Introduction
Campylobacter species represent one of the most common causes of bacterial diarrheal illness worldwide. According to the Centers for Disease Control (CDC), there are about 1.3 million cases of Campylobacter infection each year in the United States alone. This leads to an economic cost between $1.3 to $6.8 billion annually in the United States.[1]
Campylobacter infection is associated with consuming raw milk, undercooked poultry, and contaminated water. Patients typically experience a self-limited diarrheal illness lasting 5 to 7 days. Immunocompromised and elderly patients are at the highest risk for morbidity, mortality, and prolonged illness. There has been a dramatic increase in cases in developed and developing regions of the world despite having effective treatment and eradication modalities in animal reservoirs.[1]
The major Campylobacter species considered human pathogens include C. jejuni, C. coli, C. fetus, and C. lari. These species and others can cause bacteremia and systemic illness. C. jejuni infection causes serious bacteremic conditions in patients with acquired immune deficiency syndrome (AIDS). Most other reported cases of bacteremia are from C. fetus infection. Campylobacter hyointestinalis, with biochemical characteristics similar to those of C. fetus, causes occasional bacteremia in immunocompromised patients. Campylobacter upsaliensis can cause diarrhea or bacteremia.
Campylobacter organisms are also a significant cause of traveler's diarrhea, particularly in Thailand and other areas of Southeast Asia.[2] In an observational study conducted on American military personnel deployed in Thailand, over half of those with diarrhea were observed to be infected with Campylobacter species.[3]
Healthy seagulls harbor C. lari, which is reported to cause mild recurrent diarrhea in children.
Etiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Etiology
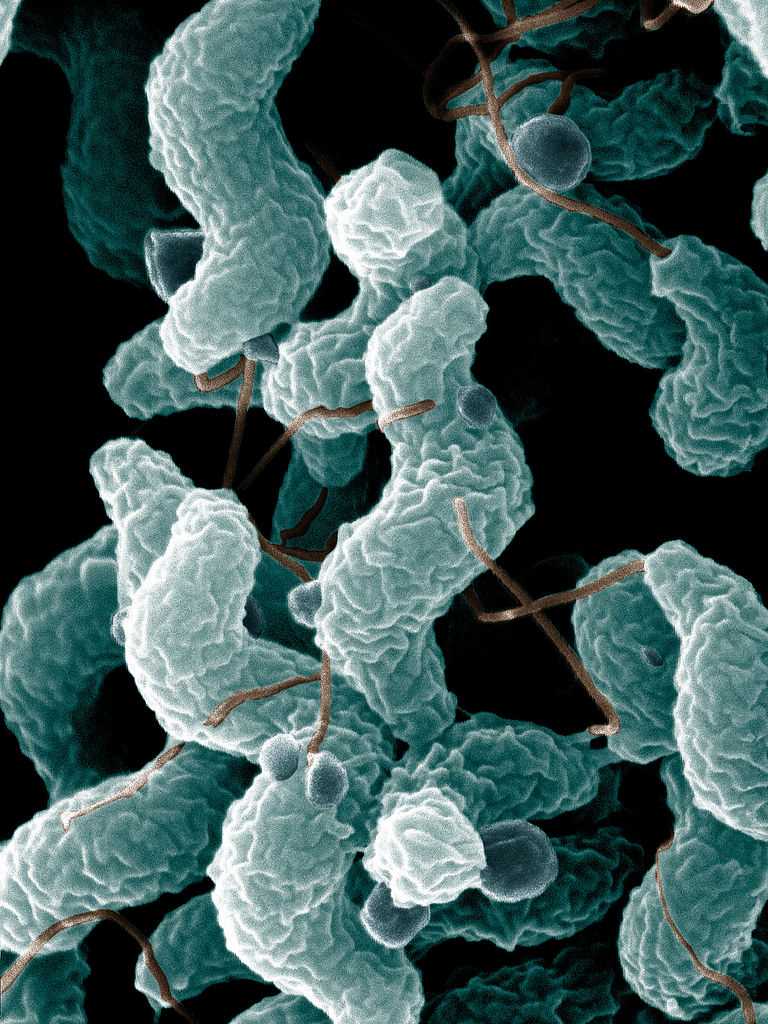
Campylobacter bacteria is a motile, corkscrew-shaped gram-negative rod. These bacteria range in size from 0.5 to 5 microns in length by 0.2 to 0.9 microns in width. There are dozens of species. C. jejuni is the most commonly implicated species in human infections.[1]
Campylobacter is prevalent in food animals, including cattle, poultry, pigs, ostriches, sheep, and pets, such as cats and dogs.[4] Transmission to humans occurs via infected animals and food product consumption. Most human infections result from consuming contaminated or improperly cooked foodstuffs. Chicken accounts for 50% to 70% of Campylobacter infections in humans.
Transmission routes to humans include consumption of contaminated food and water, contact with animals, and person-to-person contact.[5] Exposure to sick pets, particularly puppies, is also associated with Campylobacter outbreaks.[6] Campylobacter has also been discovered in shellfish.[7]
Volunteer-controlled studies have highlighted the relative virulence of Campylobacter. Infection with doses as low as 800 CFU can cause symptoms.[8] This virulence highlights the need for cleanliness in those who come into contact with infection reservoirs such as poultry, contaminated water, and raw dairy.
Epidemiology
Campylobacter is a leading cause of diarrhea in both the developing and developed world and has become increasingly prevalent. In March 2013, the CDC reported a 14% increase in C. jejuni cases in the United States. One percent of the human population in Europe is affected annually by campylobacteriosis. In developing countries, Campylobacter isolation rates for food-borne illnesses are between 5% to 20%.
Campylobacteriosis can occur in all age groups, though it predominantly affects pediatric patients in developing countries. In the developed world, 90% of cases occur during the summer months, which is believed to be related to undercooked meat at outdoor facilities. The condition most commonly affects children younger than 4 years old and individuals between 15 to 44 years of age.[9]
The worldwide prevalence of the disease highlights the ability of Campylobacter to survive in a wide range of environments. With the increasing popularity of international travel of humans and livestock, the spread of Campylobacter is challenging to control. The CDC has made efforts to educate the public about the signs and symptoms of disease and disease prevention for those traveling to countries with a high risk of contact with infected sources. These efforts include hand hygiene, separating raw meats from other food when cooking, thoroughly cooking food, and avoiding raw dairy and untreated water.
Pathophysiology
Symptoms of Campylobacter infection begin to appear after an incubation period of approximately 1 week. The affected body sites include the jejunum and the ileum and can extend to the colon and rectum. C jejuni invades and destroys epithelial cells. It is attracted to mucus; the flagella are essential in chemotaxis and adherence to mucus or epithelial cells. Such adherence promotes gut colonization.
Some strains of C jejuni release a heat-labile, cholera-like enterotoxin that plays an important role in infectious watery diarrhea. Infection with Campylobacter causes diffuse, bloody, exudative, and edematous enteritis. The inflammatory infiltrate contains neutrophils, eosinophils, and mononuclear cells. Crypt abscesses appear in the epithelial glands, and ulcers are seen in the mucosal epithelium.[10]
Patients with bloody diarrhea due to Campylobacter infection have been found to demonstrate cytotoxin production. Rarely the infection is associated with thrombotic thrombocytopenic purpura and hemolytic uremic syndrome through a poorly understood mechanism.[11] Endotoxins and immune complexes cause endothelial cell injury, followed by thrombotic microangiopathy and intravascular coagulation in the glomerulus and the gastrointestinal tract.
History and Physical
Infection from Campylobacter typically manifests as enteritis with voluminous diarrhea. Individuals may experience a prodromal phase of symptoms lasting 1 to 3 days. Symptoms of the prodromal phase include high fever, rigors, dizziness, and body aches. This prodromal phase is associated with a more severe disease course. The onset of symptoms typically occurs 24 to 72 hours following ingestion of the bacteria, though it may take longer to develop in individuals infected with smaller amounts of bacteria. The peak of illness generally lasts 24 to 48 hours.
The average time for the acute diarrheal phase of Campylobacter enteritis is approximately 7 days. It is not uncommon for symptoms of abdominal pain to persist for days to weeks after the resolution of diarrhea.[12] Patients may continue to excrete organisms in the feces for several weeks following clinical improvement, though patients receiving antimicrobial therapy appear to have a reduced likelihood of continued excretion.[13]
During the acute phase of the illness, abdominal cramping and multiple episodes of diarrhea are common. Greater than 10 stools per day may occur. Bloody and mucous-like stools are common and result from the invasion of the epithelium by the bacteria in the intestine. This leads to inflammatory lesions and mucosal damage. Abdominal pain may mimic appendicitis due to acute ileocolitis.[14] Though pathogenesis is poorly understood, the plasmid pVir is associated with more invasive disease and an increased likelihood of bloody diarrhea.[15]
Patients with Campylobacter infection can appear quite ill on examination. Abdominal examination may reveal diffuse tenderness, particularly in the right and left lower quadrants.
Evaluation
Stool culture, enzyme immunoassay (EIA), or polymerase chain reaction (PCR) can establish a diagnosis of Campylobacter infection. For a stool culture, colonies of Campylobacter species are grown using selective culture techniques designed to improve the isolation of C. jejuni and C. coli. Both C. jejuni and C. coli are thermophilic, growing best at 42 °C, and are microaerophilic, growing best in an environment containing 5% to 10% oxygen.[9]
EIA and PCR have shown increased sensitivity as compared to stool culture.[16] Recent developments using reverse transcription-polymerase chain reaction (RT-PCR) have allowed for more sensitive tests to screen potentially infected food sources.[17] Real-time PCR can quickly and accurately detect C. jejuni in the diarrheal stool.[18] Novel techniques, such as immunoenzymatic and molecular biology, are more sensitive than traditional culture.[19][20] Culture-independent tests, such as rapid testing for detecting antigens in stool, are available.[21] However, their utility as stand-alone tests is questionable.[22]
Treatment / Management
Infection with Campylobacter is typically self-limiting and mild. The focus of interventions in healthy patients is hydration and electrolyte repletion. Hydration may be oral or parenteral, depending on the severity of the illness and the degree of dehydration. Anti-motility agents should be avoided as they can impede the resolution of the infection.[23] Antibiotics are not a mainstay of treatment in healthy patients. Antibiotics should be considered for high-risk patients such as immunocompromised and older individuals. Those with more severe disease exhibiting fever, bloody stools, or severe abdominal pain may also warrant antibiotics. Immunocompromised patients may require multiple courses of antibiotics.[24] When antibiotics are indicated, Campylobacter infections are best treated with macrolide antibiotics.[25] This is consistent with multiple patterns of antibiotic resistance that have emerged in recent years.(B2)
Antibiotic use in farm animals is believed to be the source of resistant strains. Campylobacter strains in Canada showed an 8.6% resistance to tetracyclines in 1981, increasing to 56% in more recent studies.[15] A significant resistance pattern to fluoroquinolone therapy has also emerged. While in the early 1990s, there was almost no fluoroquinolone resistance described in the literature, recent data in the United States shows a 40% fluoroquinolone resistance in one area of Pennsylvania.[26] (B3)
While resistance to macrolide antibiotics has remained low at 1% to 3% in the United States, there is a concern about the eventual development of resistant strains.[26] Patients failing antibiotics therapy require drug susceptibility testing. Research suggests that resistant strains emerge from widespread antibiotic use in farm animals. Advocacy for judicious antibiotic usage in large-scale farming environments has increased efforts to control antibiotic resistance.[27](B3)
Differential Diagnosis
The differential diagnosis of Campylobacter infection includes other sources of infectious bacterial diarrhea, such as:
- Salmonella
- Shigella
- Escherichia coli
- Listeria
- Clostridium difficile
- Yersinia Enterocolitica
Viral gastroenteritis, such as norovirus, may also present similarly.[28] Toxin-mediated food poisoning can cause similar symptoms. Parasitic causes of diarrhea, including Giardia lamblia, Cryptosporidium parvum, and Entamoeba histolytica, may result in prolonged diarrhea in travelers and immunocompromised patients mimicking Campylobacter infection.[29]
The abdominal pain from Campylobacter may mimic appendicitis and require advanced imaging such as ultrasound, CT scan, or MRI to differentiate. Inflammatory bowel disease, Crohn disease and ulcerative colitis may also present with abdominal pain and bloody stools. Stool culture, diagnostic imaging, biopsy, and colonoscopy diagnose these chronic conditions.[30]
Prognosis
Generally, Campylobacter infection carries an excellent prognosis. It is almost always self-limiting, with or without specific therapy. The overall mortality for Campylobacter infection is 24 per 10,000 culture-confirmed cases. In healthy patients, Campylobacter infections have a self-limited course with excellent clinical outcomes. These patients require supportive care such as electrolyte repletion and oral and parenteral rehydration. Antibiotics should be considered in immunocompromised patients and those with fever, bloody diarrhea, numerous stools, or worsening symptoms. Approximately 10.8% of patients require hospitalization, making Campylobacter infection the third most common cause of hospitalization for gastroenteritis. Immunocompromised and older patients have the highest risks of prolonged and fatal illness. Most patients experience a complete resolution of illness without complications.[24]
Fewer than 1% of patients experience transient bacteremia, which occurs during the acute infection. Bacteremia is associated with a mortality between 2.5% to 12.5%.[31] Immunocompromised and older patients are more likely to experience bacteremia.[32] HIV/AIDS patients have an increased incidence of infection with Campylobacter and a more virulent course. These patients may require multiple doses of antibiotic therapy. They are more likely to experience extraintestinal symptoms and higher mortality.[33]
Complications
Numerous complications are associated with Campylobacter infection, many of which have a worse prognosis than the acute infection. In addition, Campylobacter infections can cause extraintestinal diseases involving the neurologic, cardiac, pulmonary, and immune systems.
In 1859, the French scientist Jean-Baptiste Octave Landry first reported the disease known today as Guillain-Barre syndrome (GBS). GBS is an autoimmune disease that presents with symmetrical ascending flaccid paralysis due to demyelination of peripheral nerves. However, it was not until the 20th century that a strong association between GBS and Campylobacter was made. Today, it is estimated that Campylobacter, particularly C. jejuni, provokes approximately 30% of all GBS. Approximately 1 in every 1000 people infected with Campylobacter will develop GBS. It occurs 1 to 3 weeks after infection with Campylobacter.
The mechanism of the autoimmune disease is thought to be through molecular mimicry, creating autoantibodies that react with peripheral nerves. The clinical course for those who suffer from GBS varies. Approximately 20% of patients with GBS have a complicated clinical course requiring prolonged intensive care. Despite the advances in the critical care management of GBS, it has a mortality between 3% to 7%.[17]
Case reports have emerged of Campylobacter-associated myocarditis and pericarditis. This is most commonly described in young, healthy males with a history of traveling-associated diarrheal illness. Patients may experience symptoms ranging from mild chest discomfort to severe chest pain that mimics myocardial infarction. Laboratory testing often shows elevated troponin, creatine kinase, brain natriuretic peptide, and C-reactive protein. Increased levels of troponins have been associated with a worse prognosis. However, a normal value does not exclude the diagnosis of myocarditis. Echocardiography is used to identify associated pericardial effusions and wall motion abnormalities associated with the condition. In most patients, the clinical evolution of symptoms and outcomes is benign. Approximately 30% of those affected develop dilated cardiomyopathy. In severe cases, life-threatening arrhythmias and cardiovascular collapse requiring extracorporeal membrane oxygenation (ECMO) have been described.[34]
Approximately 1% to 5% of patients will experience acute reactive arthritis, with an annual incidence of 4.3 per 100,000 cases. Young adults are the most commonly affected group. Both C. jejuni and C. coli are associated with the condition. Symptoms begin within the 4 weeks following acute infection, and the predominant symptom is sterile joint inflammation. Symptoms vary from mild oligoarthralgia to disabling polyarthritis. Musculoskeletal symptoms are the most common presentation, though patients can also have ocular, dermatological, and urinary symptoms. While symptoms commonly affect the knees and ankles, small joints and tendon involvement are also seen. The duration of symptoms in acute reactive arthritis is variable. Patients may experience symptoms lasting several months to 1 year. There is no association with HLA B27 on a population level.[35][36]
In the gastrointestinal (GI) system, there has been an increased association of esophageal and colorectal diseases with Campylobacter infections. In the acute phase of the disease, severe infections can manifest as colitis, leading to significant morbidity, occasionally requiring surgical intervention. Toxic megacolon is a rare complication of Campylobacter colitis and should be considered in critically ill patients. Patients that develop toxic megacolon have high morbidity and mortality despite interventions.[37]
Multiple other gastroenterological manifestations are associated with Campylobacter, including gastroesophageal reflux disease (GERD), Barrett esophagus (BE), and esophageal adenocarcinoma. Research has also shown an increased risk for irritable bowel disease and colorectal carcinoma following Campylobacter infections.[38]
A form of lymphoma, immunoproliferative small intestinal disease, has shown an association with Campylobacter infections. Biopsy specimens of several patients noted the presence of C. jejuni, and antimicrobial therapy targeting this organism led to the rapid remission of the immunoproliferative small intestinal disease.[39]
Deterrence and Patient Education
The most commonly reported sources of Campylobacter infections are under-cooked poultry in the developed world and contaminated water in the developing world. Ensuring poultry and meat are thoroughly cooked at 70 °C before ingestion decreases the risk of infection. The CDC recommends washing items that come into contact with raw poultry. They also recommend separate cutting boards for raw meat and cleaning utensils after coming in contact with raw meat.[1]
Unpasteurized milk and cheese should be avoided. Untreated water should not be consumed. Practicing basic hand hygiene also decreases the risk of infection.[40]
Pearls and Other Issues
Key facts to keep in mind regarding Campylobacter infection are as follows:
- Campylobacter is a leading cause of foodborne illness worldwide. C jejuni is identified as the most common culprit in human infections.
- Campylobacter is a robust species of bacteria that can survive over a wide range of environments. Small numbers of bacteria can cause infection.
- Those at most significant risk for infection include international travelers, people who come in frequent contact with livestock, and consumers of untreated water or unpasteurized milk.
- Clinically, Campylobacter infection typically manifests as diarrhea (sometimes bloody) that typically peaks 1 to 2 days following onset. The acute enteritis phase of the infection is typically self-limiting and resolves after 7 days.
- Stool culture, PCR, or EIA establish the diagnosis.
- Campylobacter is typically self-limiting and, in some cases, requires supportive fluid and electrolyte replenishment. When antibiotics are needed, macrolides such as azithromycin are the treatment of choice.
- Multiple complications are related to Campylobacter infection; most notably, there is an association between infection and Guillain-Barre syndrome.[41]
Enhancing Healthcare Team Outcomes
Recent outbreaks of Campylobacter underline the importance of epidemiologic reporting, preventative measures, and interprofessional team collaboration. In 2018, there was a multi-state outbreak of Campylobacter with 113 laboratory-confirmed cases spanning over 17 states. This outbreak led to 23 hospitalizations with no reported deaths. During interviews with the patients, those affected answered a survey of questions regarding recent contact with animals and food. Questionnaires revealed 87% of those affected reported contact with a puppy from the same store branch. Biologists isolated genetic sequences that proved the samples were related using a gene sequencing method called whole genome sequencing.[42] This established a common source. Isolates were tested for antibiotic sensitivities, and a resistance pattern was identified. The infection was contained by identifying the pathogen, tracing its source, and providing treatment recommendations.
Healthcare professionals need clinical expertise in diagnosing and treating Campylobacter infections, including proficiency in interpreting diagnostic tests and assessing patient presentations. Competence in evidence-based practices and the ability to adapt to emerging trends in Campylobacter epidemiology are crucial. Healthcare professionals are responsible for providing patient-centered care, emphasizing timely and accurate diagnosis, appropriate treatment, and ongoing monitoring. This includes addressing the unique challenges Campylobacter infections pose in immunocompromised and high-risk populations.
Ensuring patient safety involves minimizing the risk of Campylobacter-related complications, adverse reactions to treatments, and preventing the spread of infections. Adherence to infection control protocols and vigilant monitoring contribute to a safer healthcare environment. Continuous monitoring and evaluation of patient outcomes related to Campylobacter infections are essential. This includes assessing the effectiveness of interventions, identifying areas for improvement, and implementing changes to optimize care delivery and enhance overall patient outcomes.
Campylobacter infection must be prevented by practicing proper hygiene and food handling. The role of the interprofessional team in managing patients with Campylobacter is critical. This team should involve primary care providers, community nurses, preventative medicine, and gastroenterologists if indicated. If a patient develops complications, an appropriate specialist should be consulted, such as a neurologist in the case of GBS.
Coordinated efforts among healthcare team members are crucial for comprehensive Campylobacter care. This involves seamless collaboration in planning, implementing, and monitoring treatment, as well as facilitating patient education and follow-up care. Developing and implementing effective strategies for Campylobacter prevention, management, and patient education is essential. This includes creating protocols for early detection, treatment algorithms, and public health initiatives to reduce the overall burden of Campylobacter infections.
Health professionals must work collaboratively to enhance team performance. Regular training, ongoing education, and fostering a culture of mutual respect and communication contribute to a high-functioning, cohesive healthcare team focused on providing optimal care for patients with Campylobacter infections.
Media
(Click Image to Enlarge)
References
Kaakoush NO, Castaño-Rodríguez N, Mitchell HM, Man SM. Global Epidemiology of Campylobacter Infection. Clinical microbiology reviews. 2015 Jul:28(3):687-720. doi: 10.1128/CMR.00006-15. Epub [PubMed PMID: 26062576]
Serichantalergs O, Ruekit S, Pandey P, Anuras S, Mason C, Bodhidatta L, Swierczewski B. Incidence of Campylobacter concisus and C. ureolyticus in traveler's diarrhea cases and asymptomatic controls in Nepal and Thailand. Gut pathogens. 2017:9():47. doi: 10.1186/s13099-017-0197-6. Epub 2017 Aug 17 [PubMed PMID: 28824712]
Level 3 (low-level) evidenceSanders JW, Isenbarger DW, Walz SE, Pang LW, Scott DA, Tamminga C, Oyofo BA, Hewitson WC, Sanchez JL, Pitarangsi C, Echeverria P, Tribble DR. An observational clinic-based study of diarrheal illness in deployed United States military personnel in Thailand: presentation and outcome of Campylobacter infection. The American journal of tropical medicine and hygiene. 2002 Nov:67(5):533-8 [PubMed PMID: 12479558]
Newell DG, Fearnley C. Sources of Campylobacter colonization in broiler chickens. Applied and environmental microbiology. 2003 Aug:69(8):4343-51 [PubMed PMID: 12902214]
Level 3 (low-level) evidenceSibanda N, McKenna A, Richmond A, Ricke SC, Callaway T, Stratakos AC, Gundogdu O, Corcionivoschi N. A Review of the Effect of Management Practices on Campylobacter Prevalence in Poultry Farms. Frontiers in microbiology. 2018:9():2002. doi: 10.3389/fmicb.2018.02002. Epub 2018 Aug 24 [PubMed PMID: 30197638]
Kuhn KG, Hvass AK, Christiansen AH, Ethelberg S, Cowan SA. Sexual Contact as Risk Factor for Campylobacter Infection, Denmark. Emerging infectious diseases. 2021 Apr:27(4):1133-1140. doi: 10.3201/eid2704.202337. Epub [PubMed PMID: 33754996]
Rincé A, Balière C, Hervio-Heath D, Cozien J, Lozach S, Parnaudeau S, Le Guyader FS, Le Hello S, Giard JC, Sauvageot N, Benachour A, Strubbia S, Gourmelon M. Occurrence of Bacterial Pathogens and Human Noroviruses in Shellfish-Harvesting Areas and Their Catchments in France. Frontiers in microbiology. 2018:9():2443. doi: 10.3389/fmicb.2018.02443. Epub 2018 Oct 11 [PubMed PMID: 30364306]
Black RE, Levine MM, Clements ML, Hughes TP, Blaser MJ. Experimental Campylobacter jejuni infection in humans. The Journal of infectious diseases. 1988 Mar:157(3):472-9 [PubMed PMID: 3343522]
Epps SV, Harvey RB, Hume ME, Phillips TD, Anderson RC, Nisbet DJ. Foodborne Campylobacter: infections, metabolism, pathogenesis and reservoirs. International journal of environmental research and public health. 2013 Nov 26:10(12):6292-304. doi: 10.3390/ijerph10126292. Epub 2013 Nov 26 [PubMed PMID: 24287853]
Murphy H, Cogan T, Humphrey T. Direction of neutrophil movements by Campylobacter-infected intestinal epithelium. Microbes and infection. 2011 Jan:13(1):42-8. doi: 10.1016/j.micinf.2010.09.007. Epub 2010 Oct 8 [PubMed PMID: 20934530]
Hadden RD, Karch H, Hartung HP, Zielasek J, Weissbrich B, Schubert J, Weishaupt A, Cornblath DR, Swan AV, Hughes RA, Toyka KV, Plasma Exchange/Sandoglobulin Guillain-Barré Syndrome Trial Group. Preceding infections, immune factors, and outcome in Guillain-Barré syndrome. Neurology. 2001 Mar 27:56(6):758-65 [PubMed PMID: 11274311]
Level 1 (high-level) evidenceSmith KE, Besser JM, Hedberg CW, Leano FT, Bender JB, Wicklund JH, Johnson BP, Moore KA, Osterholm MT. Quinolone-resistant Campylobacter jejuni infections in Minnesota, 1992-1998. Investigation Team. The New England journal of medicine. 1999 May 20:340(20):1525-32 [PubMed PMID: 10332013]
Level 3 (low-level) evidenceKapperud G, Lassen J, Ostroff SM, Aasen S. Clinical features of sporadic Campylobacter infections in Norway. Scandinavian journal of infectious diseases. 1992:24(6):741-9 [PubMed PMID: 1287808]
Level 2 (mid-level) evidenceBlaser MJ. Epidemiologic and clinical features of Campylobacter jejuni infections. The Journal of infectious diseases. 1997 Dec:176 Suppl 2():S103-5 [PubMed PMID: 9396691]
Level 3 (low-level) evidenceTracz DM, Keelan M, Ahmed-Bentley J, Gibreel A, Kowalewska-Grochowska K, Taylor DE. pVir and bloody diarrhea in Campylobacter jejuni enteritis. Emerging infectious diseases. 2005 Jun:11(6):838-43 [PubMed PMID: 15963277]
Platts-Mills JA, Liu J, Gratz J, Mduma E, Amour C, Swai N, Taniuchi M, Begum S, Peñataro Yori P, Tilley DH, Lee G, Shen Z, Whary MT, Fox JG, McGrath M, Kosek M, Haque R, Houpt ER. Detection of Campylobacter in stool and determination of significance by culture, enzyme immunoassay, and PCR in developing countries. Journal of clinical microbiology. 2014 Apr:52(4):1074-80. doi: 10.1128/JCM.02935-13. Epub 2014 Jan 22 [PubMed PMID: 24452175]
Bonilauri P, Bardasi L, Leonelli R, Ramini M, Luppi A, Giacometti F, Merialdi G. Detection of Food Hazards in Foods: Comparison of Real Time Polymerase Chain Reaction and Cultural Methods. Italian journal of food safety. 2016 Jan 18:5(1):5641 [PubMed PMID: 27800434]
Lin S, Wang X, Zheng H, Mao Z, Sun Y, Jiang B. Direct detection of Campylobacter jejuni in human stool samples by real-time PCR. Canadian journal of microbiology. 2008 Sep:54(9):742-7. doi: 10.1139/w08-064. Epub [PubMed PMID: 18772937]
Bessède E, Delcamp A, Sifré E, Buissonnière A, Mégraud F. New methods for detection of campylobacters in stool samples in comparison to culture. Journal of clinical microbiology. 2011 Mar:49(3):941-4. doi: 10.1128/JCM.01489-10. Epub 2011 Jan 5 [PubMed PMID: 21209172]
Buss JE, Cresse M, Doyle S, Buchan BW, Craft DW, Young S. Campylobacter culture fails to correctly detect Campylobacter in 30% of positive patient stool specimens compared to non-cultural methods. European journal of clinical microbiology & infectious diseases : official publication of the European Society of Clinical Microbiology. 2019 Jun:38(6):1087-1093. doi: 10.1007/s10096-019-03499-x. Epub 2019 Feb 19 [PubMed PMID: 30783889]
Couturier BA, Couturier MR, Kalp KJ, Fisher MA. Detection of non-jejuni and -coli Campylobacter species from stool specimens with an immunochromatographic antigen detection assay. Journal of clinical microbiology. 2013 Jun:51(6):1935-7. doi: 10.1128/JCM.03208-12. Epub 2013 Apr 3 [PubMed PMID: 23554192]
Fitzgerald C, Patrick M, Gonzalez A, Akin J, Polage CR, Wymore K, Gillim-Ross L, Xavier K, Sadlowski J, Monahan J, Hurd S, Dahlberg S, Jerris R, Watson R, Santovenia M, Mitchell D, Harrison C, Tobin-D'Angelo M, DeMartino M, Pentella M, Razeq J, Leonard C, Jung C, Achong-Bowe R, Evans Y, Jain D, Juni B, Leano F, Robinson T, Smith K, Gittelman RM, Garrigan C, Nachamkin I. Multicenter Evaluation of Clinical Diagnostic Methods for Detection and Isolation of Campylobacter spp. from Stool. Journal of clinical microbiology. 2016 May:54(5):1209-15. doi: 10.1128/JCM.01925-15. Epub 2016 Mar 9 [PubMed PMID: 26962088]
Nolan CM, Johnson KE, Coyle MB, Faler K. Campylobacter jejuni enteritis: efficacy of antimicrobial and antimotility drugs. The American journal of gastroenterology. 1983 Oct:78(10):621-6 [PubMed PMID: 6624735]
Level 2 (mid-level) evidenceRuiz-Palacios GM. The health burden of Campylobacter infection and the impact of antimicrobial resistance: playing chicken. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2007 Mar 1:44(5):701-3 [PubMed PMID: 17278063]
Luangtongkum T, Jeon B, Han J, Plummer P, Logue CM, Zhang Q. Antibiotic resistance in Campylobacter: emergence, transmission and persistence. Future microbiology. 2009 Mar:4(2):189-200. doi: 10.2217/17460913.4.2.189. Epub [PubMed PMID: 19257846]
Level 3 (low-level) evidenceGupta A, Nelson JM, Barrett TJ, Tauxe RV, Rossiter SP, Friedman CR, Joyce KW, Smith KE, Jones TF, Hawkins MA, Shiferaw B, Beebe JL, Vugia DJ, Rabatsky-Ehr T, Benson JA, Root TP, Angulo FJ, NARMS Working Group. Antimicrobial resistance among Campylobacter strains, United States, 1997-2001. Emerging infectious diseases. 2004 Jun:10(6):1102-9 [PubMed PMID: 15207064]
Level 3 (low-level) evidenceAllos BM. Campylobacter jejuni Infections: update on emerging issues and trends. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2001 Apr 15:32(8):1201-6 [PubMed PMID: 11283810]
Level 3 (low-level) evidenceRohner P, Pittet D, Pepey B, Nije-Kinge T, Auckenthaler R. Etiological agents of infectious diarrhea: implications for requests for microbial culture. Journal of clinical microbiology. 1997 Jun:35(6):1427-32 [PubMed PMID: 9163457]
Level 2 (mid-level) evidenceSlack A. Parasitic causes of prolonged diarrhoea in travellers - diagnosis and management. Australian family physician. 2012 Oct:41(10):782-6 [PubMed PMID: 23210100]
Mee AS, Shield M, Burke M. Campylobacter colitis: differentiation from acute inflammatory bowel disease. Journal of the Royal Society of Medicine. 1985 Mar:78(3):217-23 [PubMed PMID: 3973886]
Nielsen H, Hansen KK, Gradel KO, Kristensen B, Ejlertsen T, Østergaard C, Schønheyder HC. Bacteraemia as a result of Campylobacter species: a population-based study of epidemiology and clinical risk factors. Clinical microbiology and infection : the official publication of the European Society of Clinical Microbiology and Infectious Diseases. 2010 Jan:16(1):57-61. doi: 10.1111/j.1469-0691.2009.02900.x. Epub [PubMed PMID: 19673969]
Level 2 (mid-level) evidencePacanowski J, Lalande V, Lacombe K, Boudraa C, Lesprit P, Legrand P, Trystram D, Kassis N, Arlet G, Mainardi JL, Doucet-Populaire F, Girard PM, Meynard JL, CAMPYL Study Group. Campylobacter bacteremia: clinical features and factors associated with fatal outcome. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2008 Sep 15:47(6):790-6. doi: 10.1086/591530. Epub [PubMed PMID: 18699745]
Tee W, Mijch A. Campylobacter jejuni bacteremia in human immunodeficiency virus (HIV)-infected and non-HIV-infected patients: comparison of clinical features and review. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 1998 Jan:26(1):91-6 [PubMed PMID: 9455515]
Inayat F, Ali NS, Riaz I, Virk HUH. From the Gut to the Heart: Campylobacter jejuni Enteritis Leading to Myopericarditis. Cureus. 2017 Jun 9:9(6):e1326. doi: 10.7759/cureus.1326. Epub 2017 Jun 9 [PubMed PMID: 28690959]
Pope JE, Krizova A, Garg AX, Thiessen-Philbrook H, Ouimet JM. Campylobacter reactive arthritis: a systematic review. Seminars in arthritis and rheumatism. 2007 Aug:37(1):48-55 [PubMed PMID: 17360026]
Level 1 (high-level) evidenceHannu T, Mattila L, Rautelin H, Pelkonen P, Lahdenne P, Siitonen A, Leirisalo-Repo M. Campylobacter-triggered reactive arthritis: a population-based study. Rheumatology (Oxford, England). 2002 Mar:41(3):312-8 [PubMed PMID: 11934969]
Kwok M, Maurice A, Lisec C, Brown J. Campylobacter colitis: Rare cause of toxic megacolon. International journal of surgery case reports. 2016:27():141-143. doi: 10.1016/j.ijscr.2016.08.030. Epub 2016 Aug 25 [PubMed PMID: 27614338]
Level 3 (low-level) evidenceMacfarlane S, Furrie E, Macfarlane GT, Dillon JF. Microbial colonization of the upper gastrointestinal tract in patients with Barrett's esophagus. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2007 Jul 1:45(1):29-38 [PubMed PMID: 17554697]
Lecuit M, Abachin E, Martin A, Poyart C, Pochart P, Suarez F, Bengoufa D, Feuillard J, Lavergne A, Gordon JI, Berche P, Guillevin L, Lortholary O. Immunoproliferative small intestinal disease associated with Campylobacter jejuni. The New England journal of medicine. 2004 Jan 15:350(3):239-48 [PubMed PMID: 14724303]
Level 3 (low-level) evidenceFacciolà A, Riso R, Avventuroso E, Visalli G, Delia SA, Laganà P. Campylobacter: from microbiology to prevention. Journal of preventive medicine and hygiene. 2017 Jun:58(2):E79-E92 [PubMed PMID: 28900347]
Nyati KK, Nyati R. Role of Campylobacter jejuni infection in the pathogenesis of Guillain-Barré syndrome: an update. BioMed research international. 2013:2013():852195. doi: 10.1155/2013/852195. Epub 2013 Aug 13 [PubMed PMID: 24000328]
Level 3 (low-level) evidenceLindsey RL, Gladney LM, Huang AD, Griswold T, Katz LS, Dinsmore BA, Im MS, Kucerova Z, Smith PA, Lane C, Carleton HA. Rapid identification of enteric bacteria from whole genome sequences using average nucleotide identity metrics. Frontiers in microbiology. 2023:14():1225207. doi: 10.3389/fmicb.2023.1225207. Epub 2023 Dec 14 [PubMed PMID: 38156000]