Introduction
Traditionally, carotid endarterectomy (CEA) has been the primary method of treating high-grade asymptomatic and symptomatic carotid artery stenosis. A carotid endarterectomy involves exposure of the carotid artery and removal of plaque, most typically from the carotid bulb and the proximal internal carotid artery, via a neck incision. However, in vascular surgery, as in many other surgical specialties, minimally invasive techniques have evolved over the years. These techniques offer the advantages of smaller incisions, reduced postoperative pain, reduced potential for postoperative wound complications, and a shorter length of stay in the hospital. Carotid artery stenting (CAS) is one such technique, which can be performed via a transfemoral approach or a transcarotid approach.[1][2][3]
Anatomy and Physiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Anatomy and Physiology
The common carotid artery arises from the aortic arch (left) or the brachiocephalic trunk (right). It divides into internal and external carotid arteries. The internal carotid artery provides blood to the brain while the external carotid artery provides blood to the face, scalp and the neck. The internal and external carotid arteries form collaterals at several locations. This is helpful for the maintenance of blood flow through collateral circulation in case either artery becomes occluded. The aortic arch can be divided into three anatomic types. To simplify, an imaginary horizontal line is drawn through the top of the aortic arch, and imaginary parallel lines marking the origins of the great vessels coming off the arch are drawn for comparison. For a type 1 arch, all great vessels originate close to the imaginary horizontal line drawn through the top of the aortic arch. If the origins of all great vessels fall within the second parallel line, this is considered a type 2 aortic arch. For a type 3 aortic arch, the origins of all great vessels fall within the third parallel line. The angulation, as related to the type of aortic arch, has important implications for CAS.
Indications
CAS is a reasonable alternative to CEA in select patients with high-grade asymptomatic (more than 70%) or symptomatic carotid artery stenosis. Indications for CAS include high surgical patient risk, such as severe pulmonary disease, recent myocardial infarction, unstable angina, or severe congestive heart failure; a history of prior neck radiation that is anticipated to make open surgical dissection difficult; a history of damage to contralateral vocal cords; the presence of a tracheostomy, contralateral carotid occlusion; and previous CEA with recurrent stenosis.[4][5][6][7]
Contraindications
The main contraindication to CAS via a transfemoral approach is unfavorable aortic arch anatomy. This can include a heavily calcified aortic arch or a type 3 aortic arch. A relative contraindication can be a history of severe allergic reaction to intravenous contrast dye. However, patients can be pre-medicated to mitigate some of this risk. For a transcarotid approach, a common carotid artery length of less than 5 cm from the clavicle is considered inadequate.
Equipment
An ultrasound unit, a fluoroscopic imaging system (such as a C-arm), lead shielding, sterile drapes, heparinized saline, and heparin is required for both types of approaches. An ACT machine should be available to ensure that the ACT after heparinization is greater than 250 seconds.
The transfemoral approach requires the following equipment:
- micropuncture kit for percutaneous access of the common femoral artery,
- 0.035 inch and 0.014-inch wires,
- 6 French short and long sheaths,
- angled catheters,
- angioplasty balloons,
- embolic protection device or flow reversal system.
The transcarotid approach requires the following:
- a surgical tray,
- vascular clamps to perform surgical exposure of the common carotid artery at the base of the neck and allow for direct puncture of the artery using a micropuncture kit,
- additional wires, sheaths, and balloons may be used as described above.
- a flow reversal system for neuroprotection against potential embolization.
Personnel
The following personnel is usually required for the procedure: proceduralist (such as a vascular surgeon), first assistant (fellow/resident/physician assistant), anesthesiologist, radiology technician and nursing personnel (scrub nurse technician, circulator nurse).
Preparation
In case of a transfemoral approach, most physicians choose to perform CAS under local anesthesia with or without sedation to monitor the patient’s neurologic status. For the transcarotid approach, either local anesthesia with sedation or general anesthesia may be employed. If general anesthesia is employed, use of intraoperative EEG/brain mapping should be considered.
Regardless of the approach, patients should be closely monitored during the procedure. As with CEA, CAS can cause dramatic changes in the patient’s hemodynamics, leading to hypotension or bradycardia. Anesthesia personnel should have vasoactive medications available to treat such hemodynamically significant changes. It is also prudent to have an arterial line in place for invasive hemodynamic monitoring during the procedure. Patients should be on aspirin and Plavix preoperatively, and these should be continued in the postoperative period. All patients should be asked about aspirin and Plavix use in the preoperative holding area.
For the transfemoral approach, the patient should be supine and the neck should be turned to the opposite side. For the transcarotid approach, the patient should be supine with a shoulder roll placed underneath the shoulders and the neck turned to the opposite side. Full sterile precautions should be maintained throughout the procedure as with any invasive procedure. If the transfemoral approach is being used, both groins should be prepped and draped in the usual sterile fashion.
For the transcarotid approach, the neck should be prepped and draped in addition to the groins. Arms can be tucked for both approaches.
Technique or Treatment
Transfemoral approach: This technique is believed to provide better maneuverability. However, it can be limited by aortic arch anatomy and calcifications. Under ultrasound guidance, the common femoral artery is accessed percutaneously using the micropuncture kit using the Seldinger technique. A femoral angiogram is recommended at this time to identify proper anatomic entry into the common femoral artery and to assess suitability for use of a vascular access closure device at the conclusion of the procedure. A short 6 Fr sheath is then placed over a 0.035 in the wire. The wire is advanced into the aortic arch carefully. Using an angled catheter and a left anterior oblique projection on the fluoroscopic system, the common carotid artery is selectively cannulated and mapped. The guide wire is then advanced into the external carotid artery followed by advancement of the catheter into the external carotid artery. Over a stiffer wire, a shuttle sheath is advanced into the common carotid artery. Dedicated carotid arteriograms are performed in multiple views to identify the lesion. A 0.014 in wire should be used to traverse the lesion followed by placement of embolic protection device. Pre-dilation of the lesion is performed using a small profile balloon. Careful attention should be paid to the patient’s hemodynamics during this maneuver and subsequently. After pre-dilation, an appropriately sized stent is deployed. If significant residual stenosis exists after the deployment of the stent (greater than 30 percent), balloon angioplasty can be performed to rectify this. Completion carotid and a cerebral angiogram is performed, followed by the removal of catheters and sheaths if the result is satisfactory. A closure device is then used to close the arteriotomy site in the common femoral artery.
Transcarotid approach: This technique involves direct surgical exposure of the common carotid artery at the base of neck. Duplex ultrasound can be used to plan the surgical incision over the course of the common carotid artery in addition to the use of traditional anatomic landmarks. Dissection is carried through the subcutaneous tissues using bovie electrocautery. The platysma is identified and divided. The common carotid artery is then identified and dissected out circumferentially. A vessel loop or umbilical tape is then placed around the artery. Additional proximal and distal part of the common carotid artery is dissected out as well. Using a micropuncture kit, the common carotid artery is accessed directly, and an angiogram is obtained. A guidewire is then positioned in the common carotid artery, and the arterial part of the flow reversal system is introduced over it. The common femoral vein is punctured under ultrasound guidance using a micropuncture kit, and a 0.035 guidewire is then passed into the femoral vein followed by placement of the venous sheath of the flow reversal system. The venous sheath is then attached to the arterial sheath and reverse flow is established. Carotid angiography is then performed in multiple projections. A guidewire is then used to traverse the lesion, followed by placement of clamp over the proximal common carotid artery. After this maneuver, pre-dilation and stent deployment are performed to treat the carotid artery lesion. Significant residual stenosis after stent deployment (generally greater than 30%) is treated with balloon angioplasty. Following completion angiography, the wires and catheters are removed. The distal common carotid artery is clamped, and suture-mediated closure of the arteriotomy is performed using prolene suture. The neck incision is closed in multiple layers. The femoral vein sheath is removed followed by application of manual pressure to achieve hemostasis.
Complications
For the transfemoral approach, complications can occur related to femoral artery access. These include bleeding, perforation, dissection, thrombosis, and distal embolization. Procedural complications from either approach include carotid artery dissection or embolization. The stent may not deploy properly and can be associated with fracture.
Clinical Significance
There have been multiple studies comparing CAS with CEA in both symptomatic and asymptomatic patients. The risk of myocardial ischemia appears to be higher with CEA, while the risk of a cerebrovascular accident (CVA) appears to be higher with CAS. Restenosis rates appear to be similar between CEA and CAS after two years. CAS, as a procedure, appears to have worse outcomes in patients aged 70 years and over and in patients with a high burden of white-matter lesions on brain imaging. Therefore, patient selection is important for CAS. The transcarotid approach of CAS potentially reduces the risk of embolization in case of a hostile aortic arch. More experience with this technique is being gained in ongoing trials. Hospitalization and long-term costs of CAS and CEA appear similar. [8][9][10]
Enhancing Healthcare Team Outcomes
Carotid artery disease is often encountered by the primary care provider, nurse practitioner and the internist. Because the disorder can lead to a stroke, it is important to refer these patients to a vascular surgeon or an interventional radiologist. Both the open procedure and stenting are viable options for patients with carotid artery disease. Once treated, these patients need to be educated on prevention of atherosclerosis by changing lifestyle, reducing body weight, lowering cholesterol and participating in exercise.
Media
(Click Image to Enlarge)
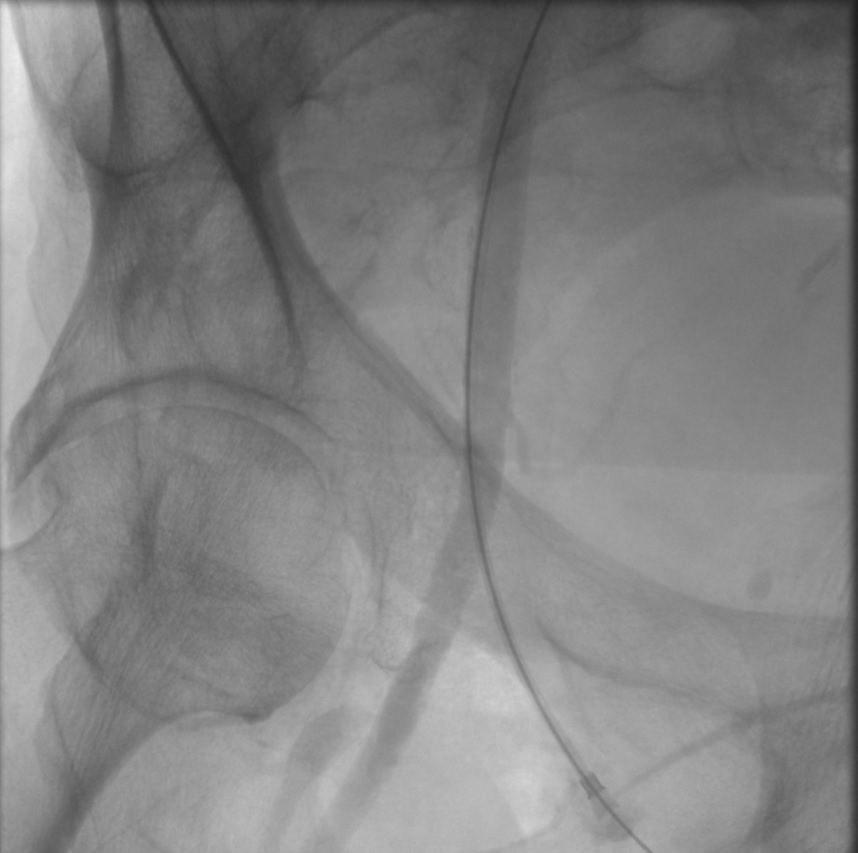
Access into Right Common Femoral Artery using micropuncture system during transfemoral carotid artery stenting. This angiogram shows adequate entry into the artery below the most superior aspect of the femoral head - a useful fluoroscopic landmark. There is no dissection in the artery.
Contributed by Taimur Saleem, MD
References
Texakalidis P, Chaitidis N, Giannopoulos S, Giannopoulos S, Machinis T, Jabbour P, Rivet D, Reavey-Cantwell J, Rangel-Castilla L. Carotid Revascularization in Older Adults: A Systematic Review and Meta-Analysis. World neurosurgery. 2019 Jun:126():656-663.e1. doi: 10.1016/j.wneu.2019.02.030. Epub 2019 Feb 22 [PubMed PMID: 30797928]
Level 1 (high-level) evidenceGaba K, Bulbulia R. Identifying asymptomatic patients at high-risk for stroke. The Journal of cardiovascular surgery. 2019 Jun:60(3):332-344. doi: 10.23736/S0021-9509.19.10912-3. Epub 2019 Feb 20 [PubMed PMID: 30785251]
Mukherjee D, Roffi M. Minimizing Distal Embolization During Carotid Artery Stenting. JACC. Cardiovascular interventions. 2019 Feb 25:12(4):404-405. doi: 10.1016/j.jcin.2018.12.024. Epub [PubMed PMID: 30784648]
Rasheed AS, White RS, Tangel V, Storch BM, Pryor KO. Carotid Revascularization Procedures and Perioperative Outcomes: A Multistate Analysis, 2007-2014. Journal of cardiothoracic and vascular anesthesia. 2019 Jul:33(7):1963-1972. doi: 10.1053/j.jvca.2019.01.022. Epub 2019 Jan 9 [PubMed PMID: 30773439]
Lan Y, Lyu J, Ma X, Ma L, Lou X. Longitudinal assessment of cerebral blood flow changes following carotid artery stenting and endarterectomy. La Radiologia medica. 2019 Jul:124(7):636-642. doi: 10.1007/s11547-018-00986-7. Epub 2019 Feb 15 [PubMed PMID: 30771219]
Chung JW, Kim SJ, Hwang J, Lee MJ, Lee J, Lee KY, Park MS, Sung SM, Kim KH, Jeon P, Bang OY. Comparison of Clopidogrel and Ticlopidine/Ginkgo Biloba in Patients With Clopidogrel Resistance and Carotid Stenting. Frontiers in neurology. 2019:10():44. doi: 10.3389/fneur.2019.00044. Epub 2019 Jan 30 [PubMed PMID: 30761076]
Lim S, Javorski MJ, Nassoiy SP, Park Y, Halandras PM, Bechara CF, Aulivola B, Crisostomo PR. Long-Term Hemodynamic Effects After Carotid Artery Revascularization. Vascular and endovascular surgery. 2019 May:53(4):297-302. doi: 10.1177/1538574419828088. Epub 2019 Feb 11 [PubMed PMID: 30744510]
Xin WQ, Li MQ, Li K, Li QF, Zhao Y, Wang WH, Gao YK, Wang HY, Yang XY. Systematic and Comprehensive Comparison of Incidence of Restenosis Between Carotid Endarterectomy and Carotid Artery Stenting in Patients with Atherosclerotic Carotid Stenosis. World neurosurgery. 2019 May:125():74-86. doi: 10.1016/j.wneu.2019.01.118. Epub 2019 Jan 31 [PubMed PMID: 30710719]
Level 1 (high-level) evidenceHintze AJ, Greenleaf EK, Schilling AL, Hollenbeak CS. Thirty-day Readmission Rates for Carotid Endarterectomy Versus Carotid Artery Stenting. The Journal of surgical research. 2019 Mar:235():270-279. doi: 10.1016/j.jss.2018.10.011. Epub 2018 Nov 1 [PubMed PMID: 30691806]
Clavel P, Hebert S, Saleme S, Mounayer C, Rouchaud A, Marin B. Cumulative incidence of restenosis in the endovascular treatment of extracranial carotid artery stenosis: a meta-analysis. Journal of neurointerventional surgery. 2019 Sep:11(9):916-923. doi: 10.1136/neurintsurg-2018-014534. Epub 2019 Jan 31 [PubMed PMID: 30705056]
Level 1 (high-level) evidence