Introduction
Antibiotic resistance is a growing problem. Resistance exists for all known antibiotics, and multi-drug resistant (MDR) pathogens are becoming more common.[1][2] There are fewer new antimicrobials in the pipeline to combat antimicrobials that have developed resistance.[3] Gram-negative bacteria have developed the broadest spectrum of resistance due to multiple structural adaptations and antibiotic degradation enzymes called beta-lactamases.[4] Some of these antibiotic degradation enzymes include extended-spectrum beta-lactamases (ESBL), which are enzymes that hydrolyze penicillins, third-generation cephalosporins, and monobactams; AmpC beta-lactamases, which can hydrolyze most penicillins, a wide array of cephalosporins, and most beta-lactam beta-lactamase combinations; and carbapenemases, which can hydrolyze carbapenems and a wide variety of other beta-lactams.[4][5] The presence of each of these has significant implications for antimicrobial selection.
Carbapenemase-producing Enterobacterales (CRE) are of particular concern as they either possess a carbapenemase, a beta-lactamase capable of hydrolyzing antibiotics in the carbapenem class, or display resistance to a carbapenem in which the organism is not intrinsically resistant.[6][4] Carbapenems are broad-spectrum and are typically reserved for treating MDR pathogens.[7] Structurally, carbapenems are similar to penicillins but have the addition of a pyrroline ring.[8] Both antibiotics degrade the bacterial cell wall at the penicillin-binding protein via a beta-lactam ring; however, adding a pyrroline ring protects the beta-lactam ring from some degrading enzymes produced by the bacterium.[8] CRE have developed resistance mechanisms against carbapenems, which has severe implications for the individual patient and hospital epidemiology.[4]
Specialized knowledge is required when diagnosing, treating, and preventing CRE infections. Antimicrobial resistance is a dynamic and rapidly changing topic, and staying informed on the latest data regarding resistance is imperative to maintain excellent patient care. Maintaining excellent antimicrobial stewardship practices is imperative to decreasing rates of CRE in our communities.[9] Bacteria may have multiple resistance mechanisms to carbapenems, but the most common is carbapenemase enzyme production.[10][11][12] These carbapenemase-producing carbapenem-resistant Enterobacterales use diverse mechanisms to degrade antibiotics.[6] Other resistance mechanisms include increased efflux pump action and specific porin blocking at the bacterial cell membrane to limit the antibiotics' permeability, rendering them ineffective as they cannot access the bacterial cell.[4]
Etiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Etiology
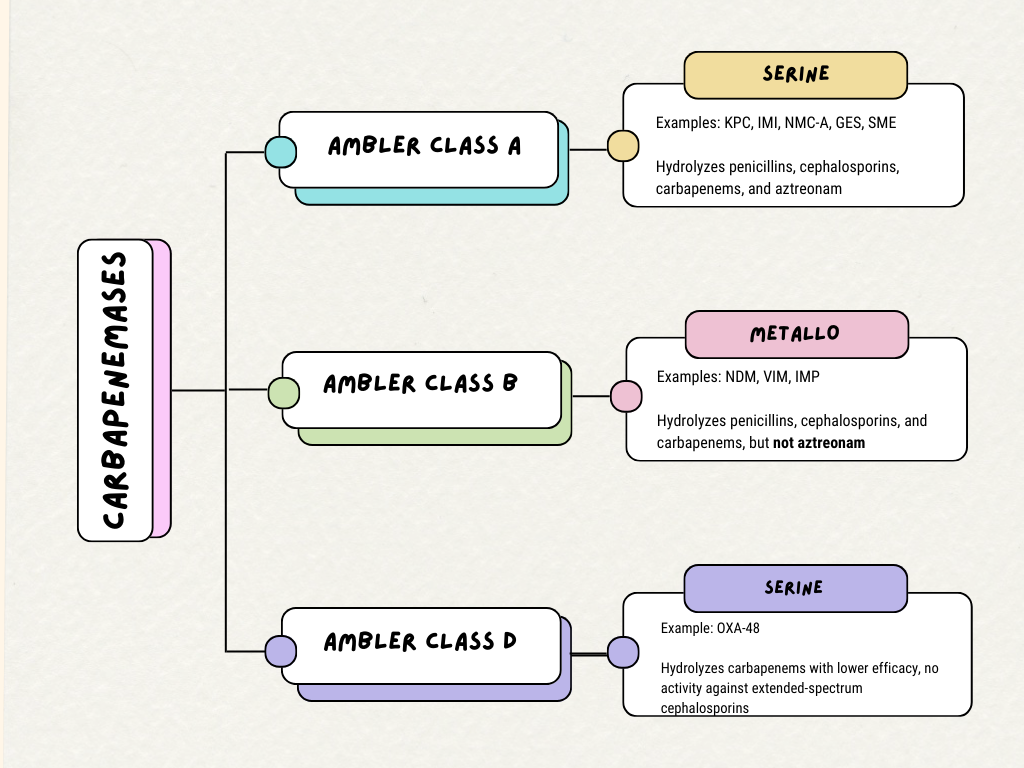
Beta-lactamases are commonly grouped according to the Ambler classification system.[13] Beta-lactamases in different classifications work via differing mechanisms.[14] Carbapenemase enzymes can be categorized into Ambler classes A, B, and D. (see Image. Carbapenemase Ambler Classes).[13]
Class A Carbapenemases
The most common class A carbapenemases include Klebsiella pneumoniae carbapenemase (KPC), imipenem-hydrolysing beta-lactamase (IMI), non-metallo carbapenemase of class A (NMC-A), Guyana extended-spectrum beta-lactamase (GES), and Serratia marscescens enzyme (SME).[14] Class A carbapenemases possess a serine amino acid at their binding sites and are characterized by their ability to hydrolyze penicillins, cephalosporins, carbapenems, and aztreonam.[14] KPC is the most prevalent carbapenemase gene amongst all carbapenemase-producing CRE and is the most commonly found in the United States.[14][15]
Class B Carbapenemases
Class B is defined by metallo-beta-lactamase (MBL) structures.[14] This class possesses amino acids in the binding site that bind to zinc.[16] These enzymes include NDM (New Delhi metallo-lactamase), IMP (Imipenem-resistant Pseudomonas), and VIM (Verona integron-encoded metallo-lactamase).[14] Most class B carbapenemases hydrolyze all beta-lactams except for aztreonam.[16] These carbapenemases are usually found on plasmid vectors and other transposable elements, allowing them to spread between bacteria.[16] Due to a high sequence variability of 15% to 70%, these enzymes are difficult to detect with molecular testing, slowing investigations to more fully understand their prevalence.[15]
Class D Carbapenemases
Oxacillin-hydrolyzing carbapenemase (OXA) enzymes make up class D carbapenemases.[14] Like class A carbapenemases, class D carbapenemases also include a serine amino acid at their binding sites.[14] They differ from class A carbapenemases as they have decreased hydrolyzing activity against carbapenems and penicillins, have no activity against extended-spectrum cephalosporins, and are not inhibited by older beta-lactamase inhibitors (eg, clavulanic acid, tazobactam, or sulbactam).[14][17][18][17] However, most are inhibited by avibactam.[17] These genes are genetically similar to ESBL genes, thus making them difficult to differentiate from other ESBL producers on molecular testing.[12] OXA-48 is the most common isolate in this class and is typically found in Klebsiella pneumoniae.
Epidemiology
KPC has emerged as the most common carbapenemase-producing CRE.[19] KPC is considered endemic in several regions, including the United States, Mexico, Portugal, Italy, Israel, and Greece, reflecting its establishment in bacterial populations of these areas.[20] Oxa-48-like carbapenemases have gained a footprint in the Middle East, North Africa, and much of Europe, while NDM carbapenemases are prevalent in South Asia, the Mediterranean, Romania, Denmark, and Poland.[18][21][22] IMP carbapenemases are notably present in the Asia-Pacific region, including Australia, China, and Japan, while Oxa-48-like carbapenemases are commonly found in Turkey.[22] The frequent movement of populations across borders can facilitate the movement of different carbapenemases to different regions.[22]
Molecular testing, such as PCR, has been the most effective way of understanding the epidemiology and spread of carbapenemase-producing CRE.[22] This testing modality can identify specific strains of CRE and map their range within a hospital or worldwide. Genetic variability in carbapenemase genes may hinder the molecular identification of CRE strains, which could cause phenotypic resistance that is missed by assays that detect specific gene sequences.[23]
Patients usually acquire carbapenemase-producing CRE infections in the hospital and long-term care facilities.[24] The majority of these patients have a long history of healthcare facility exposure due to unrelated diagnoses. If a patient is found to have a CRE infection, healthcare personnel need to utilize proper infection control protocols to prevent localized outbreaks.[25]
Pathophysiology
The prototypical beta-lactam antibiotic, penicillin G, consists of a beta-lactam ring, thiazolidine ring, and an R group, or side chain.[26] R groups help to determine the spectrum of activity of the antibiotic, help to protect against beta-lactamases, and determine some pharmacokinetic properties of the antibiotic.[26] Penicillin impairs the function of the bacterial cell wall at the penicillin-binding protein (PBP) by preventing the transpeptidation of peptidoglycan, leading to the breakdown of the cell wall and eventual destruction of the organism.[26] The synthesis of beta-lactamases can induce bacterial resistance to antibiotics.[27] These enzymes hydrolyze the amide bonds in the beta-lactam rings.[27] Other beta-lactam antibiotics, such as cephalosporins or carbapenems, have a similar mechanism of action at the PBP.[28][8] Still, the addition of different R groups on the adjacent thiazolidine ring and the addition of a pyrroline ring in the instance of carbapenems helps to shield against beta-lactamases, increase affinity for the PBP, and increase the stability of the antibiotic without hindering antimicrobial activity.[29] Carbapenemases overcome these obstacles, rendering the antibiotic susceptible to degradation.[30]
Pathogenic bacteria have gained the ability to circumvent carbapenem hydrolysis through intrinsic and acquired mechanisms.[31] These mechanisms include enzymatic inactivation, porin selectivity, and efflux pumps.[31] The earliest instances of carbapenem resistance appeared in bacteria with inherent resistance. Aeromonas hydrophilia, Serratia marcescens, and Enterobacter cloacae were some of the first environmental organisms discovered to resist carbapenem.[32] Plasmids play a prominent role in the propagation of carbapenem resistance.[33]
Carbapenems require transport into the bacterial intermembrane space via outer membrane proteins (OMPs), also known as porin channels, to reach the PBP.[29] Thus, the loss of porin channels decreases the drug's ability to reach its site of action.[31] Efflux pumps work in this space to remove the irregular or hostile molecules and antibiotics, further regulating the intramembrane environment.[31] Non-carbapenemase-producing CRE resistance mechanisms act in this space to prevent access to the PBP and do not produce a carbapenem-specific beta-lactamase.[12] Generally, efflux pumps and porin mutations do not confer high-level carbapenem resistance and usually help to confer resistance by coexisting with carbapenemases.[31] In some strains, Pseudomonas aeruginosa combines increased efflux pump expression with down-regulation of OprD porin, leading to increased carbapenem resistance.[31] OprD porin resistance to imipenem has also appeared in Enterobacter aerogenes and Klebsiella spp.[34]
History and Physical
CRE infection can involve any site infected by Enterobacterales species, including the urinary tract, lungs, abdomen, surgical site, and bloodstream. CRE infections and colonization are significant problems in hospitals and extended-care facilities.[35] Long-term and varied antibiotic exposure increases the risk of bacterial resistance.[36] Clinicians should obtain a history of hospitalizations and residence in extended-care facilities, noting any history of antimicrobial treatment, which is essential information to collect during history taking to assess a patient's risk for CRE infections. Other risk factors include the presence of a urinary catheter and previous colonization with CRE.[36] Patients hospitalized or in extended-care facilities with a history of CRE infection or colonization should be put under isolation precautions to limit the risk of the spread of CRE to other patients.[37]
Carbapenem resistance does not confer increased pathogenicity, making initial presentation similar to other infections caused by a less resistant strain of the same organism. Therapeutic failure of empiric therapy may indicate a resistant organism, possibly CRE. Identification of CRE requires the identification of the organism and either susceptibility testing or molecular detection of resistance genes in the laboratory.[37] As obtaining results can take several days, clinicians should prompt a change in empiric therapy if CRE is suspected.
Evaluation
Phenotypic diagnosis requires bacterial culture and identification.[14] Disk diffusion, E-test, or automated susceptibility testing is done to identify the carbapenem resistance phenotype.[14] Determining the minimum inhibitory concentration (MIC) for carbapenems allows one to ascertain carbapenem resistance by comparing it to predetermined carbapenem breakpoints.[14] The drawbacks of this process include the time required to perform this testing, usually several days, and the limited information obtained on specific resistance mechanisms.[14] This becomes particularly relevant as although the MIC for carbapenems may fall within the susceptible range, a carbapenemase could still be present.[14] If an isolate is suspected of harboring a carbapenemase due to susceptibility patterns, this should be confirmed with molecular testing.[14]
Another commonly employed phenotypic test is chromogenic media, which reacts and turns color in the presence of resistance enzymes produced by CRE.[14][38] The Carba NP is also a cost-effective testing modality recommended for carbapenemase detection in Enterobacterales.[14] Performing the test typically takes a few minutes to 2 hours and has high sensitivity and specificity.[14] Matrix-assisted laser desorption/ionization-time of flight (MALDI-TOF) is the newer technology for identifying CRE.[14] This technique uses laser ionization of bacterial proteins to determine their molecular weight and create a proteomic profile.[14] Hydrolysis of carbapenems can be detected via a change in the molecular weight of the compounds, which can be detected by MALDI-TOF and extrapolated to identify CRE status.[38] This method is demonstrated to have 98.9% and 97.1% sensitivity and specificity, respectively.[38] Cost, lack of access to MALDI-TOF units, and relative newness of this method are the most significant barriers to further utilization of this diagnostic modality.[14] The modified Hodge test, which detects the amount of carbapenem left in agar in the presence of a suspected carbapenemase producer, is no longer uniformly recommended as it has a low sensitivity for MBL-producing bacteria and has a high rate of both false positives and false negatives.[14]
Molecular identification can be performed in hours and quickly determine the type of resistance mechanism involved.[14] However, this method indicates the presence of a resistance gene and does not give susceptibility testing results.[14] Early detection of CRE infections helps to narrow down the best treatments and decrease the likelihood of spreading the infection. The most common modality of detection is multiplex PCR and microarray technologies.[14] Multiple PCR panels are commercially available.[14] Although these tests quickly determine the presence of pre-selected resistance genes, not all genes that confer resistance are available on these panels, and they are not used extensively due to the cost of these tests.[14]
The decision on which testing modality to employ is determined by the resources of the individual laboratory and the clinical picture, which determines the urgency of diagnosis. Available susceptibilities typically suggest CRE, and confirmatory testing, such as a Carba NP test or molecular identification, is employed for further information. In more critical situations, a combination of phenotypic and molecular identification tests can be utilized simultaneously so empiric therapy can be appropriately tailored once susceptibility testing is available.
Treatment / Management
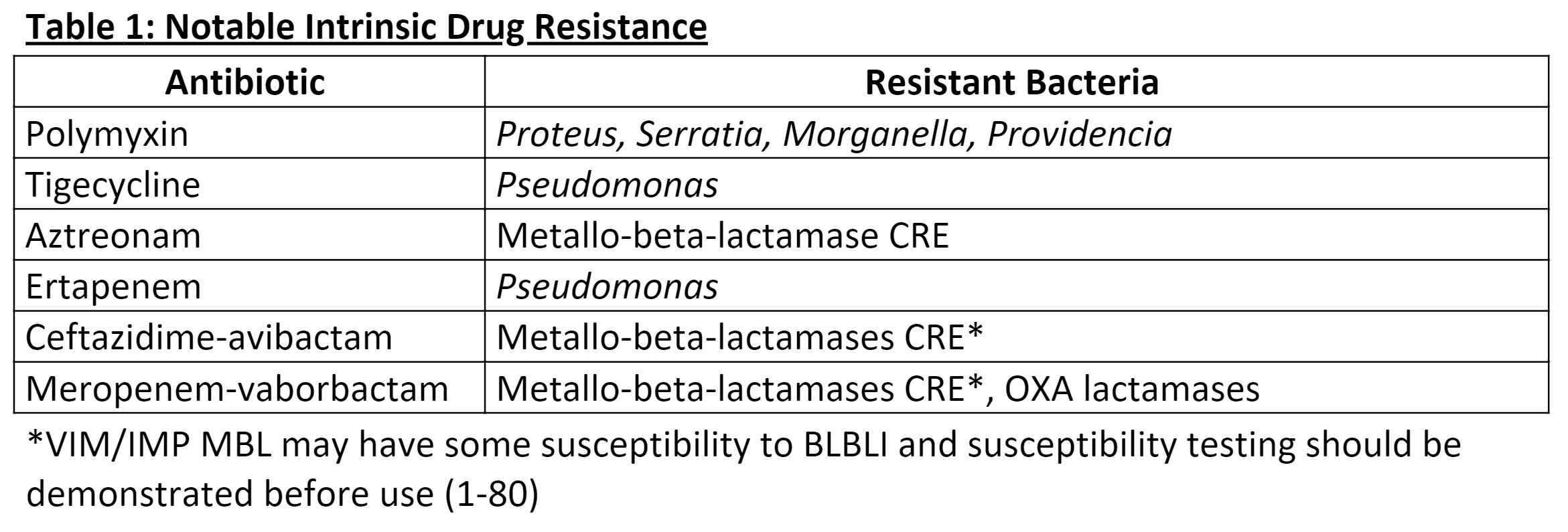
Treatment of CRE depends on the infection site, the isolated pathogen, the resistance profile, and species-specific native resistances (see Table. Intrinsic Drug Resistance). Due to the complexity and variability of possible treatments, plans should be driven by in vitro susceptibility testing.[39] The duration of therapy should be directed by guidelines relating to the specific infection being treated. Additionally, infectious disease specialists and microbiologists should be consulted for further recommendations. Antibiotics that may have activity against CRE include carbapenems, cefiderocol, a siderophore cephalosporin, polymyxins, aminoglycosides, tigecycline, fluoroquinolones, trimethoprim-sulfamethoxazole, and beta-lactam/beta-lactamase inhibitors (BLBLI).[40][41][40](B3)
Uncomplicated Cystitis
For uncomplicated cystitis, trimethoprim/sulfamethoxazole, nitrofurantoin, a dose of an aminoglycoside, or a fluoroquinolone (eg, ciprofloxacin or levofloxacin) are recommended if susceptible.[40] Fosfomycin could be considered in E. coli CRE isolates.[40] If meropenem MIC is ≤1 and carbapenemase testing is negative or unavailable, meropenem can be used as it is unlikely that a carbapenemase is present when meropenem susceptibility is preserved.[40] If other agents are resistant and the isolate is not susceptible to meropenem or a carbapenemase is present, other agents such as ceftazidime-avibactam, imipenem-cilastatin-relebactam, cefiderocol, or meropenem-vaborbactam should be used, guided by susceptibility testing.[40] Colistin should be considered if no other option is available.[40]
Complicated Urinary Tract Infections
Complicated urinary tract infections (cUTI) with CRE usually affect patients with recurrent UTIs who have had repeated exposure to multiple antibiotics. Susceptibility and urine drug concentrations should drive effective antibiotic therapy. Fluoroquinolones and trimethoprim-sulfamethoxazole are preferred therapies for cUTI if susceptible.[40] In isolates with a meropenem MIC ≤1, extended infusion meropenem may be considered in isolates without a carbapenemase or when carbapenemase testing is unavailable.[40] Otherwise, other agents such as ceftazidime-avibactam, imipenem-cilastatin-relebactam, cefiderocol, or meropenem-vaborbactam should be used, as guided by susceptibility testing.[40] Aminoglycoside therapy could additionally be considered while monitoring for nephrotoxicity.[40]
Infections Outside the Urinary Tract
Infections outside the urinary tract with meropenem MICs ≤1, no carbapenemase noted, or no carbapenemase testing is unavailable should be treated with extended-infusion meropenem.[40] If the meropenem cannot be used, ceftazidime-avibactam therapy is recommended.[40]
In meropenem-resistant infections, ceftazidime-avibactam, imipenem-cilastatin-relebactam, or meropenem-vaborbactam can be used.[40] If the patient has received medical care in an area of the world that has a high prevalence of metallo-beta-lactamases or has been previously infected or colonized, either ceftazidime-avibactam plus aztreonam or cefiderocol is recommended if carbapenemase testing is unavailable due to increased risk for a metallo-beta-lactamase.[40] Previously, combination therapy with polymixins and aminoglycosides was recommended, but this is no longer recommended due to the severe nephrotoxicity of this regimen.[40] Effective BLBLIs and cefiderocol have been developed that have better side effect profiles.[40]
Carbapenemase-producing Enterobacterales
For carbapenemase-producing isolates outside of the urinary tract, the recommended treatment differs based on the carbapenemase present. For KPC isolates, either ceftazidime-avibactam, imipenem-cilastatin-relebactam, or meropenem-vaborbactam is recommended.[40] However, for metallo-beta-lactamase producers, cefiderocol or ceftazidime-avibactam plus aztreonam is recommended.[40] BLBLIs (eg, ceftazidime-avibactam, meropenem-vaborbactam, and imipenem-cilastatin-relebactam) are ineffective against metallo-beta-lactamase producers.[40] Conversely, aztreonam is typically effective.[40] Combining treatment with ceftazidime-avibactam and aztreonam is recommended as ESBL-producing isolates and OXA-48-like carbapenemases often coexist in metallo-beta-lactamase producers.[40] For OXA-48-like carbapenemases, ceftazidime-avibactam is preferred, with cefiderocol as an alternative agent.[40]
Tigecycline and eravacycline could be considered alternative agents in patients with isolates that are not susceptible to beta-lactams or have contraindications to them.[40] Still, they should not be used as monotherapy for bloodstream infections or urinary infections due to low serum and urinary concentrations.[40] Additionally, tigecycline has been shown to have increased mortality when used for monotherapy for severe infections.[42] Colistin and other polymixins are not recommended for infections other than uncomplicated cystitis.[40](A1)
Differential Diagnosis
Possible causes of carbapenem antibiotic resistance:
- Carbapenemase production
- Decreased porin expression
- Increased efflux pump action [43]
Prognosis
The severity of the infection determines the prognosis of the patient. Prognosis can be adversely affected if access to an effective antibiotic is absent or limited. Effective antibiotics tend to be newer and more expensive, limiting access to therapy.
Complications
Complications that result from antibiotic resistance include severe illnesses, adverse effects secondary to medical treatments, and death. A complicating factor during the diagnosis and treatment of CRE infections is identifying resistance to previously discussed drug regimens. Commonly induced resistance mechanisms in CRE to other active antimicrobials include aminoglycosides, fosfomycin, and BLBLI. Acquiring a single-point mutation in the 16S rRNA methyltransferase leads to resistance of all aminoglycosides.[44] This mutation can occur de novo or in combination with other resistance mechanisms and is best studied in NDM carbapenemase-producing CRE but can also be present in many other carbapenemase producers.[45] This mutation can develop in the setting of aminoglycoside use and confers resistance. Fosfomycin resistance is common in E.coli, but many other Enterobacterales, including ESBL and CRE, do not demonstrate resistance. The development of resistance makes treating these infections more challenging. Resistance to this drug is typically seen by selecting resistant microbes through fosfomycin monotherapy.[46] Ceftazidime-avibactam is generally used for OXA-48 and KPC CRE, but when resistance appears among these isolates, mutations in blaOxa48 and blaKPC induce resistance, respectively.[47]
Consultations
For patients who demonstrate CRE infection, an infectious disease consult is necessary. Microbiologists should also be involved because specialized tests may need to be performed for definitive diagnosis and treatment planning.
Deterrence and Patient Education
Any CRE infection is considered an urgent complication, and patients need to understand the risks of their disease. Patients also need to be counseled regarding the likelihood of being colonized with CRE after their initial infections. This adds complexity to their medical treatment, as they are at increased risk of passing CRE colonization or infection to others and are at increased general risk of infections in the future.[48] Infection prevention practices such as contact isolation should be employed with CRE infection or colonization patients.[49]
Enhancing Healthcare Team Outcomes
CRE outbreaks are usually uncommon and isolated to institutional or geographic regions. The prevalence of community-acquired CRE has been reported to be 0% to 29.5%, and early screening detection of CRE colonization in patients can help prevent or limit outbreaks via proper isolation methods.[12] Long-term care facilities have been shown to have lower rates of CRE than acute care hospitals, but this may be due to poor detection and surveillance methods.[50] Any healthcare facility should be aware of the risk CRE poses to patients and should take part in regular institutional, local, and national surveillance. An epidemiological assessment should accompany any case of CRE into etiology to limit further spread.
An interprofessional team approach is critical when caring for patients with CRE infections. An infectious disease specialist, as well as an infectious disease board-certified pharmacist, can help to direct appropriate antimicrobial therapy. Infection control personnel are crucial team members as they quickly ensure that contact precautions are appropriately implemented and that patients are promptly isolated to protect other hospitalized patients. Laboratory personnel, who are well-trained on when to suspect CRE and the appropriate testing to perform, play a crucial role. However, bedside nurses are among the most pivotal team members because they are responsible for correctly administering antimicrobial therapy and ensuring that contact precautions are followed by hospital personnel and visitors. They also help to provide crucial education to patients and their family members. As the team plays its role, it ensures that the patient and other hospitalized patients get the care and that the risk of hospital-acquired infections is minimized.
Media
References
van Duin D, Paterson DL. Multidrug-Resistant Bacteria in the Community: An Update. Infectious disease clinics of North America. 2020 Dec:34(4):709-722. doi: 10.1016/j.idc.2020.08.002. Epub 2020 Sep 30 [PubMed PMID: 33011046]
Darby EM, Trampari E, Siasat P, Gaya MS, Alav I, Webber MA, Blair JMA. Molecular mechanisms of antibiotic resistance revisited. Nature reviews. Microbiology. 2023 May:21(5):280-295. doi: 10.1038/s41579-022-00820-y. Epub 2022 Nov 21 [PubMed PMID: 36411397]
Al-Tawfiq JA, Momattin H, Al-Ali AY, Eljaaly K, Tirupathi R, Haradwala MB, Areti S, Alhumaid S, Rabaan AA, Al Mutair A, Schlagenhauf P. Antibiotics in the pipeline: a literature review (2017-2020). Infection. 2022 Jun:50(3):553-564. doi: 10.1007/s15010-021-01709-3. Epub 2021 Oct 4 [PubMed PMID: 34606056]
Munita JM, Arias CA. Mechanisms of Antibiotic Resistance. Microbiology spectrum. 2016 Apr:4(2):. doi: 10.1128/microbiolspec.VMBF-0016-2015. Epub [PubMed PMID: 27227291]
Jacoby GA. AmpC beta-lactamases. Clinical microbiology reviews. 2009 Jan:22(1):161-82, Table of Contents. doi: 10.1128/CMR.00036-08. Epub [PubMed PMID: 19136439]
Tamma PD, Aitken SL, Bonomo RA, Mathers AJ, van Duin D, Clancy CJ. Infectious Diseases Society of America 2023 Guidance on the Treatment of Antimicrobial Resistant Gram-Negative Infections. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2023 Jul 18:():. pii: ciad428. doi: 10.1093/cid/ciad428. Epub 2023 Jul 18 [PubMed PMID: 37463564]
Vardakas KZ, Tansarli GS, Rafailidis PI, Falagas ME. Carbapenems versus alternative antibiotics for the treatment of bacteraemia due to Enterobacteriaceae producing extended-spectrum β-lactamases: a systematic review and meta-analysis. The Journal of antimicrobial chemotherapy. 2012 Dec:67(12):2793-803. doi: 10.1093/jac/dks301. Epub 2012 Aug 21 [PubMed PMID: 22915465]
Level 1 (high-level) evidenceDe Rosa M, Verdino A, Soriente A, Marabotti A. The Odd Couple(s): An Overview of Beta-Lactam Antibiotics Bearing More Than One Pharmacophoric Group. International journal of molecular sciences. 2021 Jan 9:22(2):. doi: 10.3390/ijms22020617. Epub 2021 Jan 9 [PubMed PMID: 33435500]
Level 3 (low-level) evidenceWong D, Spellberg B. Leveraging antimicrobial stewardship into improving rates of carbapenem-resistant Enterobacteriaceae. Virulence. 2017 May 19:8(4):383-390. doi: 10.1080/21505594.2016.1188234. Epub 2016 May 17 [PubMed PMID: 27187821]
Tzouvelekis LS, Markogiannakis A, Psichogiou M, Tassios PT, Daikos GL. Carbapenemases in Klebsiella pneumoniae and other Enterobacteriaceae: an evolving crisis of global dimensions. Clinical microbiology reviews. 2012 Oct:25(4):682-707. doi: 10.1128/CMR.05035-11. Epub [PubMed PMID: 23034326]
Level 3 (low-level) evidenceMunoz-Price LS, Poirel L, Bonomo RA, Schwaber MJ, Daikos GL, Cormican M, Cornaglia G, Garau J, Gniadkowski M, Hayden MK, Kumarasamy K, Livermore DM, Maya JJ, Nordmann P, Patel JB, Paterson DL, Pitout J, Villegas MV, Wang H, Woodford N, Quinn JP. Clinical epidemiology of the global expansion of Klebsiella pneumoniae carbapenemases. The Lancet. Infectious diseases. 2013 Sep:13(9):785-96. doi: 10.1016/S1473-3099(13)70190-7. Epub [PubMed PMID: 23969216]
Codjoe FS, Donkor ES. Carbapenem Resistance: A Review. Medical sciences (Basel, Switzerland). 2017 Dec 21:6(1):. doi: 10.3390/medsci6010001. Epub 2017 Dec 21 [PubMed PMID: 29267233]
Hammoudi Halat D, Ayoub Moubareck C. The Current Burden of Carbapenemases: Review of Significant Properties and Dissemination among Gram-Negative Bacteria. Antibiotics (Basel, Switzerland). 2020 Apr 16:9(4):. doi: 10.3390/antibiotics9040186. Epub 2020 Apr 16 [PubMed PMID: 32316342]
Rabaan AA, Eljaaly K, Alhumaid S, Albayat H, Al-Adsani W, Sabour AA, Alshiekheid MA, Al-Jishi JM, Khamis F, Alwarthan S, Alhajri M, Alfaraj AH, Tombuloglu H, Garout M, Alabdullah DM, Mohammed EAE, Yami FSA, Almuhtaresh HA, Livias KA, Mutair AA, Almushrif SA, Abusalah MAHA, Ahmed N. An Overview on Phenotypic and Genotypic Characterisation of Carbapenem-Resistant Enterobacterales. Medicina (Kaunas, Lithuania). 2022 Nov 19:58(11):. doi: 10.3390/medicina58111675. Epub 2022 Nov 19 [PubMed PMID: 36422214]
Level 3 (low-level) evidenceMarsik FJ, Nambiar S. Review of carbapenemases and AmpC-beta lactamases. The Pediatric infectious disease journal. 2011 Dec:30(12):1094-5. doi: 10.1097/INF.0b013e31823c0e47. Epub [PubMed PMID: 22105420]
Boyd SE, Livermore DM, Hooper DC, Hope WW. Metallo-β-Lactamases: Structure, Function, Epidemiology, Treatment Options, and the Development Pipeline. Antimicrobial agents and chemotherapy. 2020 Sep 21:64(10):. doi: 10.1128/AAC.00397-20. Epub 2020 Sep 21 [PubMed PMID: 32690645]
Hirvonen VHA, Spencer J, van der Kamp MW. Antimicrobial Resistance Conferred by OXA-48 β-Lactamases: Towards a Detailed Mechanistic Understanding. Antimicrobial agents and chemotherapy. 2021 May 18:65(6):. doi: 10.1128/AAC.00184-21. Epub 2021 May 18 [PubMed PMID: 33753332]
Level 3 (low-level) evidencevan Duin D, Doi Y. The global epidemiology of carbapenemase-producing Enterobacteriaceae. Virulence. 2017 May 19:8(4):460-469. doi: 10.1080/21505594.2016.1222343. Epub 2016 Aug 11 [PubMed PMID: 27593176]
Cui X, Zhang H, Du H. Carbapenemases in Enterobacteriaceae: Detection and Antimicrobial Therapy. Frontiers in microbiology. 2019:10():1823. doi: 10.3389/fmicb.2019.01823. Epub 2019 Aug 20 [PubMed PMID: 31481937]
Livermore DM, Nicolau DP, Hopkins KL, Meunier D. Carbapenem-Resistant Enterobacterales, Carbapenem Resistant Organisms, Carbapenemase-Producing Enterobacterales, and Carbapenemase-Producing Organisms: Terminology Past its "Sell-By Date" in an Era of New Antibiotics and Regional Carbapenemase Epidemiology. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2020 Oct 23:71(7):1776-1782. doi: 10.1093/cid/ciaa122. Epub [PubMed PMID: 32025698]
Poirel L, Benouda A, Hays C, Nordmann P. Emergence of NDM-1-producing Klebsiella pneumoniae in Morocco. The Journal of antimicrobial chemotherapy. 2011 Dec:66(12):2781-3. doi: 10.1093/jac/dkr384. Epub 2011 Sep 19 [PubMed PMID: 21930570]
Level 3 (low-level) evidencePotter RF, D'Souza AW, Dantas G. The rapid spread of carbapenem-resistant Enterobacteriaceae. Drug resistance updates : reviews and commentaries in antimicrobial and anticancer chemotherapy. 2016 Nov:29():30-46. doi: 10.1016/j.drup.2016.09.002. Epub 2016 Sep 19 [PubMed PMID: 27912842]
Level 3 (low-level) evidenceHaeili M, Barmudeh S, Omrani M, Zeinalzadeh N, Kafil HS, Batignani V, Ghodousi A, Cirillo DM. Whole-genome sequence analysis of clinically isolated carbapenem resistant Escherichia coli from Iran. BMC microbiology. 2023 Feb 27:23(1):49. doi: 10.1186/s12866-023-02796-y. Epub 2023 Feb 27 [PubMed PMID: 36850019]
Arnold RS, Thom KA, Sharma S, Phillips M, Kristie Johnson J, Morgan DJ. Emergence of Klebsiella pneumoniae carbapenemase-producing bacteria. Southern medical journal. 2011 Jan:104(1):40-5. doi: 10.1097/SMJ.0b013e3181fd7d5a. Epub [PubMed PMID: 21119555]
Salamzade R, Manson AL, Walker BJ, Brennan-Krohn T, Worby CJ, Ma P, He LL, Shea TP, Qu J, Chapman SB, Howe W, Young SK, Wurster JI, Delaney ML, Kanjilal S, Onderdonk AB, Bittencourt CE, Gussin GM, Kim D, Peterson EM, Ferraro MJ, Hooper DC, Shenoy ES, Cuomo CA, Cosimi LA, Huang SS, Kirby JE, Pierce VM, Bhattacharyya RP, Earl AM. Inter-species geographic signatures for tracing horizontal gene transfer and long-term persistence of carbapenem resistance. Genome medicine. 2022 Apr 5:14(1):37. doi: 10.1186/s13073-022-01040-y. Epub 2022 Apr 5 [PubMed PMID: 35379360]
Wright AJ. The penicillins. Mayo Clinic proceedings. 1999 Mar:74(3):290-307 [PubMed PMID: 10090000]
Tooke CL, Hinchliffe P, Bragginton EC, Colenso CK, Hirvonen VHA, Takebayashi Y, Spencer J. β-Lactamases and β-Lactamase Inhibitors in the 21st Century. Journal of molecular biology. 2019 Aug 23:431(18):3472-3500. doi: 10.1016/j.jmb.2019.04.002. Epub 2019 Apr 5 [PubMed PMID: 30959050]
Gustaferro CA, Steckelberg JM. Cephalosporin antimicrobial agents and related compounds. Mayo Clinic proceedings. 1991 Oct:66(10):1064-73 [PubMed PMID: 1921490]
Papp-Wallace KM, Endimiani A, Taracila MA, Bonomo RA. Carbapenems: past, present, and future. Antimicrobial agents and chemotherapy. 2011 Nov:55(11):4943-60. doi: 10.1128/AAC.00296-11. Epub 2011 Aug 22 [PubMed PMID: 21859938]
Jeon JH, Lee JH, Lee JJ, Park KS, Karim AM, Lee CR, Jeong BC, Lee SH. Structural basis for carbapenem-hydrolyzing mechanisms of carbapenemases conferring antibiotic resistance. International journal of molecular sciences. 2015 Apr 29:16(5):9654-92. doi: 10.3390/ijms16059654. Epub 2015 Apr 29 [PubMed PMID: 25938965]
Lepe JA, Martínez-Martínez L. Resistance mechanisms in Gram-negative bacteria. Medicina intensiva. 2022 Jul:46(7):392-402. doi: 10.1016/j.medine.2022.05.004. Epub 2022 May 31 [PubMed PMID: 35660283]
Franco MR, Caiaffa-Filho HH, Burattini MN, Rossi F. Metallo-beta-lactamases among imipenem-resistant Pseudomonas aeruginosa in a Brazilian university hospital. Clinics (Sao Paulo, Brazil). 2010:65(9):825-9 [PubMed PMID: 21049207]
Sotgiu G, Are BM, Pesapane L, Palmieri A, Muresu N, Cossu A, Dettori M, Azara A, Mura II, Cocuzza C, Aliberti S, Piana A. Nosocomial transmission of carbapenem-resistant Klebsiella pneumoniae in an Italian university hospital: a molecular epidemiological study. The Journal of hospital infection. 2018 Aug:99(4):413-418. doi: 10.1016/j.jhin.2018.03.033. Epub 2018 Apr 3 [PubMed PMID: 29621600]
Level 2 (mid-level) evidenceDoumith M, Ellington MJ, Livermore DM, Woodford N. Molecular mechanisms disrupting porin expression in ertapenem-resistant Klebsiella and Enterobacter spp. clinical isolates from the UK. The Journal of antimicrobial chemotherapy. 2009 Apr:63(4):659-67. doi: 10.1093/jac/dkp029. Epub 2009 Feb 20 [PubMed PMID: 19233898]
Chen HY, Jean SS, Lee YL, Lu MC, Ko WC, Liu PY, Hsueh PR. Carbapenem-Resistant Enterobacterales in Long-Term Care Facilities: A Global and Narrative Review. Frontiers in cellular and infection microbiology. 2021:11():601968. doi: 10.3389/fcimb.2021.601968. Epub 2021 Apr 23 [PubMed PMID: 33968793]
Level 3 (low-level) evidencePérez-Galera S, Bravo-Ferrer JM, Paniagua M, Kostyanev T, de Kraker MEA, Feifel J, Sojo-Dorado J, Schotsman J, Cantón R, Daikos GL, Carevic B, Dragovac G, Tan LK, Raka L, Hristea A, Viale P, Akova M, Reguera JM, Valiente de Santis L, Torre-Cisneros J, Cano Á, Roilides E, Radulovic L, Kirakli C, Shaw E, Falagas ME, Pintado V, Goossens H, Bonten MJ, Gutiérrez-Gutiérrez B, Rodriguez-Baño J, COMBACTE-CARE-EURECA Team. Risk factors for infections caused by carbapenem-resistant Enterobacterales: an international matched case-control-control study (EURECA). EClinicalMedicine. 2023 Mar:57():101871. doi: 10.1016/j.eclinm.2023.101871. Epub 2023 Feb 27 [PubMed PMID: 36895801]
Level 2 (mid-level) evidenceZeng M, Xia J, Zong Z, Shi Y, Ni Y, Hu F, Chen Y, Zhuo C, Hu B, Lv X, Li J, Liu Z, Zhang J, Yang W, Yang F, Yang Q, Zhou H, Li X, Wang J, Li Y, Ren J, Chen B, Chen D, Wu A, Guan X, Qu J, Wu D, Huang X, Qiu H, Xu Y, Yu Y, Wang M, Society of Bacterial Infection and Resistance of Chinese Medical Association, Expert Committee on Clinical Use of Antimicrobial Agents and Evaluation of Antimicrobial Resistance of the National Health Commission, Infectious Diseases Society of Chinese Medical Education Association. Guidelines for the diagnosis, treatment, prevention and control of infections caused by carbapenem-resistant gram-negative bacilli. Journal of microbiology, immunology, and infection = Wei mian yu gan ran za zhi. 2023 Aug:56(4):653-671. doi: 10.1016/j.jmii.2023.01.017. Epub 2023 Feb 18 [PubMed PMID: 36868960]
Level 2 (mid-level) evidenceSparbier K, Schubert S, Weller U, Boogen C, Kostrzewa M. Matrix-assisted laser desorption ionization-time of flight mass spectrometry-based functional assay for rapid detection of resistance against β-lactam antibiotics. Journal of clinical microbiology. 2012 Mar:50(3):927-37. doi: 10.1128/JCM.05737-11. Epub 2012 Jan 11 [PubMed PMID: 22205812]
Rodríguez-Baño J, Cisneros JM, Gudiol C, Martínez JA. Treatment of infections caused by carbapenemase-producing Enterobacteriaceae. Enfermedades infecciosas y microbiologia clinica. 2014 Dec:32 Suppl 4():49-55. doi: 10.1016/S0213-005X(14)70174-0. Epub [PubMed PMID: 25542052]
Level 3 (low-level) evidenceTamma PD, Aitken SL, Bonomo RA, Mathers AJ, van Duin D, Clancy CJ. Infectious Diseases Society of America 2022 Guidance on the Treatment of Extended-Spectrum β-lactamase Producing Enterobacterales (ESBL-E), Carbapenem-Resistant Enterobacterales (CRE), and Pseudomonas aeruginosa with Difficult-to-Treat Resistance (DTR-P. aeruginosa). Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2022 Aug 25:75(2):187-212. doi: 10.1093/cid/ciac268. Epub [PubMed PMID: 35439291]
Kazmierczak KM, Tsuji M, Wise MG, Hackel M, Yamano Y, Echols R, Sahm DF. In vitro activity of cefiderocol, a siderophore cephalosporin, against a recent collection of clinically relevant carbapenem-non-susceptible Gram-negative bacilli, including serine carbapenemase- and metallo-β-lactamase-producing isolates (SIDERO-WT-2014 Study). International journal of antimicrobial agents. 2019 Feb:53(2):177-184. doi: 10.1016/j.ijantimicag.2018.10.007. Epub 2018 Oct 26 [PubMed PMID: 30395986]
Yahav D, Lador A, Paul M, Leibovici L. Efficacy and safety of tigecycline: a systematic review and meta-analysis. The Journal of antimicrobial chemotherapy. 2011 Sep:66(9):1963-71. doi: 10.1093/jac/dkr242. Epub 2011 Jun 18 [PubMed PMID: 21685488]
Level 1 (high-level) evidenceMeletis G. Carbapenem resistance: overview of the problem and future perspectives. Therapeutic advances in infectious disease. 2016 Feb:3(1):15-21. doi: 10.1177/2049936115621709. Epub [PubMed PMID: 26862399]
Level 3 (low-level) evidenceDoi Y, Wachino JI, Arakawa Y. Aminoglycoside Resistance: The Emergence of Acquired 16S Ribosomal RNA Methyltransferases. Infectious disease clinics of North America. 2016 Jun:30(2):523-537. doi: 10.1016/j.idc.2016.02.011. Epub [PubMed PMID: 27208771]
Li JJ, Sheng ZK, Deng M, Bi S, Hu FS, Miao HF, Ji ZK, Sheng JF, Li LJ. Epidemic of Klebsiella pneumoniae ST11 clone coproducing KPC-2 and 16S rRNA methylase RmtB in a Chinese University Hospital. BMC infectious diseases. 2012 Dec 23:12():373. doi: 10.1186/1471-2334-12-373. Epub 2012 Dec 23 [PubMed PMID: 23259910]
Level 2 (mid-level) evidenceKarageorgopoulos DE, Miriagou V, Tzouvelekis LS, Spyridopoulou K, Daikos GL. Emergence of resistance to fosfomycin used as adjunct therapy in KPC Klebsiella pneumoniae bacteraemia: report of three cases. The Journal of antimicrobial chemotherapy. 2012 Nov:67(11):2777-9. doi: 10.1093/jac/dks270. Epub 2012 Jul 10 [PubMed PMID: 22782489]
Level 3 (low-level) evidenceBoth A, Büttner H, Huang J, Perbandt M, Belmar Campos C, Christner M, Maurer FP, Kluge S, König C, Aepfelbacher M, Wichmann D, Rohde H. Emergence of ceftazidime/avibactam non-susceptibility in an MDR Klebsiella pneumoniae isolate. The Journal of antimicrobial chemotherapy. 2017 Sep 1:72(9):2483-2488. doi: 10.1093/jac/dkx179. Epub [PubMed PMID: 28637339]
Hassoun-Kheir N, Hussien K, Karram M, Saffuri M, Badaan S, Peleg S, Aboelhega W, Warman S, Alon T, Pollak D, Szwarcwort Cohen M, Paul M. Clinical significance and burden of carbapenem-resistant Enterobacterales (CRE) colonization acquisition in hospitalized patients. Antimicrobial resistance and infection control. 2023 Nov 20:12(1):129. doi: 10.1186/s13756-023-01323-y. Epub 2023 Nov 20 [PubMed PMID: 37986092]
Mills JP, Marchaim D. Multidrug-Resistant Gram-Negative Bacteria: Infection Prevention and Control Update. Infectious disease clinics of North America. 2021 Dec:35(4):969-994. doi: 10.1016/j.idc.2021.08.001. Epub [PubMed PMID: 34752228]
Gniadek TJ, Carroll KC, Simner PJ. Carbapenem-Resistant Non-Glucose-Fermenting Gram-Negative Bacilli: the Missing Piece to the Puzzle. Journal of clinical microbiology. 2016 Jul:54(7):1700-1710. doi: 10.1128/JCM.03264-15. Epub 2016 Feb 24 [PubMed PMID: 26912753]