Neuroanatomy, Pupillary Light Reflexes and Pathway
Neuroanatomy, Pupillary Light Reflexes and Pathway
Introduction
The visual (retino-thalamocortical) pathway and pupillary light reflex pathway are the two essential ways through which the eye perceives and responds to changes in the environment. Multiple relays of information processing efficiently proceed from the cornea to the brain, and any lesion in the visual or the pupillary reflex pathway may result in visual pathology.
Structure and Function
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Structure and Function
Visual System and Pathway
Cornea
The cornea is the first anterior part of the eye that refracts light and helps transfer neural signals to the brain via the visual pathway. While the air-cornea interface is responsible for approximately 65% of the total refractive power of the eye, the corneal power has a fixed focus.[1][2]
Iris and Pupil
The iris is the colored section located posterior to the anterior chamber of the eye. It surrounds the opening known as the pupil and allows the light to pass through the hole and reach the retina for phototransduction. The pupil either constricts or dilates to respond to light and near stimuli. The pupillary sphincter and dilator muscles are the two involuntary iris muscles that are required to control the amount of light traveling to the retina. Images displayed onto the retina are upside-down and backward.[3]
Lens
The lens and the neighboring ciliary muscle are responsible for accommodation and focusing light onto the retina from different distances. The accommodation reflex occurs when the ciliary muscle contraction leads to the relaxation of the zonular fibers attached to the lens. As a result, the lens shape becomes more spherical, increasing its axial thickness and dioptric power, and bringing the focus, which is initially behind the retina, back onto the retina. The ciliary muscle receives parasympathetic inputs from the short ciliary nerve, and its contraction allows the eye to accommodate to nearer objects. When the ciliary muscle relaxes, an increase in zonular tension causes the lens to flatten and its dioptric power to decrease. The ability to accommodate progressively declines with age. Presbyopia is a common condition in the aging population where their lens becomes denser and more rigid. With its decreasing ability to change shape, the lens can no longer focus on nearer objects.[4][5][6]
Retina
When the light reaches the retina, it must initially pass through multiple retinal layers before interacting with rods and cones in the photoreceptor outer segments. These two photoreceptors translate the light signal into a bioelectric message. Upon photon absorption, the activated rods and cones lead to the initiation of a phototransduction cascade and result in cell hyperpolarization, which decreases the release of neurotransmitter glutamate from the photoreceptors. The glutamate receptors on bipolar cells propagate signals to ganglion cells, which have axons in the retinal nerve fiber layer that travel through the optic nerve.[1]
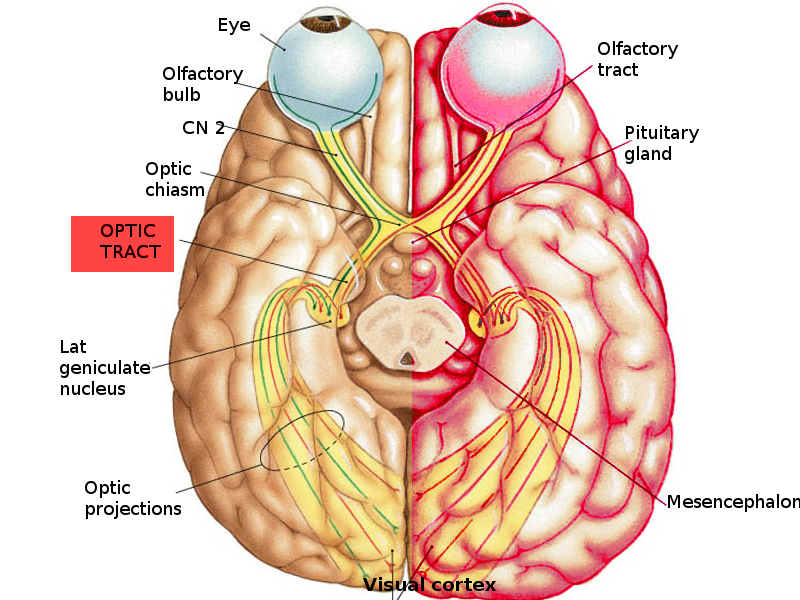
Optic Nerve, Chiasm, and Tracts
The optic nerve of each eye is composed of a group of unmyelinated axons of the retinal ganglion cells which emerge from the optic disc. The absence of retinal photoreceptors in the optic disc leads to a blind spot in the visual field of each eye. The optic nerve passes posteriorly from the disc through a mesh-like structure known as the lamina cribrosa, after which it becomes myelinated by oligodendrocytes.
The optic nerves traveling from both eyes meet at the optic chiasm anterior to the pituitary gland. The nerve fibers continue after the optic chiasm as the optic tracts. The retinal fibers which come from the nasal side of each eye decussate at the optic chiasm into the contralateral optic tract, whereas the retinal fibers from the temporal side of each eye do not cross but instead proceed through the ipsilateral optic tract; thus, each optic tract is composed of the ipsilateral temporal fibers and the contralateral nasal fibers. This decussation of the nasal, but not the retinal, fibers allows the right half of the visual field to be processed by the left cerebral hemisphere and the left half of the field to proceed to the right hemisphere.[7][8]
Lateral Geniculate Nucleus
Most of the optic tract fibers synapse in the ipsilateral lateral geniculate nucleus (LGN) of the thalamus for image processing. A small number of the optic tract fibers arrive at the pretectal nucleus in the midbrain instead of the LGN and participate in the pupillary light reflex. The axons of the LGN project to the calcarine (also known as striate) cortex as two bundles called temporal and parietal radiations. The temporal radiations, also known as Meyer’s loop, are fibers that relay information from the contralateral superior visual field. The parietal radiations, also known as Baum’s loop, are fibers that carry information from the contralateral inferior visual field.[1]
Calcarine Cortex
The electrochemical signal finally arrives at the calcarine cortex (Brodmann area 17), which is the primary visual cortex in the occipital lobe. The visual information is further processed by the higher-order visual cortex to identify an object.[1]
Pupillary Light Reflexes and Pathway
The pupillary light reflex pathway is similar to the visual pathway; however, the optic tract fibers involved in pupillary light reflexes terminate at the pretectal nucleus in the midbrain and not at the LGN of the thalamus. The nasally aligned fibers decussate at the optic chiasm and transfer the signal to the contralateral pretectal nucleus, whereas the temporally aligned fibers relay the information to the ipsilateral pretectal nucleus.
When each pretectal nucleus projects bilaterally and synapses in both Edinger-Westphal nuclei (cranial nerve III), the activated Edinger-Westphal nuclei begin the efferent limb of the reflex by generating action potentials. The axons of these preganglionic parasympathetic neurons send the signals along the oculomotor nerve to the post-ganglionic nerve fibers of the ciliary ganglion. Subsequently, the short ciliary nerves arising from the ciliary ganglion stimulate the pupillary sphincter muscle and cause pupillary constriction. Even when the light is shined in only one eye, a consensual response occurs in the other eye since the nasal retinal fibers cross at the optic chiasm to reach the contralateral pretectal nucleus. Also, the projection of each pretectal nucleus to both Edinger-Westphal nuclei contributes to the consensual pupillary reflex in the contralateral eye. Each activated Edinger-Westphal nucleus is responsible for the ipsilateral pupillary constriction, and these stimulated nuclei together allow the bilateral pupillary reflex to occur.
In the dim light, pupillary dilator muscle fibers contract and widen the size of the pupil. The postganglionic sympathetic fibers from the long ciliary nerve innervate the dilator muscle.[9][4][10][11]
Embryology
The ocular development of an embryo begins at approximately three weeks into gestation and continues until week 10. The optic cup and optic stalk are the invaginations from the optic vesicle, which develops from the neuroectoderm of the diencephalon of the brain. The retina, iris, and ciliary body all arise from the optic cup, whereas the optic nerve forms from the optic stalk. The lens placode develops from the thickened surface ectoderm, and this precursor later gives rise to the lens and anterior corneal epithelium. The mesodermal layer forms the white area of the external eye known as the sclera, part of the vitreous body, and extraocular muscles. The neural crest cells participate in the development of the pupillary muscles of the iris and the ciliary muscle of the lens. The hyaloid artery and vein are the primitive blood vessels that later form the central artery and vein of the retina.[4]
Blood Supply and Lymphatics
The ophthalmic artery is the first arterial branch of the internal carotid artery that provides the blood supply to the majority of components in the eye. Together with the optic nerve, the ophthalmic artery travels through the optic canal to enter the orbit of the eye. The central retinal artery, the anterior ciliary arteries, and posterior ciliary arteries are the essential vascular branches that arise from the ophthalmic artery. The central retinal artery pierces the meninges from the inferior aspect of the optic nerve. It emerges from the optic cup of the disc to supply blood to the inner layers of the retina. The posterior ciliary arteries provide blood supply to the head of the optic nerve, choroid, and the outer layers of the retina. The anterior ciliary arteries are the crucial vasculatures for the iris and ciliary body.
Venous drainage of the retina is carried out by the central retinal vein, which travels temporally to the central retinal artery near the head of the optic nerve. From the central vein, the blood drains into the superior ophthalmic vein that projects to the cavernous sinus. The choroidal veins drain into vortex veins, which project to the superior and inferior ophthalmic veins and then to the cavernous sinus.[12][1]
Nerves
The optic nerve (cranial nerve II) is the essential nerve for relaying visual signals to the brain. Pupillary light reflexes require both optic and oculomotor nerve (cranial nerve III) to constrict pupils upon light exposure. The optic nerve mediates the afferent limb of the pupillary reflex, whereas the oculomotor nerve involves the efferent limb of the reflex. The oculomotor nerve is also responsible for innervation of the ciliary muscle, which mediates lens accommodation, and the sphincter pupillae muscle, which controls pupillary constriction. The postganglionic sympathetic fibers from the superior cervical ganglion innervate the dilator pupillae muscle. These sympathetic fibers “hitchhike” on the long ciliary nerve that arises from the nasociliary nerve of the ophthalmic branch (cranial nerve V). The trigeminal nerve branches give rise to the ophthalmic nerve.[11][10]
Clinical Significance
The visual pathway and pupillary light reflex pathway are complex coordinated systems in which multiple components participate with precision. Even one lesion in the pathway can severely deteriorate the quality of vision. The location of the lesion is associated with the extent and type of vision deficit.
Optic Nerve
A lesion in the optic nerve anterior to the optic chiasm leads to vision loss in the ipsilateral eye. Damage to the right optic nerve will result in the right anopia and compromise the entire visual field in the right eye. Damage to the optic nerve will also affect the pupillary reflex pathway. When a light is shined in the left eye with a damaged optic nerve, the light will not project to the retina for further response and pupillary constriction will be absent in both eyes. When a light is shined in the other eye without a damaged optic nerve, bilateral pupillary constriction will occur, which confirms that the optic nerve, not the oculomotor nerve, is damaged in the left eye.
Oculomotor Nerve
Damage to the oculomotor nerve, which presents parasympathetic outputs, results in sympathetic actions that are not balanced by the parasympathetic. These sympathetic symptoms include pupillary dilation, reduced or loss of pupillary light reflex, and compromised accommodation of the lens. When a light is shined in the left eye with a damaged oculomotor nerve, the light will project onto the retina, and the signal will arrive at bilateral Edinger-Westphal nuclei. However, only the right eye will show the pupillary constriction because the left eye’s efferent pathway has suffered disruption. A light shining in the right eye will also lead to a monocular pupillary constriction in the right eye and not the left eye.
Visual System
Damage at the optic chiasm leads to the bitemporal hemianopia. This condition frequently results from a pituitary lesion due to the proximity of the optic chiasm to the pituitary gland or an aneurysm of the anterior communicating artery. When the optic chiasm injury is present, the nasal retinal fibers that decussate at the chiasm get damaged. The temporal retinal fibers that do not cross but ipsilaterally project to the lateral geniculate nucleus will be unaffected. Because the images displayed on the back of the eye are upside-down and backward, the temporal half of the visual field projected onto the nasal portion of the retina would be lost. When the optic tract is damaged, the same side of the visual field is affected on both eyes. Each optic tract relays the visual signal from both eyes by projecting the ipsilateral temporal fibers and the contralateral nasal fibers. When the right optic tract gets injured, the left side of the visual field would be lost in both eyes and result in the left homonymous hemianopia.
Media
References
Prasad S, Galetta SL. Anatomy and physiology of the afferent visual system. Handbook of clinical neurology. 2011:102():3-19. doi: 10.1016/B978-0-444-52903-9.00007-8. Epub [PubMed PMID: 21601061]
Level 3 (low-level) evidenceSridhar MS. Anatomy of cornea and ocular surface. Indian journal of ophthalmology. 2018 Feb:66(2):190-194. doi: 10.4103/ijo.IJO_646_17. Epub [PubMed PMID: 29380756]
Bouffard MA. The Pupil. Continuum (Minneapolis, Minn.). 2019 Oct:25(5):1194-1214. doi: 10.1212/CON.0000000000000771. Epub [PubMed PMID: 31584534]
Smith AM, Czyz CN. Neuroanatomy, Cranial Nerve 2 (Optic). StatPearls. 2023 Jan:(): [PubMed PMID: 29939684]
Hejtmancik JF, Shiels A. Overview of the Lens. Progress in molecular biology and translational science. 2015:134():119-27. doi: 10.1016/bs.pmbts.2015.04.006. Epub 2015 May 27 [PubMed PMID: 26310153]
Level 3 (low-level) evidenceWolffsohn JS, Davies LN. Presbyopia: Effectiveness of correction strategies. Progress in retinal and eye research. 2019 Jan:68():124-143. doi: 10.1016/j.preteyeres.2018.09.004. Epub 2018 Sep 19 [PubMed PMID: 30244049]
Kidd D. The optic chiasm. Clinical anatomy (New York, N.Y.). 2014 Nov:27(8):1149-58. doi: 10.1002/ca.22385. Epub 2014 May 14 [PubMed PMID: 24824063]
Ireland AC, Carter IB. Neuroanatomy, Optic Chiasm. StatPearls. 2023 Jan:(): [PubMed PMID: 31194427]
Belliveau AP, Somani AN, Dossani RH. Pupillary Light Reflex. StatPearls. 2023 Jan:(): [PubMed PMID: 30725865]
McDougal DH, Gamlin PD. Autonomic control of the eye. Comprehensive Physiology. 2015 Jan:5(1):439-73. doi: 10.1002/cphy.c140014. Epub [PubMed PMID: 25589275]
Level 3 (low-level) evidenceRuskell GL. Access of autonomic nerves through the optic canal, and their orbital distribution in man. The anatomical record. Part A, Discoveries in molecular, cellular, and evolutionary biology. 2003 Nov:275(1):973-8 [PubMed PMID: 14533171]
Hayreh SS. Orbital vascular anatomy. Eye (London, England). 2006 Oct:20(10):1130-44 [PubMed PMID: 17019411]