Introduction
Melanoacanthoma is a rare, benign cutaneous neoplasm. The lesion is more common in older adults, with a median age of 65 and a male predominance. Cutaneous melanoacanthoma has an unclear etiology, though some argue the presentation is a heavily pigmented seborrheic keratosis variant.[1] The initial presentation is typically a solitary, slow-growing black nodule on the head, neck, trunk, or extremities (see Image. Cutaneous Melanoacanthoma on the Right Preauricular Cheek).[2] Clinical examination, dermoscopy, and reflectance confocal microscopy of melanoacanthoma may be equivocal compared with malignant disease, so a biopsy with histopathologic evaluation is essential.[3] Microscopic examination shows an epithelial lesion with hyperkeratosis, acanthosis, and papillomatosis. The epidermis has mixed melanocytes with large dendritic processes and keratinocytes. Further staining with Fontana-Masson, S100 protein, or Melan-A (ie, MART-1) may be required to pinpoint a diagnosis.[4] Conservative excision of cutaneous melanoacanthoma is the treatment of choice since incomplete removal of the tumor can result in persistence and continued growth or recurrence of the lesion. Follow-up for patients with cutaneous melanoacanthoma should include evaluating any new or recurring lesions.
Melanoacanthoma, called oral melanoacanthoma, may be present in the mouth (or other mucosal epithelium) and is thought to be reactive without tumor-like progression.[5] See the StatPearls companion resource, "Oral Melanoacanthoma," for more information.[6] In contrast, the neoplasm can also occur on non-mucosal sites as a benign neoplasm and is designated as a cutaneous melanoacanthoma.[7][8][9] Considerable debate exists if pigmented seborrheic keratosis and cutaneous melanoacanthoma are 2 distinct entities, but they are considered separate, given their unique clinical features and management.[10][11][12][13]
Melanoacanthoma is associated with other syndromes. Metachronous oral melanoacanthomas were found in patients with Laugier-Hunziker syndrome, and melanoacanthoma on the lower lip has been associated with plasma cell cheilitis and Langerhans cell hyperplasia of the lower lip.[14][15]
Etiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Etiology
The etiology of melanoacanthoma has not been definitively established, though some have proposed a reactive process.[15] The secondary colonization by dendritic melanocytes of nonmelanocytic lesions has been observed, a common feature in reactive processes (eg, lichen simplex chronicus, verrucae, squamous cell carcinoma). Dendritic melanocyte colonization may account for these pigmented cells found at all epidermal levels in a melanoacanthoma. The colonization may be observed in sites of trauma, which is postulated to lead to irritation, lesion formation, and rapid augmentation of the lesion.[10][16][17] Superficial changes, like ulceration, may occur related to poor blood flow, friction, pressure when lying down, and trauma.[12] However, although features are similar to reactive processes, cutaneous melanoacanthoma is a neoplasm, differing from oral melanoacanthoma, which is thought to be purely reactive. Therefore, some etiologic factors are unexplored.
Epidemiology
The results of studies are limited regarding melanoacanthoma to establish definitive epidemiologic trends. The incidence of melanoacanthoma has been shown to range from 1 in 100,000 pathology specimens to between approximately 1% and 38% of seborrheic keratoses. Studies vary on the incidence, from 0.8% to 38%. However, many of these results have varying amounts of inclusion of pigmented seborrheic keratosis; earlier evaluations of seborrheic keratoses may have neglected the diagnosis of cutaneous melanoacanthoma.
Melanoacanthoma appears in older adults (usually aged greater than 60 years) with a median age of 65 but can appear across all adults. Most cases have been reported in men, but no clear gender predominance is apparent. Of 52 melanoacanthomas in 1 study, 28 were in men. Melanoacanthoma occurs worldwide. Reports of patients originate from many countries, including Belgium, Brazil, Canada, India, Italy, Japan, South Korea, Spain, and the United States of America.[2][7][2][9][13][9][18] The patient’s race or nationality was described for 43 individuals. Melanoacanthoma has been most described in white patients, who comprise 42% of reported cases.[19][18][20] Other reported patients were Hispanic (8 patients), Indian (6 patients), Asian (4 patients: 3 Japanese and 1 Korean), Black (4 patients), Brazilian, Haitian-Creole, and Italian.[8][10][11][12][17][21][22][23]
Pathophysiology
The pathophysiology of melanoacanthoma is unclear. Unlike seborrheic keratosis, trauma-induced cutaneous injury and partial inhibition of melanin pigment transfer from large melanocytes with numerous dendritic processes to adjacent keratinocytes may have a role in the pathophysiology of melanoacanthoma.[24] In 1 study, 20% to 50% croton oil placement or excision (to replicate trauma) was performed on half of a seborrheic keratosis to see if the formation of melanoacanthoma would occur. In contrast to melanoacanthomas, where the melanocytes are dispersed throughout the epidermis, the melanocytes of the irritated and traumatized seborrheic keratoses predominantly remained in the epidermal basal layers at the junction between the epidermis and dermis (see Image. Cutaneous Melanoacanthoma, Histopathology).[25] These findings implicate a pathogenesis different from seborrheic keratosis and more related to reactive or other pathogenesis.[21]
Histopathology
Microscopic features of an H&E-stained melanoacanthoma (see Image. Cutaneous Melanoacanthoma, Microscopic Features) include:
- Thickening of the stratum corneum (hyperkeratosis)
- Thickening of the entire epidermis (acanthosis)
- Absent cellular atypia of the keratinocytes
- Undulation of the epidermis (papillomatosis)
- Hyperpigmentation in all epidermis layers (correlating with melanocytes found in all epidermal layers)
- Keratin-filled pseudocysts in the epidermis
- Perivascular lymphocytic inflammation in the dermis [8][19][26][27][28]
Some researchers consider melanoacanthoma to have 2 histologic variants, either with melanocytes unevenly scattered throughout the epidermis or in nests with keratinocytes in the epidermis.[12][17] Stains may be required to pinpoint a diagnosis, including Fontana-Masson, S100 protein, or Melan-A (ie, MART-1). Combining these stains should differentiate the lesion from melanoma, which may appear similar clinically.[7][13] Because melanoacanthoma may have nests of melanocytes throughout the epidermis, careful evaluation for any atypical nests consistent with melanoma should be elucidated through stains, including cytokeratin stains (eg, cytokeratin 7), which may highlight keratinocytes to distinguish the lesion from melanoma.[18][23] If brown chromagen is used to identify the positive-staining cells when immunoperoxidase stains are performed, differentiating the brown-staining melanocytes from the brown-appearing melanin-laden keratinocytes is imperative. However, if red chromagen is used, this problem is resolved since only the melanocytes show the red staining.
Electron microscopy confirms the presence of highly dendritic melanocytes and reveals the defective transfer of melanin from these melanocytes to the adjacent keratinocytes. Also, Langerhans cells may be present throughout the epidermis except in the basal layer.[10]
History and Physical
Melanoacanthoma is a solitary, pigmented, asymptomatic, slow-growing papule, nodule, or plaque. Although uncommon, they can grow rapidly, itch, be tender, or become irritated.[22] Melanoacanthomas may be present for weeks to years before diagnosis; they range from 0.2 to 15 cm in size, with a median size of 2 cm.[11][17][11][19] Most importantly, half of the lesions identified are black and may express multiple colors (eg, black and blue, black and gray, black and brown), which resembles melanoma.[23][29] Occasionally, the surface of a melanoacanthoma may be lobulated or verrucous.[8][10][18][10][8][21][20] Usually, melanoacanthoma presents on the head, neck, or trunk, though some have been reported on extremities, genitals, or buttocks. Multiple lesions are rare, unlike oral melanoacanthomas.[5] Rarely, a melanoacanthoma presents as a cutaneous horn or exophytic, mimicking verrucae or a squamous cell carcinoma (see Image. Cutaneous Melanoacanthoma, Left Lower Abdomen).[12]
Evaluation
A partial or excisional biopsy is the most appropriate approach for evaluating a lesion when diagnosing melanoacanthoma. An adequate biopsy specimen should be obtained so that the pathologist has sufficient tissue to examine and thereby be able to provide an accurate diagnosis.[3] Other tools that may show promise include dermoscopy and reflectance confocal microscopy, though these alone, based on the available literature, are insufficient to diagnose the condition.
Dermoscopy
Dermoscopic features of melanoacanthoma vary from mimicking malignant lesions to benign lesions. The results of some studies show that the dermoscopy of melanoacanthoma is similar to a pigmented Spitz nevus, including a starburst pattern consisting of symmetrically distributed pigmented streaks on the periphery. This pattern has been considered diagnostic for a pigmented Spitz nevus. Features of melanoma may be visualized when performing a dermoscopy of melanoacanthoma, including gray to whitish hyperkeratotic areas, multiple black-to-brown dots, multiple globules, blue-white veil, polymorphous vessels, and granularity. Dermoscopy may visualize benign lesions, with a regular pigmentary network showing a cribriform pattern of ridges and fissures, comedo-like openings, hairpin vessels, milia-like cysts, moth-eaten border, and sharp demarcations.[11][20]
Reflectance Confocal Microscopy
Reflectance confocal microscopy of melanoacanthoma has been reviewed in multiple studies. Findings included keratotic debris-filled surface depressions and ridges at the level of the stratum corneum of the epidermis, which correlates with the verrucous lesion. Numerous tangled bright dendritic cells may be noted in the suprabasal spinous and granular layers of the hyperplastic epidermis.[18] At the spinous-granular layer, a typical honeycomb pattern may be present with epidermal projections, keratin-filled invaginations, and widespread dendritic pagetoid cells. Dendritic cell infiltration can occur at the dermal-epidermal junction between the dermal papillae. Melanophages may be noted at the level of the papillary dermis.[22]
Treatment / Management
Sometimes confused for melanoma on visual examination, most melanoacanthomas are traditionally surgically removed either at the time of biopsy or subsequently by simple excision. Partial removal during the biopsy of a melanoacanthoma has resulted in the persistence of the preauricular lesion in at least 1 patient.[21] Also, the recurrence of a nasal melanoacanthoma was observed 6 months after removal in one reported case.[10] Other therapeutic interventional modalities may be considered, including cryotherapy with liquid nitrogen or topical 5-fluorouracil 5% cream.[11][12](B3)
Differential Diagnosis
While benign, melanoacanthomas have a wide differential that should include:
- Actinic keratosis: Pigmented actinic keratoses should show atypia during the evaluation of a skin biopsy specimen.
- Basal cell carcinoma: Pigmented basal cell carcinoma may present similar to melanoacanthoma but should be distinguished by histopathology.
- Melanocytic nevus: If the lesion is raised, distinguishing melanoacanthoma without histopathologic evaluation is challenging. However, some melanocytic nevi are flat and can be distinguished clinically from melanoacanthoma.
- Melanoma: Clinically, melanoma (including lentigo maligna) appears nearly identical to melanoacanthoma. However, if an H&E-stained specimen is nondiagnostic, histopathologic evaluation with stains may need to be used to distinguish the 2 entities.
- Pigmented spindle cell (Spitz) nevus
- Seborrheic keratosis: In seborrheic keratosis, the pigment and melanocytes are predominantly present and restricted to the basal or lower epidermis layers. In contrast, in a melanoacanthoma, the pigment results from the dendritic melanocytes that are present throughout not only the lower layers but also the upper layers of the epidermis.
- Solar lentigo: Solar lentigo is usually flat, while melanoacanthoma is generally raised.
- Squamous cell carcinoma: The absence of cellular atypia and numerous mitoses excludes squamous cell carcinoma.
- Verruca vulgaris: The absence of koilocytes or koilocytic change excludes the diagnosis of verrucae.[2][19][22]
Prognosis
The prognosis for an individual with melanoacanthoma is excellent, as the lesion is benign and not associated with systemic manifestations. Follow-up of lesion excision usually reveals no recurrence, though scarring and other sequelae of excision may be present.[17][23] Some lesions may persist or recur, but this is less likely; lesions are unlikely to progress to malignancy.[10][21]
Complications
Melanoacanthoma is a benign epithelial lesion without any observation of malignant transformation. However, the lesion may occur adjacent to a malignant lesion, which may obfuscate diagnosis. Although most melanoacanthomas grow slowly, they can become giant, measuring over 10 cm in greatest diameter. Also, albeit less common, some melanoacanthomas have been noted to grow rapidly. In either of these settings, the lesion can become irritated and tender from rubbing against the adjacent skin or the overlying clothing. Rarely, a larger melanoacanthoma may spontaneously ulcerate or superficially infect.[12]
Deterrence and Patient Education
Melanoacanthomas are benign but mimic other benign and malignant lesions, like seborrheic keratosis or melanoma, respectively. Therefore, older adults with lesions resembling cutaneous melanoacanthoma must seek appropriate evaluation. Noninvasive examination methods, including clinical inspection, dermoscopy, and reflectance confocal microscopy, can be performed but are insufficient to confirm a diagnosis. Therefore, when a melanoacanthoma is possible, an appropriate lesion biopsy should be performed to confirm the diagnosis and exclude other conditions.
Pearls and Other Issues
Melanoacanthoma is a rare benign epidermis lesion; however, the actual incidence remains to be established. The clinician who evaluates the patient seldom considers melanoacanthoma. Most often, when a biopsy specimen is submitted, either melanoma or seborrheic keratosis or both are suggested. Complete removal of the melanoacanthoma is recommended since the lesion may persist following partial removal.
Enhancing Healthcare Team Outcomes
The clinical morphology of a melanoacanthoma can mimic that of another benign skin tumor, a seborrheic keratosis. However, for many patients in whom this epithelial neoplasm has been described, the initial impression was a melanoma. Therefore, the patient’s primary clinician should consider performing a complete skin check on the patient. Detecting a new or progressively enlarging asymptomatic black lesion should prompt the primary clinician to refer the patient to a dermatologist or surgeon for evaluation, including a lesion biopsy. Periodic follow-up by the clinician to confirm healing and ensure no persistence or recurrence of the melanoacanthoma is recommended.
Media
(Click Image to Enlarge)
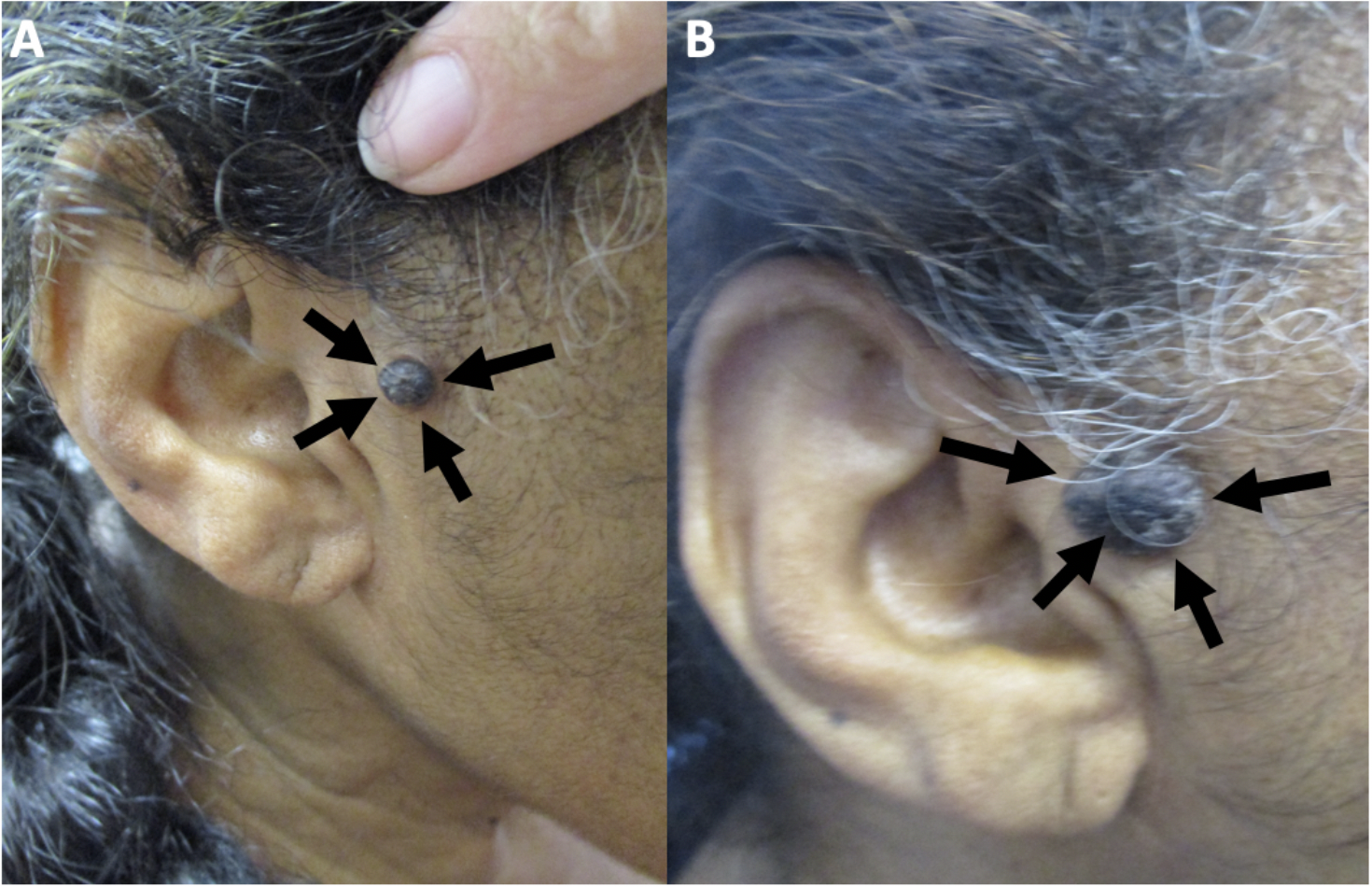
Cutaneous Melanoacanthoma on the Right Preauricular Cheek. Initial (A) and recurrent (B) lesion in a 65-year-old woman.
Guiterrez N, Erickson CP, Calame A, Sateesh BR, Cohen PR: Melanoacanthoma masquerading as melanoma: case reports and literature review. Cureus. 2019 Jun 25:11(6):e4998) and are republished with permission. CC licensing.
(Click Image to Enlarge)
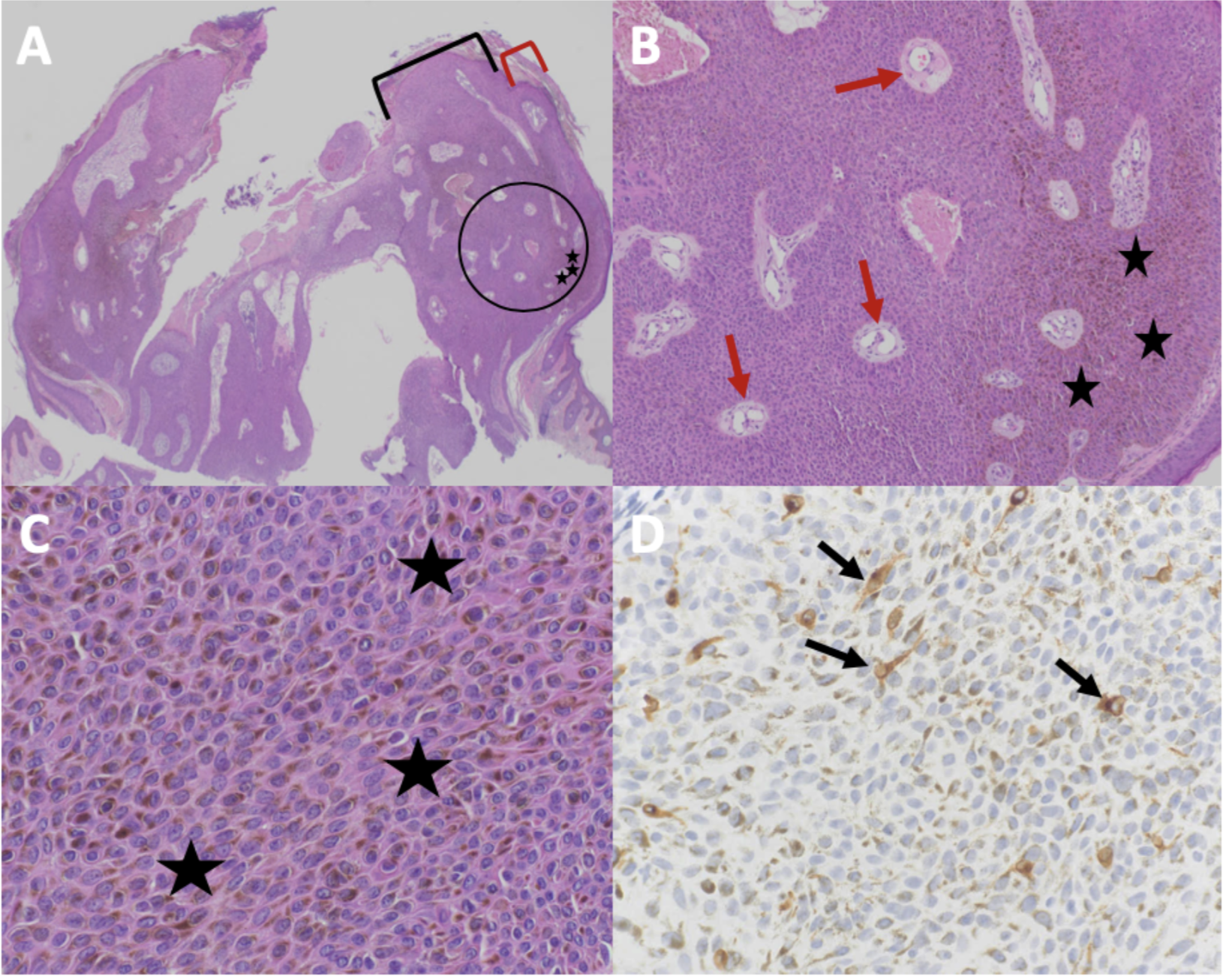
Cutaneous Melanoacanthoma, Histopathology. H&E-stained sections magnified at 2x (A), 10x (B; magnified black circle from 2x power image), and 40x (C) showing hyperpigmentation at all epidermal layers (black stars) and epithelial lined vessels and erythrocytes within the epithelium (red arrows); MART-1–stained section at 40x magnification (D) showing dendritic melanocytes at all epidermal layers (black arrows).
Guiterrez N, Erickson CP, Calame A, Sateesh BR, Cohen PR: Melanoacanthoma masquerading as melanoma: case reports and literature review. Cureus. 2019 Jun 25:11(6):e4998) and are republished with permission. CC Licensing.
(Click Image to Enlarge)
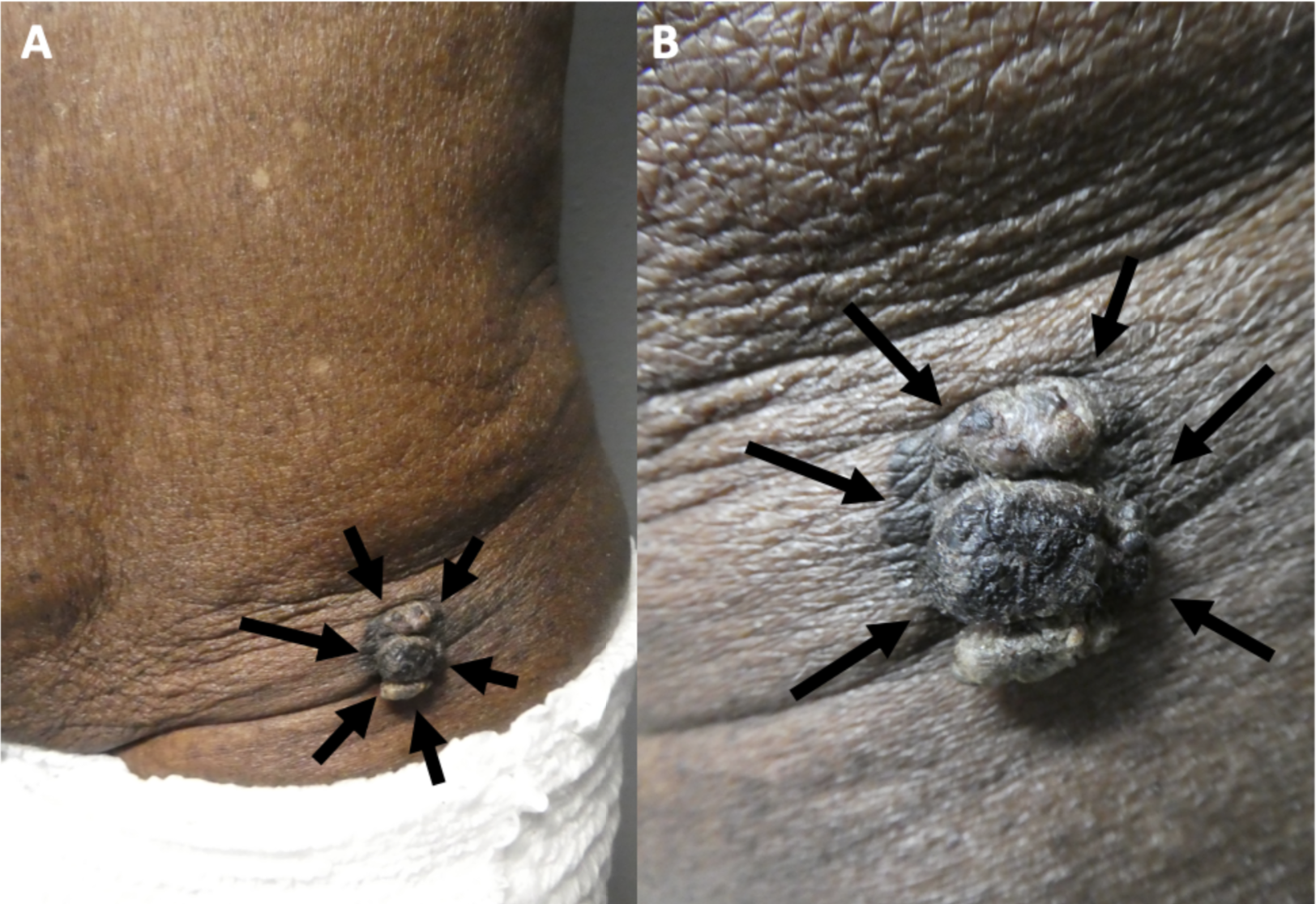
Cutaneous Melanoacanthoma, Left Lower Abdomen. Exophytic appearance (black arrows) of cutaneous melanoacanthoma at distant (A) and close (B) views.
Guiterrez N, Erickson CP, Calame A, Sateesh BR, Cohen PR: Melanoacanthoma masquerading as melanoma: case reports and literature review. Cureus. 2019 Jun 25:11(6):e4998) and are republished with permission. CC Licensing.
(Click Image to Enlarge)
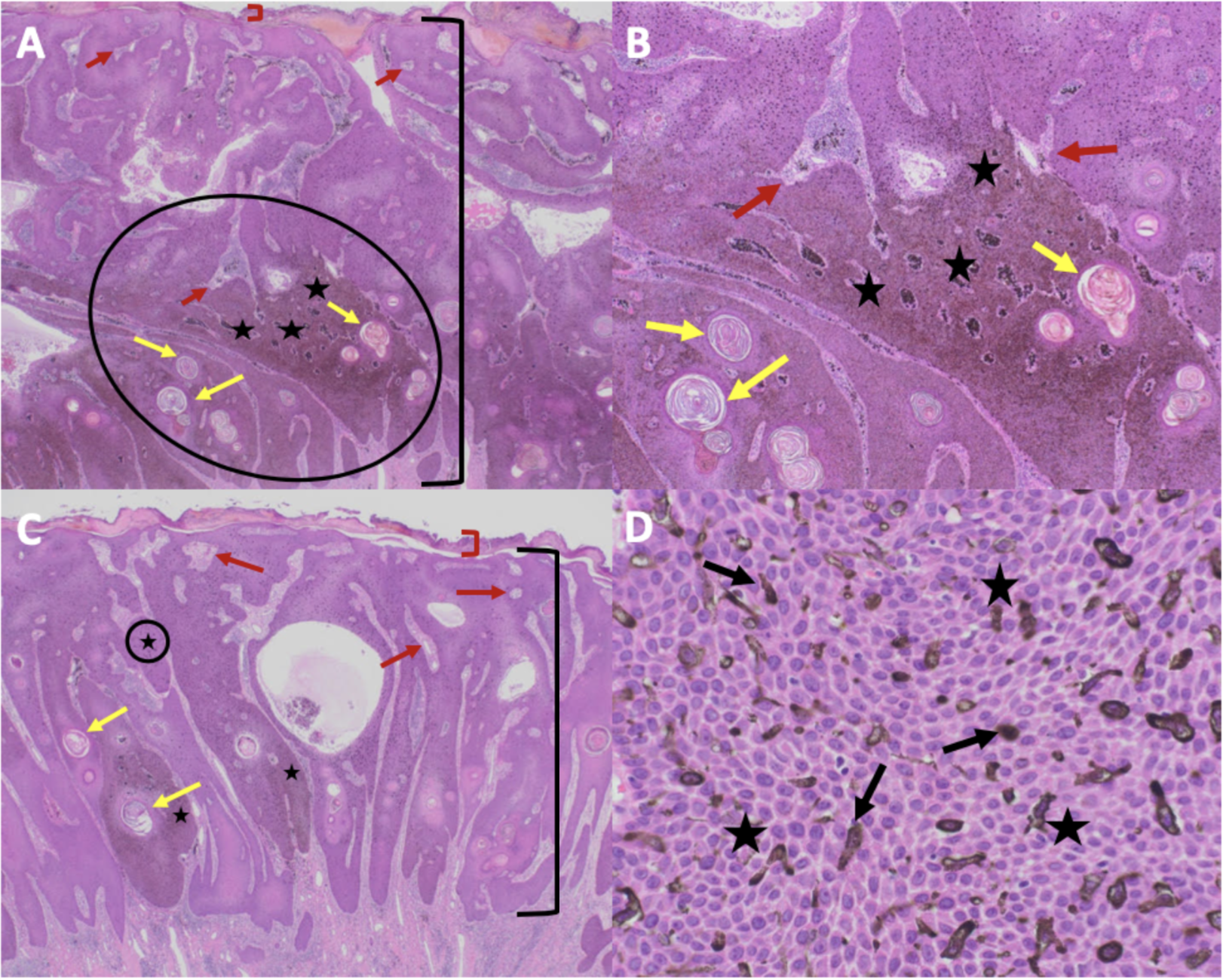
Cutaneous Melanoacanthoma, Microscopic Features. Microscopic views of exophytic (A, B) and endophytic (C, D) portions of cutaneous melanoacanthoma on an H&E-stained specimen showing hyperkeratosis (red bracket), acanthosis (black bracket), hyperpigmentation at all epidermal layers (black stars), keratin-filled pseudocysts (yellow arrows), epithelial-lined vessels with erythrocytes in the dermis (red arrows), and dendritic melanocytes in all epidermal layers (black arrows) at 2x (A), 10x (B), and 40x (C, D) power; circled portions in (A) and (C) are shown in (B) and (D), respectively.
Guiterrez N, Erickson CP, Calame A, Sateesh BR, Cohen PR: Melanoacanthoma masquerading as melanoma: case reports and literature review. Cureus. 2019 Jun 25:11(6):e4998) and are republished with permission. CC Licensing.
References
Hunziker MF, Guaraldo GR, Michalany NS, Michalany A, de Andrade ACH, Fagundes Sortino AM. Melanoacanthoma on the nipple of a middle-aged woman: A diagnostic challenge. JAAD case reports. 2021 Mar:9():93-96. doi: 10.1016/j.jdcr.2021.01.026. Epub 2021 Feb 2 [PubMed PMID: 33665288]
Level 3 (low-level) evidenceChung E, Marghoob AA, Carrera C, Marchetti MA. Clinical and Dermoscopic Features of Cutaneous Melanoacanthoma. JAMA dermatology. 2015 Oct:151(10):1129-30. doi: 10.1001/jamadermatol.2015.1453. Epub [PubMed PMID: 26083708]
Ticknor IL, Cassarino DS. Malignant Melanoacanthoma. The American Journal of dermatopathology. 2023 Dec 1:45(12):822-824. doi: 10.1097/DAD.0000000000002572. Epub [PubMed PMID: 37883954]
Choi JE, Bae EJ, Kim AR, Son SW, Song HJ, Oh CH. A Case of Melanoacanthoma: Immunohistochemical Staining Using VECTOR® NovaRED™ to Distinguish Melanocytes from the Cutaneous Pigment. Annals of dermatology. 2008 Mar:20(1):18-21. doi: 10.5021/ad.2008.20.1.18. Epub 2008 Mar 31 [PubMed PMID: 27303152]
Level 3 (low-level) evidenceDatta A, Lamba AK, Tandon S, Urs A, Lnu M. A Unique Presentation of Gingival Melanoacanthoma: Case Report and Review of Literature. Cureus. 2020 Mar 18:12(3):e7315. doi: 10.7759/cureus.7315. Epub 2020 Mar 18 [PubMed PMID: 32313756]
Level 3 (low-level) evidenceJimenez M, Brizuela M. Oral Melanoacanthoma. StatPearls. 2024 Jan:(): [PubMed PMID: 33351414]
Sanchez Yus E, Simon Huarte P. [Seborrheic warts and melanoacanthoma; are both tumors the same? (Sainz de Aja's prize 1968)]. Actas dermo-sifiliograficas. 1969 Mar-Apr:60(3):73-87 [PubMed PMID: 4184385]
Prince C, Mehregan AH, Hashimoto K, Plotnick H. Large melanoacanthomas: a report of five cases. Journal of cutaneous pathology. 1984 Aug:11(4):309-17 [PubMed PMID: 6491009]
Level 3 (low-level) evidenceRoh NK, Hahn HJ, Lee YW, Choe YB, Ahn KJ. Clinical and Histopathological Investigation of Seborrheic Keratosis. Annals of dermatology. 2016 Apr:28(2):152-8. doi: 10.5021/ad.2016.28.2.152. Epub 2016 Mar 31 [PubMed PMID: 27081260]
MISHIMA Y, PINKUS H. Benign mixed tumor of melanocytes and malpighian cells. Melanoacanthoma: Its relationship to Bloch's benign non-nevoid melanoepithelioma. Archives of dermatology. 1960 Apr:81():539-50 [PubMed PMID: 14422903]
Shankar V, Nandi J, Ghosh K, Ghosh S. Giant melanoacanthoma mimicking malignant melanoma. Indian journal of dermatology. 2011 Jan:56(1):79-81. doi: 10.4103/0019-5154.77559. Epub [PubMed PMID: 21572799]
Jain S, Barman KD, Garg VK, Sharma S, Dewan S, Mahajan N. Multifocal cutaneous melanoacanthoma with ulceration: a case report with review of literature. Indian journal of dermatology, venereology and leprology. 2011 Nov-Dec:77(6):699-702. doi: 10.4103/0378-6323.86485. Epub [PubMed PMID: 22016278]
Level 3 (low-level) evidenceSimón P, Requena L, Sánchez Yus E. How rare is melanoacanthoma? Archives of dermatology. 1991 Apr:127(4):583-4 [PubMed PMID: 2006888]
Level 3 (low-level) evidenceZaki H, Sabharwal A, Kramer J, Aguirre A. Laugier-Hunziker Syndrome Presenting with Metachronous Melanoacanthomas. Head and neck pathology. 2019 Jun:13(2):257-263. doi: 10.1007/s12105-018-0897-3. Epub 2018 Feb 15 [PubMed PMID: 29450847]
Silva EV, Silveira HA, Nogueira GM, Campos AC, F da Silva AT, León JE. Melanoacanthoma, Plasma Cell Cheilitis and Langerhans Cell Hyperplasia on the Lower Lip: An Unusual Association and Immunohistochemical Analysis. Indian journal of dermatology. 2022 Jul-Aug:67(4):404-408. doi: 10.4103/ijd.ijd_380_22. Epub [PubMed PMID: 36578708]
Albagieh H, Aloyouny A, Alshagroud R, Alwakeel A, Alkait S, Almufarji F, Almutairi G, Alkhalaf R. Habitual khat chewing and oral melanoacanthoma: A case report. World journal of clinical cases. 2023 Jan 16:11(2):449-455. doi: 10.12998/wjcc.v11.i2.449. Epub [PubMed PMID: 36686340]
Level 3 (low-level) evidenceMishra DK, Jakati S, Dave TV, Agrawal M. A Rare Pigmented Lesion of the Eyelid. International journal of trichology. 2019 Jul-Aug:11(4):167-169. doi: 10.4103/ijt.ijt_51_19. Epub [PubMed PMID: 31523108]
Shahriari N, Grant-Kels JM, Rabinovitz HS, Oliviero M, Scope A. In vivo reflectance confocal microscopy features of a melanoacanthoma. Dermatology practical & conceptual. 2016 Oct:6(4):27-30 [PubMed PMID: 27867743]
Schlappner OL, Rowden G, Philips TM, Rahim Z. Melanoacanthoma. Ultrastructural and immunological studies. Journal of cutaneous pathology. 1978 Jun:5(3):127-41 [PubMed PMID: 210197]
Level 3 (low-level) evidenceRossiello L, Zalaudek I, Ferrara G, Docimo G, Giorgio CM, Argenziano G. Melanoacanthoma simulating pigmented spitz nevus: an unusual dermoscopy pitfall. Dermatologic surgery : official publication for American Society for Dermatologic Surgery [et al.]. 2006 May:32(5):735-7 [PubMed PMID: 16706772]
Level 3 (low-level) evidenceGutierrez N, Erickson CP, Calame A, Sateesh BR, Cohen PR. Melanoacanthoma Masquerading as Melanoma: Case Reports and Literature Review. Cureus. 2019 Jun 25:11(6):e4998. doi: 10.7759/cureus.4998. Epub 2019 Jun 25 [PubMed PMID: 31497429]
Level 3 (low-level) evidencePorto ACS, Blumetti TP, Macedo MP, Braga JCT. Melanoacanthoma: a potential pitfall of reflectance confocal microscopy. Anais brasileiros de dermatologia. 2019 Nov-Dec:94(6):747-750. doi: 10.1016/j.abd.2019.01.002. Epub 2019 Oct 26 [PubMed PMID: 31789261]
Pierard GE. [Clear cell melanoacanthoma]. Annales de dermatologie et de venereologie. 1986:113(3):253-5 [PubMed PMID: 3752865]
Level 3 (low-level) evidenceCantudo-Sanagustín E, Gutiérrez-Corrales A, Vigo-Martínez M, Serrera-Figallo MÁ, Torres-Lagares D, Gutiérrez-Pérez JL. Pathogenesis and clinicohistopathological caractheristics of melanoacanthoma: A systematic review. Journal of clinical and experimental dentistry. 2016 Jul:8(3):e327-36. doi: 10.4317/jced.52860. Epub 2016 Jul 1 [PubMed PMID: 27398186]
Level 1 (high-level) evidenceMevorah B, Mishima Y. Cellular response of seborrheic keratosis following croton oil irritation and surgical trauma with special reference to melanoacanthoma. Dermatologica. 1965:131(6):452-64 [PubMed PMID: 4222277]
Santana T, Queiroz A, Gonçales LMC, Andrade NS, Trierveiler M. Focal melanocytic lesions of the oral mucosa: An epidemiological and morphological study. Oral diseases. 2023 Oct:29(7):2723-2733. doi: 10.1111/odi.14482. Epub 2023 Jan 4 [PubMed PMID: 36565435]
Level 2 (mid-level) evidenceTavares TS, da Costa AAS, da Mata Camargos F, Gomez RS, Nonaka CFW, Alves PM, Mendonça EF, Sousa-Neto SS, Vasconcelos ACU, Tarquínio SBC, Caldeira PC. Oral melanoacanthoma: Clinicopathological and immunohistochemical features of a case series and a scoping review. Journal of oral pathology & medicine : official publication of the International Association of Oral Pathologists and the American Academy of Oral Pathology. 2023 Nov:52(10):1013-1020. doi: 10.1111/jop.13495. Epub 2023 Oct 18 [PubMed PMID: 37852303]
Level 2 (mid-level) evidenceHeppt MV, Heinzerling L. [Pigmented lesions of the mucosa]. Dermatologie (Heidelberg, Germany). 2022 Sep:73(9):682-691. doi: 10.1007/s00105-022-05039-9. Epub 2022 Aug 23 [PubMed PMID: 35997969]
Roh D, Ha DL, Kim Y, Shin K, Kim HS, Ko HC, Kim BS, Kim MB. Comparison of Dermoscopic Features Between Melanoacanthomas and Melanoacanthoma-Like Malignant Melanomas in Korean Patients. Journal of cutaneous medicine and surgery. 2021 May-Jun:25(3):243-248. doi: 10.1177/1203475420977470. Epub 2020 Nov 26 [PubMed PMID: 33242988]
