Introduction
Brucellosis is an infectious disease caused by Brucella species. It is known by many other names, including remitting fever, undulant fever, Mediterranean fever, Maltese fever, Gibraltar fever, Crimean fever, goat fever, and Bang disease. The disease was described by George Cleghorn, a British army surgeon stationed on Minorca, in his 1751 work Observations on the Epidemical Diseases in Minorca from the Year 1744 to 1749. Brucellosis again came to the attention of medical officers of the British army on the island of Malta following the Crimean War. The genus Brucella is named for Major-General Sir David Bruce, who, in 1886, led the Malta Fever Commission that identified Brucella melitensis as the organism responsible for the disease. In 1905, Sir Themistocles Zammit identified that infected goats transmitted brucellosis in their milk.[1][2][3]
Brucella organisms are small aerobic intracellular coccobacilli. These are found in the reproductive organs of host animals, causing abortions and sterility. They are shed in urine, milk, placental fluid, and other fluids of the animals. Many species have been identified, but the following four have moderate-to-significant human pathogenicity:
- Brucella melitensis (from sheep)
- Brucella suis (from pigs)
- Brucella abortus (from cattle)
- Brucella canis (from dogs)
Brucella melitensis and suis have the highest pathogenicity, while Brucella abortus and canis have moderate pathogenicity.
Etiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Etiology
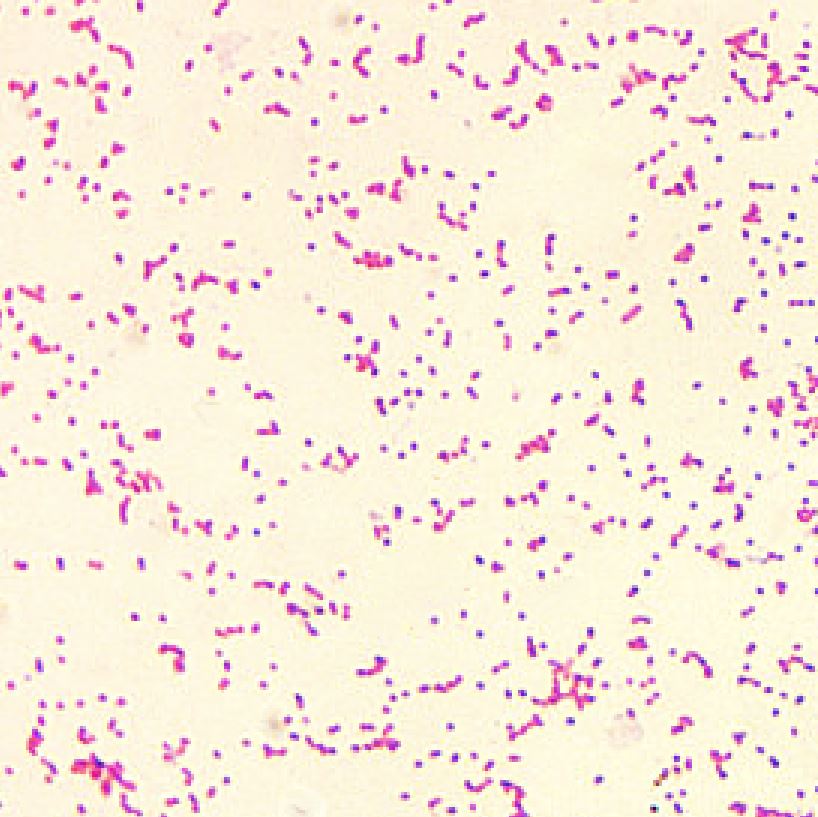
Brucellosis is a zoonotic disease that can be caused by four different Brucella species in humans: B. suis, B. melitensis, B. abortus, and B. canis. As few as 10 to 100 organisms can cause the disease in humans. All Brucella species are gram-negative, nonmotile, facultative intracellular coccobacilli. Brucella species do not form spores or toxins. The animal host of B. suis is swine; the hosts of B. melitensis are sheep and goats; the host of B. abortus is cattle, and the hosts of B. canis are dogs.[4][5]
Among these, B. melitensis is the most virulent, followed by B. suis. B. melitensis causes the most severe cases of brucellosis and is the most prevalent worldwide. B melitensis is acquired by exposure to animals or animal products. Ingestion of unpasteurized goat milk and related dairy products is the main route by which B. melitensis is transmitted to humans. Slaughterhouse workers become inoculated with brucellae through aerosolization of fluids, splashing of mucous membranes, and contamination of skin abrasions. Veterinarians are usually infected by inadvertent inoculation of animal vaccines against B. abortus and B. melitensis. In laboratories, it can infect technicians from specimens of animals (including humans) whose tissues are operated upon or submitted for culture or pathologic analysis. B. abortus is more widely distributed, but it is less pathogenic for both animals and humans and rarely causes complications. It has been the most common cause of brucellosis in North America. B. suis has been the second most common cause of brucellosis in North America. It causes a prolonged course of illness associated with suppurative destructive lesions. B. canis infection causes a disease similar to that of B abortus.
Epidemiology
The disease is transmitted from animals to humans by consuming unpasteurized milk and dairy products, consuming undercooked meat, or skin penetration of those in contact with livestock.
United State Statistics
It is one of the most common laboratory-acquired bacterial infections in the United States. It also is transmitted by inhalation of contaminated aerosols, conjunctival inoculation, blood transfusions, transplacentally from mother to fetus, and rarely from person to person. Brucella causes more than 500,000 infections per year worldwide. In 2010, 115 cases of brucellosis were diagnosed in the United States and reported to the Centers for Disease Control and Prevention. California and Texas were the states with the highest number of cases in 2010, and most cases in the United States are related to occupational exposure of those who work with animals. The number of reported cases in the United States has dropped significantly because of animal vaccination programs and milk pasteurization.[6][7]
Age
Brucellosis is more prevalent in the young population, with 60% of cases in persons aged 13-40 years, 16% in those aged 40-60 years, and 2.5% in 60 years and older.[8] Brucellosis may be more common in children in developing countries because of a lack of pasteurization.
Sex
Brucellosis is more common in males as compared to females. Occupational exposure to animals is the most likely cause of this gender vulnerability.
Pathophysiology
The incubation period may be as few as three days or as long as several weeks. When ingested, Brucella organisms are phagocytosed and enter the intestinal submucosa, where they are transported to lymphoid tissue by macrophages. In the bloodstream, the organisms are quickly contained within circulating polymorphonuclear cells (PMNs) and macrophages, making use of numerous mechanisms to avoid or suppress bactericidal responses. The bacteria do not activate the alternative complement system. Brucellae are transported into the lymphatic system and may replicate there locally; they also may replicate in the liver, spleen, kidney, breast tissue, or joints, causing both localized and systemic infection. Symptoms frequently last for several months, and chronic infections with brucellosis may last for years.
History and Physical
A detailed history is a very important tool in the diagnosis of brucellosis. A dietary history is important in evaluating the possibility of brucellosis among individuals who live in non-endemic regions because the disease may be acquired through the ingestion of infected foods shipped from the endemic regions. Symptoms of brucellosis include a headache, cyclical fever, migratory arthralgia, myalgia, asthenia, anorexia, fatigue, malaise, weakness, sweating, vomiting, diarrhea, abdominal pain, and miscarriage. Brucella has been shown to cause sacroiliitis, osteomyelitis, spondylodiscitis, septic arthritis, and epidural abscess. Hepatic abscesses and granulomas occur in some cases. Spontaneous bacterial peritonitis and infection of a ventriculoperitoneal shunt have been reported. Respiratory symptoms, including a cough, dyspnea, and pleurisy, may occur; however, in most cases, chest radiography is normal, although focal abscesses, effusions, granulomas, and abscesses have occurred. Neurologic symptoms and Guillain-Barre syndrome have been associated with acute neurobrucellosis. Subarachnoid hemorrhage and myelitis also have been observed. Immune thrombocytopenic purpura has occurred. Death occurs in 2% of cases. Patients have been reported to have particularly foul-smelling perspiration that has been described as similar to mold or wet hay.
The physical examination is most often normal, although lymphadenopathy, splenomegaly, and hepatomegaly may be found. Epididymo-orchitis can occur in association with brucellosis; a tender, swollen scrotum with erythema is present in these patients. Endocarditis may present with new or changing murmurs, and a pericardial rub is present in patients with pericarditis. Endocarditis rarely occurs; however, it is the most common cause of death from brucellosis. Aortic fistulas have also been documented. Neurologic findings vary according to the presentation of neurologic disease. In the case of meningitis, nuchal rigidity, Kerning sign, and Brudzinski sign are present. Increased intracranial pressure (ICP) or brain abscess will manifest as papilledema, cranial nerve palsy, and focal neurologic deficits. Skin lesions such as maculopapular eruptions, erythema nodosum, abscesses, and panniculitis have been reported. Ocular findings can include uveitis, keratoconjunctivitis, iridocyclitis, optic neuritis, and cataracts.
Evaluation
Symptoms and signs of brucellosis are nonspecific; cultures and serology are usually necessary for diagnosis. Some general laboratory findings might suggest the diagnosis. Following laboratory tests are ordered in patients suspected of having brucellosis:
Complete Blood Count (CBC)
CBC typically shows neutropenia and anemia. Thrombocytopenia is also reported (secondary to hepatosplenomegaly or from immune thrombocytopenia). Pancytopenia can be seen in a few patients.
Inflammatory Markers
Laboratory testing on the patient may show elevated C-reactive protein, erythrocyte sedimentation rate, serum lactate dehydrogenase, and alkaline phosphatase.
Liver Enzymes
Elevation in liver enzymes is very common. These elevated levels may reflect the severity of hepatic involvement and correlate clinically with hepatomegaly.
Culture
Brucellosis may be diagnosed by blood cultures in tryptose medium; however, because of the slow-growing nature of Brucella, the cultures may take a week or more to become positive. Bone marrow culture has a higher yield than blood cultures because the reticuloendothelial system holds a high concentration of brucellae. Because of the ease of aerosol transmission, any potential Brucella specimens should be handled under a biohazard hood.
Serology
Standard agglutination testing is the most common method of diagnosis in endemic areas. Indirect enzyme-linked immunosorbent assay (ELISA) and Rose Bengal testing also may be used. Repeated serologic testing is recommended if the initial titer is low. Titers higher than 1:160 in conjunction with a compatible clinical presentation are suggestive of infection. Titers higher than 1:320 are considered to be more specific, especially in endemic areas. Seroconversion and evolution of the titers can also be used for diagnosis.
Polymerase Chain Reaction
Polymerase chain reaction (PCR) tests have been developed for the rapid detection and diagnosis of Brucella species in human blood specimens.[9] Two major genetic targets are the Brucella gene BCSP31 and the 16S-23S rRNA operon.
Radiography
Pedro Pons sign, erosion of the anterior superior aspect of lumbar vertebrae with osteophytosis, is associated with spondylodiscitis caused by Brucella. Disc space narrowing, bone destruction, and sclerosis may be seen on imaging in patients with spondylitis. Nonspecific hepatitis and granulomas may be observed on liver biopsy.[10]
Biopsy
Bone marrow aspiration and biopsy may be required to establish a diagnosis in certain patients. A percutaneous liver biopsy may be needed in the patient with liver granulomas to obtain a specimen for diagnosis. Analysis of liver biopsy specimens may reveal granulomatous hepatitis and hepatic microabscesses.
Treatment / Management
The goal of medical and surgical therapy in brucellosis is to control the disease as quickly as possible in order to prevent complications and relapses.
Antibiotics
Doxycycline is used in conjunction with either streptomycin, rifampin, gentamicin, or sulfamethoxazole/trimethoprim to treat brucellosis; because the bacteria reside within cells, several weeks of treatment are needed. Treating with a single antibiotic should be avoided, as there is a high rate of relapse when a combination regimen is not used. For uncomplicated infections, doxycycline (100 mg PO twice daily for 6 weeks) may be the most appropriate monotherapy; however, relapse rates with such monotherapy approach 40%; hence rifampin (600-900 mg/day) is usually added. In children, a regimen of trimethoprim-sulfamethoxazole and rifampin for 4 to 6 weeks may be used since children should not be treated with doxycycline. Pregnant women may be treated with rifampin during pregnancy, with trimethoprim-sulfamethoxazole added postpartum. In spondylitis or sacroiliitis, doxycycline and rifampin combined with an aminoglycoside (gentamicin) for the initial 2-3 weeks, followed by 6 weeks of rifampin and doxycycline, is usually recommended.
Additional Drugs
Symptomatic treatment with antipyretics and analgesics must also be administered. Corticosteroids may be used in patients with brucella meningitis, but the evidence is lacking.
Surgical Intervention
The main roles of surgery in patients with brucellosis lie in the treatment of endocarditis and in the drainage of pyogenic joint effusions or paraspinal abscesses.[11][12] The infected heart valves of patients with endocarditis may need to be replaced, and repair of aortic and aortopulmonary fistulas has been performed. Surgical debridement and bone grafting of patients with spondylitis also have been performed. Dairy herds in the United States are tested for brucellosis yearly, and cows are vaccinated against the disease; accidental exposure to the cow vaccine has been shown to cause the disease in humans.[13][14](A1)
Differential Diagnosis
The clinical presentation of brucellosis mimics other bacterial and viral infections. Following differential diagnoses should be considered in patients suspected of having brucellosis:
- Epstein bar virus infectious mononucleosis
- Infective endocarditis
- Influenza
- Leptospirosis
- Malaria
- Mechanical back pain
- Meningitis
- Mycoplasmal pneumonia
- Viral hepatitis
- Enteric fever
- Acute epididymitis
- Urinary tract infection
Prognosis
Although it may take time to obtain control of the symptoms, the prognosis for most patients with brucellosis is excellent. The overall risk of relapse is very low. However, in patients with comorbid disorders like heart or lung disease, the condition can be disabling. The prognosis is poor in patients who present with congestive heart failure due to endocarditis. Unlike many other infection disorders, the condition tends to improve with physical activity rather than bed rest. The recovery usually takes 3-6 months. When recurrence does occur, it may be either local or systemic. Overall mortality in acute or chronic cases of brucellosis is very low, certainly less than 5% and probably less than 2%.
Complications
Complications are rare if the patient is treated timely and appropriately, though relapse of infection may occur. The risk for the development of focal complications increases if symptom duration is greater than one month before diagnosis. The following are the most common complication of brucellosis:
- Cardiovascular
- Endocarditis
- Myocarditis
- Pericarditis
- Mycotic aneurysms of the aorta and cerebral vessels
- Genitourinary
- Orchitis
- Epididymo-orchitis
- Glomerulonephritis
- Pyelonephritis
- Abortion
- CNS
- Meningitis
- Meningoencephalitis
- Papilledema
- Radiculopathy
- Stroke
- Optic neuropathy
- Hematologic
- Disseminated intravascular coagulation
- Hemophagocytic syndrome
- Musculoskeletal
- Spondylitis
- Sacroiliitis
- Arthritis
- Osteomyelitis
- Bursitis
- Tenosynovitis
- Gastrointestinal or hepatobiliary
- Hepatitis
- Hepatic abscess
- Acute cholecystitis.
- Ileitis
- Colitis
- Spontaneous peritonitis
- Abscess in the spinal cord, spleen, or thyroid
Postoperative and Rehabilitation Care
The majority of patients are managed as outpatients unless there are complications. Avoiding contact with the source of infection is important. To ensure the patient is responding to treatment, serological monitoring is required. Compliance with medical treatment is essential for a full recovery.
Deterrence and Patient Education
Patients should be educated regarding the nature of the disease, its transmission, symptoms, complications, and treatment. Other issues for which patients should be educated include:
- The adverse effects of the medications
- Compliance with medications until full recovery
- Avoid infected animals, use gloves and masks when dealing with an infected animal, and avoid potentially contaminated foods
- Immunization of cattle, sheep, and goats
- Adequate cooking of meat products and pasteurization of dairy products
- Laboratory workers maintain an appropriate level of containment
Pearls and Other Issues
Brucella species have been successfully developed into biological weapons by the United States and several countries, although it is not known to have been used in war. B. suis (agent US), B. melitensis (agent AM), and B. abortus (agent AB) were all researched by the United States for its biological warfare program; however, their lethality of less than 5% made them less than ideal for military leaders who were seeking lethal agents. Brucella is easily aerosolized and survives well after aerosolization. In the event of a biological attack or a laboratory accident involving a Brucella species, treatment with one of the antibiotic regimens mentioned above should be considered. Antibiotic prophylaxis is not generally recommended for exposure to endemic Brucella species, and prevention is best achieved by wearing appropriate protection when working with infected animals, adequately cooking meat products, and pasteurization of dairy products. Although vaccines are available for animals, there is no human vaccine to prevent Brucella infection.
Enhancing Healthcare Team Outcomes
Brucellosis is a common zoonotic disease that is transmitted to humans from ruminants. While the disease is on the decline, epidemiological surveillance is vital to detect any outbreak or local emergence in humans or animals. Expert evidence indicates that brucellosis is best managed by an interprofessional team of health care providers that includes an epidemiologist, an infectious disease consultant, internists, a pharmacist, and a nurse.[15][16] [Level 5]
Because the disease is acquired from wildlife, it is vital to educate the patient and family on avoidance of exposure. Since brucellosis can present with the involvement of almost any organ system, high suspicion should be maintained. The medical laboratory is essential to help make the diagnosis of the disease. Once the infection is treated, the pharmacist should ensure that the patient remains compliant with drug therapy. In addition, if there is any evidence of recurrence, the patient should be referred to an internist for serological studies. Countless studies show that an interprofessional approach to brucellosis results in a complete cure within 4-6 months, with excellent outcomes.[17][18] [Level 5]
Media
(Click Image to Enlarge)
(Click Image to Enlarge)
References
Głowacka P, Żakowska D, Naylor K, Niemcewicz M, Bielawska-Drózd A. Brucella - Virulence Factors, Pathogenesis and Treatment. Polish journal of microbiology. 2018 Jun 30:67(2):151-161. doi: 10.21307/pjm-2018-029. Epub [PubMed PMID: 30015453]
Hull NC, Schumaker BA. Comparisons of brucellosis between human and veterinary medicine. Infection ecology & epidemiology. 2018:8(1):1500846. doi: 10.1080/20008686.2018.1500846. Epub 2018 Jul 24 [PubMed PMID: 30083304]
Bakri FG, AlQadiri HM, Adwan MH. The Highest Cited Papers in Brucellosis: Identification Using Two Databases and Review of the Papers' Major Findings. BioMed research international. 2018:2018():9291326. doi: 10.1155/2018/9291326. Epub 2018 Apr 11 [PubMed PMID: 29850587]
Bukhari EE. Pediatric brucellosis. An update review for the new millennium. Saudi medical journal. 2018 Apr:39(4):336-341. doi: 10.15537/smj.2018.4.21896. Epub [PubMed PMID: 29619483]
Brown VR, Bowen RA, Bosco-Lauth AM. Zoonotic pathogens from feral swine that pose a significant threat to public health. Transboundary and emerging diseases. 2018 Jun:65(3):649-659. doi: 10.1111/tbed.12820. Epub 2018 Jan 31 [PubMed PMID: 29388363]
Craighead L, Meyer A, Chengat B, Musallam I, Akakpo J, Kone P, Guitian J, Häsler B. Brucellosis in West and Central Africa: A review of the current situation in a changing landscape of dairy cattle systems. Acta tropica. 2018 Mar:179():96-108. doi: 10.1016/j.actatropica.2017.12.026. Epub 2017 Dec 26 [PubMed PMID: 29287761]
Peck D, Bruce M. The economic efficiency and equity of government policies on brucellosis: comparative insights from Albania and the United States of America. Revue scientifique et technique (International Office of Epizootics). 2017 Apr:36(1):291-302. doi: 10.20506/rst.36.1.2629. Epub [PubMed PMID: 28926008]
Level 2 (mid-level) evidenceFallatah SM, Oduloju AJ, Al-Dusari SN, Fakunle YM. Human brucellosis in Northern Saudi Arabia. Saudi medical journal. 2005 Oct:26(10):1562-6 [PubMed PMID: 16228056]
Level 2 (mid-level) evidenceMitka S, Anetakis C, Souliou E, Diza E, Kansouzidou A. Evaluation of different PCR assays for early detection of acute and relapsing brucellosis in humans in comparison with conventional methods. Journal of clinical microbiology. 2007 Apr:45(4):1211-8 [PubMed PMID: 17267626]
Guzmán-Hernández RL, Contreras-Rodríguez A, Ávila-Calderón ED, Morales-García MR. [Brucellosis: a zoonosis of importance in Mexico]. Revista chilena de infectologia : organo oficial de la Sociedad Chilena de Infectologia. 2016 Dec:33(6):656-662. doi: 10.4067/S0716-10182016000600007. Epub [PubMed PMID: 28146190]
Quiroga J, Miralles A, Farinola T, Obi C, Granados J, Fontanillas C, Saura E, Benito M, Calbet JM, Castells E. Surgical treatment of Brucella endocarditis. Cardiovascular surgery (London, England). 1996 Apr:4(2):227-30 [PubMed PMID: 8861443]
Keshtkar-Jahromi M,Razavi SM,Gholamin S,Keshtkar-Jahromi M,Hossain M,Sajadi MM, Medical versus medical and surgical treatment for brucella endocarditis. The Annals of thoracic surgery. 2012 Dec; [PubMed PMID: 23102495]
Zamri-Saad M, Kamarudin MI. Control of animal brucellosis: The Malaysian experience. Asian Pacific journal of tropical medicine. 2016 Dec:9(12):1136-1140. doi: 10.1016/j.apjtm.2016.11.007. Epub 2016 Nov 9 [PubMed PMID: 27955740]
Level 3 (low-level) evidenceMeng F, Pan X, Tong W. Rifampicin versus streptomycin for brucellosis treatment in humans: A meta-analysis of randomized controlled trials. PloS one. 2018:13(2):e0191993. doi: 10.1371/journal.pone.0191993. Epub 2018 Feb 20 [PubMed PMID: 29462155]
Level 1 (high-level) evidenceButtigieg SC, Savic S, Cauchi D, Lautier E, Canali M, Aragrande M. Brucellosis Control in Malta and Serbia: A One Health Evaluation. Frontiers in veterinary science. 2018:5():147. doi: 10.3389/fvets.2018.00147. Epub 2018 Jul 3 [PubMed PMID: 30018972]
Mailles A, Garin-Bastuji B, Lavigne JP, Jay M, Sotto A, Maurin M, Pelloux I, O'Callaghan D, Mick V, Vaillant V, De Valk H. Human brucellosis in France in the 21st century: Results from national surveillance 2004-2013. Medecine et maladies infectieuses. 2016 Dec:46(8):411-418. doi: 10.1016/j.medmal.2016.08.007. Epub 2016 Oct 4 [PubMed PMID: 27717526]
Arenas-Gamboa AM, Rossetti CA, Chaki SP, Garcia-Gonzalez DG, Adams LG, Ficht TA. Human Brucellosis and Adverse Pregnancy Outcomes. Current tropical medicine reports. 2016 Dec:3(4):164-172. doi: 10.1007/s40475-016-0092-0. Epub 2016 Oct 1 [PubMed PMID: 29226068]
Jia B, Zhang F, Lu Y, Zhang W, Li J, Zhang Y, Ding J. The clinical features of 590 patients with brucellosis in Xinjiang, China with the emphasis on the treatment of complications. PLoS neglected tropical diseases. 2017 May:11(5):e0005577. doi: 10.1371/journal.pntd.0005577. Epub 2017 May 1 [PubMed PMID: 28459811]