Introduction
Since ancient times, children around the world have been afflicted with cystic fibrosis that leads to shortened lifespans. In medieval Europe, these children were believed to be cursed by witches and doomed to die. The curse that became folklore pronounced, “Woe to the child who tastes salty from a kiss on the brow, for he is cursed and soon will die.” Salty skin was a sign of an impending illness without cause or cure. Until relatively modern times, cystic fibrosis was poorly understood. In 1949, Lowe et al. postulated that cystic fibrosis must be caused by a genetic defect from the autosomal recessive pattern of inheritance of the disease. High levels of salt in the sweat of patients with cystic fibrosis suggested an abnormality in electrolyte transport from the sweat gland. Quinton postulated that sweat ducts in these patients were impermeable to chloride. Further studies led to the hypothesis that the faulty chloride channel must be situated in the apical membranes of the lung surface or glandular epithelium to explain the respiratory and systemic organ failure associated with cystic fibrosis. Researchers now know that cystic fibrosis is an autosomal recessive disorder of exocrine gland function most commonly affecting persons of Northern European descent at a rate of 1 in 3500. It is a chronic disease that frequently leads to chronic sinopulmonary infections and pancreatic insufficiency. The most common cause of death is end-stage lung disease.[1][2][3][4]
Etiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Etiology
CF is caused by a genetic mutation in a gene on chromosome 7 that codes for a protein transmembrane conductance regulator (CFTR) protein, which functions as a transmembrane cAMP-activated chloride channel. Both copies of the gene are mutated in clinical disease.
There are over 2000 different mutations in the CFTR gene that can cause disease. These mutations are divided into five classes:
- Defective protein synthesis
- Defective protein processing
- Disordered regulation
- Defective chloride conductance
- Accelerated channel turnover[5]
Epidemiology
The most common mutation is delta F508, which is found in 70% of American white patients with CF and two-thirds of all cases worldwide. This mutation is a class 2 mutation of abnormal folding of the CFTR protein, leading to premature destruction within the Golgi apparatus. The delta F508 mutation commonly leads to exocrine pancreatic insufficiency and a higher likelihood of meconium ileus.[6]
Pathophysiology
Class 1 dysfunction is the result of nonsense, frameshift, or splice-site mutation, which leads to premature termination of the mRNA sequence. This fails to translate the genetic information into a protein product with a subsequent total absence of CFTR protein, and approximately 2% to 5% of cystic fibrosis cases result.
Class 2 dysfunction results in abnormal post-translational processing of the CFTR protein. This step in protein processing is essential for the proper intracellular transit of the protein. As a result, CFTR is unable to be moved to the correct cellular location.
Class 3 dysfunction is characterized by diminished protein activity in response to intracellular signaling. The result is a fully formed protein channel in the cellular membrane that is non-functional.
Class 4 dysfunction is when the protein is produced and correctly localized to the cell surface. However, the rate of chloride ion flow and the duration of channel activation after stimulation is decreased from normal.
Class 5 dysfunction is the net decreased concentration of CFTR channels in the cellular membrane as a result of rapid degradation by cellular processes. It includes mutations that alter the stability of mRNA and others that alter the stability of the mature CFTR protein.
The result of all mutations is decreased secretion of chloride and consequently increased resorption of sodium into the cellular space. The increased sodium reabsorption leads to increased water resorption and manifests as thicker mucus secretions on epithelial linings and more viscous secretions from exocrine tissues. Thickened mucus secretions in nearly every organ system involved result in mucous plugging with obstruction pathologies. The most commonly affected organs include the sinuses, lungs, pancreas, biliary and hepatic systems, intestines, and sweat glands.
Sinus disease occurs when secretion viscosity increases which obstruct the sinus ostia. Additional processes often exist here. These include ciliary dysfunction, increased inflammatory mediators, and increased bacterial colonization with pathogens such as Pseudomonas aeruginosa. The result of this syndrome is impaired sinus secretion clearance. Subsequently, chronic sinusitis occurs, and secondary structural damage may occur.
Lung disease occurs as a spectrum obstruction from thickened mucous secretion. It is important to note that the lungs of a CF patient are normal in utero, at birth, and after birth. Disease results as a cascade effect following infection and the subsequent inflammatory process. Mucus plugging in the bronchioles results in an obstructive lung disease clinical picture. As a result of obstruction, an environment optimal for bacterial growth is created within the airways. Bronchiectasis and further thick purulent sputum production occur. Part of the inflammatory reaction includes the production of the neutrophil interleukin-8 from epithelial cells, which functions as a secretagogue, increasing mucous secretion, thereby creating a positive feedback loop of mucous secretion with persistence of inflammation, infection, and structural damage. The result of this cascade is obstruction of the airways with the resulting failure of ventilation of the lung. Poorly managed pulmonary manifestations are the primary cause of death in patients with CF.
Pancreatic manifestations of CF are due in large part to obstruction of the pancreatic ductules by thickened secretions. On the passage of gastric contents into the proximal duodenum, the pancreatic exocrine glands are triggered to excrete pancreatic enzymes into the luminal space of the small intestines. However, increased viscosity of excretions and obstruction of the pancreatic ductules inhibits this process. The net pH of the secretions lessens due to decreased sodium bicarbonate composition lending to a lesser neutralization of the acidic stomach chyme. The lower pH chyme effectively degrades what pancreatic enzymes reach the intestinal lumen. As a result, intestinal chyme is not enzymatically processed in the intestines resulting in the pathognomonic greasy stools, colicky abdominal pain, and malabsorption of nutrients from foods. Specifically, fat-soluble vitamins A, D, E, and K are notably deficient.
Furthermore, autodigestion of the pancreas may occur as these enzymes target the pancreatic tissues. This results in pancreatitis. In severe, chronic cases, this can lead to endocrine pancreatic failure when the islets of Langerhans begin to be digested by trapped pancreatic enzymes. The lasting impacts of this spectrum of disease mimic type-1 diabetes mellitus.
Increased secretion viscosity does not spare the biliary and hepatic systems. The biliary ductules may be plugged with secretions. Obstructive cirrhosis and post-hepatic hyperbilirubinemia can occur. Secondary to this, esophageal varices, splenomegaly, and hypersplenism may occur as a result of increased hepatic portal vein pressures. Gallbladder disease is more likely to occur as a spectrum of this manifestation of CF, with up to 15% of those with cystic fibrosis having gallstones.
Intestinal involvement is typically seen in children with meconium ileus at birth and intestinal obstruction later in life. The cause of meconium ileus is multifactorial. Likely, it is due to increased fluid absorption as a result of the faulty CFTR channel with dehydration of the intestinal contents leading to constipation coupled with a change in luminal contents from normal secondary to pancreatic insufficiency as discussed above. Mechanical obstruction chronically leads to inflammation and eventual scarring and stricture formation. This may lead to further intestinal obstruction by fecal impaction or intussusception later in life.
Sweat glands offer an interesting contrast to all other tissues containing CFTR channels in that the flow of chloride is reversed. Normally, sweat glands move chloride from the extracellular space into the intracellular space. Thus, sodium and water are reabsorbed from the sweat gland tissues into the body. However, failure of the chloride channel to reabsorb chloride leads to a loss of sodium onto the skin surface and a subsequent fluid loss. This causes the pathognomonic salty skin seen with cystic fibrosis. In prolonged or warm environments or more severe cases, this may lead to hyponatremic dehydration.
In addition to acting as a chloride transport protein, other interactions of CFTR have been postulated. In the apical plasma membrane, CFTR is part of a multiprotein assembly where three of its amino acids, threonine, arginine, and leucine, act to root the protein to a region known as PDZ-type receptors. These PDZ regions have been observed to occur in multiple intracellular signaling proteins associated with the plasma membrane as well. This also roots CFTR closely to other transporters, ion channels, receptors, kinases, phosphatases, signaling molecules, and cytoskeletal elements. These interactions between CFTR and its binding proteins have been shown to be critically involved in regulating CFTR-mediated transepithelial ion transport in vitro and in vivo. It seems that these close associations may allow for CFTR to play an essential role in epithelial cells beyond as an ion channel. While not yet fully understood, modulation of CFTR in animal studies proved that inflammatory responses, maturational processing, non-chloride ion transport, and intracellular signaling are related to its function. These other interacting proteins are potential modifiers of the cystic fibrosis phenotype and may help explain the substantial differences in clinical severity between similar genotypic patients with CF.[7][8][9]
History and Physical
Newborns with CF may present with meconium ileus, prolonged neonatal jaundice, or early lung infection. Infants and children with CF may present with failure to thrive and poor weight gain, anemia, undescended testicles in boys, recurrent sinopulmonary infections, and a distal intestinal obstructive syndrome with or without pancreatic insufficiency. The median age of diagnosis is 6 to 8 months; although, individuals may not exhibit clinical signs and symptoms until later.
Adults with CF often present with exacerbations of one or more symptoms. Lung manifestations of CF include chronic bronchitis, abnormal pulmonary function tests, bronchiectasis, atypical asthma, allergic bronchopulmonary aspergillosis, and colonization with Pseudomonas aeruginosa. Sinus manifestations of CF include chronic rhinosinusitis, chronic post-nasal drip, nasal polyposis, and panopacification of the paranasal sinuses. Pancreatic manifestations include pancreatic insufficiency, recurrent pancreatitis, and early-onset diabetes. Hepatobiliary manifestations include focal biliary cirrhosis, cholelithiasis, periportal fibrosis, liver cirrhosis, portal hypertension, and variceal bleeding. Musculoskeletal manifestations include kyphoscoliosis, osteopenia/osteoporosis, and arthropathy. Hematologic manifestations include iron-deficiency anemia or anemia of chronic disease leading to splenomegaly. Nephrogenic manifestations include nephrolithiasis, nephrocalcinosis, hyperoxaluria, and hypocitraturia. Dermatologic manifestations include “salty sweat,” digital clubbing, and cyanosis. Additional dermatologic conditions due to malabsorption include acrodermatitis enteropathica due to zinc deficiency and scaly dermatitis associated with fatty acid deficiency. Finally, males may be infertile due to absent vas deferens, while females have reduced fertility due to thickened cervical mucus.
Evaluation
In the United States, newborns are screened for CF as part of a standard newborn screening panel. Some cases of CF are discovered on prenatal ultrasound, which may demonstrate meconium peritonitis, bowel dilation, or absent gallbladder. Such findings often lead to prenatal CF carrier screening.[10][11][12][13]
To diagnose CF, the following criteria must be met:
Suspicion for Cystic Fibrosis
- Sibling with cystic fibrosis
- Positive newborn screen
- Clinical symptoms consistent with CF in 1 or more organ systems
- Chronic sinopulmonary disease
- Gastrointestinal or nutritional abnormalities
- Salt loss syndromes
- Obstructive azoospermia
Evidence of CFTR Dysfunction
- Elevated sweat chloride 2 than 60 mEq/L on two occasions
- Two disease-causing CFTR mutations
- Abnormal nasal potential difference
The diagnostic pathway starts with a sweat chloride test. If normal but still symptomatic, a repeat sweat chloride test is indicated. If the test is abnormal, DNA testing is indicated. If one or less CFTR mutations are found, expanded DNA analysis is indicated. However, the finding of 2 CF-related mutations confirms the diagnosis of cystic fibrosis.
The test for immunoreactive trypsinogen (IRT), a pancreatic enzyme, increases sensitivity and specificity in screening newborns with meconium ileus for CF. IRT monitoring can be correlated with the severity of CF, and when it drops below detectable levels can indicate the need to start pancreatic enzyme replacement.
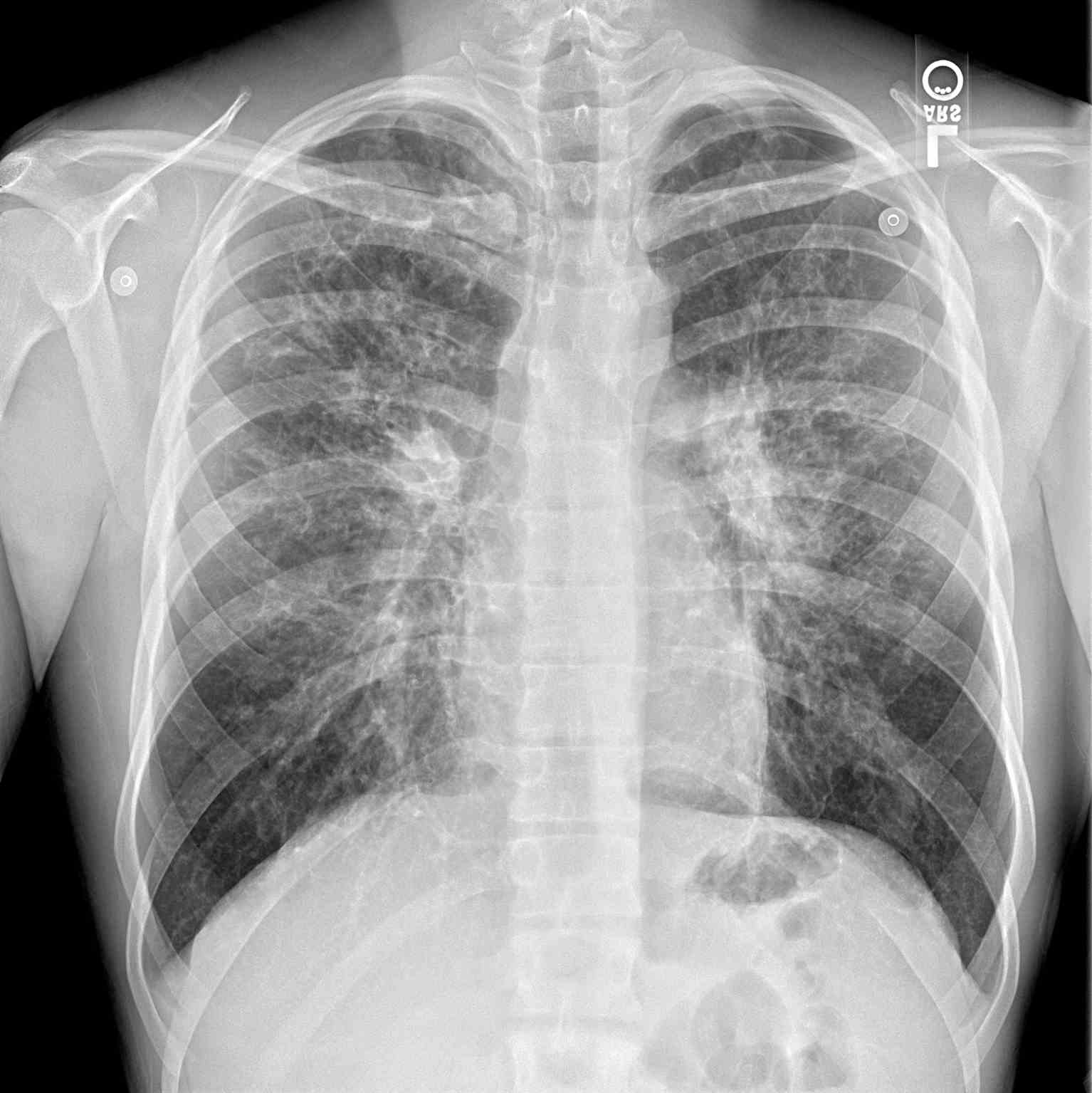
Additional diagnostics may be indicated depending on the presenting symptoms. A chest radiograph may help identify hyperinflation, bronchiectasis, abscesses, or atelectasis. Sinus radiography may demonstrate panopacification of the paranasal sinuses. Abdominal radiology may be helpful in neonates who present with meconium ileus. Bronchoalveolar lavage typically shows many neutrophils, and microbiology is commonly positive for Haemophilus influenza, Staphylococcus aureus, Pseudomonas aeruginosa, Burkholderia cepacia, Escherichia coli, or Klebsiella pneumoniae.
Pulmonary function testing is a major tool for evaluating and monitoring disease state and progression in CF. Spirometry is the commonly used pulmonary function test. It measures the volume of air exhaled during a forceful and complete exhalation after a maximal inhalation. The total exhaled volume, known as the forced vital capacity (FVC), the volume exhaled in the first second, known as the forced expiratory volume in one second (FEV1), and their ratio (FEV1/FVC) are the most critical variables reported. These values allow for interpretation of the status of the lung ventilation function. These values are compared to an expected normal for age, height, and gender to generate an expected normal value. The measured value is then calculated as a percent of normal where normal equals 100%. A normal or high FEV1 and/or a low FVC may indicate restrictive lung disease. A low FEV1 with a high FVC indicates obstructive lung disease with airway trapping. Cystic fibrosis can be expected to show air trapping patterns with low FEV1 values proportional to the severity of the disease.[14]
Treatment / Management
Cystic fibrosis is a systemic illness that has broad implications for both quality and quantity of life when poorly controlled. Therefore, treatment should focus on optimizing function to avoid acute illness events. This should target maintaining lung function by aggressively controlling respiratory infection and clearing airways of mucus, optimizing nutritional status with pancreatic enzyme supplements and multivitamins, and finally, by managing any other health complications that may arise. This is best performed when using a team approach of specialists who are experienced in managing cystic fibrosis.[15][16][17][18](A1)
As stated previously, pulmonary disease is the most common cause of mortality in cystic fibrosis. As such, it is extremely vital to have a low threshold for diagnosis and intervention in pulmonary illness exacerbations. A pulmonary exacerbation is the worsening of lung function due to an infection. Often this is characterized by shortness of breath, fatigue, productive cough, and fever. Pulmonary function testing will worsen from baseline during an exacerbation. Any acute illness should prompt admission to a hospital facility familiar with cystic fibrosis management.
Pulmonary illness should be managed with two primary goals: treat the infection and improve oxygenation. P. aeruginosa typically induces infectious etiologies, and antibiotic therapy should have spectrum coverage against this pathogen. However, sputum culture should be obtained and a sensitivity profile obtained for the pathogens present. CF guidelines recommend at least one antibiotic to cover each pathogenic bacteria cultured from respiratory secretions and two antibiotics for P. aeruginosa infections. Mild exacerbations may be amenable to oral antibiotics, but more severe exacerbations require intravenous medication use. Inhaled antibiotics are not recommended whenever an intravenous alternative is possible. Ventilation and oxygenation should be supported through the use of inhaled bronchodilators, including albuterol and ipratropium bromide. Agents such as inhaled dornase alfa or inhaled hypertonic saline are prescribed to promote airway secretion clearance in conjunction with chest physiotherapy. Anti-inflammatory medicines such as glucocorticoids are also used to assist in opening airways to relieve the obstruction. The work of breathing should be optimized, utilizing nasal cannula oxygen when appropriate. Bilevel positive airway pressure (BiPAP) ventilation may be necessary to overcome airway trapping. Intubation with mechanical ventilation is an option but should be avoided whenever possible and used only when respiratory failure is imminent.
Chronic, supportive therapy for patients with CF includes regular pancreatic enzymes, fat-soluble vitamins (A, D, E, K), mucolytics, bronchodilators, antibiotics, and anti-inflammatory agents.
A new class of medications known as CFTR modulator therapies is designed to correct the dysfunction by improving production, intracellular processing, or function of the CFTR protein caused by the mutated gene. Each medication is targeted at a specific dysfunction caused by a specific gene mutation. Ivacaftor is used in the treatment of class 3 dysfunctions, where a mutation at G551D is the primary aberration. It acts by binding the defective CFTR protein at the cell surface and opening the chloride channel, thus restoring the proper function of the protein. This was the first medication to directly impact the protein channel rather than treating the effects of CF. Dosing for patients older than six years is 150 mg by mouth every 12 hours. Younger patients should receive weight-based dosing where those with less than 14 kg bodyweight receive 50 mg by mouth every 12 hours, and those with greater than 14 kg bodyweight should receive 75 mg by mouth every 12 hours. Lumacaftor is a chaperone molecule that was designed to move the defective CFTR protein from the intracellular organelles where it is processed and onto the cell surface. As such, it is effective in delta F508 homozygous mutation genotypes. Clinically, this medicine has no benefit when given alone. However, when combined as lumacaftor/ivacaftor has been shown in the 92-week PROGRESS study to have modest benefits for patients who tolerate the medicine in both pulmonary function testing and body mass index (BMI). This medicine combination was subsequently approved for use in a patient older than six years old. Dosing is two tablets containing lumacaftor 200 mg/ivacaftor 125 mg taken orally every 12 hours. Another medication combination currently in clinical trials is tezacaftor/ivacaftor. Tezacaftor, as a medicine, is very similar to lumacaftor in that it is a CFTR protein chaperone that improves the intracellular processing and trafficking of CFTR. Likewise, its clinical efficacy is only seen when placed in combination with ivacaftor. This medicine is showing a good safety profile, and preliminary EVOLVE and EXPAND trials are indicating less side effect profiles compared to lumacaftor/ivacaftor.
Despite major advancements in medical therapies for CF, the disease process continues to advance, and the lungs will ultimately fail prematurely from the disease burden without surgical intervention. Lung transplant is the treatment of choice for end-stage lung disease. The timing of the transplant is multifactorial. The International Society of Heart and Lung Transplantation published a list of conditions to be used when considering transplant referral and takes into consideration the 5-year predicted survival of less than 50%, the FEV1 that has fallen to 30% of predicted values, rapidly falling FEV1 despite optimal therapy, a 6-minute walk distance of less than 400 meters, the development of pulmonary hypertension in the absence of a hypoxemic exacerbation, clinical decline characterized by increasing frequency of exacerbations including acute respiratory failure requiring noninvasive ventilation, a pattern of poor clinical recovery from successive exacerbations, worsening nutritional status despite supplementation, pneumothorax, or life-threatening hemoptysis despite bronchial artery embolization. Virtually all lung transplants for CF will need replacement of both lungs. This is because a native, diseased lung would act as a source of infected secretions that would threaten the transplanted lung and possibly induce respiratory failure. It is important to note that transplantation is not a cure for CF, but it confers a prolongation of life and offers significant symptomatic relief.
Diet and Exercise
Individuals with CF are encouraged to consume a high-fat diet with supplemental fat-soluble vitamins to compensate for malabsorption. Additionally, patients living with CF are encouraged to consume a high-calorie diet to maintain a healthy weight and combat chronic inflammation and frequent infections that are commonly encountered. According to the Cystic Fibrosis Foundation, women should consume 2500 to 3000 calories a day, while men should consume 3000 to 3700 calories a day. Those living in hot climates or who participate in activities that cause sweating are encouraged to consume additional sodium in their diet. Oral feedings are preferred; however, if the intake does not meet metabolic demand as determined by continued decreases in BMI, enteral (tube) feedings should be considered. These are typically in the form of gastric tube feedings or jejunal tube feedings. Multiple control studies of enteral nutrition in patients with CF have shown benefit in the form of improved, or neutral lung function following exacerbations of illness that directly correlate with BMI. With that noted, however, no randomized studies of enteral nutrition have been performed in patients with CF to date. Parenteral nutrition may be considered only when oral or enteral nutrition is not meeting metabolic needs. Parenteral nutrition has been linked to increased risk for sepsis events and should be used sparingly. Regular exercise is encouraged in patients with CF to maintain and support lung function.
Differential Diagnosis
- Asthma
- Bronchiolitis
- Bronchiectasis
- Celiac disease
- Nutritional considerations in failure to thrive
- Pediatric Aspergillosis
- Primary ciliary dyskinesia
- Sinusitis
Prognosis
Patients with CF are estimated to live until about the fourth decade of life before requiring lung transplantation. Lung transplantation confers a median survival of 8.5 years.
Enhancing Healthcare Team Outcomes
Cystic fibrosis is a systemic illness that has broad implications for both quality and quantity of life when poorly controlled. Therefore, treatment should focus on optimizing function to avoid acute illness events. This should target maintaining lung function by aggressively controlling respiratory infection and clearing airways of mucus, optimizing nutritional status with pancreatic enzyme supplements and multivitamins, and finally, by managing any other health complications that may arise. This is best performed when using a team approach of specialists who are experienced in managing cystic fibrosis. Specialties that may be part of the team include pulmonologists, infectious disease, and gastroenterology. Nurses monitor patients and facilitate team communication. Pharmacists review all medications, check for drug-drug interactions, and educate patients and their families about the importance of compliance and potential side effects. [Level 5] Lung infections continue to have the highest mortality rates. For children who undergo a lung transplant, survival is increased for 6-8 years, but without a lung transplant, most children are dead within a few years.
Media
References
Lyamin AV, Ismatullin DD, Zhestkov AV, Kondratenko OV. [The laboratory diagnostic in patients with mucoviscidosis: A review.]. Klinicheskaia laboratornaia diagnostika. 2018:63(5):315-320. doi: 10.18821/0869-2084-2018-63-5-315-320. Epub [PubMed PMID: 30689329]
Poncin W, Lebecque P. [Lung clearance index in cystic fibrosis]. Revue des maladies respiratoires. 2019 Mar:36(3):377-395. doi: 10.1016/j.rmr.2018.03.007. Epub 2019 Jan 25 [PubMed PMID: 30686561]
Bianconi I,D'Arcangelo S,Esposito A,Benedet M,Piffer E,Dinnella G,Gualdi P,Schinella M,Baldo E,Donati C,Jousson O, Persistence and Microevolution of {i}Pseudomonas aeruginosa{/i} in the Cystic Fibrosis Lung: A Single-Patient Longitudinal Genomic Study. Frontiers in microbiology. 2018 [PubMed PMID: 30692969]
Eschenhagen P, Schwarz C. [Patients with cystic fibrosis become adults : Treatment hopes and disappointments]. Der Internist. 2019 Jan:60(1):98-108. doi: 10.1007/s00108-018-0536-9. Epub [PubMed PMID: 30627755]
Awatade NT, Wong SL, Hewson CK, Fawcett LK, Kicic A, Jaffe A, Waters SA. Human Primary Epithelial Cell Models: Promising Tools in the Era of Cystic Fibrosis Personalized Medicine. Frontiers in pharmacology. 2018:9():1429. doi: 10.3389/fphar.2018.01429. Epub 2018 Dec 7 [PubMed PMID: 30581387]
Pallin M. Cystic fibrosis vigilance in Arab countries: The role of genetic epidemiology. Respirology (Carlton, Vic.). 2019 Feb:24(2):93-94. doi: 10.1111/resp.13461. Epub 2018 Dec 13 [PubMed PMID: 30548951]
Bush A, Floto RA. Pathophysiology, causes and genetics of paediatric and adult bronchiectasis. Respirology (Carlton, Vic.). 2019 Nov:24(11):1053-1062. doi: 10.1111/resp.13509. Epub 2019 Feb 25 [PubMed PMID: 30801930]
García-Clemente M, Enríquez-Rodríguez AI, Iscar-Urrutia M, Escobar-Mallada B, Arias-Guillén M, López-González FJ, Madrid-Carbajal C, Pérez-Martínez L, Gonzalez-Budiño T. Severe asthma and bronchiectasis. The Journal of asthma : official journal of the Association for the Care of Asthma. 2020 May:57(5):505-509. doi: 10.1080/02770903.2019.1579832. Epub 2019 Feb 20 [PubMed PMID: 30784336]
Blasi F, Elborn JS, Palange P. Adults with cystic fibrosis and pulmonologists: new training needed to recruit future specialists. The European respiratory journal. 2019 Jan:53(1):. pii: 1802209. doi: 10.1183/13993003.02209-2018. Epub 2019 Jan 17 [PubMed PMID: 30655450]
Rajapaksha IG, Angus PW, Herath CB. Current therapies and novel approaches for biliary diseases. World journal of gastrointestinal pathophysiology. 2019 Jan 5:10(1):1-10. doi: 10.4291/wjgp.v10.i1.1. Epub [PubMed PMID: 30622832]
Mandalia A, Wamsteker EJ, DiMagno MJ. Recent advances in understanding and managing acute pancreatitis. F1000Research. 2018:7():. pii: F1000 Faculty Rev-959. doi: 10.12688/f1000research.14244.2. Epub 2018 Jun 28 [PubMed PMID: 30026919]
Level 3 (low-level) evidenceFenker DE, McDaniel CT, Panmanee W, Panos RJ, Sorscher EJ, Sabusap C, Clancy JP, Hassett DJ. A Comparison between Two Pathophysiologically Different yet Microbiologically Similar Lung Diseases: Cystic Fibrosis and Chronic Obstructive Pulmonary Disease. International journal of respiratory and pulmonary medicine. 2018:5(2):. pii: 098. doi: 10.23937/2378-3516/1410098. Epub 2018 Nov 29 [PubMed PMID: 30627668]
Kiedrowski MR, Bomberger JM. Viral-Bacterial Co-infections in the Cystic Fibrosis Respiratory Tract. Frontiers in immunology. 2018:9():3067. doi: 10.3389/fimmu.2018.03067. Epub 2018 Dec 20 [PubMed PMID: 30619379]
Stern M, Picard C, Roux A. [Lung transplantation]. La Revue du praticien. 2018 Feb:68(2):189-194 [PubMed PMID: 30801151]
Declercq D, Van Meerhaeghe S, Marchand S, Van Braeckel E, van Daele S, De Baets F, Van Biervliet S. The nutritional status in CF: Being certain about the uncertainties. Clinical nutrition ESPEN. 2019 Feb:29():15-21. doi: 10.1016/j.clnesp.2018.10.009. Epub 2018 Nov 7 [PubMed PMID: 30661680]
Radovanovic D, Santus P, Blasi F, Sotgiu G, D'Arcangelo F, Simonetta E, Contarini M, Franceschi E, Goeminne PC, Chalmers JD, Aliberti S. A comprehensive approach to lung function in bronchiectasis. Respiratory medicine. 2018 Dec:145():120-129. doi: 10.1016/j.rmed.2018.10.031. Epub 2018 Nov 2 [PubMed PMID: 30509700]
Lapp V, Chase SK. How Do Youth with Cystic Fibrosis Perceive Their Readiness to Transition to Adult Healthcare Compared to Their Caregivers' Views? Journal of pediatric nursing. 2018 Nov-Dec:43():104-110. doi: 10.1016/j.pedn.2018.09.012. Epub 2018 Sep 28 [PubMed PMID: 30473151]
Pawlaczyk-Kamieńska T, Borysewicz-Lewicka M, Śniatała R, Batura-Gabryel H, Cofta S. Dental and periodontal manifestations in patients with cystic fibrosis - A systematic review. Journal of cystic fibrosis : official journal of the European Cystic Fibrosis Society. 2019 Nov:18(6):762-771. doi: 10.1016/j.jcf.2018.11.007. Epub 2018 Nov 23 [PubMed PMID: 30473190]
Level 1 (high-level) evidence