Values and Long-Term Ischemic Events Among Medically Managed Patients With Acute Coronary Syndromes. Journal of the American Heart Association. 2017 Apr 11:6(4):. doi: 10.1161/JAHA.116.005334. Epub 2017 Apr 11
[PubMed PMID: 28400368]
[10]
Eakins F,Harford JJ,Knupp C,Roessle M,Squire JM, Different Myosin Head Conformations in Bony Fish Muscles Put into Rigor at Different Sarcomere Lengths. International journal of molecular sciences. 2018 Jul 18
[PubMed PMID: 30022010]