Introduction
Heart valves undergo approximately 100,000 cycles daily, culminating in around 3 billion cycles over a 75-year life span, facing numerous stresses. Traditionally, the aortic valve was viewed as a "static" structure with the primary role of ensuring unidirectional blood flow from the left ventricle (LV) and assisting coronary perfusion. However, recent insights have transformed this view, recognizing the aortic valve as a functionally intricate regulatory system essential for the heart's optimal mechanobiological coupling (see Image. The Aortic Valve).
The aortic valve is a complex regulatory system crucial for the heart's optimal function, and it comprises several components, including the annulus, cusps, sinuses of Valsalva, and the sinotubular junction (STJ). These components form a biomechanical unit that efficiently manages the forces exerted during the cardiac cycle. The crown-shaped geometry of the semilunar valves enhances their efficiency and responsiveness during the cardiac cycle, allowing them to effectively manage the varying forces exerted upon them (see Video. Aortic Valve Movement).[1] Aortic valve pathologies can severely affect the body due to perfusion deficits. Aortic valve insufficiency is primarily caused due to the underlying shear forces that lead to stretching, dilation, and valve rupture.[2] Moreover, conditions of the aortic root, including coarctation, dissection, and aneurysm, can exacerbate aortic valve pathology by increasing the shear force of blood at the aortic valve, disrupting the laminar flow.[3]
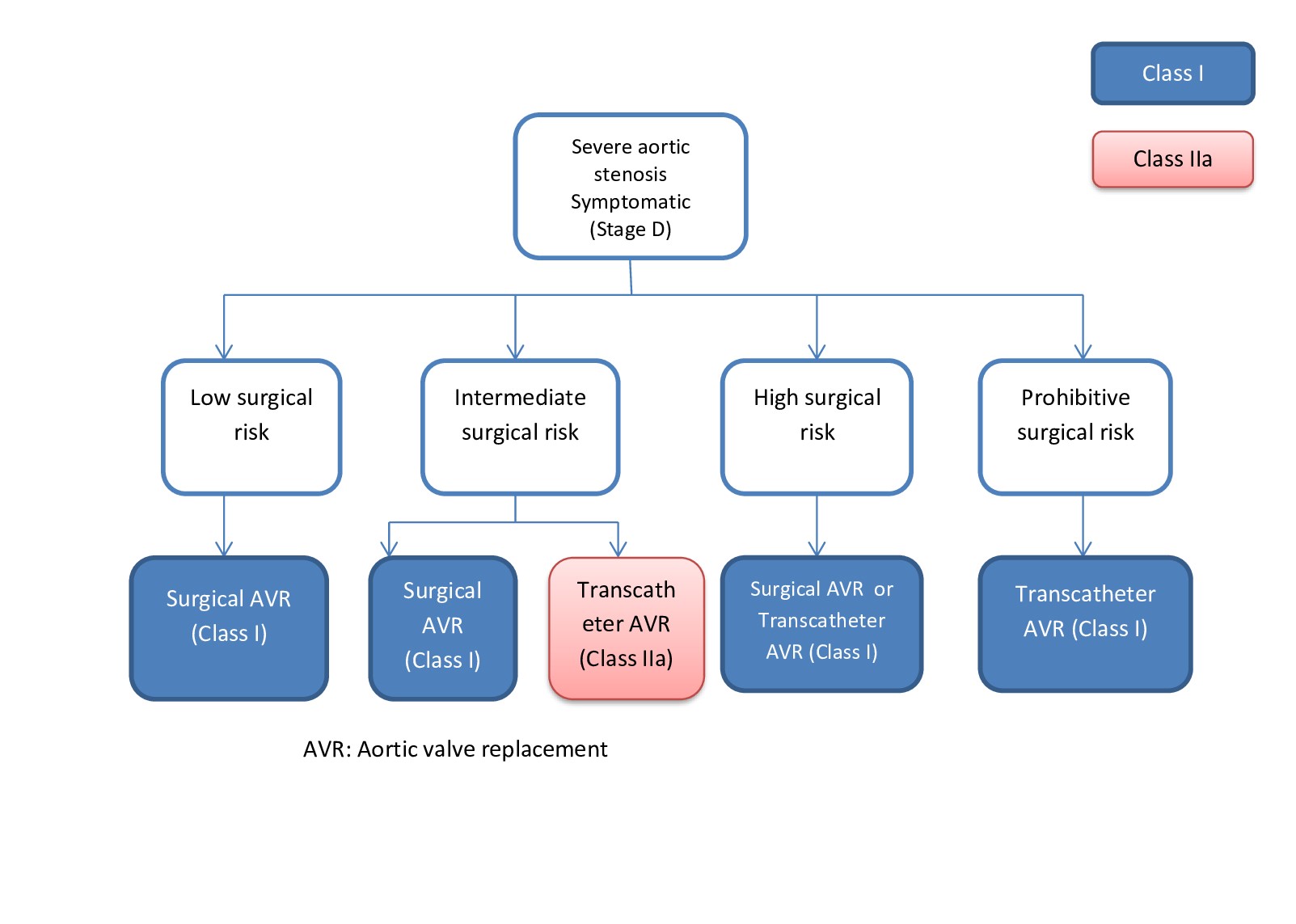
Managing aortic valve pathologies often involves either repair or replacement. Aortic valve repair is a surgical procedure designed to preserve the native valve and restore its function in patients with aortic valve insufficiency or disease. Aortic valve replacement with a biological or prosthetic valve has long been the cornerstone of treatment for these conditions. However, there is ongoing debate regarding the relative merits of aortic valve repair versus replacement in specific patient subsets. While aortic valve repair may offer advantages in certain scenarios, its efficacy and suitability must be carefully weighed against the established benefits of valve replacement (see Image. Recommendations for Aortic Valve Replacement).
Over the last decade, aortic valve repair and preservation have gained significant importance in treating aortic root disease, both with and without insufficiencies. These procedures have been successfully performed worldwide, although they account for less than 2% of all valve procedures due to the high level of expertise required.[4] The low prevalence of aortic valve repair compared to mitral valve repair is partly because calcific aortic stenosis is the most common valvular pathology, making aortic valve replacement a more frequent intervention.
Aortic valve replacement, while seemingly simpler than repair, carries long-term complications such as structural deterioration, restenosis, infection, and bleeding due to the need for anticoagulation.[5][6] These complications have led to a growing interest in aortic valve preservation procedures, which maintain the normal anatomy and physiology of the aortic valve functional unit and potentially avoid the complications associated with replacement. Consequently, aortic valve preservation is becoming a reasonable alternative to replacement, aiming to prevent long-term issues and improve patient outcomes.
Anatomy and Physiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Anatomy and Physiology
Understanding the aortic valve's surgical anatomy is crucial to understanding the valve repair and preservation procedures. The aortic valve functional unit comprises the ventriculo-arterial junction (VAJ), STJ, aortic leaflets, and sinuses. The origin of the aorta is close to the cardiac valve, bridging the LV and the systemic circulation. Within the internal part of the aortic root are the leaflets, interleaflet triangles, and commissures. Typically, the coaptation height is midlevel between the STJ and VAJ.[7]
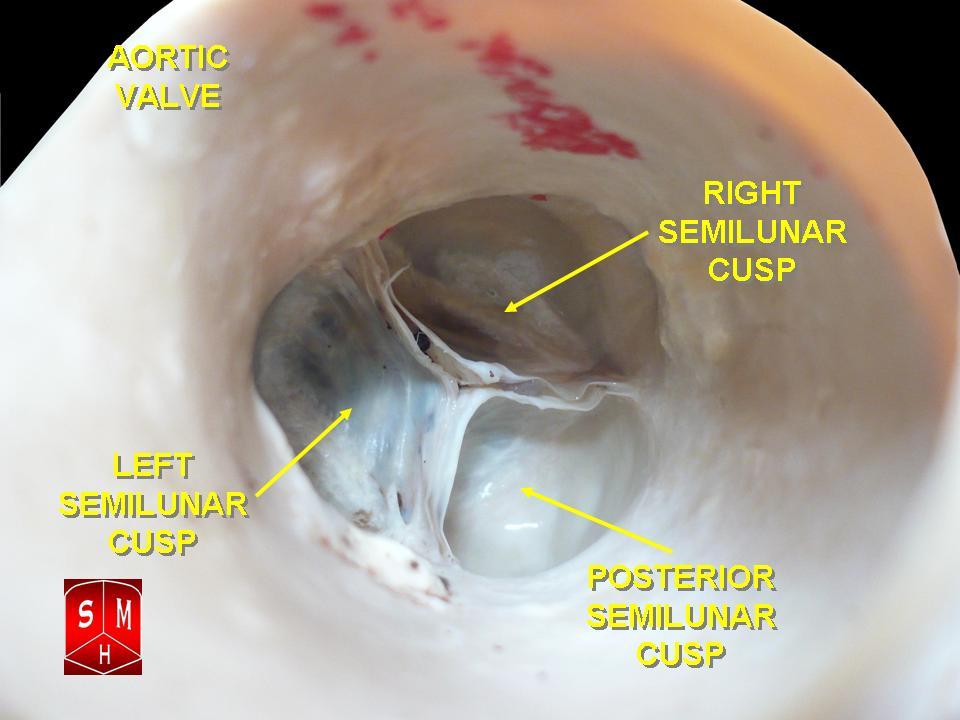
The aortic root is the basal attachment of the aortic valve leaflets to the STJ. German pathologist and anatomist Friedrich Henle was the first to define the arterial root instead of the ring. The aortic root bulges to form the 3 sinuses—2 of these sinuses give rise to the main coronary arteries, while the third one, furthest from the pulmonary trunk, is a noncoronary aortic sinus. Anatomically, the sinuses are named anterior (right coronary), left posterior (left coronary), and right dorsal (noncoronary) (see Image. The Anatomy of the Aortic Valve).[8]
Critical Parts of the Aortic Valve
Ventriculo-arterial junction: The VAJ is an elliptical junction marking the transition between the LV outflow tract and the aortic root. The average diameter of the VAJ is 23 mm in adults with a normal aortic valve.[9][10] Anatomically, it represents the point where the ventricular muscles terminate and begin to connect to the aortic sleeve wall. Surgically, it signifies the leaflet hinge lines.[11] The crown shape of the aortic cusps represents the line of insertion. The virtual basal ring or annulus is the line passing through the nadir of each adjacent cusp and is defined as a commissure. The functional unit is the 3-dimensional crown-shaped attachment of the cusps, known as the aortic annulus. The intraluminal region of the annulus joins the nadir of the 3 cusps. The circumferential plane of the annulus joins the 3 commissures and represents an STJ. Moreover, to understand how the external dissection of the aorta can extend lower to the basal ring on the posterior portion of the root, the left coronary sinus and noncoronary sinus extend inferiorly to the right coronary sinus.[12][13]
Sinotubular junction: This is a well-defined circular ridge found between the superior or tubular segment of the ascending aorta and the inferior, bulbous segment of the ascending aorta, known as the aortic root. The STJ follows the superior contour of the 3 sinuses of Valsalva that forms the bulbs of the aortic root and intersects the commissures of the aortic valve. The diameter of STJ is 5 mm more than the base of the valve, creating a ratio of 1.3 between the STJ and the virtual basal annulus.[12]
Aortic leaflets: Evaluating the size of the aortic leaflets is crucial for assessing prolapse or tissue deficiency. Intraoperatively, measuring cusp size and height is essential. The geometric height is the longest distance from the aortic insertion to the cusp's free central margin, while the effective height is measured from the basal plane to the central coaptation point. Successful aortic valve repair is often indicated by the retracted cusp's geometric height being less than 19 mm in bicuspid valves and less than 16 mm in tricuspid valves (see Image. Schematic Illustration of Geometric and Effective Height).[14][15]
Aortic sinuses of Valsalva: These sinuses are the bulging regions of the aortic root that house the leaflets and provide openings for the coronary arteries. At the STJ, the sinuses transition the elliptical base to a circular shape, stabilizing the central valve leaflet.
Understanding the detailed anatomy of the aortic valve, including the VAJ, STJ, aortic leaflets, and sinuses, is essential for successful aortic valve repair and preservation. This knowledge helps maintain the valve's normal structure and function, potentially preventing complications associated with valve replacement.
Indications
The indications for aortic valve repair align closely with those for valve replacement but with the added advantage of lower perioperative complications (see Image. Indications for Aortic Valve Replacement).[16]
These indications include:
- Symptomatic or asymptomatic aortic insufficiency: Patients with significant LV dilation, characterized by an LV end-systolic diameter greater than 50 mm and an LV end-diastolic diameter greater than 70 mm, or those with LV dysfunction, are prime candidates for aortic valve repair.[17]
- Aortic root disease or ascending aorta pathology: Aortic valve repair is indicated in patients with aortic root disease or ascending aorta issues accompanied by valvular pathology.[18][19]
- Aortic valve regurgitation and insufficiency: Experts recommend aortic valve repair as the first-line intervention for patients with aortic valve regurgitation and insufficiency.[20]
This procedure can potentially preserve native valve function and reduce complications compared to replacement. The outcomes of aortic valve repair in aortic valve insufficiency depend significantly on the size and quality of the cusp available for repair. Patients with significant destruction of the aortic valve, extensive calcifications, or mechanical damage due to rheumatic heart disease are less likely to benefit from aortic valve repair.[21] However, aortic valve repair has demonstrated positive outcomes in patients with unicuspid, bicuspid, and quadricuspid aortic valves.[22][23]
Contraindications
A common contraindication to aortic valve repair surgery is a valvular apparatus that is not amenable to repair. Before planning the repair, the valvular apparatus should be carefully evaluated. Factors precluding a repair procedure include excessively pliable or densely calcified, sclerotic, stiff leaflets, or valvular leaflet retraction.[24] Surgical repair should not be offered to patients with a valvular apparatus that is not amenable to repair to prevent such complications.
A transesophageal echocardiogram (TEE) should be performed preoperatively to evaluate the entire valvular apparatus, including the aortic annulus, aortic sinus, and STJ, as well as a thorough assessment of the aortic cusps. Valve repair should be conducted in a center with appropriate physician expertise and collaboration among cardiologists, cardiac surgeons, and anesthesiologists, emphasizing that large-volume centers are best suited for these reparative procedures.
Equipment
Aortic valve-sparing surgery or valve repair is performed under general anesthesia, with all patients requiring antegrade cardioplegia either through the aortic root or directly into the coronary ostia if severe aortic insufficiency is present. Patients needing concomitant ascending aortic replacement will require the addition of a Dacron prosthesis. The surgical technique regarding arterial cannulation may need to be adjusted based on the condition of the aortic arch. The axillary or femoral artery will be used for cannulation in cases where an aneurysm extends to the aortic arch.[25]
Personnel
Aortic valve repair should be carried out in high-volume centers. As these procedures are not commonly performed, having a well-trained surgical team in place is important when planning a valve repair. This ensures the healthcare team can work together effectively as a unit rather than as individual performers.
Anesthesiologists and cardiologists with extensive TEE experience are crucial team members assisting with valve repair procedures. In addition to the primary cardiothoracic surgeon performing the procedure, 1 or 2 assistant surgeons and a surgical technician should be present during the surgery. Highly trained nursing staff is also essential for the anesthesiologist and the operative team.
Preparation
The preoperative assessment includes a baseline electrocardiogram (ECG), a chest radiograph, a cardiac computed tomography (CT) scan or coronary angiogram, a transthoracic echocardiogram (TTE), and a chest CT scan for patients undergoing redo sternotomies. The patient should also be clinically assessed, and any medical issues should be addressed. A pulmonologist and cardiologist should evaluate the patient to optimize lung and cardiac function. The goals and objectives of the aortic valve repair must be clearly defined and discussed with the patient, and informed consent must be obtained before surgery.
In the operating room, 1 of the team members should perform a surgical timeout, affirming the patient, stating the procedure, and reviewing all essential medications and allergies. After sedating the patient, the patient should be prepped and draped using a sterile technique.
A TEE is performed in the operating room. Measurements are taken for the aortic annulus, sinus of Valsalva, STJ, and ascending aorta. Notably, 2-dimensional long-axis measurements are verified with short-axis views to ensure accuracy and avoid projection artifacts. In addition, 3-dimensional TEE with multiplanar reconstruction provides even more detailed information. Surgical collaboration with cardiologists and anesthesiologists experienced in TEE is essential during these cases.
Technique or Treatment
Aortic valve repair has been reported to have good intermediate and long-term results. A perfected and standardized technique ensures the success of the procedure. Surgical repair techniques are divided into aortic root and annular remodeling and cusp repair techniques. Knowing the 3 types of aortic insufficiency is essential to understand these techniques better. The types are:
- Type 1 corresponds to a dilated aortic root, which causes cusps to stretch and tether, resulting in a central regurgitation jet.
- Type 2 is due to excessive valve leaflet tissue with prolapse and presents as an eccentric jet.
- Type 3 is the least amenable to surgical repair and involves fibrosis and valve calcifications leading to cusp retraction.
In most cases, all 3 of these coexist to some degree. The European Society of Cardiology and the European Association of Cardiothoracic Surgery have further categorized the types of aortic insufficiency (see Table 1. Types of Aortic Insufficiency).[26]
Table 1. Types of Aortic Insufficiency
| Type | Valve Leaflet | Aortic Pathology | Regurgitation Jet |
| 1a | Normal | STJ/ascending aorta dilation | Central |
| 1b | Normal | Sinuses of Valsalva/STJ dilation | Central |
| 1c | Normal | Aortic annulus dilation | Central |
| 1d | Cusp perforation | Aortic annulus dilation | Central |
| 2 |
Excessive leaflet Prolapse leaflet Commissural disruption |
No aortic pathology | Eccentric |
| 3 |
Cusp retraction Cusp restricted in motion |
No aortic pathology | Central or eccentric |
Aortic Root and Annular Techniques
Type 1 aortic lesions in aortic sufficiency include pathology of any component of the functional annulus leading to a central jet. These type 1 lesions can present alone or can accompany cusp pathology. In such cases, subcommissural annuloplasty can improve cusp coaptation.[27] Type 1a aortic insufficiency is associated with ascending aortic aneurysm extending to the STJ. Type 1b is associated with dilation of the sinuses of Valsalva and STJ. Type 1c is associated with aortic annular dilation. Type 1d is aortic annular dilation with cusp perforation. Repairs must be tailored to address the specific aortic dilation pathology.[28]
Valve-Sparing Root Replacement
First described in 1983, this technique has gradually gained popularity in patients with aortic root dilation and resultant aortic insufficiency.[28] This technique consists of either aortic root remodeling with annuloplasty or reimplanting the aortic root with valve cusp assessment. This technique, particularly in high-volume centers, is utilized in patients with tricuspid aortic valves and aortic insufficiency, thereby causing a central regurgitation jet. The success of this technique has also been demonstrated in bicuspid aortic valve patients with eccentric regurgitation jets.
Repair of Isolated Aortic Valve–Related Aortic Insufficiency
This promising technique has been practiced in patients with aortic insufficiency types 1 and 2 with preserved valvular apparatus. Cusp prolapse, generations, and other deformities leading to aortic insufficiency can be effectively repaired. Additionally, a low threshold to repair involved structures must be considered in patients with annulus and STJ abnormalities. The procedure may involve circumferential subvalvular annuloplasty, and in patients with suspicion of supravalvular pathology, STJ annuloplasty or root replacement should be performed concomitantly.[28] This technique might not be successful in type 3 aortic insufficiency, which involves less pliable valves.
Complications can arise promptly in the operating room during all procedures, underscoring the importance of ongoing echocardiographic monitoring until sternal closure. With the patient still on the operating table, the possibility of promptly returning to bypass to address any issues that arise is present.
Complications
Aortic valve repair demands a substantial case volume, a proficient interdisciplinary team for thorough evaluation, and expertise in patient selection. Despite these measures, complications stemming from insufficient repair of the original valvular defect may endure.[29] They are listed as follows:
Early Complications
Early complications typically result from technical errors.
- Low coaptation: Inadequate valvular coaptation observed during intraoperative or postoperative TEE should prompt the surgeon to continue the procedure and further plicate the cusps to prevent aortic insufficiency due to poor coaptation.
- Residual prolapse: Any remaining prolapse following completion of valve repair surgery, whether due to the surgical process or primary prolapse unrelated to surgery, should be addressed before concluding the procedure.
- Cusp perforation and missed fenestration: Iatrogenic perforation occurring during manipulation of the valvular cusps should be promptly repaired. If the valve's fenestration is significant, it should also be addressed before completing the procedure.
- Patch dehiscence: In some cases, when a pericardial patch is utilized to reimplant the aortic valve and aortic root, the patch may dehisce during the early recovery phase, necessitating reoperation and patch fixation.
- Rupture of the anterior leaflet of the mitral valve: Due to the proximity of the anterior leaflet of the mitral valve's base, an inadvertent perforation may lead to mitral regurgitation. This should be addressed promptly to prevent hemodynamic compromise related to mitral insufficiency.
- Hematomas: Localized bleeding at the repair site may lead to small hematomas that usually reabsorb within a few days.
- Complications related to subcommissural annuloplasties: Subcommissural annuloplasties can disrupt the aortic valve apparatus at the subvalvular level, causing fistula formation and pseudoaneurysms. These are severe complications that require fixation either during the index procedure or may require reoperation.
Delayed Complications
Delayed complications manifest months to years after the valve replacement.
- Late-onset persistent aortic insufficiency: Despite a successful procedure, some patients may develop a small prolapse or mild malcoaptation of the valve, which can progress to become hemodynamically significant over the years. Similarly, gradual aortic root dilation can lead to progressive aortic insufficiency over months or years.
- Endocarditis: Reports of endocarditis involving the repaired valve are rare in the literature.
- Bleeding: Although rare, bleeding may be observed late after surgery in patients who have undergone aortic valve repair. The underlying mechanism is not well understood.
- Thromboembolism: Late-onset thromboembolic events are also infrequently reported in these patients.
- Progressive valve fibrosis and calcification: While attempts to repair type 3 aortic insufficiency may be initially successful, they can eventually lead to further fibrosis and calcification of the valve.[30]
A recent meta-analysis revealed that the outcomes of valve-sparing root replacement are comparable between patients with a bicuspid aortic valve and those with a tricuspid aortic valve. Although there is a higher likelihood of reinterventions among patients with a bicuspid aortic valve after valve-sparing root replacement, this procedure remains a dependable and effective treatment for aortic root dilation, irrespective of aortic valve insufficiency. While patients with a tricuspid aortic valve experience a slightly lower long-term reintervention rate over 10 years, the disparity is not statistically significant, indicating that patients with a bicuspid aortic valve may face a greater clinical risk for reintervention.[31]
Clinical Significance
Aortic valve repair procedures are becoming more common in patients with tricuspid and bicuspid aortic valves, involving tailored surgical techniques. These surgeries offer opportunities to address both subvalvular and supravalvular structures, including aortic root repair, facilitating native valve repair or remodeling. With careful patient selection and treatment at expert centers, outcomes comparable to valve replacement have been observed, highlighting the potential of this approach.[32]
The Aortic Valve Insufficiency and Ascending Aorta Aneurysm International Registry (AVIATOR) was created as part of the Heart Valve Society's Valve Research Networks. The adult surgical AVIATOR database has been actively enrolling patients since August 2013. AVIATOR tracks a longitudinal, disease-specific patient cohort, including individuals with isolated aortic valve insufficiency (such as congenital mixed aortic valve disease) and ascending aorta aneurysm (including root and supracoronary aorta and aortic dissection). This registry includes all patients, regardless of whether they undergo aortic valve repair or replacement, and welcomes participation from any center treating these conditions, not just highly specialized centers. The primary goal is to chart the entire disease trajectory of aortic regurgitation from diagnosis to death through regular clinical and echocardiographic follow-ups. This initiative represents a distinctive global endeavor to share data and advance the understanding and treatment of these conditions. This international collaboration promises to yield invaluable insights into long-term outcomes in the near future.[33]
Midterm results from centers worldwide are now being published. A collaborative group in Croatia, comprising cardiologists, anesthesiologists, and cardiac surgeons, has reported that performing surgical repair of a bicuspid aortic valve alongside extra-aortic annuloplasty is linked to low perioperative morbidity and mortality. The midterm outcomes of this procedure are reported as excellent, comparable to those achieved with tricuspid aortic valve repair.[34]
The repair of the bicuspid aortic valve warrants special attention. Found in 1% to 2% of the patient population, a bicuspid aortic valve can manifest as aortic insufficiency at a relatively young age. Over the past 2 decades, repair techniques for significant aortic regurgitation and aneurysms associated with the bicuspid aortic valve have advanced considerably. Initially, while valve repair showed excellent short-term outcomes, some patients required reoperations at midterm follow-ups. Over time, several predictors and mechanisms of failure have been identified and addressed during surgery, enhancing the durability of repairs. Additionally, correcting concurrent aortic dilatation has proven crucial in improving long-term results.[35]
Enhancing Healthcare Team Outcomes
In the context of aortic valve repair, a multidisciplinary team approach is essential to ensure comprehensive patient-centered care and optimize outcomes. Physicians, advanced practitioners, nurses, pharmacists, and other health professionals play crucial roles throughout the patient's journey. During the preoperative phase, collaborative discussions and planning sessions involving all healthcare members help ensure thorough evaluation and preparation of the patient. Advanced practitioners and physicians assess the patient's medical history, perform physical examinations, and determine the appropriateness of aortic valve repair. Nurses provide support and education to patients and their families, helping to alleviate anxiety and ensure compliance with preoperative instructions. Pharmacists play a vital role in medication reconciliation, ensuring patients receive appropriate preoperative pharmacotherapy and optimizing perioperative medication management.
In the postoperative period, effective interprofessional communication and care coordination are paramount. Nurses closely monitor patients for signs of complications, including pain, bleeding, respiratory distress, and thromboembolic events, while pharmacists collaborate with healthcare team members to manage pain and optimize medication regimens. Advanced practitioners and physicians provide ongoing assessment and management of the patient's condition, promptly addressing any concerns or complications. This collaborative approach enhances patient safety, reduces the risk of adverse events, and facilitates optimal recovery and long-term outcomes for patients undergoing aortic valve repair. Through meticulous planning, open communication, and coordinated efforts, the multidisciplinary team strives to achieve the best possible outcomes and improve the overall quality of care for patients with aortic insufficiency.[32]
Media
(Click Image to Enlarge)
The Aortic Valve. The aortic valve resides centrally between the ascending aorta and the left ventricular outflow tract.
Anatomist90, Public Domain, via Wikimedia Commons
<p>Valveguru, <a href="https://creativecommons.org/licenses/by-sa/3.0">Public Domain</a>, via Wikimedia Commons</p>
(Click Image to Enlarge)
(Click Image to Enlarge)
(Click Image to Enlarge)
References
Notenboom ML, Van Hoof L, Schuermans A, Takkenberg JJM, Rega FR, Taverne YJHJ. Aortic Valve Embryology, Mechanobiology, and Second Messenger Pathways: Implications for Clinical Practice. Journal of cardiovascular development and disease. 2024 Feb 1:11(2):. doi: 10.3390/jcdd11020049. Epub 2024 Feb 1 [PubMed PMID: 38392263]
Yacoub MH, Cohn LH. Novel approaches to cardiac valve repair: from structure to function: Part II. Circulation. 2004 Mar 9:109(9):1064-72 [PubMed PMID: 15007015]
El Khoury G, de Kerchove L. Principles of aortic valve repair. The Journal of thoracic and cardiovascular surgery. 2013 Mar:145(3 Suppl):S26-9. doi: 10.1016/j.jtcvs.2012.11.071. Epub 2012 Dec 20 [PubMed PMID: 23260436]
Iung B, Baron G, Butchart EG, Delahaye F, Gohlke-Bärwolf C, Levang OW, Tornos P, Vanoverschelde JL, Vermeer F, Boersma E, Ravaud P, Vahanian A. A prospective survey of patients with valvular heart disease in Europe: The Euro Heart Survey on Valvular Heart Disease. European heart journal. 2003 Jul:24(13):1231-43 [PubMed PMID: 12831818]
Level 3 (low-level) evidenceCarr JA, Savage EB. Aortic valve repair for aortic insufficiency in adults: a contemporary review and comparison with replacement techniques. European journal of cardio-thoracic surgery : official journal of the European Association for Cardio-thoracic Surgery. 2004 Jan:25(1):6-15 [PubMed PMID: 14690726]
Aicher D, Fries R, Rodionycheva S, Schmidt K, Langer F, Schäfers HJ. Aortic valve repair leads to a low incidence of valve-related complications. European journal of cardio-thoracic surgery : official journal of the European Association for Cardio-thoracic Surgery. 2010 Jan:37(1):127-32. doi: 10.1016/j.ejcts.2009.06.021. Epub 2009 Jul 29 [PubMed PMID: 19643618]
Level 2 (mid-level) evidenceAnderson RH. Clinical anatomy of the aortic root. Heart (British Cardiac Society). 2000 Dec:84(6):670-3 [PubMed PMID: 11083753]
Underwood MJ, El Khoury G, Deronck D, Glineur D, Dion R. The aortic root: structure, function, and surgical reconstruction. Heart (British Cardiac Society). 2000 Apr:83(4):376-80 [PubMed PMID: 10722531]
Crooke PS, Beavan LA, Griffin CD, Mazzitelli D, Rankin JS. A geometric model of the normal human aortic root and design of a fully anatomic aortic root graft. Innovations (Philadelphia, Pa.). 2015 Jan-Feb:10(1):57-62. doi: 10.1097/IMI.0000000000000125. Epub [PubMed PMID: 25628255]
Kim M, Roman MJ, Cavallini MC, Schwartz JE, Pickering TG, Devereux RB. Effect of hypertension on aortic root size and prevalence of aortic regurgitation. Hypertension (Dallas, Tex. : 1979). 1996 Jul:28(1):47-52 [PubMed PMID: 8675263]
de Kerchove L, El Khoury G. Anatomy and pathophysiology of the ventriculo-aortic junction: implication in aortic valve repair surgery. Annals of cardiothoracic surgery. 2013 Jan:2(1):57-64. doi: 10.3978/j.issn.2225-319X.2012.12.05. Epub [PubMed PMID: 23977560]
Khelil N, Sleilaty G, Palladino M, Fouda M, Escande R, Debauchez M, Di Centa I, Lansac E. Surgical anatomy of the aortic annulus: landmarks for external annuloplasty in aortic valve repair. The Annals of thoracic surgery. 2015 Apr:99(4):1220-6. doi: 10.1016/j.athoracsur.2014.12.034. Epub 2015 Feb 27 [PubMed PMID: 25728963]
de Kerchove L, Jashari R, Boodhwani M, Duy KT, Lengelé B, Gianello P, Mosala Nezhad Z, Astarci P, Noirhomme P, El Khoury G. Surgical anatomy of the aortic root: implication for valve-sparing reimplantation and aortic valve annuloplasty. The Journal of thoracic and cardiovascular surgery. 2015 Feb:149(2):425-33. doi: 10.1016/j.jtcvs.2014.09.042. Epub 2014 Sep 18 [PubMed PMID: 25439782]
Schäfers HJ, Schmied W, Marom G, Aicher D. Cusp height in aortic valves. The Journal of thoracic and cardiovascular surgery. 2013 Aug:146(2):269-74. doi: 10.1016/j.jtcvs.2012.06.053. Epub 2012 Jul 31 [PubMed PMID: 22853942]
Giebels C, Ehrlich T, Schäfers HJ. Aortic root remodeling. Annals of cardiothoracic surgery. 2023 Jul 31:12(4):369-376. doi: 10.21037/acs-2023-avs2-12. Epub 2023 Jul 6 [PubMed PMID: 37554714]
Gillinov AM, Mihaljevic T, Blackstone EH, George K, Svensson LG, Nowicki ER, Sabik JF 3rd, Houghtaling PL, Griffin B. Should patients with severe degenerative mitral regurgitation delay surgery until symptoms develop? The Annals of thoracic surgery. 2010 Aug:90(2):481-8. doi: 10.1016/j.athoracsur.2010.03.101. Epub [PubMed PMID: 20667334]
American College of Cardiology/American Heart Association Task Force on Practice Guidelines, Society of Cardiovascular Anesthesiologists, Society for Cardiovascular Angiography and Interventions, Society of Thoracic Surgeons, Bonow RO, Carabello BA, Kanu C, de Leon AC Jr, Faxon DP, Freed MD, Gaasch WH, Lytle BW, Nishimura RA, O'Gara PT, O'Rourke RA, Otto CM, Shah PM, Shanewise JS, Smith SC Jr, Jacobs AK, Adams CD, Anderson JL, Antman EM, Faxon DP, Fuster V, Halperin JL, Hiratzka LF, Hunt SA, Lytle BW, Nishimura R, Page RL, Riegel B. ACC/AHA 2006 guidelines for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (writing committee to revise the 1998 Guidelines for the Management of Patients With Valvular Heart Disease): developed in collaboration with the Society of Cardiovascular Anesthesiologists: endorsed by the Society for Cardiovascular Angiography and Interventions and the Society of Thoracic Surgeons. Circulation. 2006 Aug 1:114(5):e84-231 [PubMed PMID: 16880336]
Level 1 (high-level) evidenceHiratzka LF, Bakris GL, Beckman JA, Bersin RM, Carr VF, Casey DE Jr, Eagle KA, Hermann LK, Isselbacher EM, Kazerooni EA, Kouchoukos NT, Lytle BW, Milewicz DM, Reich DL, Sen S, Shinn JA, Svensson LG, Williams DM, American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines, American Association for Thoracic Surgery, American College of Radiology, American Stroke Association, Society of Cardiovascular Anesthesiologists, Society for Cardiovascular Angiography and Interventions, Society of Interventional Radiology, Society of Thoracic Surgeons, Society for Vascular Medicine. 2010 ACCF/AHA/AATS/ACR/ASA/SCA/SCAI/SIR/STS/SVM guidelines for the diagnosis and management of patients with Thoracic Aortic Disease: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines, American Association for Thoracic Surgery, American College of Radiology, American Stroke Association, Society of Cardiovascular Anesthesiologists, Society for Cardiovascular Angiography and Interventions, Society of Interventional Radiology, Society of Thoracic Surgeons, and Society for Vascular Medicine. Circulation. 2010 Apr 6:121(13):e266-369. doi: 10.1161/CIR.0b013e3181d4739e. Epub 2010 Mar 16 [PubMed PMID: 20233780]
Level 3 (low-level) evidenceSvensson LG, Adams DH, Bonow RO, Kouchoukos NT, Miller DC, O'Gara PT, Shahian DM, Schaff HV, Akins CW, Bavaria J, Blackstone EH, David TE, Desai ND, Dewey TM, D'Agostino RS, Gleason TG, Harrington KB, Kodali S, Kapadia S, Leon MB, Lima B, Lytle BW, Mack MJ, Reece TB, Reiss GR, Roselli E, Smith CR, Thourani VH, Tuzcu EM, Webb J, Williams MR. Aortic valve and ascending aorta guidelines for management and quality measures: executive summary. The Annals of thoracic surgery. 2013 Apr:95(4):1491-505. doi: 10.1016/j.athoracsur.2012.12.027. Epub 2013 Jan 2 [PubMed PMID: 23291103]
Level 2 (mid-level) evidenceSharma V, Suri RM, Dearani JA, Burkhart HM, Park SJ, Joyce LD, Li Z, Schaff HV. Expanding relevance of aortic valve repair-is earlier operation indicated? The Journal of thoracic and cardiovascular surgery. 2014 Jan:147(1):100-7. doi: 10.1016/j.jtcvs.2013.08.015. Epub 2013 Sep 29 [PubMed PMID: 24084289]
Level 2 (mid-level) evidenceBoodhwani M, de Kerchove L, Glineur D, Poncelet A, Rubay J, Astarci P, Verhelst R, Noirhomme P, El Khoury G. Repair-oriented classification of aortic insufficiency: impact on surgical techniques and clinical outcomes. The Journal of thoracic and cardiovascular surgery. 2009 Feb:137(2):286-94. doi: 10.1016/j.jtcvs.2008.08.054. Epub 2008 Dec 27 [PubMed PMID: 19185138]
Level 2 (mid-level) evidenceAicher D, Kunihara T, Abou Issa O, Brittner B, Gräber S, Schäfers HJ. Valve configuration determines long-term results after repair of the bicuspid aortic valve. Circulation. 2011 Jan 18:123(2):178-85. doi: 10.1161/CIRCULATIONAHA.109.934679. Epub 2011 Jan 3 [PubMed PMID: 21200006]
Level 2 (mid-level) evidenceBoodhwani M, de Kerchove L, Glineur D, Rubay J, Vanoverschelde JL, Noirhomme P, El Khoury G. Repair of regurgitant bicuspid aortic valves: a systematic approach. The Journal of thoracic and cardiovascular surgery. 2010 Aug:140(2):276-284.e1. doi: 10.1016/j.jtcvs.2009.11.058. Epub 2010 May 20 [PubMed PMID: 20488466]
Level 2 (mid-level) evidenceSadovnick AD, Armstrong H, Rice GP, Bulman D, Hashimoto L, Paty DW, Hashimoto SA, Warren S, Hader W, Murray TJ. A population-based study of multiple sclerosis in twins: update. Annals of neurology. 1993 Mar:33(3):281-5 [PubMed PMID: 8498811]
Vendramin I, Bortolotti U, De Manna DN, Lechiancole A, Sponga S, Livi U. Combined Replacement of Aortic Valve and Ascending Aorta-A 70-Year Evolution of Surgical Techniques. Aorta (Stamford, Conn.). 2021 Jun:9(3):118-123. doi: 10.1055/s-0041-1729913. Epub 2021 Oct 11 [PubMed PMID: 34634836]
Vahanian A, Beyersdorf F, Praz F, Milojevic M, Baldus S, Bauersachs J, Capodanno D, Conradi L, De Bonis M, De Paulis R, Delgado V, Freemantle N, Gilard M, Haugaa KH, Jeppsson A, Jüni P, Pierard L, Prendergast BD, Sádaba JR, Tribouilloy C, Wojakowski W, ESC/EACTS Scientific Document Group. 2021 ESC/EACTS Guidelines for the management of valvular heart disease. European heart journal. 2022 Feb 12:43(7):561-632. doi: 10.1093/eurheartj/ehab395. Epub [PubMed PMID: 34453165]
Boodhwani M, de Kerchove L, Glineur D, Rubay J, Vanoverschelde JL, Van Dyck M, Noirhomme P, El Khoury G. Aortic valve repair with ascending aortic aneurysms: associated lesions and adjunctive techniques. European journal of cardio-thoracic surgery : official journal of the European Association for Cardio-thoracic Surgery. 2011 Aug:40(2):424-8. doi: 10.1016/j.ejcts.2010.11.053. Epub 2011 Jan 13 [PubMed PMID: 21236695]
Level 2 (mid-level) evidenceDavid TE, El Khoury G. Future directions on aortic valve-sparing operations. Annals of cardiothoracic surgery. 2023 Jul 31:12(4):366-368. doi: 10.21037/acs-2023-avs2-17. Epub 2023 Jul 19 [PubMed PMID: 37554708]
Level 3 (low-level) evidencePrice J, De Kerchove L, Glineur D, Vanoverschelde JL, Noirhomme P, El Khoury G. Risk of valve-related events after aortic valve repair. The Annals of thoracic surgery. 2013 Feb:95(2):606-12; discussion 613. doi: 10.1016/j.athoracsur.2012.07.016. Epub 2012 Sep 7 [PubMed PMID: 22959573]
Aicher D, Langer F, Lausberg H, Bierbach B, Schäfers HJ. Aortic root remodeling: ten-year experience with 274 patients. The Journal of thoracic and cardiovascular surgery. 2007 Oct:134(4):909-15 [PubMed PMID: 17903506]
Level 2 (mid-level) evidenceZuo Y, Tan R, Qin C. Outcomes of valve-sparing aortic root replacement in patients with bicuspid aortic valve and tricuspid aortic valve: a systematic review and meta-analysis. Journal of cardiothoracic surgery. 2023 Jul 3:18(1):206. doi: 10.1186/s13019-023-02329-8. Epub 2023 Jul 3 [PubMed PMID: 37400892]
Level 1 (high-level) evidenceBoodhwani M, El Khoury G. Aortic valve repair: a glimpse into the future. European journal of cardio-thoracic surgery : official journal of the European Association for Cardio-thoracic Surgery. 2012 Jan:41(1):2-3. doi: 10.1093/ejcts/ezr013. Epub [PubMed PMID: 22219496]
de Heer F, Lansac E, El-Hamamsy I, Pibarot P, De Kerchove L, El Khoury G, Schäfers HJ, Takkenberg JJM, Kluin J. The AVIATOR registry: the importance of evaluating long-term patient outcomes. Annals of cardiothoracic surgery. 2019 May:8(3):393-395. doi: 10.21037/acs.2019.04.08. Epub [PubMed PMID: 31240184]
Baric D, Sliskovic N, Sestan G, Gjorgjievska S, Unic D, Kusurin M, Varvodic J, Safaric Oremus Z, Jurin I, Bulj N, Susnjar D, Rudez I. Aortic Valve Repair with External Annuloplasty in Bicuspid versus Tricuspid Aortic Valve Patients. Journal of cardiovascular development and disease. 2024 Jan 6:11(1):. doi: 10.3390/jcdd11010017. Epub 2024 Jan 6 [PubMed PMID: 38248887]
Ram D, Bouhout I, Karliova I, Schneider U, El-Hamamsy I, Schäfers HJ. Concepts of Bicuspid Aortic Valve Repair: A Review. The Annals of thoracic surgery. 2020 Apr:109(4):999-1006. doi: 10.1016/j.athoracsur.2019.09.019. Epub 2019 Oct 11 [PubMed PMID: 31610163]