Introduction
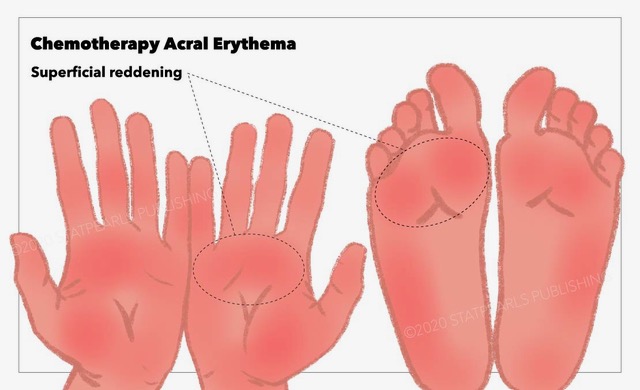
Acral erythema, also known as Palmar-plantar erythrodysesthesia (PPE), palmoplantar erythrodysesthesia, toxic erythema of the palms and soles, hand-foot syndrome (HFS), or Burgdorf reaction. This adverse event is caused by many classic chemotherapeutic agents and newer molecular-targeted therapies. It is characterized by intense, painful erythema of the palms and soles that can progress to the formation of vesicles or bullae.[1][2][3]. See Image. Chemotherapy Acral Erythema.
Etiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Etiology
Doxorubicin (most common with pegylated lysosomal preparation), cytarabine, docetaxel, capecitabine, or 5-fluorouracil (mostly infusional regimen) are the most frequently implicated agents[1][4].
Other drugs causing acral erythema are bleomycin, cisplatin, cyclophosphamide, daunorubicin, doxifluridine, etoposide, fludarabine, gemcitabine, hydroxyurea, idarubicin, ixabepilone, methotrexate, mitotane, paclitaxel, tegafur, thiotepa, and vinorelbine.[5][6][7][8].
It is also postulated that genetic polymorphisms of enzymes involved in the metabolism of some fluoropyrimidines like Capecitabine are the culprit in developing PPE [9]
PPE seems to be dose-dependent, and both peak drug concentration and total cumulative dose determine its occurrence. Drug formulation and administration schedules that result in sustained serum levels of cytotoxic agents are more frequently associated with acral erythema[10][11][1].
Multitargeted tyrosine kinase inhibitors and others that target angiogenesis are associated with hand-foot skin reactions (This is not discussed in the current article). However, clinical and histologic patterns differ from the typical acral erythema that develops with standard cytotoxic agents.
Epidemiology
Overall Incidence of HFS is not well documented. However, the incidence of developing HFS in patients treated with capecitabine is around 60%.
Pathophysiology
Pathogenesis is not very well understood. Researchers think that the accumulation of chemotherapeutic drugs in eccrine glands, which are more numerous in palms and soles, may cause eccrine squamous syringometaplasia (ESS). ESS is characterized by metaplasia and focal necrosis of the epithelium of the eccrine duct. ESS is occasionally detected in conjunction with acral erythema, but such occurrences are relatively uncommon.
Data suggests that Capecitabine (fluoropyrimidine) toxicity may be associated with genetic polymorphisms of dihydropyrimidine dehydrogenase (DPYD) and thymidylate synthase (TYMS), enzymes involved in its metabolism[9]. Data also suggest that palmar skin has a high expression of capecitabine-activating enzyme thymidine phosphorylase leading to a high concentration of active component levels [12].
Histopathology
Histologic changes include dermatitis with necrotic and dyskeratotic keratinocytes, dermal edema, vacuolar degeneration and perivascular lymphocytic infiltrate[5]. However, the biopsy is not usually done promptly for diagnostic purposes.
History and Physical
The condition typically begins with dysesthesias like a tingling sensation in the palms and/or soles and produces symmetric, well-demarcated painful erythema that can progress to blistering with desquamation, erosion, and ulceration. An especially severe bullous variant that advances to full-thickness epidermal necrosis and sloughing has been reported following cytarabine or high-dose intravenous methotrexate, particularly in children.
Although rare, acral erythema involving the penis and scrotum has been reported.
A presumed variant of acral erythema, called fixed erythrodysesthesia plaque (FEP), is characteristic of intravenous (IV) injections of docetaxel. This lesion develops as a fixed, single plaque, proximal to the infusion site, which does not include the palms or soles. It resolves with desquamation and leaves an area of hyperpigmented skin in 5 to 6 weeks.
Temporary Loss of fingerprint is seen with capecitabine-associated, chronic acral erythema and can cause identification issues.
Evaluation
Painful red swelling of the feet and hands in a patient receiving chemotherapy suggests the diagnosis. A high degree of suspicion is the key to diagnosis in patients receiving chemotherapeutic agents. The problem may also occur in patients with bone marrow transplants, as the clinical and histologic features may mimic cutaneous manifestations of graft-versus-host disease.
Dangerous graft-versus-host disease must be differentiated from benign palmar-plantar erythrodysesthesia. Patients with graft-versus-host disease will have progression to other body parts whereas PPE will be limited to hands and feet. Serial biopsies every 3 to 5 days may help in differentiating the two disorders.[5]
Treatment / Management
The mainstay of treatment is the discontinuation of the drug and symptomatic treatment to provide pain relief, decrease edema, and prevent superinfection. Symptomatic treatment includes wound care, alcohol-free emollients, elevation, and pain medication. Healing includes superficial desquamation of affected areas. Often chemotherapy dose intensity modification or reduction is needed if the drug cannot be discontinued or substituted for another cancer drug or cancer treatment.
Usually, it resolves within 2 to 4 weeks of drug cessation.
In the case of severe (grade 3) acral erythema, further chemotherapy doses should be reduced to prevent a recurrence. Depending on overall clinical status switching to an alternative regimen may be necessary if the risk of recurrence is thought to be high.
Differential Diagnosis
- Graft versus host disease (GVHD)
- Steven johnson syndrome (SJS)
- Toxic epidermal necrolysis (TEN)
- Fixed drug eruption
Treatment Planning
HFS is dose-related. The treatment for HFS is dose modification or drug interruption.
HFS resolves within two to four weeks of discontinuation of the chemotherapy. In patients who are getting capecitabine, topical urea 10% cream can be used for the prevention of HFS from capecitabine.
For severe grade 3 HFS, subsequent doses of chemotherapy need to be reduced.
Toxicity and Adverse Effect Management
Regional cooling using ice packs to wrists and ankles during therapy may help in decreasing the frequency and severity of symptoms by decreasing blood flow [13][14]. It is usually a well-tolerated prevention strategy.
Randomized clinical trials using topical urea preparation to hands and feet revealed contradictory results depending on the dosage and duration of treatment in patients receiving capecitabine. Further studies need to validate the use to be applied in clinical practice[15][16].
Studies including meta-analysis conducted using pyridoxine (Vitamin B6) reveal varying results[17][18][19]
Oral Celecoxib 200 to 400 mg twice a day for 12 to 18 weeks significantly reduced the risk of grades 2 and 3 HFS (odds ratio 0.37, 95% CI 0.19-0.71) in a meta-analysis of randomized trials[20]. However, celecoxib is known for its potential cardiovascular adverse effects with long-term use and upper gastrointestinal (GI) risk of bleeding. The benefit-to-risk ratio is not favorable for the extensive use of celecoxib for the prevention or treatment of HFS [3].
Studies using systemic corticosteroids in the management of HFS are also not convincing resulting in the need for large randomized clinical trials[21]
Medical Oncology
Tyrosine kinase inhibitors like sunitinib, sorafenib, bevacizumab which target angiogenesis are also associated with hand-foot skin reaction (HFSR) which is different from HFS from conventional chemotherapy. Studies have shown that HFSR showed an association with tumor response and overall survival. [22] Combined use of chemotherapy and TKI has increased in the recent past. It is crucial to differentiate the side effects between side effects from both the agents to guide the reduction in the appropriate therapy if needed. HFS from cytotoxic chemotherapy causes diffuse erythema and desquamation compared to hyperkeratotic lesions in HSFR.
Staging
According to the national cancer institute, common terminology criteria for adverse events (NCI CTCAE version 5), Grading of Severity of HFS is classified as follows
- Grade 1: Minimal skin changes or dermatitis (e.g., erythema or edema) without pain
- Grade 2: Skin changes (e.g., peeling, blisters, bleeding, or hyperkeratosis) with pain leading to limitation of instrumental activities of daily living (ADL)
- Grade 3: Severe skin changes (e.g., peeling, blisters, bleeding, edema, or hyperkeratosis) with pain, limiting basic self-care ADL [3]
Complications
PPE can cause a significant impact on a patient's quality of life. There are usually no long-term after-effects; however, palmoplantar keratoderma may develop as a result of long-standing acral erythema. Fingerprint loss is reversible after treatment discontinuation.
Enhancing Healthcare Team Outcomes
Acral erythema is also known as palmar-plantar erythrodysesthesia (PPE), palmoplantar erythrodysesthesia, hand-foot syndrome (HFS), or Burgdorf reaction. This is an adverse event caused by many classic chemotherapeutic agents and newer molecular targeted therapies. It is an intense, painful erythema of the palms and soles that can progress to the formation of vesicles or bullae. Healthcare professionals should be familiar with acral erythema while managing patients on chemotherapy. The condition is best managed by an interprofessional team that includes the dermatologist, oncologist, oncology nurse, pharmacist, and primary care provider. Patients and families need to be educated about the condition to be vigilant in monitoring the symptoms. When the condition is diagnosed, depending on the overall clinical condition and severity of symptoms decisions regarding decreasing the dose or discontinuation of treatment need to be made with shared decision making.
Regional cooling with ice packs around wrists and ankles is a well-tolerated strategy with no side effects and should be tried. There is a need for large randomized controlled trials to formulate appropriate management options to treat the condition effectively. Overall, the quality of life is poor if the condition is not adequately treated [Level V][5].
Media
(Click Image to Enlarge)
References
Ozmen S, Dogru M, Bozkurt C, Kocaoglu AC. Probable cytarabine-induced acral erythema: report of 2 pediatric cases. Journal of pediatric hematology/oncology. 2013 Jan:35(1):e11-3. doi: 10.1097/MPH.0b013e3182580ba0. Epub [PubMed PMID: 22767133]
Level 3 (low-level) evidenceGiacchero D, Monpoux F, Chiavérini C, Lacour JP. [6-mercaptopurine-related hand-foot syndrome in a four-year-old child]. Annales de dermatologie et de venereologie. 2008 Aug-Sep:135(8-9):580-3. doi: 10.1016/j.annder.2008.02.021. Epub 2008 May 27 [PubMed PMID: 18789293]
Level 3 (low-level) evidenceDegen A, Alter M, Schenck F, Satzger I, Völker B, Kapp A, Gutzmer R. The hand-foot-syndrome associated with medical tumor therapy - classification and management. Journal der Deutschen Dermatologischen Gesellschaft = Journal of the German Society of Dermatology : JDDG. 2010 Sep:8(9):652-61. doi: 10.1111/j.1610-0387.2010.07449.x. Epub 2010 May 6 [PubMed PMID: 20482685]
Chidharla A,Kasi A, Cancer, Chemotherapy Acral Erythema (Palmar-Plantar Erythrodysesthesia, Palmoplantar Erythrodysesthesia, Hand-Foot Syndrome) 2018 Jan; [PubMed PMID: 29083584]
Hueso L, Sanmartín O, Nagore E, Botella-Estrada R, Requena C, Llombart B, Serra-Guillén C, Alfaro-Rubio A, Guillén C. [Chemotherapy-induced acral erythema: a clinical and histopathologic study of 44 cases]. Actas dermo-sifiliograficas. 2008 May:99(4):281-90 [PubMed PMID: 18394404]
Level 3 (low-level) evidenceGupta A, Sardana K, Bhardwaj M, Singh A. Methotrexate Cutaneous Toxicity following a Single Dose of 10 mg in a Case of Chronic Plaque Psoriasis: A Possible Idiosyncratic Reaction. Indian dermatology online journal. 2018 Sep-Oct:9(5):328-330. doi: 10.4103/idoj.IDOJ_316_17. Epub [PubMed PMID: 30258802]
Level 3 (low-level) evidenceJalandhara P, Kaeley G. Cutaneous toxicity of oral low-dose methotrexate. Proceedings (Baylor University. Medical Center). 2018 Jul:31(3):364-366. doi: 10.1080/08998280.2018.1463039. Epub 2018 May 22 [PubMed PMID: 29904314]
Amjad MT, Chidharla A, Kasi A. Cancer Chemotherapy. StatPearls. 2024 Jan:(): [PubMed PMID: 33232037]
Rosmarin D, Palles C, Church D, Domingo E, Jones A, Johnstone E, Wang H, Love S, Julier P, Scudder C, Nicholson G, Gonzalez-Neira A, Martin M, Sargent D, Green E, McLeod H, Zanger UM, Schwab M, Braun M, Seymour M, Thompson L, Lacas B, Boige V, Ribelles N, Afzal S, Enghusen H, Jensen SA, Etienne-Grimaldi MC, Milano G, Wadelius M, Glimelius B, Garmo H, Gusella M, Lecomte T, Laurent-Puig P, Martinez-Balibrea E, Sharma R, Garcia-Foncillas J, Kleibl Z, Morel A, Pignon JP, Midgley R, Kerr D, Tomlinson I. Genetic markers of toxicity from capecitabine and other fluorouracil-based regimens: investigation in the QUASAR2 study, systematic review, and meta-analysis. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2014 Apr 1:32(10):1031-9. doi: 10.1200/JCO.2013.51.1857. Epub 2014 Mar 3 [PubMed PMID: 24590654]
Level 1 (high-level) evidenceGambichler T, Strutzmann S, Tannapfel A, Susok L. Paraneoplastic acral vascular syndrome in a patient with metastatic melanoma under immune checkpoint blockade. BMC cancer. 2017 May 12:17(1):327. doi: 10.1186/s12885-017-3313-6. Epub 2017 May 12 [PubMed PMID: 28499411]
Caccavale S, Ruocco E. Acral manifestations of systemic diseases: Drug-induced and infectious diseases. Clinics in dermatology. 2017 Jan-Feb:35(1):55-63. doi: 10.1016/j.clindermatol.2016.09.008. Epub 2016 Sep 10 [PubMed PMID: 27938813]
Levine LE, Medenica MM, Lorincz AL, Soltani K, Raab B, Ma A. Distinctive acral erythema occurring during therapy for severe myelogenous leukemia. Archives of dermatology. 1985 Jan:121(1):102-4 [PubMed PMID: 3855356]
Level 3 (low-level) evidenceMolpus KL, Anderson LB, Craig CL, Puleo JG. The effect of regional cooling on toxicity associated with intravenous infusion of pegylated liposomal doxorubicin in recurrent ovarian carcinoma. Gynecologic oncology. 2004 May:93(2):513-6 [PubMed PMID: 15099971]
Level 2 (mid-level) evidenceMangili G, Petrone M, Gentile C, De Marzi P, Viganò R, Rabaiotti E. Prevention strategies in palmar-plantar erythrodysesthesia onset: the role of regional cooling. Gynecologic oncology. 2008 Feb:108(2):332-5 [PubMed PMID: 18083217]
Wolf SL, Qin R, Menon SP, Rowland KM Jr, Thomas S, Delaune R, Christian D, Pajon ER Jr, Satele DV, Berenberg JL, Loprinzi CL, North Central Cancer Treatment Group Study N05C5. Placebo-controlled trial to determine the effectiveness of a urea/lactic acid-based topical keratolytic agent for prevention of capecitabine-induced hand-foot syndrome: North Central Cancer Treatment Group Study N05C5. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2010 Dec 10:28(35):5182-7. doi: 10.1200/JCO.2010.31.1431. Epub 2010 Nov 8 [PubMed PMID: 21060036]
Level 1 (high-level) evidenceHofheinz RD,Gencer D,Schulz H,Stahl M,Hegewisch-Becker S,Loeffler LM,Kronawitter U,Bolz G,Potenberg J,Tauchert F,Al-Batran SE,Schneeweiss A, Mapisal Versus Urea Cream as Prophylaxis for Capecitabine-Associated Hand-Foot Syndrome: A Randomized Phase III Trial of the AIO Quality of Life Working Group. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2015 Aug 1 [PubMed PMID: 26124485]
Level 2 (mid-level) evidenceKang YK,Lee SS,Yoon DH,Lee SY,Chun YJ,Kim MS,Ryu MH,Chang HM,Lee JL,Kim TW, Pyridoxine is not effective to prevent hand-foot syndrome associated with capecitabine therapy: results of a randomized, double-blind, placebo-controlled study. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2010 Aug 20 [PubMed PMID: 20625131]
Level 1 (high-level) evidenceChalermchai T, Tantiphlachiva K, Suwanrusme H, Voravud N, Sriuranpong V. Randomized trial of two different doses of pyridoxine in the prevention of capecitabine-associated palmar-plantar erythrodysesthesia. Asia-Pacific journal of clinical oncology. 2010 Sep:6(3):155-60. doi: 10.1111/j.1743-7563.2010.01311.x. Epub [PubMed PMID: 20887495]
Level 1 (high-level) evidenceChen M, Zhang L, Wang Q, Shen J. Pyridoxine for prevention of hand-foot syndrome caused by chemotherapy: a systematic review. PloS one. 2013:8(8):e72245. doi: 10.1371/journal.pone.0072245. Epub 2013 Aug 20 [PubMed PMID: 23977264]
Level 1 (high-level) evidenceMacedo LT, Lima JP, dos Santos LV, Sasse AD. Prevention strategies for chemotherapy-induced hand-foot syndrome: a systematic review and meta-analysis of prospective randomised trials. Supportive care in cancer : official journal of the Multinational Association of Supportive Care in Cancer. 2014 Jun:22(6):1585-93. doi: 10.1007/s00520-014-2129-z. Epub 2014 Jan 26 [PubMed PMID: 24463616]
Level 1 (high-level) evidenceDrake RD, Lin WM, King M, Farrar D, Miller DS, Coleman RL. Oral dexamethasone attenuates Doxil-induced palmar-plantar erythrodysesthesias in patients with recurrent gynecologic malignancies. Gynecologic oncology. 2004 Aug:94(2):320-4 [PubMed PMID: 15297168]
Jain L, Sissung TM, Danesi R, Kohn EC, Dahut WL, Kummar S, Venzon D, Liewehr D, English BC, Baum CE, Yarchoan R, Giaccone G, Venitz J, Price DK, Figg WD. Hypertension and hand-foot skin reactions related to VEGFR2 genotype and improved clinical outcome following bevacizumab and sorafenib. Journal of experimental & clinical cancer research : CR. 2010 Jul 14:29(1):95. doi: 10.1186/1756-9966-29-95. Epub 2010 Jul 14 [PubMed PMID: 20630084]
Level 2 (mid-level) evidence