Introduction
Diabetic retinopathy (DR) is a microvascular disorder occurring due to the long-term effects of diabetes mellitus. Diabetic retinopathy may lead to vision-threatening damage to the retina, eventually leading to blindness. It is the most common cause of severe vision loss in adults of working age groups in the western world.[1] Early detection and timely intervention are the keys to avoiding blindness due to diabetic retinopathy. The number of patients with diabetic retinopathy in America is estimated to reach 16.0 million by 2050, with vision-threatening complications affecting around 3.4 million of them.[2] The usefulness of strict glycemic control was clearly seen in clinical trials like the UK Prospective Diabetes Study (UKPDS) and Diabetes Control and Complication Trial (DCCT).[3][4]
Uncontrolled diabetes can lead to many ocular disorders like cataracts, glaucoma, ocular surface disorders, recurrent stye, non-arteritic anterior ischemic optic neuropathy, diabetic papillopathy, and diabetic retinopathy. Diabetic retinopathy may lead to vision-threatening damage to the retina, eventually leading to blindness; it is the most common and severe ocular complication.[5][6][7] Poor glycemic control, uncontrolled hypertension, dyslipidemia, nephropathy, male sex, and obesity are associated with worsening diabetic retinopathy.[8][9] Typical fundus features of diabetic retinopathy include microaneurysms, hard exudates, macular edema (diabetic macular edema or DME), and new vessels (in proliferative DR or PDR). The management options include strict control of the systemic conditions, intravitreal pharmacotherapy, and laser photocoagulation. With early diagnosis and prompt management, good final visual acuity may be achieved in most patients with DR.
Etiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Etiology
Diabetic retinopathy affects people with diagnosed or undiagnosed diabetes mellitus. The propensity to develop diabetic retinopathy is directly proportional to the patient's age and duration of diabetes, as well as poor glycemic control and fluctuating blood pressure levels.[10]
Risk factors for diabetic retinopathy can be classified into:
- Non-modifiable
- Puberty
- Pregnancy
- Modifiable
- Hypertension
- Obesity
- Dyslipidemia
- Poor glycemic control
- Nephropathy
- Newer risk factors
- Inflammation
- Apolipoprotein
- Hormonal influence - leptin and adiponectin
- Vitamin D
- Oxidative stress
- Genetic factors[11]
Epidemiology
Diabetic retinopathy is one of the major neurovascular complications of diabetes and is a leading cause of blindness in adults of the working-age group. According to the recent epidemiological data shared by the American Academy of Ophthalmology, the global burden of diabetes mellitus is 387 million, which is estimated to increase to 592 million by 2035. Ninety-three million people are globally affected by diabetic retinopathy. Prevalence of diabetic retinopathy is 77.3% in type 1 diabetes patients and 25.1% in type 2 diabetes patients, out of which approximately 25% to 30% are expected to develop vision-threatening diabetic macular edema.[12] Between 5% and 8% of patients with diabetic retinopathy need laser treatment.[13] As many as 5% of patients will require vitrectomy surgery.[14]
Pathophysiology
Chronic hyperglycemia is considered to be the primary pathogenic agent in DR (as described by UKPDS and DCCT).[4][3] Hyperglycemia leads to the activation of alternative pathways of glucose metabolism, including the polyol pathway. The oxidative stress, protein kinase C activation, and non-enzymatic protein glycation lead to advanced glycation endproducts (AGEs). The result of these alternative pathways is the activation of cytokines along with the growth factors and vascular endothelial dysfunction, which eventually leads to increased vascular permeability and microvascular occlusion. Retinal ischemia, which occurs as a consequence of microvascular occlusion, leads to the formation of IRMA (intraretinal microvascular abnormalities) and neovascularization.[15][16]
In the polyol pathway, glucose is reduced to sorbitol by the aldose reductase enzyme. The impermeability of sorbitol leads to its accumulation in all retinal cells leading to osmotic damage to the cells. Also, the use of NADPH (reduced nicotinamide adenine dinucleotide phosphate) during the reduction process leads to further oxidative damage.
Oxidative stress results from increased levels of reactive oxygen species (ROS), leading to cell and tissue damage.
Protein kinase C is involved in signal transduction. Its activation leads to basement membrane alterations and vascular changes like increased vascular permeability, the release of angiogenic growth factors, vascular stasis, and capillary occlusion.
In non-enzymatic protein glycation, reducing sugars react with free amino acids of nucleic acids, proteins, and lipids leading to the formation of advanced glycation endproducts responsible for the alterations in extracellular matrix proteins.
The morphological changes seen in small retinal vessels in DR include early loss of pericytes, basement membrane thickening, loss of endothelial cells, increased vascular permeability, platelet aggregation, leukostasis, and capillary dropout.[17][18]
Diabetic retinopathy does not only affect the microvessels of the retina but also the Müller cells, which are the primary glial cells of the retina. The functions of Müller cells are maintaining the structural integrity of the retina, regulation of the blood-retinal barrier and retinal blood flow, uptake and recycling of various neurotransmitters, retinoic acid compounds, and ions (such as potassium), regulation of metabolism and supply of nutrients to the retina.[19]
In diabetes, there is downregulation of the Kir 4.1 channel because of which there is continued potassium uptake leading to swelling of Müller cells, which leads to Muller cell dysfunction.[20] Fluid accumulation inside the Muller cells is responsible for DME.[21][20] Early inner retinal neuronal and Müller cell involvement may be noted in preclinical and early clinical DR.[22] Activation of Müller cells and overexpression of glial fibrillary acidic protein (GFAP) are noted in DR.[19]
Hyperglycemia leads to the release of growth factors and cytokines/chemokines.[23]
- Growth factors:
- Vascular endothelial growth factor (VEGF)
- Pigment epithelium-derived factor (PEDF)[24]
- Platelet-derived growth factor(PDGF)[25]
- Basic fibroblast growth factor ( bFGF or FGF2)
- Insulin-like growth factor (IGF)
- Hepatocyte growth factor/scatter factor (HGF/SF)
- Placental growth factor (PIGF)[26]
- Erythropoietin
- Angiopoietin-2
- Cytokines and chemokines:
Histopathology
Histologically diabetic retinopathy presents with microangiopathy associated with vascular changes like focal capillary closure, dilatation of venules, hyalinization of arterioles, and capillary changes like basement membrane thickening, pericyte degeneration, and focal outpouchings (microaneurysms). Capillary closure leads to cotton wool spots and ischemic areas, leading to the development of IRMA (intraretinal microvascular abnormalities) and neovascularization, followed by fibrosis and contracture of the retina.[29][30]
In diabetic retinopathy, the uptake of glutamate by Müller cells decreases, leading to glutamate toxicity.[31] Also, potassium conductance is decreased in proliferative diabetic retinopathy.[32]
History and Physical
Proper history taking should be done. This includes the duration of diabetes, type of diabetes mellitus, past glycemic control (HbA1C), medication, and associated systemic disorders like obesity, kidney disease, hypertension, pregnancy, dyslipidemia, and nephropathy.
Patients might be asymptomatic in the early stages and might be discovered incidentally on fundus examination. As the disease progresses, the symptoms include blurred vision, distorted vision, floaters, and partial or total vision loss.
Signs seen in diabetic retinopathy on fundus examination include:
Microaneurysms
Microaneuryms are the earliest clinically detectable lesions.[33] Microaneurysms initially appear temporal to the fovea and may disappear with time. The diameter of a microaneurysm is 15- 60 µm (less than 125 µm). Microaneurysms with a diameter of less than 30 µm may not be detectable clinically.[34] The features of microaneurysms include:
- Focal saccular dilatation of the capillary wall
- Location in the inner nuclear layer
- Clinically identified by ophthalmoscopy as tiny, round, red dots with a sharp regular margin
Microaneurysms are differentiated from dot hemorrhages by FFA (fundus fluorescein angiogram), wherein microaneurysms show tiny hyperfluorescent points, whereas dot hemorrhages show blocked fluorescence. Dot hemorrhages are clinically larger and may have an irregular margin.[35]
Hemorrhages
Weakened capillary wall ruptures lead to intraretinal dot hemorrhages. Superficial or flame-shaped hemorrhages arise from the precapillary arterioles located in the retinal nerve fiber layer. Deep hemorrhages or dot and blot hemorrhages are located in the inner nuclear and outer plexiform layers of the retina.
Hard Exudates
They are composed of lipoprotein and lipid-filled macrophages located in the outer plexiform layer. They develop at the junction of the edematous and non-edematous retina. The hard exudates typically form a circinate ring around a leaking microaneurysm.
Cotton Wool Spots/Soft Exudates
They are located in the retinal nerve fiber layer (axoplasmic debris) and represent focal infarcts of the precapillary arterioles.
IRMA (intraretinal microvascular abnormalities)
IRMAs are intercommunications between retinal arteriole and venules, which bypass the capillaries and are seen near the areas of capillary closure. IRMAs are intraretinal in location, do not cross the major vessels, and do not leak on fluorescein angiography.
Venous Changes
- Dilatation
- Looping
- Beading
- Sausage-like segmentation
Arterial Changes
- Peripheral narrowing
- Silver-wiring
- Obliteration
Neovascularization
Neovascularization (new vessels) at the disc (NVD) is defined as neovascularization at or within one disc diameter of the optic disc. Neovascularization elsewhere (NVE) is defined as a new vessel away from one disc diameter of the optic disc. Neovascularization of Iris (NVI) is a marker of poor prognosis and is associated with the propensity to develop neovascular glaucoma.
Early Treatment Diabetic Retinopathy Study (ETDRS) Classification
Nonproliferative Diabetic Retinopathy
- No retinopathy: No retinal lesions
- Very mild NPDR: Microaneurysms only
- Mild NPDR: A few microaneurysms, retinal hemorrhages & hard exudates
- Moderate NPDR: Retinal hemorrhages (about 20 medium-large per quadrant) in 1-3 quadrant + cotton wool spots (between the grades mild and severe NPDR)
- Severe NPDR: fulfilling one rule of the 4-2-1 rule.
- 4-2-1 rule
- Severe hemorrhages in all four quadrants
- Venous beading in 2 or more quadrants
- Moderate IRMA in 1 or more quadrants
- 4-2-1 rule
- Very Severe NPDR: fulfilling two or more rules of the 4-2-1 rule.[36][37]
Proliferative Diabetic Retinopathy
- Mild to moderate PDR- NVD or NVE insufficient to meet high-risk characteristics
- High-risk PDR-
- NVD greater than ETDRS standard photograph 10A (about 1/3 disc area).
- Any NVD with vitreous hemorrhage.
- NVE greater than 1/2 disc area with vitreous hemorrhage.
Advanced Diabetic Eye Disease is the end-stage vision-threatening complication of diabetic retinopathy in patients whose treatment is inadequate or unsuccessful. It may present as pre-retinal or intragel hemorrhage, tractional retinal detachment, or rubeosis iridis.
Diabetic Macular Edema (DME) can be classified into the following groups
- Focal exudative and diffuse maculopathy
- Ischemic and non-ischemic maculopathy
- Tractional and non-tractional maculopathy
- Center involving macular edema and non-center involving macular edema
ETDRS Definition of Clinically Significant Macular Edema (CSME)
- Retinal edema within 500 µm of the center of the fovea
- Hard exudates within 500 µm of the center of the fovea if associated with adjacent retinal thickening (which may be outside the 500 µm limit)
- Retinal edema one disc area (1500 µm) or larger, any part of which is within one disc diameter of the center of the fovea[38][39]
OCT (optical coherence tomography) Classification of Diabetic Macular Edema
- Sponge-like thickening of retinal layers
- Large cystoid spaces
- Serous detachment of the retina
- Tractional detachment of the fovea or vitreomacular traction
- Taut posterior hyaloid membrane.
International Clinical Diabetic Retinopathy Disease Severity Scale
- No apparent retinopathy-No abnormality
- Mild NPDR- Microaneurysms only
- Moderate NPDR -More than just microaneurysms and less than severe disease
- Severe NPDR -No signs of PDR and any of the following:
- 20 intraretinal hemorrhages in each of the four quadrants
- Venous beading in ≥2 quadrants
- Prominent IRMA ≥1 quadrant
- PDR - One or more of the following:
- Neovascularization
- Vitreous or pre-retinal hemorrhage[40]
With regards to diabetic macular edema, the DME may be:
- 'DME apparently absent'- Apparent retinal thickening and hard exudates at the posterior pole are absent.
- 'DME apparently present'- There is some 'apparent retinal thickening and hard exudates at the posterior pole.' It can further be classified into mild, moderate, and severe based on the distance of thickening and hard exudates from the center of the fovea.
- Mild DME: The retinal thickening or hard exudates are located far from the center of the fovea.
- Moderate DME: Retinal thickening or hard exudates are approaching the center of the macula but not involving the center
- Severe DME: Hard exudate and thickening involve the center of the fovea.
Evaluation
Laboratory Examination
The following are the diabetic markers crucial for all the patients getting screened for diabetic retinopathy:
- Fasting blood Glucose- The fasting blood glucose level of less than 110 mg/dl is normal. A fasting blood sugar (FBS) level of 110 to 125 mg/dl is prediabetes. In at least two samples, FBS of more than 126 mg/dl denotes diabetes.[41]
- HbA1C (glycosylated hemoglobin)- The normal range of HbA1c is between 4% to 5.6%. HbA1c level should be kept below 7%.
Diabetes affects almost all the major organs of the body. Hence a thorough systemic examination should be done to rule out diabetic complications. Especially kidney function test, cardiac function test, liver function test, thyroid markers, blood pressure, and a neurological examination should be carried out. For this, interprofessional collaboration is required.
Ocular Examination
On presentation, the patient should be thoroughly tested for:
- Visual acuity,
- IOP (intraocular pressure) measurement,
- Gonioscopy (for neovascularization of iris/angles),
- Slit-lamp examination (to rule out other ocular manifestations of diabetes mellitus), and
- Dilated fundus examination with +78 or +90 D lens on slit lamp and with +20 or +28 D lens with an indirect ophthalmoscope - the grading of diabetic retinopathy should be done
Additional posterior segment investigations for the evaluation of diabetic retinopathy include:
Fundus Photography
Color fundus photographs are taken in the instances listed below.[42]
- Documentation and record purposes
- Patient education with counseling
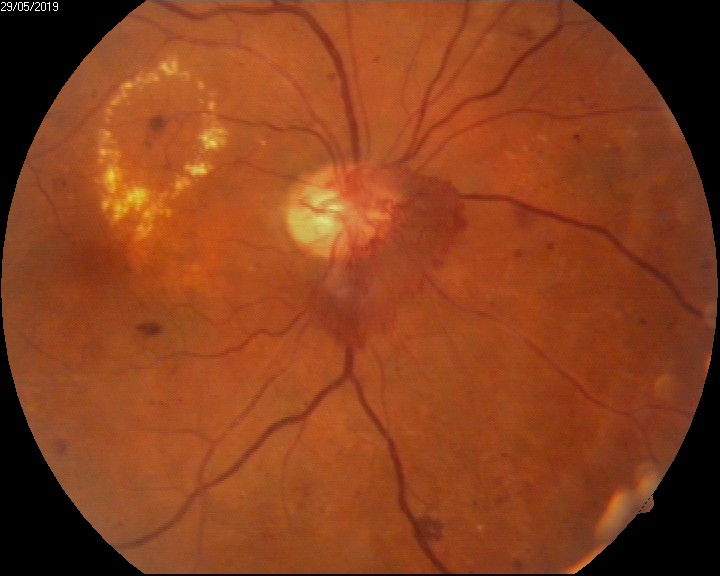
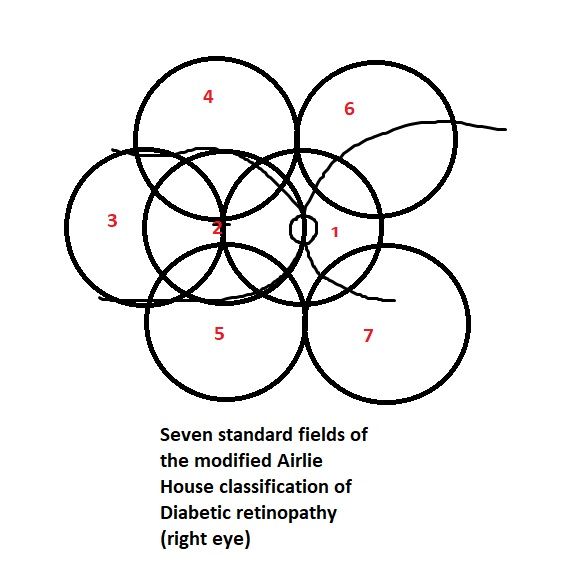
The standard seven fields of modified Airlie House classification should be taken (Figure).[36][43] Each image has a 30-degree field.
Fundus Fluorescein Angiography (FFA)
FFA is indicated in the following situations.[35]
- For the diagnosis of ischemic maculopathy
- To locate capillary dropout areas
- To differentiate IRMA from neovascularization
- To differentiate disc collaterals from disc neovascularization
- To reveal occult new vessels that could not be detected on clinical examination
- To find out the cause of unexplained visual loss
- To differentiate pseudophakic cystoid macular edema and diabetic macular edema
Ultrawide Field Fundus Imaging (UWFI) and Fundus Fluorescein Angiogram (UWFA)
UWFI and UWFA document the changes in the posterior segment in a single frame. Commercially available UWFI (at least a 100-degree field of view) includes Optos/California, Mirante, Clarus, Retcam, and Spectralis. UWFI using a scanning laser ophthalmoscope and ellipsoid mirror, captures images in small pupils and hazy media.[44][45] Peripheral retinal capillary nonperfusion areas may be better delineated and planned for 'targeted retinal photocoagulation' in proliferative diabetic retinopathy or diabetic macular edema.[46][47][48] Peripheral vascular changes may be more common in DR than was previously thought.
Optical Coherence Tomography (OCT)
OCT of the macula helps
- To evaluate retinal thickening
- For the assessment and monitoring of edema after initiation of treatment, especially to plan for the next sitting of intravitreal injections
- To diagnose vitreomacular traction (VMT) and the epiretinal membrane (ERM), which might require surgery (pars plans vitrectomy)
Ultrasonography (B Scan)
It is a very useful tool in hazy media to evaluate
- Vitreous hemorrhage
- Tractional retinal detachment
- Subhyaloid hemorrhage
- Posterior vitreous detachment
Optical Coherence Tomography Angiography (OCTA)
OCTA employs motion contrast imaging to retinal blood flow, generating images similar to fluorescein angiography without the need to inject the dye invasively.
- It provides detailed information on the retinal vasculature
- It is helpful for the demarcation of the foveal avascular zone, helping to find out foveal ischemia
- It enables accurate detection of even mild IRMA
- It helps to delineate capillary dropout areas
Vascular signs like looping, beading, and dilatation can very well be appreciated on OCTA. OCTA may help the earliest detection of microvascular changes (before the visibility of microaneurysm).[49] OCTA is especially useful in patients with kidney disease and patients prone to an anaphylactic reaction to the dyes.
Artificial Intelligence-based Devices
FDA has approved a software program IDx-DR for the detection of 'more than mild diabetic retinopathy' or its absence in 2018. In 2020, EyeArt (Eyenuk) received FDA clearance for the autonomous detection of 'more than mild diabetic retinopathy and vision-threatening diabetic retinopathy.'
Treatment / Management
General Systemic Control of Diabetes
The systemic status should be optimized by
- Strict metabolic control of diabetes,
- Maintaining HbA1C levels under 7%,
- Lifestyle modifications like routine exercises and a proper diabetic food diet,
Patients should visit diabetologists for proper follow-up visits and take timely antidiabetic medications. Other systemic ailments like hypertension, dyslipidemia, hypoproteinemia, anemia, nephropathy, neuropathy, cardiac ailments, and others should also be taken care of by respective medications and excellent interprofessional collaboration.
Management of Non-Proliferative Diabetic Retinopathy (NPDR)
Strict glycemic control and strict compliance of patients towards antidiabetic medication is the key to managing a case of NPDR. Stress should be on proper systemic control of diabetes mellitus, keeping a watch on the progression of ocular findings. Associated comorbidities should be addressed. A detailed systemic investigation by a physician or an endocrinologist should be done to rule out any other systemic side-effects of diabetes mellitus.
The suggested follow-up schedule is given.
- Very mild NPDR- follow-up every year
- Mild to moderate NPDR - follow-up 6-12 monthly. Sixteen percent of cases of mild NPDR and 23% of cases of moderate NPDR progress to proliferative stages within four years[40]
- Severe to Very Severe NPDR- Close follow-up within 2-4 months. Fifty percent of severe NPDR and 75% of cases of very severe NPDR progress to PDR within one year[40][50] (A1)
Management of Diabetic Macular Edema (DME)
Center-involving diabetic macular edema- Nowadays, anti-VEGF agents have become the first line of treatment for center-involving diabetic macular edema. Bevacizumab, ranibizumab, and aflibercept have beneficial effects in patients with baseline better visual acuity. Aflibercept is shown to have better visual outcomes in patients with worse baseline visual acuity on presentation.[51] Recently, aflibercept and ranibizumab have received FDA approval for use in both DME and diabetic retinopathy.[52] Intravitreal steroid implants also are used in recalcitrant cases.[53] (B3)
Non-center involving macular edema - Focal or Grid laser-guided by ETDRS is the treatment of choice.
Nowadays, in the era of Anti-VEGF medications, laser treatment is usually avoided in the center involving macular edema. It may be added as adjuvant therapy in patients not responding to anti-VEGF therapy alone. As stated by ETDRS, laser helped in halting the progression of moderate visual loss, but it did not improve the visual acuity. Bevacizumab intravitreal injection is also effective in the center-involving DME, but it is not FDA approved. Hence it is available only for off-label use. Cluster endophthalmitis due to fake drugs or other reasons is another concern.[54] DRCR (diabetic retinopathy clinical research) network studied the role of aflibercept in DME, and it was found to be useful in patients with initial worse visual acuity (Protocol T of DRCR.net).[51] Other notable studies other than the DRCR network on intravitreal Anti-VEGF injections included RISE, RIDE, READ, RESTORE, BOLT, and RESOLVE. (B3)
Doses of commonly used intravitreal injections:
- Bevacizumab- 1.25mg/0.05 ml (PACORES, DRCR.net PROTOCOL H, BOLT, READ-2)[55][56][57][58][59]
- Ranibizumab - 0.5 mg or 0.3 mg /0.05 ml(RESOLVE, DRCR.net PROTOCOL I, RESTORE, RISE/RIDE[60][61])[62][63][64]
- Aflibercept -2 mg/0.05 ml (DA VINCI, VISTA/VIVID, DRCR.net PROTOCOL T)[65][66][67]
- Pegaptanib- 0.3mg/ 0.09 ml (not FDA approved for this indication)[68][69]
- Brolucizumab 6mg/0.05 ml (not approved by FDA for this indication as of May 2022)[70] (A1)
Long-acting corticosteroid implants like dexamethasone implants and fluocinolone acetonide implants (Retisert and Iluvien) are also used in diabetic macular edema. Though they are not considered the first line of treatment. They are reserved for cases not responding to multiple injections of Anti-VEGF medications (according to protocol U of DRCR.net).[71] Their main advantage is long-term control of inflammation and a lesser frequency of administration. The high cost is also one of the factors for its limited use. Intravitreal injections of triamcinolone are not FDA approved, have poorer results, and are linked with more complications than anti-VEGF medications. Glaucoma is a significant side effect of intravitreal steroids. A new port-delivery system for sustained release of ranibizumab, has been approved to manage wet age-related macular degeneration by the FDA in 2021. Its promises to be a new therapy for DME.(A1)
Tractional diabetic macular edema can occur because of VMT (vitreomacular traction) or ERM (epiretinal membrane). Vitrectomy surgery should be considered in such cases. Intravitreal injections might worsen the traction component. Laser spots, too, should be given away from the tractional area to avoid further worsening.
Refractory Diabetic macular edema- The definition of refractory or persistent DME varies, including macular edema not responding to multiple injections of anti-VEGF and, at least one sitting of laser, and suboptimal response (central macular thickness on OCT of more than 250 microns with associated visual loss) to 6 consecutive monthly injections of anti-VEGF agents.[72][73] A possible approach to such cases includes evaluating systemic control, higher dosing or frequency of anti-VEGF medications, use of steroid implants (according to protocol U of DRCR.net), rechecking of the diagnosis, ruling out any unfavorable anatomical causes, and ultrawide field angiography to look for extreme peripheral ischemic areas and to laser as far in the periphery as possible.[74][71][75](A1)
Ischemic type of diabetic macular edema is associated with poor visual prognosis. The patient can be considered for an extreme peripheral laser to kill out the ischemic areas responsible for VEGF release.[76] Steroid implants could be tried. However, the prognosis is guarded despite all the measures.[77](B3)
Treatment of Proliferative Diabetic Retinopathy (PDR)
Panretinal photocoagulation (PRP) has been considered the standard treatment of proliferative diabetic retinopathy for ages. But because of its destructive properties, intense research was done to find an alternative non-destructive mode of management of diabetic retinopathy. Various studies evaluated the role of intravitreal anti-VEGF injections in PDR cases. Because of the limited half-life of these injections, repeated injections at timely intervals become mandatory. Hence cost-effectivity of this regime is questionable. A concoction of intravitreal injection and pan-retinal photocoagulation, depending on the severity of the disease and taking into account the presence or absence of macular edema, may help manage diabetic retinopathy patients.[78]
- Non-high-risk PDR without DME - Careful 2 to 4 monthly follow-up and immediate pan-retinal photocoagulation should be done if high-risk PDR occurs. Some physicians perform PRP in all PDR cases.
- High-risk PDR without macular edema- Pan-retinal photocoagulation should be initiated.[79]
- PDR with DME- Combined intravitreal anti-VEGF injections and PRP sessions should be considered.
- High-risk PDR in which complete photocoagulation is not possible- Alternatives like cryopexy or vitrectomy should be considered.[79]
In Protocol S of DRCR.net, a comparison of safety and efficacy of PRP to intravitreal injection of 0.5 mg ranibizumab was studied. The primary outcome of protocol S ranibizumab 0.5 mg was non-inferior to PRP. Hence, ranibizumab might compete with PRP to be the first line of management in the near future. High cost, repeated injection, multiple follow-up visits, distortion of the vitreous anatomy and physiology because of the repeated injections, chances of endophthalmitis, and tachyphylaxis are hindering factors.[80]
Treatment of Advanced Diabetic Eye Disease
Prognosis is guarded in patients with advanced diabetic eye disease. Persistent vitreous hemorrhage can initially be given the trial of observation or intravitreal injection of the anti-VEGF agent, following which, if the hemorrhage resolves, then scattered pan-retinal photocoagulation in the visible areas can be tried.[81] Ultrasnogram must exclude retinal traction if such an approach is considered.
If the hemorrhage does not resolve with the above management, pars plana vitrectomy should be considered. Also, vitrectomy is the treatment of choice in cases presenting with tractional retinal detachment.
Indications of vitrectomy in diabetic patients include:
- Non-clearing vitreous hemorrhage (vitreous/subhyaloid/pre-macular)
- Tractional retinal detachment with macula off
- Combined tractional with rhegmatogenous retinal detachment
- Anterior segment neovascularization with the invisibility of the posterior segment
- Ghost cell glaucoma
- Thick epiretinal membrane
- Vitreomacular traction
In vitrectomy surgery, three pars plana ports are made, one for the infusion line, one for the light pipe, and the third for the microinstruments. A fourth port can be made if a chandelier light source is used in cases of bimanual vitrectomy.[82] Core vitrectomy is done. Meticulous separation of posterior vitreous from the retina is tried with the use of triamcinolone. The fibrovascular proliferation at the optic disc should be trimmed as pulling it off may cause massive intraocular hemorrhage and is not recommended. Tractional components are managed by segmentation or delamination.[83] All bleeders are cauterized. Fluid- Air exchange is done. If required, ERM peeling with or without ILM (internal limiting membrane) peeling is done with due precautions. Pan-retinal endophotocoagulation is done. At the end of the surgery, tamponade in the form of air, fluid, gases (C3F8, SF6), or silicone oil, can be injected.
In cases of rubeosis iridis, pan-retinal photocoagulation with or without anti-VEGF medication might be helpful in the early phases. In the advanced stages, the new vessels lead to fibrosis and contracture, leading to permanent distortion of the angles causing refractory glaucoma, which might eventually require a glaucoma drainage implant). The prognosis is, however, poor in such cases. Painful blind eyes may need diode laser cyclophotocoagulation or cyclocryotherapy.
Treatment of Diabetic Retinopathy (without DME)
This is an FDA-approved indication of ranibizumab and aflibercept, which can reduce the severity of diabetic retinopathy.
Differential Diagnosis
The differential diagnosis of diabetic macular edema includes:
- Hypertensive retinopathy
- Central retinal vein occlusion
- Branch retinal vein occlusion[84]
- Irvine Gass syndrome
- Post uveitic macular edema
- Ruptured microaneurysm[85]
- Macular edema secondary to epiretinal membrane
- Choroidal neovascular membrane.
The diseases which can be mistaken as diabetic retinopathy based on the general fundus appearance include:
- Central retinal vein occlusion[86]
- Hypertensive retinopathy[87]
- Sickle cell retinopathy[88]
- Terson syndrome[89][90][89]
- Ocular ischemic syndrome[91][92][91]
- Branch retinal vein occlusion[93]
- Hemiretinal vein occlusion[94]
- Valsalva retinopathy[95]
- Post-traumatic retinal bleed[96]
- Retinal macroaneurysm[97]
- Retinopathy in thalassemia[98]
Prognosis
The prognosis of diabetic retinopathy depends on the duration of diabetes, glycemic control, associated comorbid conditions, and patient compliance to the appropriate line of treatment.[99][100] Proper patient counseling is needed about their retinal condition and making patients aware that delay in proper follow-up could lead to permanent, irreversible vision loss. The initial stages of diabetic retinopathy are reversible if proper glycemic control is achieved. Many patients with diabetic macular edema require long-term support of repeated injections of intravitreal anti-VEGF medications. Patients treated with pan-retinal photocoagulation may require additional supplementation of anti-VEGF medications if there is persistent macular edema and neovascularization. Once there is tractional macular detachment for a longer duration, the visual prognosis is usually guarded as the macular anatomy is markedly distorted.
OCT biomarkers of prognosis of diabetic retinopathy include refractile bodies, disorganization of inner layers of the retina (DRIL), disruption of outer layers of the retina (DORL), choroidal thickness, epiretinal membrane, vitreomacular adhesions, subretinal fluid, macular thickness, and integrity of ellipsoid zone.[42][101]
Complications
Vision-threatening complications associated with poorly controlled diabetic retinopathy include diabetic macular edema, tractional retinal detachment, and vitreous hemorrhage[102] as a late sequela of proliferative diabetic retinopathy.[103][104]
Management of Diabetic retinopathy with anti-VEGF therapy, laser photocoagulation, or vitrectomy surgery is also not free of complications.
Complications Related to Anti-VEGF Medications
- IOP spike
- Cataract formation
- Iatrogenic retinal/posterior capsular tear
- Vitreous hemorrhage
- Rhegmatogenous Retinal detachment
- Worsening of traction over the macula if given in patients with tractional retinal detachment
- Endophthalmitis
Complications Related to Laser Photocoagulation
- Vitreous hemorrhage
- Exudative retinal detachment and choroidal detachment if too many laser shots are given in a single sitting
- Reduction of contrast sensitivity, peripheral vision, and night vision after pan-retinal photocoagulation
- Permanent scotomas in the visual field
- Worsening of preexisting macular edema or development of macular edema after laser/PRP
- Chances of worsening optic disc pallor
Complications Related to Vitrectomy
- Cataract formation
- IOP spike because of gas or oil tamponade
- Iatrogenic secondary tears leading to rhegmatogenous retinal detachment
- In chronic macular edema, the macular roof is very thin, so there are chances of deroofing at the macular region leading to a macular hole
Postoperative and Rehabilitation Care
Laser photocoagulation is performed as an OPD (outpatient department) procedure. Maintaining proper glycemic control and taking care of other associated systemic illnesses are necessary. There are chances of worsening macular edema and moderate vision reduction after pan-retinal photocoagulation (PRP). If that happens, the patient should report to the retinal surgeon for documentation and for planning a further line of management.
It is common to have an acute spike of IOP after intravitreal injections for a few hours. It usually returns to normal within 3-6 hours. It may be prudent to routinely use a single tablet of acetazolamide 250 mg stat after intravitreal injection. After intravitreal injections, patients should be called for follow-up the next day; then, according to the stability of the ocular condition, follow-up at regular intervals should be planned. Sequential OCT and/or OCTA parameters are assessed in each follow-up visit, and depending on their findings, further management with repeat injections, observation, and/or switching to laser /vitrectomy surgery is planned.
Post-operative Care
After surgery, the eye is patched. After the patch is removed, the eye is cleaned, and topical administration of medications like antibiotics with steroid eye drops and cycloplegic eye drops are started. After injecting gases like sf6/c3f8 or silicone oil, there are chances of IOP rise post-operatively, which needs to be managed with appropriate antiglaucoma medications. Patients can then be called for regular, timely follow-ups to screen their post-operative outcomes. If silicone oil tamponade is done, the patient should be explained the need for a mandatory second surgery to remove oil.
Deterrence and Patient Education
The American Academy of Ophthalmology's current recommendation states that people with diabetes mellitus type 1 should have yearly screening for diabetic retinopathy starting five years after the onset of diabetes. Patients with diabetes mellitus type 2 should have fundus evaluation at the time of diagnosis and annually thereafter.[105][106]
The patient should be counseled properly regarding the prognosis of diabetic retinopathy. Early diagnosis, management, and follow-up visits at timely intervals depending on the stage of presentation of diabetic retinopathy are mandatory. Systemic dysregulation of diabetes can lead to an exponential worsening of diabetic retinopathy. Lifestyle modification should be done along with proper systemic and topical medications to halt the progression of diabetic retinopathy.
Enhancing Healthcare Team Outcomes
Any patient presenting with diabetic retinopathy should not receive treatment from an ophthalmologist in isolation. It requires a multispecialty evaluation by an endocrinologist, nephrologist, cardiologist, and neurologist. Interprofessional communication can lead to better patient management. The patient most often presents to the primary health care provider or nurse practitioner, and these professionals should be aware of the condition as it is treatable. Prompt referral to an ophthalmologist is necessary. These patients can then be followed by their primary clinicians and should ensure correct dosing on the medication management aspect of the condition.
Nursing is the first department to come in contact with the patients on follow-up. The nurse can assess the treatment progress, evaluate compliance with both medication and lifestyle measures, and report any issue to the primary care physician. This collaborative, interprofessional approach to care can ensure optimal patient outcomes.
Media
(Click Image to Enlarge)
(Click Image to Enlarge)
References
Eisma JH, Dulle JE, Fort PE. Current knowledge on diabetic retinopathy from human donor tissues. World journal of diabetes. 2015 Mar 15:6(2):312-20. doi: 10.4239/wjd.v6.i2.312. Epub [PubMed PMID: 25789112]
Hendrick AM, Gibson MV, Kulshreshtha A. Diabetic Retinopathy. Primary care. 2015 Sep:42(3):451-64. doi: 10.1016/j.pop.2015.05.005. Epub [PubMed PMID: 26319349]
Diabetes Control and Complications Trial Research Group, Nathan DM, Genuth S, Lachin J, Cleary P, Crofford O, Davis M, Rand L, Siebert C. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. The New England journal of medicine. 1993 Sep 30:329(14):977-86 [PubMed PMID: 8366922]
Level 1 (high-level) evidence. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). UK Prospective Diabetes Study (UKPDS) Group. Lancet (London, England). 1998 Sep 12:352(9131):837-53 [PubMed PMID: 9742976]
Level 1 (high-level) evidenceSayin N, Kara N, Pekel G. Ocular complications of diabetes mellitus. World journal of diabetes. 2015 Feb 15:6(1):92-108. doi: 10.4239/wjd.v6.i1.92. Epub [PubMed PMID: 25685281]
Vieira-Potter VJ, Karamichos D, Lee DJ. Ocular Complications of Diabetes and Therapeutic Approaches. BioMed research international. 2016:2016():3801570. doi: 10.1155/2016/3801570. Epub 2016 Mar 28 [PubMed PMID: 27119078]
Khan A, Petropoulos IN, Ponirakis G, Malik RA. Visual complications in diabetes mellitus: beyond retinopathy. Diabetic medicine : a journal of the British Diabetic Association. 2017 Apr:34(4):478-484. doi: 10.1111/dme.13296. Epub 2016 Dec 20 [PubMed PMID: 27917530]
Nittala MG, Keane PA, Zhang K, Sadda SR. Risk factors for proliferative diabetic retinopathy in a Latino American population. Retina (Philadelphia, Pa.). 2014 Aug:34(8):1594-9. doi: 10.1097/IAE.0000000000000117. Epub [PubMed PMID: 24662751]
Level 2 (mid-level) evidencePorta M, Sjoelie AK, Chaturvedi N, Stevens L, Rottiers R, Veglio M, Fuller JH, EURODIAB Prospective Complications Study Group. Risk factors for progression to proliferative diabetic retinopathy in the EURODIAB Prospective Complications Study. Diabetologia. 2001 Dec:44(12):2203-9 [PubMed PMID: 11793022]
Wang Y, Lin Z, Zhai G, Ding XX, Wen L, Li D, Zou B, Feng KM, Liang YB, Xie C. Prevalence of and Risk Factors for Diabetic Retinopathy and Diabetic Macular Edema in Patients with Early- and Late-Onset Diabetes Mellitus. Ophthalmic research. 2022:65(3):293-299. doi: 10.1159/000508335. Epub 2020 Apr 30 [PubMed PMID: 32353847]
Schreur V, van Asten F, Ng H, Weeda J, Groenewoud JMM, Tack CJ, Hoyng CB, de Jong EK, Klaver CCW, Jeroen Klevering B. Risk factors for development and progression of diabetic retinopathy in Dutch patients with type 1 diabetes mellitus. Acta ophthalmologica. 2018 Aug:96(5):459-464. doi: 10.1111/aos.13815. Epub [PubMed PMID: 30188024]
Wilkinson-Berka JL, Miller AG. Update on the treatment of diabetic retinopathy. TheScientificWorldJournal. 2008 Feb 6:8():98-120. doi: 10.1100/tsw.2008.25. Epub 2008 Feb 6 [PubMed PMID: 18264628]
Moutray T, Evans JR, Lois N, Armstrong DJ, Peto T, Azuara-Blanco A. Different lasers and techniques for proliferative diabetic retinopathy. The Cochrane database of systematic reviews. 2018 Mar 15:3(3):CD012314. doi: 10.1002/14651858.CD012314.pub2. Epub 2018 Mar 15 [PubMed PMID: 29543992]
Level 1 (high-level) evidenceGupta V, Arevalo JF. Surgical management of diabetic retinopathy. Middle East African journal of ophthalmology. 2013 Oct-Dec:20(4):283-92. doi: 10.4103/0974-9233.120003. Epub [PubMed PMID: 24339677]
Behl T, Kotwani A. Exploring the various aspects of the pathological role of vascular endothelial growth factor (VEGF) in diabetic retinopathy. Pharmacological research. 2015 Sep:99():137-48. doi: 10.1016/j.phrs.2015.05.013. Epub 2015 Jun 6 [PubMed PMID: 26054568]
Kador PF, Wyman M, Oates PJ. Aldose reductase, ocular diabetic complications and the development of topical Kinostat(®). Progress in retinal and eye research. 2016 Sep:54():1-29. doi: 10.1016/j.preteyeres.2016.04.006. Epub 2016 Apr 19 [PubMed PMID: 27102270]
Kern TS, Antonetti DA, Smith LEH. Pathophysiology of Diabetic Retinopathy: Contribution and Limitations of Laboratory Research. Ophthalmic research. 2019:62(4):196-202. doi: 10.1159/000500026. Epub 2019 Jul 30 [PubMed PMID: 31362288]
Wang W, Lo ACY. Diabetic Retinopathy: Pathophysiology and Treatments. International journal of molecular sciences. 2018 Jun 20:19(6):. doi: 10.3390/ijms19061816. Epub 2018 Jun 20 [PubMed PMID: 29925789]
Coughlin BA, Feenstra DJ, Mohr S. Müller cells and diabetic retinopathy. Vision research. 2017 Oct:139():93-100. doi: 10.1016/j.visres.2017.03.013. Epub 2017 Sep 5 [PubMed PMID: 28866025]
Curtis TM, Hamilton R, Yong PH, McVicar CM, Berner A, Pringle R, Uchida K, Nagai R, Brockbank S, Stitt AW. Müller glial dysfunction during diabetic retinopathy in rats is linked to accumulation of advanced glycation end-products and advanced lipoxidation end-products. Diabetologia. 2011 Mar:54(3):690-8. doi: 10.1007/s00125-010-1971-x. Epub 2010 Nov 30 [PubMed PMID: 21116609]
Level 3 (low-level) evidencePannicke T, Iandiev I, Wurm A, Uckermann O, vom Hagen F, Reichenbach A, Wiedemann P, Hammes HP, Bringmann A. Diabetes alters osmotic swelling characteristics and membrane conductance of glial cells in rat retina. Diabetes. 2006 Mar:55(3):633-9 [PubMed PMID: 16505225]
Level 3 (low-level) evidenceVujosevic S, Midena E. Retinal layers changes in human preclinical and early clinical diabetic retinopathy support early retinal neuronal and Müller cells alterations. Journal of diabetes research. 2013:2013():905058. doi: 10.1155/2013/905058. Epub 2013 Jun 12 [PubMed PMID: 23841106]
Cai J, Boulton M. The pathogenesis of diabetic retinopathy: old concepts and new questions. Eye (London, England). 2002 May:16(3):242-60 [PubMed PMID: 12032713]
Mori K, Duh E, Gehlbach P, Ando A, Takahashi K, Pearlman J, Mori K, Yang HS, Zack DJ, Ettyreddy D, Brough DE, Wei LL, Campochiaro PA. Pigment epithelium-derived factor inhibits retinal and choroidal neovascularization. Journal of cellular physiology. 2001 Aug:188(2):253-63 [PubMed PMID: 11424092]
Level 3 (low-level) evidenceFreyberger H, Bröcker M, Yakut H, Hammer J, Effert R, Schifferdecker E, Schatz H, Derwahl M. Increased levels of platelet-derived growth factor in vitreous fluid of patients with proliferative diabetic retinopathy. Experimental and clinical endocrinology & diabetes : official journal, German Society of Endocrinology [and] German Diabetes Association. 2000:108(2):106-9 [PubMed PMID: 10826517]
Ziche M, Maglione D, Ribatti D, Morbidelli L, Lago CT, Battisti M, Paoletti I, Barra A, Tucci M, Parise G, Vincenti V, Granger HJ, Viglietto G, Persico MG. Placenta growth factor-1 is chemotactic, mitogenic, and angiogenic. Laboratory investigation; a journal of technical methods and pathology. 1997 Apr:76(4):517-31 [PubMed PMID: 9111514]
Level 3 (low-level) evidenceVincent JA, Mohr S. Inhibition of caspase-1/interleukin-1beta signaling prevents degeneration of retinal capillaries in diabetes and galactosemia. Diabetes. 2007 Jan:56(1):224-30 [PubMed PMID: 17192486]
Level 3 (low-level) evidenceLei X, Zhang J, Shen J, Hu LM, Wu Y, Mou L, Xu G, Li W, Xu GT. EPO attenuates inflammatory cytokines by Muller cells in diabetic retinopathy. Frontiers in bioscience (Elite edition). 2011 Jan 1:3(1):201-11 [PubMed PMID: 21196299]
Level 3 (low-level) evidenceGarner A. Histopathology of diabetic retinopathy in man. Eye (London, England). 1993:7 ( Pt 2)():250-3 [PubMed PMID: 7607344]
Crăiţoiu S. [The morphopathological aspects of diabetic retinopathy]. Oftalmologia (Bucharest, Romania : 1990). 1992 Apr-Jun:36(2):141-8 [PubMed PMID: 1525142]
Pannicke T, Iandiev I, Uckermann O, Biedermann B, Kutzera F, Wiedemann P, Wolburg H, Reichenbach A, Bringmann A. A potassium channel-linked mechanism of glial cell swelling in the postischemic retina. Molecular and cellular neurosciences. 2004 Aug:26(4):493-502 [PubMed PMID: 15276152]
Level 3 (low-level) evidenceReichenbach A, Wurm A, Pannicke T, Iandiev I, Wiedemann P, Bringmann A. Müller cells as players in retinal degeneration and edema. Graefe's archive for clinical and experimental ophthalmology = Albrecht von Graefes Archiv fur klinische und experimentelle Ophthalmologie. 2007 May:245(5):627-36 [PubMed PMID: 17219109]
Level 3 (low-level) evidenceKanski JJ. Diabetic retinopathy--a preventable cause of blindness. The Practitioner. 1985 Apr:229(1402):343-8 [PubMed PMID: 3991450]
Ezra E, Keinan E, Mandel Y, Boulton ME, Nahmias Y. Non-dimensional analysis of retinal microaneurysms: critical threshold for treatment. Integrative biology : quantitative biosciences from nano to macro. 2013 Mar:5(3):474-80. doi: 10.1039/c3ib20259c. Epub [PubMed PMID: 23371018]
Ruia S, Tripathy K. Fluorescein Angiography. StatPearls. 2024 Jan:(): [PubMed PMID: 35015403]
. Grading diabetic retinopathy from stereoscopic color fundus photographs--an extension of the modified Airlie House classification. ETDRS report number 10. Early Treatment Diabetic Retinopathy Study Research Group. Ophthalmology. 1991 May:98(5 Suppl):786-806 [PubMed PMID: 2062513]
Level 1 (high-level) evidence. Classification of diabetic retinopathy from fluorescein angiograms. ETDRS report number 11. Early Treatment Diabetic Retinopathy Study Research Group. Ophthalmology. 1991 May:98(5 Suppl):807-22 [PubMed PMID: 2062514]
Level 1 (high-level) evidenceTripathy K, Sharma YR, R K, Chawla R, Gogia V, Singh SK, Venkatesh P, Vohra R. Recent advances in management of diabetic macular edema. Current diabetes reviews. 2015:11(2):79-97 [PubMed PMID: 25801496]
Level 3 (low-level) evidenceJorge EC, Jorge EN, Botelho M, Farat JG, Virgili G, El Dib R. Monotherapy laser photocoagulation for diabetic macular oedema. The Cochrane database of systematic reviews. 2018 Oct 15:10(10):CD010859. doi: 10.1002/14651858.CD010859.pub2. Epub 2018 Oct 15 [PubMed PMID: 30320466]
Level 1 (high-level) evidenceWilkinson CP, Ferris FL 3rd, Klein RE, Lee PP, Agardh CD, Davis M, Dills D, Kampik A, Pararajasegaram R, Verdaguer JT, Global Diabetic Retinopathy Project Group. Proposed international clinical diabetic retinopathy and diabetic macular edema disease severity scales. Ophthalmology. 2003 Sep:110(9):1677-82 [PubMed PMID: 13129861]
Zhang Q, Zhao G, Yang N, Zhang L. Fasting blood glucose levels in patients with different types of diseases. Progress in molecular biology and translational science. 2019:162():277-292. doi: 10.1016/bs.pmbts.2019.01.004. Epub 2019 Mar 6 [PubMed PMID: 30905457]
Kwan CC, Fawzi AA. Imaging and Biomarkers in Diabetic Macular Edema and Diabetic Retinopathy. Current diabetes reports. 2019 Aug 31:19(10):95. doi: 10.1007/s11892-019-1226-2. Epub 2019 Aug 31 [PubMed PMID: 31473838]
Tripathy K, Chawla R, Wadekar BR, Venkatesh P, Sharma YR. Evaluation of rhegmatogenous retinal detachments using Optos ultrawide field fundus fluorescein angiography and comparison with ETDRS 7 field overlay. Journal of current ophthalmology. 2018 Sep:30(3):263-267. doi: 10.1016/j.joco.2018.06.006. Epub 2018 Jul 3 [PubMed PMID: 30197958]
Tripathy K, Chawla R, Venkatesh P, Sharma YR, Vohra R. Ultrawide Field Imaging in Uveitic Non-dilating Pupils. Journal of ophthalmic & vision research. 2017 Apr-Jun:12(2):232-233. doi: 10.4103/2008-322X.205360. Epub [PubMed PMID: 28540019]
Tripathy K, Chawla R, Vohra R. Evaluation of the fundus in poorly dilating diabetic pupils using ultrawide field imaging. Clinical & experimental optometry. 2017 Nov:100(6):735-736. doi: 10.1111/cxo.12484. Epub 2016 Oct 5 [PubMed PMID: 27704602]
Muqit MM, Marcellino GR, Henson DB, Young LB, Patton N, Charles SJ, Turner GS, Stanga PE. Optos-guided pattern scan laser (Pascal)-targeted retinal photocoagulation in proliferative diabetic retinopathy. Acta ophthalmologica. 2013 May:91(3):251-8. doi: 10.1111/j.1755-3768.2011.02307.x. Epub 2011 Dec 16 [PubMed PMID: 22176513]
Level 2 (mid-level) evidenceNikkhah H, Ghazi H, Razzaghi MR, Karimi S, Ramezani A, Soheilian M. Extended targeted retinal photocoagulation versus conventional pan-retinal photocoagulation for proliferative diabetic retinopathy in a randomized clinical trial. International ophthalmology. 2018 Feb:38(1):313-321. doi: 10.1007/s10792-017-0469-7. Epub 2017 Feb 6 [PubMed PMID: 28168567]
Level 1 (high-level) evidenceBrown DM, Ou WC, Wong TP, Kim RY, Croft DE, Wykoff CC, DAVE Study Group. Targeted Retinal Photocoagulation for Diabetic Macular Edema with Peripheral Retinal Nonperfusion: Three-Year Randomized DAVE Trial. Ophthalmology. 2018 May:125(5):683-690. doi: 10.1016/j.ophtha.2017.11.026. Epub 2018 Jan 11 [PubMed PMID: 29336896]
Level 1 (high-level) evidenceLajmi H, Hmaied W, Othmen AB, Chelly Z, El Fekih L. Optical coherence tomography angiography microvascular changes in diabetics without diabetic retinopathy. Saudi journal of ophthalmology : official journal of the Saudi Ophthalmological Society. 2020 Jul-Sep:34(3):156-159. doi: 10.4103/1319-4534.310404. Epub 2021 Feb 27 [PubMed PMID: 34085005]
. Early photocoagulation for diabetic retinopathy. ETDRS report number 9. Early Treatment Diabetic Retinopathy Study Research Group. Ophthalmology. 1991 May:98(5 Suppl):766-85 [PubMed PMID: 2062512]
Level 1 (high-level) evidenceCai S, Bressler NM. Aflibercept, bevacizumab or ranibizumab for diabetic macular oedema: recent clinically relevant findings from DRCR.net Protocol T. Current opinion in ophthalmology. 2017 Nov:28(6):636-643. doi: 10.1097/ICU.0000000000000424. Epub [PubMed PMID: 28837425]
Level 3 (low-level) evidenceVaziri K,Schwartz SG,Relhan N,Kishor KS,Flynn HW Jr, New Therapeutic Approaches in Diabetic Retinopathy. The review of diabetic studies : RDS. 2015 Spring-Summer [PubMed PMID: 26676668]
Lattanzio R, Cicinelli MV, Bandello F. Intravitreal Steroids in Diabetic Macular Edema. Developments in ophthalmology. 2017:60():78-90. doi: 10.1159/000459691. Epub 2017 Apr 20 [PubMed PMID: 28427068]
Kumar A, Tripathy K, Chawla R. Intraocular use of bevacizumab in India: An issue resolved? The National medical journal of India. 2017 Nov-Dec:30(6):345-347. doi: 10.4103/0970-258X.239079. Epub [PubMed PMID: 30117450]
Stefanini FR, Arevalo JF, Maia M. Bevacizumab for the management of diabetic macular edema. World journal of diabetes. 2013 Apr 15:4(2):19-26. doi: 10.4239/wjd.v4.i2.19. Epub [PubMed PMID: 23593532]
Diabetic Retinopathy Clinical Research Network, Scott IU, Edwards AR, Beck RW, Bressler NM, Chan CK, Elman MJ, Friedman SM, Greven CM, Maturi RK, Pieramici DJ, Shami M, Singerman LJ, Stockdale CR. A phase II randomized clinical trial of intravitreal bevacizumab for diabetic macular edema. Ophthalmology. 2007 Oct:114(10):1860-7 [PubMed PMID: 17698196]
Level 3 (low-level) evidenceMichaelides M, Kaines A, Hamilton RD, Fraser-Bell S, Rajendram R, Quhill F, Boos CJ, Xing W, Egan C, Peto T, Bunce C, Leslie RD, Hykin PG. A prospective randomized trial of intravitreal bevacizumab or laser therapy in the management of diabetic macular edema (BOLT study) 12-month data: report 2. Ophthalmology. 2010 Jun:117(6):1078-1086.e2. doi: 10.1016/j.ophtha.2010.03.045. Epub 2010 Apr 22 [PubMed PMID: 20416952]
Level 1 (high-level) evidenceRajendram R, Fraser-Bell S, Kaines A, Michaelides M, Hamilton RD, Esposti SD, Peto T, Egan C, Bunce C, Leslie RD, Hykin PG. A 2-year prospective randomized controlled trial of intravitreal bevacizumab or laser therapy (BOLT) in the management of diabetic macular edema: 24-month data: report 3. Archives of ophthalmology (Chicago, Ill. : 1960). 2012 Aug:130(8):972-9 [PubMed PMID: 22491395]
Level 1 (high-level) evidenceNguyen QD, Shah SM, Heier JS, Do DV, Lim J, Boyer D, Abraham P, Campochiaro PA, READ-2 Study Group. Primary End Point (Six Months) Results of the Ranibizumab for Edema of the mAcula in diabetes (READ-2) study. Ophthalmology. 2009 Nov:116(11):2175-81.e1. doi: 10.1016/j.ophtha.2009.04.023. Epub 2009 Aug 22 [PubMed PMID: 19700194]
Level 1 (high-level) evidenceBrown DM, Nguyen QD, Ehrlich JS, RISE and RIDE Research Group. Ranibizumab for diabetic macular edema. Author reply. Ophthalmology. 2013 Jan:120(1):221-2. doi: 10.1016/j.ophtha.2012.07.070. Epub [PubMed PMID: 23283201]
Level 3 (low-level) evidenceBrown DM, Nguyen QD, Marcus DM, Boyer DS, Patel S, Feiner L, Schlottmann PG, Rundle AC, Zhang J, Rubio RG, Adamis AP, Ehrlich JS, Hopkins JJ, RIDE and RISE Research Group. Long-term outcomes of ranibizumab therapy for diabetic macular edema: the 36-month results from two phase III trials: RISE and RIDE. Ophthalmology. 2013 Oct:120(10):2013-22. doi: 10.1016/j.ophtha.2013.02.034. Epub 2013 May 22 [PubMed PMID: 23706949]
Level 1 (high-level) evidenceMassin P, Bandello F, Garweg JG, Hansen LL, Harding SP, Larsen M, Mitchell P, Sharp D, Wolf-Schnurrbusch UE, Gekkieva M, Weichselberger A, Wolf S. Safety and efficacy of ranibizumab in diabetic macular edema (RESOLVE Study): a 12-month, randomized, controlled, double-masked, multicenter phase II study. Diabetes care. 2010 Nov:33(11):2399-405. doi: 10.2337/dc10-0493. Epub [PubMed PMID: 20980427]
Level 1 (high-level) evidenceDiabetic Retinopathy Clinical Research Network, Elman MJ, Aiello LP, Beck RW, Bressler NM, Bressler SB, Edwards AR, Ferris FL 3rd, Friedman SM, Glassman AR, Miller KM, Scott IU, Stockdale CR, Sun JK. Randomized trial evaluating ranibizumab plus prompt or deferred laser or triamcinolone plus prompt laser for diabetic macular edema. Ophthalmology. 2010 Jun:117(6):1064-1077.e35. doi: 10.1016/j.ophtha.2010.02.031. Epub 2010 Apr 28 [PubMed PMID: 20427088]
Level 1 (high-level) evidenceMitchell P, Bandello F, Schmidt-Erfurth U, Lang GE, Massin P, Schlingemann RO, Sutter F, Simader C, Burian G, Gerstner O, Weichselberger A, RESTORE study group. The RESTORE study: ranibizumab monotherapy or combined with laser versus laser monotherapy for diabetic macular edema. Ophthalmology. 2011 Apr:118(4):615-25. doi: 10.1016/j.ophtha.2011.01.031. Epub [PubMed PMID: 21459215]
Level 1 (high-level) evidenceDo DV, Schmidt-Erfurth U, Gonzalez VH, Gordon CM, Tolentino M, Berliner AJ, Vitti R, Rückert R, Sandbrink R, Stein D, Yang K, Beckmann K, Heier JS. The DA VINCI Study: phase 2 primary results of VEGF Trap-Eye in patients with diabetic macular edema. Ophthalmology. 2011 Sep:118(9):1819-26. doi: 10.1016/j.ophtha.2011.02.018. Epub 2011 May 5 [PubMed PMID: 21546089]
Level 1 (high-level) evidenceKorobelnik JF, Do DV, Schmidt-Erfurth U, Boyer DS, Holz FG, Heier JS, Midena E, Kaiser PK, Terasaki H, Marcus DM, Nguyen QD, Jaffe GJ, Slakter JS, Simader C, Soo Y, Schmelter T, Yancopoulos GD, Stahl N, Vitti R, Berliner AJ, Zeitz O, Metzig C, Brown DM. Intravitreal aflibercept for diabetic macular edema. Ophthalmology. 2014 Nov:121(11):2247-54. doi: 10.1016/j.ophtha.2014.05.006. Epub 2014 Jul 8 [PubMed PMID: 25012934]
Level 1 (high-level) evidenceDiabetic Retinopathy Clinical Research Network, Wells JA, Glassman AR, Ayala AR, Jampol LM, Aiello LP, Antoszyk AN, Arnold-Bush B, Baker CW, Bressler NM, Browning DJ, Elman MJ, Ferris FL, Friedman SM, Melia M, Pieramici DJ, Sun JK, Beck RW. Aflibercept, bevacizumab, or ranibizumab for diabetic macular edema. The New England journal of medicine. 2015 Mar 26:372(13):1193-203. doi: 10.1056/NEJMoa1414264. Epub 2015 Feb 18 [PubMed PMID: 25692915]
Level 1 (high-level) evidenceCunningham ET Jr,Adamis AP,Altaweel M,Aiello LP,Bressler NM,D'Amico DJ,Goldbaum M,Guyer DR,Katz B,Patel M,Schwartz SD, A phase II randomized double-masked trial of pegaptanib, an anti-vascular endothelial growth factor aptamer, for diabetic macular edema. Ophthalmology. 2005 Oct [PubMed PMID: 16154196]
Level 1 (high-level) evidenceSultan MB, Zhou D, Loftus J, Dombi T, Ice KS, Macugen 1013 Study Group. A phase 2/3, multicenter, randomized, double-masked, 2-year trial of pegaptanib sodium for the treatment of diabetic macular edema. Ophthalmology. 2011 Jun:118(6):1107-18. doi: 10.1016/j.ophtha.2011.02.045. Epub 2011 May 6 [PubMed PMID: 21529957]
Level 1 (high-level) evidenceBrown DM, Emanuelli A, Bandello F, Barranco JJE, Figueira J, Souied E, Wolf S, Gupta V, Ngah NF, Liew G, Tuli R, Tadayoni R, Dhoot D, Wang L, Bouillaud E, Wang Y, Kovacic L, Guerard N, Garweg JG. KESTREL and KITE: 52-Week Results From Two Phase III Pivotal Trials of Brolucizumab for Diabetic Macular Edema. American journal of ophthalmology. 2022 Jun:238():157-172. doi: 10.1016/j.ajo.2022.01.004. Epub 2022 Jan 14 [PubMed PMID: 35038415]
Maturi RK, Glassman AR, Liu D, Beck RW, Bhavsar AR, Bressler NM, Jampol LM, Melia M, Punjabi OS, Salehi-Had H, Sun JK, Diabetic Retinopathy Clinical Research Network. Effect of Adding Dexamethasone to Continued Ranibizumab Treatment in Patients With Persistent Diabetic Macular Edema: A DRCR Network Phase 2 Randomized Clinical Trial. JAMA ophthalmology. 2018 Jan 1:136(1):29-38. doi: 10.1001/jamaophthalmol.2017.4914. Epub [PubMed PMID: 29127949]
Level 1 (high-level) evidenceHussain RM, Ciulla TA. Treatment strategies for refractory diabetic macular edema: switching anti-VEGF treatments, adopting corticosteroid-based treatments, and combination therapy. Expert opinion on biological therapy. 2016:16(3):365-74. doi: 10.1517/14712598.2016.1131265. Epub 2016 Jan 12 [PubMed PMID: 26674182]
Level 3 (low-level) evidenceShah SU, Maturi RK. Therapeutic Options in Refractory Diabetic Macular Oedema. Drugs. 2017 Apr:77(5):481-492. doi: 10.1007/s40265-017-0704-6. Epub [PubMed PMID: 28197794]
Jiang AC, Srivastava SK, Hu M, Figueiredo N, Babiuch A, Boss JD, Reese JL, Ehlers JP. Quantitative Ultra-Widefield Angiographic Features and Associations with Diabetic Macular Edema. Ophthalmology. Retina. 2020 Jan:4(1):49-56. doi: 10.1016/j.oret.2019.08.008. Epub 2019 Aug 28 [PubMed PMID: 31690541]
Khan Z, Kuriakose RK, Khan M, Chin EK, Almeida DR. Efficacy of the Intravitreal Sustained-Release Dexamethasone Implant for Diabetic Macular Edema Refractory to Anti-Vascular Endothelial Growth Factor Therapy: Meta-Analysis and Clinical Implications. Ophthalmic surgery, lasers & imaging retina. 2017 Feb 1:48(2):160-166. doi: 10.3928/23258160-20170130-10. Epub [PubMed PMID: 28195619]
Level 1 (high-level) evidenceBresnick GH. Diabetic macular edema. A review. Ophthalmology. 1986 Jul:93(7):989-97 [PubMed PMID: 3531959]
Usman M. An Overview of Our Current Understanding of Diabetic Macular Ischemia (DMI). Cureus. 2018 Jul 30:10(7):e3064. doi: 10.7759/cureus.3064. Epub 2018 Jul 30 [PubMed PMID: 30280060]
Level 3 (low-level) evidenceShah KB, Han DP. Proliferative diabetic retinopathy. International ophthalmology clinics. 2004 Fall:44(4):69-84 [PubMed PMID: 15577565]
Kansora MB, Goldhardt R. Decision Making in Proliferative Diabetic Retinopathy Treatment. Current ophthalmology reports. 2019 Mar:7(1):45-50. doi: 10.1007/s40135-019-00198-w. Epub 2019 Feb 4 [PubMed PMID: 31595210]
Sun JK, Glassman AR, Beaulieu WT, Stockdale CR, Bressler NM, Flaxel C, Gross JG, Shami M, Jampol LM, Diabetic Retinopathy Clinical Research Network. Rationale and Application of the Protocol S Anti-Vascular Endothelial Growth Factor Algorithm for Proliferative Diabetic Retinopathy. Ophthalmology. 2019 Jan:126(1):87-95. doi: 10.1016/j.ophtha.2018.08.001. Epub 2018 Aug 7 [PubMed PMID: 30096354]
El Annan J, Carvounis PE. Current management of vitreous hemorrhage due to proliferative diabetic retinopathy. International ophthalmology clinics. 2014 Spring:54(2):141-53. doi: 10.1097/IIO.0000000000000027. Epub [PubMed PMID: 24613890]
Wang ZY, Zhao KK, Li JK, Rossmiller B, Zhao PQ. Four-port bimanual 23-gauge vitrectomy for diabetic tractional retinal detachment. Acta ophthalmologica. 2016 Jun:94(4):365-72. doi: 10.1111/aos.12951. Epub 2016 Feb 8 [PubMed PMID: 26855122]
Mishra C, Tripathy K. Retinal Traction Detachment. StatPearls. 2024 Jan:(): [PubMed PMID: 32644378]
Dysli M, Rückert R, Munk MR. Differentiation of Underlying Pathologies of Macular Edema Using Spectral Domain Optical Coherence Tomography (SD-OCT). Ocular immunology and inflammation. 2019:27(3):474-483. doi: 10.1080/09273948.2019.1603313. Epub [PubMed PMID: 31184556]
Singh D, Tripathy K. Retinal Macroaneurysm. StatPearls. 2024 Jan:(): [PubMed PMID: 35015432]
Blair K, Czyz CN. Central Retinal Vein Occlusion. StatPearls. 2023 Jan:(): [PubMed PMID: 30252241]
Modi P, Arsiwalla T. Hypertensive Retinopathy. StatPearls. 2023 Jan:(): [PubMed PMID: 30252236]
Bonanomi MT, Lavezzo MM. Sickle cell retinopathy: diagnosis and treatment. Arquivos brasileiros de oftalmologia. 2013 Oct:76(5):320-7 [PubMed PMID: 24232951]
Hassan A, Lanzino G, Wijdicks EF, Rabinstein AA, Flemming KD. Terson's syndrome. Neurocritical care. 2011 Dec:15(3):554-8. doi: 10.1007/s12028-011-9555-2. Epub [PubMed PMID: 21604080]
Level 3 (low-level) evidenceTripathy K. Dissociated optic nerve fiber layer in a case of Terson syndrome. European journal of ophthalmology. 2020 Sep:30(5):NP11-NP14. doi: 10.1177/1120672119853465. Epub 2019 Jun 3 [PubMed PMID: 31155955]
Level 3 (low-level) evidenceTerelak-Borys B,Skonieczna K,Grabska-Liberek I, Ocular ischemic syndrome - a systematic review. Medical science monitor : international medical journal of experimental and clinical research. 2012 Aug; [PubMed PMID: 22847215]
Level 3 (low-level) evidenceTripathy K, Mazumdar S. Recurrent retinal and choroidal ischemia in a case of ocular ischemic syndrome. Therapeutic advances in ophthalmology. 2019 Jan-Dec:11():2515841419848926. doi: 10.1177/2515841419848926. Epub 2019 Jul 12 [PubMed PMID: 31321381]
Level 3 (low-level) evidenceCochran ML, Mahabadi N, Czyz CN. Branch Retinal Vein Occlusion. StatPearls. 2023 Jan:(): [PubMed PMID: 30570991]
Scott IU, Figueroa MJ, Oden NL, Ip MS, Blodi BA, VanVeldhuisen PC, SCORE2 Investigator Group. SCORE2 Report 5: Vision-Related Function in Patients With Macular Edema Secondary to Central Retinal or Hemiretinal Vein Occlusion. American journal of ophthalmology. 2017 Dec:184():147-156. doi: 10.1016/j.ajo.2017.10.008. Epub 2017 Oct 23 [PubMed PMID: 29074161]
Simakurthy S, Tripathy K. Valsalva Retinopathy. StatPearls. 2023 Jan:(): [PubMed PMID: 31424803]
Elyadari M, Slassi N, Ghanem A, Bekkay R, Zarrouki M, Mouine S, Benchakroun N, Berraho A. [Case report of rare post-traumatic subretinal hemorrhage]. Journal francais d'ophtalmologie. 2017 May:40(5):e153-e156. doi: 10.1016/j.jfo.2017.02.003. Epub 2017 May 6 [PubMed PMID: 28483155]
Level 3 (low-level) evidenceClemente-Tomás R,Gargallo-Benedicto A,Schilt-Catafal M,García-Ibor F, Retinal arterial macroaneurysm: A study using optical coherence tomography and retinography. Archivos de la Sociedad Espanola de Oftalmologia. 2020 Feb; [PubMed PMID: 31879142]
Heydarian S, Jafari R, Dailami KN, Hashemi H, Jafarzadehpour E, Heirani M, Yekta A, Mahjoob M, Khabazkhoob M. Ocular abnormalities in beta thalassemia patients: prevalence, impact, and management strategies. International ophthalmology. 2020 Feb:40(2):511-527. doi: 10.1007/s10792-019-01189-3. Epub 2019 Oct 10 [PubMed PMID: 31602527]
Schönfeld CL, Schneider T, Körner U, Heidenkummer HP, Kampik A. Prognostic factors in vitreous surgery for proliferative diabetic retinopathy. German journal of ophthalmology. 1994 May:3(3):137-43 [PubMed PMID: 8038681]
Fernández-Vigo J, Fandiño J, Cordido M. [Prognostic factors in the treatment of edematous diabetic retinopathy with focal photocoagulation]. Ophtalmologie : organe de la Societe francaise d'ophtalmologie. 1988 Oct:2(4):311-4 [PubMed PMID: 3247197]
Acón D, Wu L. Multimodal Imaging in Diabetic Macular Edema. Asia-Pacific journal of ophthalmology (Philadelphia, Pa.). 2018 Jan-Feb:7(1):22-27. doi: 10.22608/APO.2017504. Epub 2017 Jan 29 [PubMed PMID: 29376234]
Jena S, Tripathy K. Vitreous Hemorrhage. StatPearls. 2024 Jan:(): [PubMed PMID: 32644557]
Stewart MW, Browning DJ, Landers MB. Current management of diabetic tractional retinal detachments. Indian journal of ophthalmology. 2018 Dec:66(12):1751-1762. doi: 10.4103/ijo.IJO_1217_18. Epub [PubMed PMID: 30451175]
Gross JG, Glassman AR, Liu D, Sun JK, Antoszyk AN, Baker CW, Bressler NM, Elman MJ, Ferris FL 3rd, Gardner TW, Jampol LM, Martin DF, Melia M, Stockdale CR, Beck RW, Diabetic Retinopathy Clinical Research Network. Five-Year Outcomes of Panretinal Photocoagulation vs Intravitreous Ranibizumab for Proliferative Diabetic Retinopathy: A Randomized Clinical Trial. JAMA ophthalmology. 2018 Oct 1:136(10):1138-1148. doi: 10.1001/jamaophthalmol.2018.3255. Epub [PubMed PMID: 30043039]
Level 1 (high-level) evidenceTseng VL, Greenberg PB, Scott IU, Anderson KL. Compliance with the American Academy of Ophthalmology Preferred Practice Pattern for Diabetic Retinopathy in a resident ophthalmology clinic. Retina (Philadelphia, Pa.). 2010 May:30(5):787-94. doi: 10.1097/IAE.0b013e3181cd47a2. Epub [PubMed PMID: 20168268]
. Screening guidelines for diabetic retinopathy. American College of Physicians, American Diabetes Association, and American Academy of Ophthalmology. Annals of internal medicine. 1992 Apr 15:116(8):683-5 [PubMed PMID: 1546870]
Level 1 (high-level) evidence