Introduction
The auditory system processes how we hear and understand sounds within the environment. Peripheral and central structures comprise this organ system. The outer, middle, and inner ear are the peripheral auditory structures. Central auditory structures include the cochlear nuclei, superior olivary nuclei, lateral lemniscus, inferior colliculus, medial geniculate nuclei, and auditory cortex (see Image. Peripheral and Central Structures of the Auditory System).
Central auditory pathways encode sound frequency, attenuation, and spatial location, combinations of which help individuals understand and correctly interpret sounds. Central auditory information processing adapts continuously to environmental, attentional, and perceived importance of environmental cues. This chapter provides a basic description of audition and auditory processing.
Structure and Function
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Structure and Function
Peripheral Auditory System: How Sound Reaches the Brain
Sounds are produced by energy waves traveling through a medium, causing particle motion in that medium. The movement of particles leads to air pressure fluctuations—that is, alternating compression and rarefaction—nearby. Sound frequency is the number of compression-rarefaction cycle repetitions within a given time interval, measured in Hertz (Hz) or number of cycles per second. Humans typically hear within the 20-20,000 Hz frequency range.
Sound waves reach the outer ear and travel down the external acoustic meatus to reach the tympanic membrane, aka the eardrum. Contact with sound waves causes tympanic membrane movement, initiating vibrations of the middle ear's small bones (ossicles), the malleus, incus, and stapes. The 3 ossicles convey the tympanic membrane movements to the oval (vestibular) window. These 3 middle ear bones also serve as sound amplifiers and impedance-matching tools when sound moves from the air-filled middle ear to the fluid (perilymph) in contact with the oval window (see Image. Auditory Ossicles). The oval window is the gateway to the inner ear's vestibule.
The vestibule is a compartment connected to the cochlea on one side and the semicircular canals on the other (see Image. Inner Ear Anatomy). The cochlea spirals 2½ turns around a central pillar, the modiolus. Perilymph is the fluid inside the cochlea.
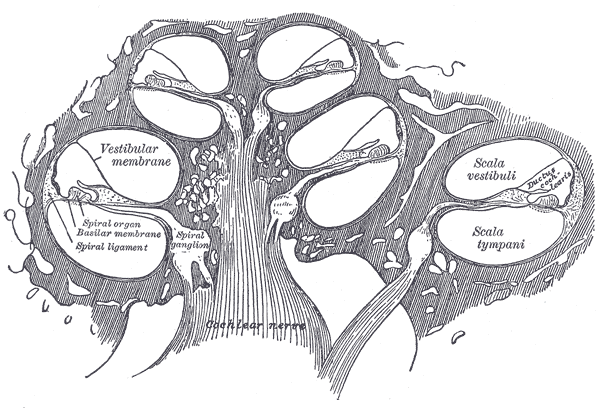
The cochlea has 3 chambers: the scala tympani, scala vestibuli, and scala media (see Image. Diagrammatic Longitudinal Section of the Cochlea). The scala tympani occupies the cochlea's outermost portion. The scala vestibuli lies in the innermost region. The scala media, aka cochlear duct, separates the two other chambers as they turn, except at the helicotrema, where the scala tympani connects with the scala vestibuli. Perilymph fills the scala tympani and scala vestibuli. The scala media contains endolymph, the basilar membrane, and the organ of Corti.
Oscillations of the oval window produce fluid waves, which pass through the scala vestibuli. The pressure changes inside the scala vestibuli transmit energy to the basilar membrane at the floor of the scala media.
The Organ of Corti lies on the basilar membrane inside the scala media (see Image. Organ of Corti). It houses mechanical receptor cells: 3 rows of outer hair cells and 1 row of inner hair cells. The base of the hair cells is embedded in the basilar membrane. At the apex of each cell, stereocilia connect to a second membrane, the tectorial membrane. Stereocilia have potassium channels on their surfaces.
The basilar membrane shifts when the scala vestibuli and scala tympani oscillate. Basilar membrane motions bend the stereocilia against the tectorial membrane. The stereocilia's potassium channels open or close depending on the bending direction. Hair cell depolarization sends an impulse toward the auditory nerve. Sound energy is thus converted to electrical energy and nerve signals within the inner ear.
The tectorial and basilar membranes' location within the cochlea influences their response to different sound pitches or frequencies. The membranes are stiffer, and stereocilia are shorter at the region close to the oval window, the cochlear base. This region responds best to high-pitched or high-frequency sounds. The membranes become more flexible and stereocilia longer, farther from the oval window, and closer to the cochlear apex. Stereocilia at the cochlear apex is more than twice as long as those in the cochlear base, making them more responsive to low-frequency sounds.[1][2] This arrangement is called "tonotopic gradient," which the brain uses to map out different sound frequencies.
Spiral ganglion peripheral axons, which comprise part of the auditory nerve, synapse at the bases of the hair cell bodies. Most (90%) auditory nerve fibers receive input from the inner hair cells. [2] Thus, the inner hair cells process most auditory stimuli. The remaining 10% comes from the outer hair cells.
The auditory cortex relies on spiral ganglion neurons to protect the inner ear from loud sounds. Stimulation of the spiral ganglion neurons contracts their cell bodies, increasing basilar membrane tension and dampening hair cell excitation.[3][3] When loud sounds are heard, the brain stimulates the spiral ganglion neurons to reduce the hair cells' response. Transient loss of hearing may develop afterward as the descending auditory pathways and peripheral auditory organs re-adjust to a quieter environment.
Central Auditory System
Peripheral auditory impulses reach the auditory cortex nuclei via the auditory nerve. From the auditory nerve, sensory information ascends along the following path: [4]
- Ipsilateral auditory nuclei
- Ipsilateral cochlear nucleus
- Superior olivary complex—most fibers cross over to the contralateral superior olivary complex; some remain on the ipsilateral superior olivary complex
- Contralateral and ipsilateral lateral lemniscus nuclei
- Contralateral and ipsilateral inferior colliculus
- Contralateral and ipsilateral medial geniculate nuclei
- Contralateral and ipsilateral transverse temporal gyrus (auditory region)
Lower levels of the central auditory pathways are "monaural" because they receive sensory inputs only from the ipsilateral ear. From the superior olivary complex upward, auditory pathway nuclei become "binaural," receiving inputs from peripheral auditory structures on both sides of the head. The binaural arrangement allows for more accurate localization of auditory stimuli (see Image. Auditory Pathways).
Types of Processing
Some of the most critical sound attributes that the central auditory pathways process are the following:
- Intensity
- Attenuation
- Spatial location
- Frequency
- Combination sensitivity
Auditory nuclei have a tonotopic arrangement, helping to map out these sound attributes in the environment.[4]
Intensity and attenuation are processed by neurons firing at different rates depending on sound intensity. Increasing sound also increases the neurons' firing rates. More specialized neurons respond maximally to environmental sounds within specific intensity ranges. [4]
The brain relies on the binaural auditory pathways to locate a sound's origin accurately. For example, a sound coming directly from the midline, ie, front or back of the head, would reach both ears simultaneously. Sensory inputs from both ears would also reach the superior olivary complex simultaneously. In contrast, sound coming from one side causes a temporal delay of sensory inputs from the contralateral ear to the superior olivary complex.[4]
Combination-sensitive neurons are a subset of auditory neurons that respond variably to different acoustic elements. Combination-sensitive neurons may be facilitatory or inhibitory and are located within the inferior colliculus, lateral lemniscus, medial geniculate, and auditory cortex.[5][6][7][8][9] These neurons are thought to help identify important sound cues necessary for communication.[10]
Descending Circuits
It was once thought that audition involved relaying sound information from the environment to the auditory cortex. However, we now know about the descending auditory pathways and their modulatory role in sound stimulus processing. The descending auditory pathways start from the auditory cortex, moving downward to its direct projections to the inferior colliculus, superior olivary complex, and cochlear nucleus.[11][12][13][14][15] Modulatory signals attune responses depending on learned behaviors, emotional state, and if attention and relevance are given to the stimulus. Such higher-order functions originate from many regions of the brain—eg, prefrontal cortex, hippocampus, nucleus basalis of Meynert, and limbic circuits—that have either direct or indirect connections with each other and the auditory cortex.[16][17][18]
Embryology
The cochlea starts to develop from the surface ectoderm at gestational day 4, beginning as the otic vesicle that later turns into the inner ear's membranous labyrinth. The dorsal aspect of the labyrinth develops into the utricle and semicircular ducts, while the ventral aspect transforms into the cochlea and saccule.
The central auditory pathway components arise from different parts of the brain:
- The auditory cortex from the telencephalon
- The medial geniculate from the diencephalon
- The inferior colliculus from the mesencephalon
- The cochlear and superior olivary nuclei from the rhombencephalon
Though highly plastic and constantly changing throughout life, these regions are fully functioning at birth.[19]
Blood Supply and Lymphatics
Blood Supply
External Ear
- Posterior auricular branch of the external carotid artery
Middle Ear
- Mastoid branches from the posterior auricular arteries
- Occipital arteries
- Deep auricular arteries
Inner Ear
- Anterior tympanic branch of the maxillary artery
- Stylomastoid branch of the posterior auricular artery
- Petrosal branch of the middle meningeal artery
- Labyrinthine artery (branch of the basilar or anterior inferior cerebellar artery) (See Image. Blood Supply to the Brain, Meninges, and Other Structures in the Skull) [20]
Lymphatics
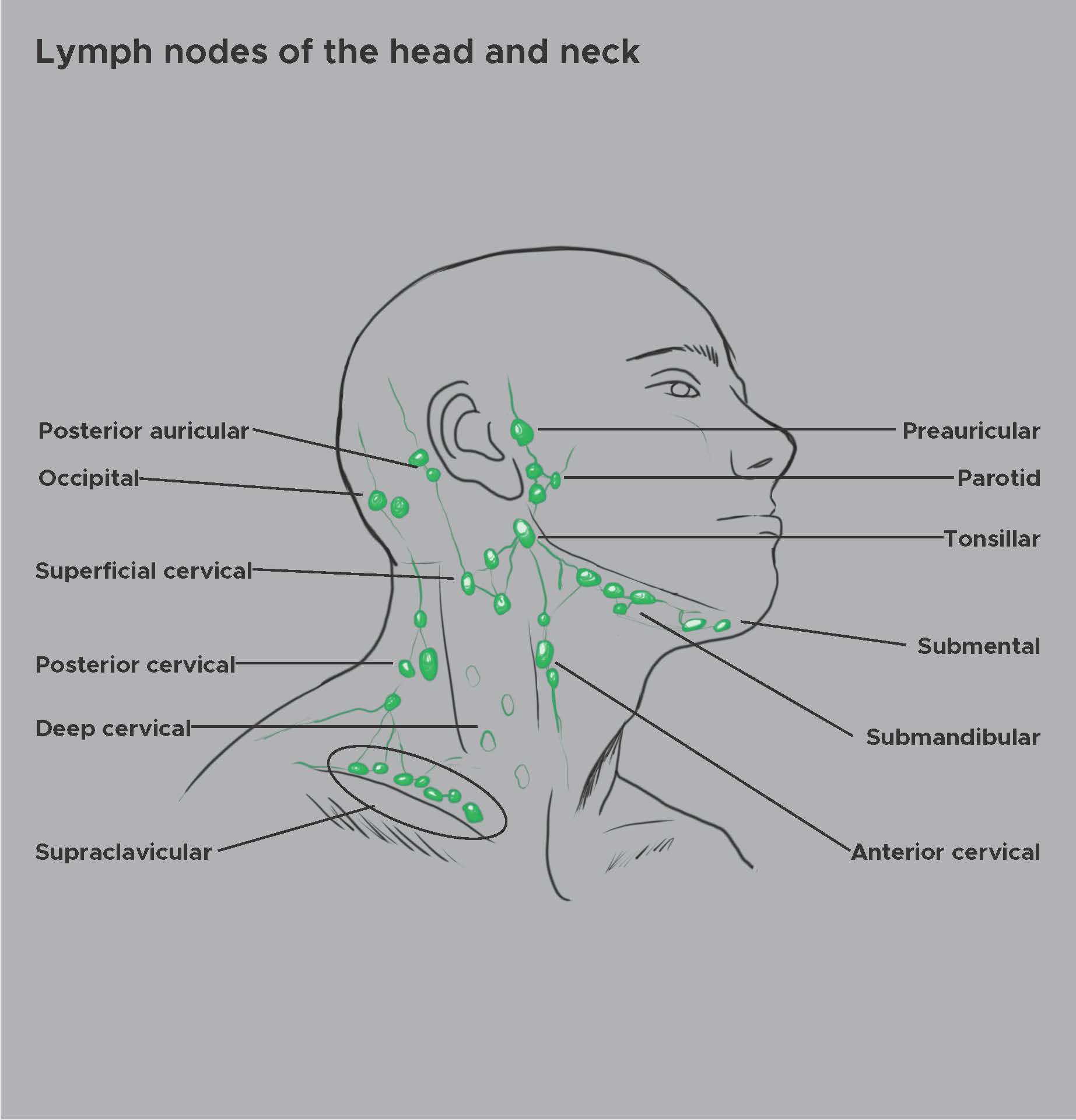
External Ear: Pre-auricular lymph nodes [21]
Middle Ear: Retroauricular and junctional lymph nodes [22]
Inner Ear: Whether or not the inner ear drains via a normal lymphatic system is unclear. Salt and Hirose proposed that the inner ear drained diffusely through the perilymph and bone.[23] (See Image. Lymph Nodes of the Head and Neck)
Muscles
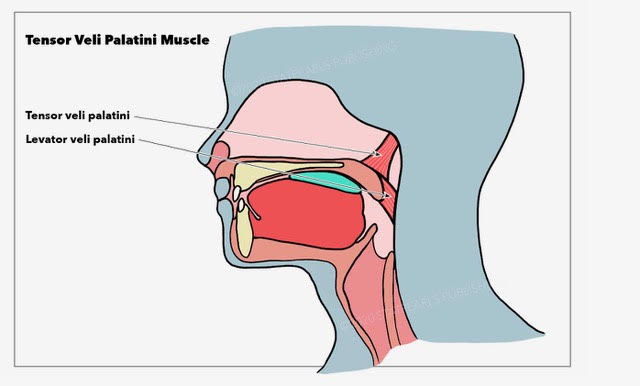
Muscles around and inside the ears influence audition. The levator veli palatini and tensor veli palatini assist in opening the auditory tube (aka eustachian or pharyngotympanic tube), helping to regulate air pressure in the middle ear (see Image. Muscles Around the Pharyngotympanic Tube).
The stapedius originates from the pyramidal eminence of the temporal bone and inserts on the neck of the stapes bone. The stapedius limits the stapes' excessive vibration and subsequent cochlear stimulation, especially on exposure to loud sounds (see Image. Schematic Illustration of the Middle Ear).
Physiologic Variants
Cauliflower Ear
Repeated trauma can cause outer ear abnormalities. Trauma can induce auricular hematoma, wherein blood accumulates between the perichondrium and auricular cartilage. Over time, the outer ear can become fibrotic and distorted. Blood flow to the auricular cartilage may also be impaired.[24]
Otitis Media
Otitis media is an inflammation of the middle ear lining. The condition is more common in children as they have horizontally oriented auditory tubes that limit fluid drainage. Otitis media becomes rarer in adulthood when the auditory tubes become more laterally and caudally oriented. Middle ear inflammation causes swelling and increased pressure against the tympanic membrane. In severe cases, the tympanic membrane can rupture and reduce auditory acuity.[25]
Surgical Considerations
Recurrent otitis media can be treated with drainage tube insertion. Tympanoplasty can repair damaged ossicles and tympanic membrane perforation and scars.
Clinical Significance
Acoustic Neuroma
The auditory nerve, which lies at the junction between the pons, medulla, and cerebellum, is the most common location of a slow-growing tumor called "acoustic neuroma."(see Image. Peripheral and Central Structures of the Auditory System) [26] Tumor enlargement can put pressure on the surrounding structures, notably cranial nerves VIII, VII, and IX, the cerebellum, and the brainstem. Hearing loss is usually the initial symptom. As the tumor grows, other nerve deficits may also manifest, with some ipsilateral to the tumor. Examples are the following:[27]
- Tinnitus
- Vertigo
- Nystagmus
- Facial drooping
- Decreased corneal reflex
- Hoarseness
- Dysphagia
- Ataxia
- Dysarthria
Tinnitus
Tinnitus is the perception of a sound not coming from the environment, typically described as a ringing or buzzing in the ear. The perception is induced by hyper-excitation within a specific region of the auditory cortex.[28][29] Patients with tinnitus may complain of inability to differentiate sounds or conversations in noisy environments. Severe tinnitus can disrupt sleep and affect an individual's social and emotional state.[29][30]
Tinnitus typically presents gradually after several years of repeated exposure to loud noises. Phantom sound perception occurs when, despite inner ear damage, the auditory cortex is activated due to false signals coming from interconnected auditory nuclei.
Tinnitus has no cure. Biofeedback therapy may help reduce but not eliminate the symptom. Biofeedback therapy uses one or more sounds at or near the patient's perceived frequency. Inhibitory limbic circuits help suppress aberrant cortical excitation.[29] Playing white noise in the background at bedtime can help individuals with tinnitus-induced sleep difficulties sleep better.[31]
Other Issues
Most people respond well to a typical hearing test. However, factors like undeveloped speech, disease, and trauma can affect one's ability to provide feedback to the examiner. The auditory brainstem response (ABR) is a more universal approach to auditory testing as it does not require patient feedback. ABR records the electrical activity changes in the brainstem's auditory areas in response to auditory cues. Deviations from normal values are evidence of auditory dysfunction. The test also assesses multiple auditory nuclei, allowing physicians to identify areas of trauma or disease.[32]
Media
(Click Image to Enlarge)
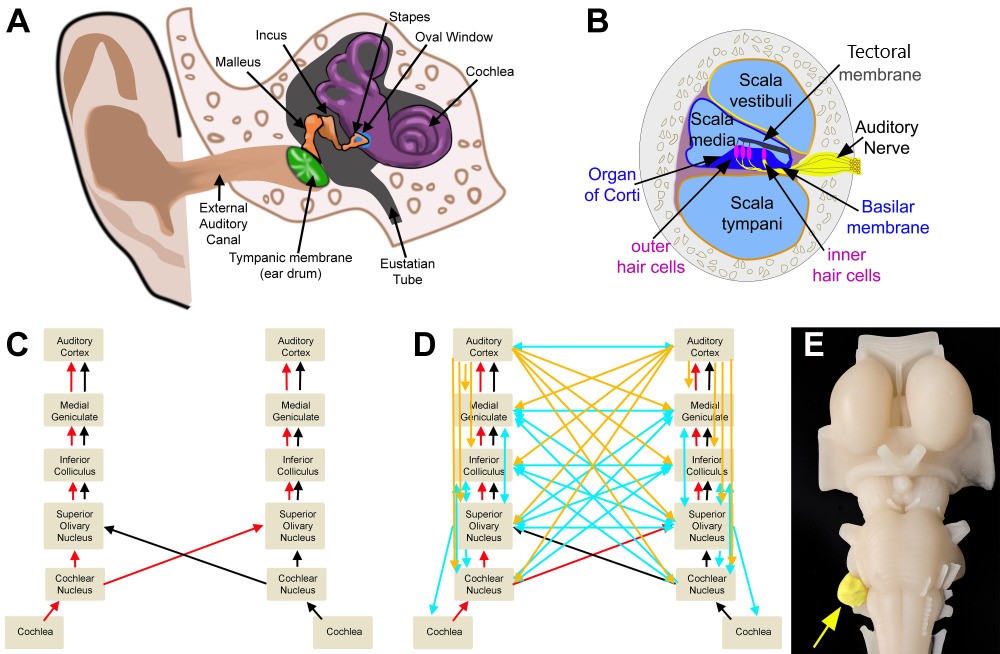
Peripheral and Central Structures of the Auditory System. A. Outer, middle, and inner ear B. Cross section of the cochlea C. Ascending auditory pathway D. Ascending (red/black), descending (cortical: orange; brain-stem: blue), and crossed (blue) auditory pathways E. Model representation of an acoustic neuroma
All imaging was created by Diana Peterson for the current manuscript.
(Click Image to Enlarge)
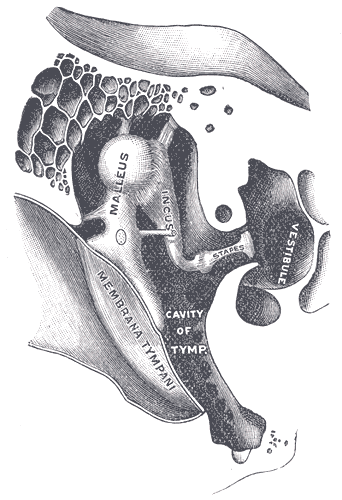
Right Auditory Ossicles. This image shows the anterior aspect of the right chain of ossicles (malleus, incus, and stapes) and their ligaments. Other structures in this illustration include the tympanic membrane, tympanic cavity, and vestibule.
Henry Vandyke Carter, Public Domain, via Wikimedia Commons
(Click Image to Enlarge)
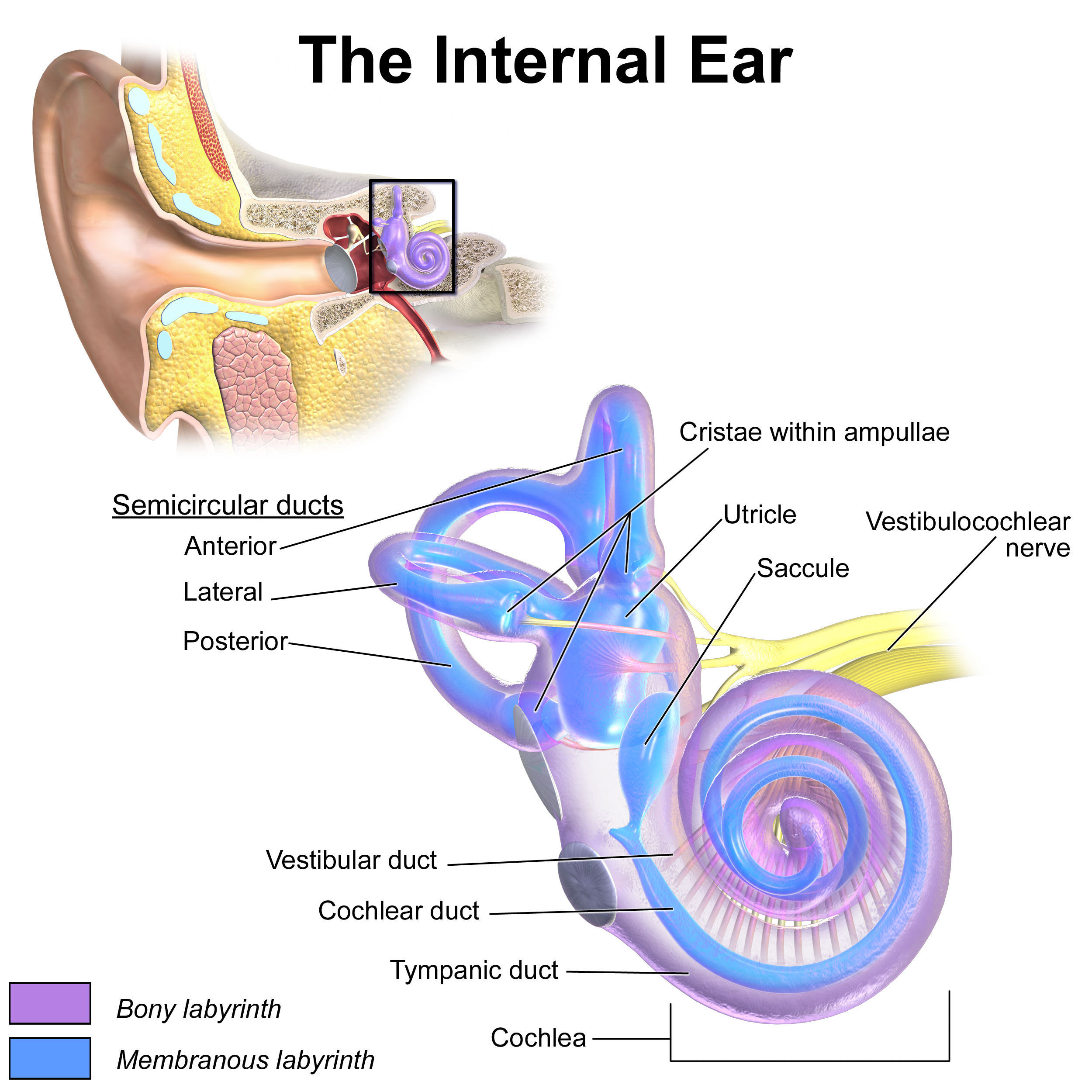
Inner Ear Anatomy. This illustration shows the semicircular ducts and parts of the cochlea.
Blausen.com staff. Medical Gallery of Blausen Medical 2014. WikiJournal of Medicine. doi: 10.15347/wjm/2014.010.
ISSN 2002-4436. [CC BY 3.0 (https://creativecommons.org/licenses/by/3.0)] via Wikimedia Commons.
(Click Image to Enlarge)
(Click Image to Enlarge)
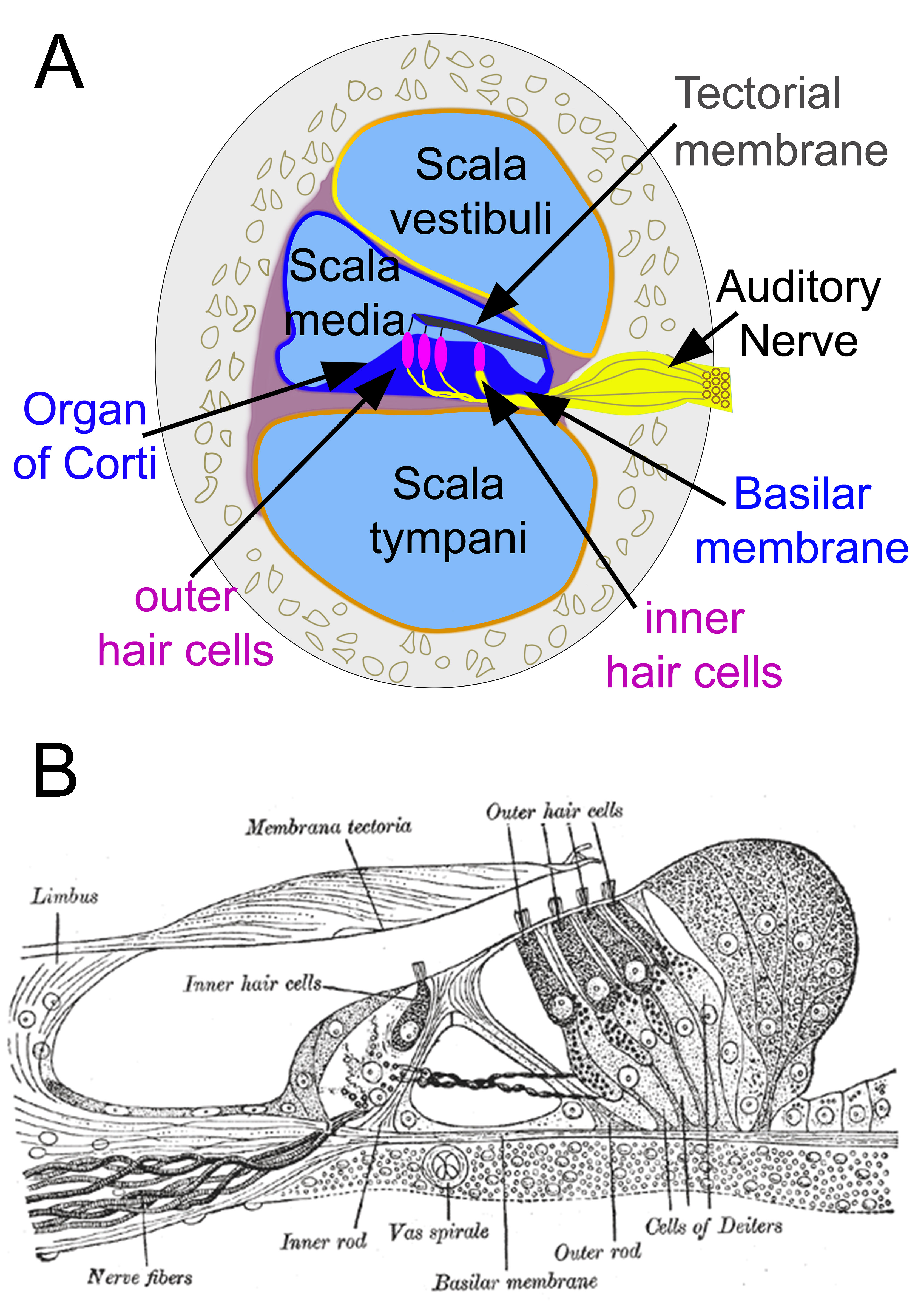
Organ of Corti. Cross-section of the cochlea (A). Section through the organ of Corti (B).
(A) Contributed by D Peterson, MD
(B) Henry Vandyke Carter, Public Domain, via Wikimedia Commons
(Click Image to Enlarge)
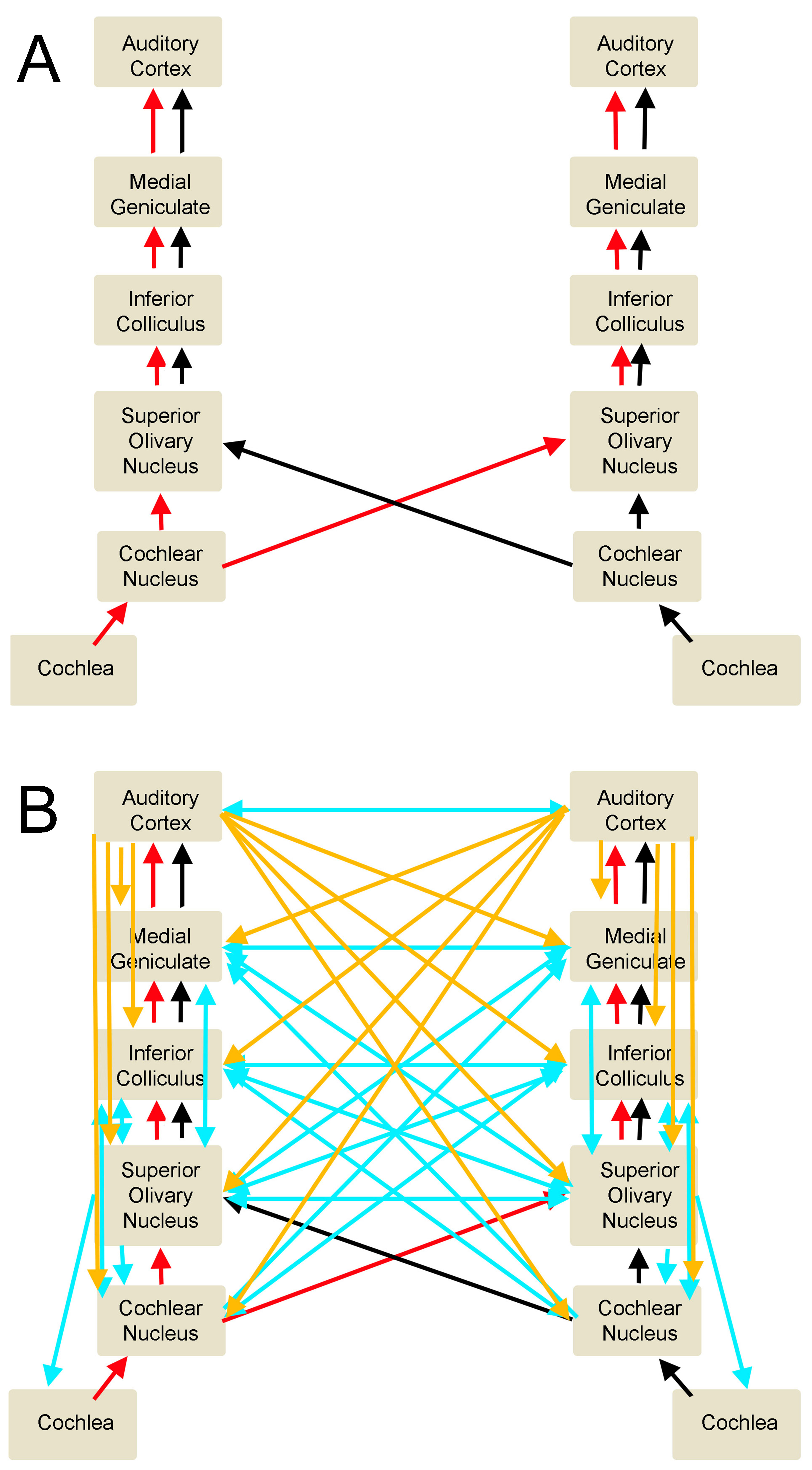
Auditory Pathways. A. This illustration represents the ascending auditory circuits. B. In this combined illustration, the ascending auditory pathway is represented by the red and black arrows, which stand for binaural ascending signals. The descending pathway is represented by the orange arrows (for cortical impulses) and blue arrows (for brainstem signals). Crossing arrows represent the interconnections between the various auditory centers.
Contributed and created by Diana Peterson.
(Click Image to Enlarge)
References
Zhao B, Müller U. The elusive mechanotransduction machinery of hair cells. Current opinion in neurobiology. 2015 Oct:34():172-9. doi: 10.1016/j.conb.2015.08.006. Epub 2015 Sep 3 [PubMed PMID: 26342686]
Level 3 (low-level) evidenceDelacroix L, Malgrange B. Cochlear afferent innervation development. Hearing research. 2015 Dec:330(Pt B):157-69. doi: 10.1016/j.heares.2015.07.015. Epub 2015 Jul 29 [PubMed PMID: 26231304]
Mulders WH, Robertson D. Evidence for direct cortical innervation of medial olivocochlear neurones in rats. Hearing research. 2000 Jun:144(1-2):65-72 [PubMed PMID: 10831866]
Level 3 (low-level) evidenceFelix RA 2nd, Gourévitch B, Portfors CV. Subcortical pathways: Towards a better understanding of auditory disorders. Hearing research. 2018 May:362():48-60. doi: 10.1016/j.heares.2018.01.008. Epub 2018 Jan 31 [PubMed PMID: 29395615]
Level 3 (low-level) evidenceWenstrup JJ. Frequency organization and responses to complex sounds in the medial geniculate body of the mustached bat. Journal of neurophysiology. 1999 Nov:82(5):2528-44 [PubMed PMID: 10561424]
Level 3 (low-level) evidencePeterson DC, Voytenko S, Gans D, Galazyuk A, Wenstrup J. Intracellular recordings from combination-sensitive neurons in the inferior colliculus. Journal of neurophysiology. 2008 Aug:100(2):629-45. doi: 10.1152/jn.90390.2008. Epub 2008 May 21 [PubMed PMID: 18497365]
Level 3 (low-level) evidencePeterson DC, Nataraj K, Wenstrup J. Glycinergic inhibition creates a form of auditory spectral integration in nuclei of the lateral lemniscus. Journal of neurophysiology. 2009 Aug:102(2):1004-16. doi: 10.1152/jn.00040.2009. Epub 2009 Jun 10 [PubMed PMID: 19515958]
Level 3 (low-level) evidenceGans D, Sheykholeslami K, Peterson DC, Wenstrup J. Temporal features of spectral integration in the inferior colliculus: effects of stimulus duration and rise time. Journal of neurophysiology. 2009 Jul:102(1):167-80. doi: 10.1152/jn.91300.2008. Epub 2009 Apr 29 [PubMed PMID: 19403742]
Level 3 (low-level) evidenceYavuzoglu A, Schofield BR, Wenstrup JJ. Circuitry underlying spectrotemporal integration in the auditory midbrain. The Journal of neuroscience : the official journal of the Society for Neuroscience. 2011 Oct 5:31(40):14424-35. doi: 10.1523/JNEUROSCI.3529-11.2011. Epub [PubMed PMID: 21976527]
Level 3 (low-level) evidencePeterson DC, Wenstrup JJ. Selectivity and persistent firing responses to social vocalizations in the basolateral amygdala. Neuroscience. 2012 Aug 16:217():154-71. doi: 10.1016/j.neuroscience.2012.04.069. Epub 2012 May 6 [PubMed PMID: 22569154]
Level 3 (low-level) evidenceCoomes DL, Schofield BR. Projections from the auditory cortex to the superior olivary complex in guinea pigs. The European journal of neuroscience. 2004 Apr:19(8):2188-200 [PubMed PMID: 15090045]
Level 3 (low-level) evidenceSchofield BR, Coomes DL. Projections from auditory cortex contact cells in the cochlear nucleus that project to the inferior colliculus. Hearing research. 2005 Aug:206(1-2):3-11 [PubMed PMID: 16080994]
Level 3 (low-level) evidenceSchofield BR, Coomes DL. Pathways from auditory cortex to the cochlear nucleus in guinea pigs. Hearing research. 2006 Jun-Jul:216-217():81-9 [PubMed PMID: 16874906]
Level 3 (low-level) evidenceSchofield BR, Coomes DL, Schofield RM. Cells in auditory cortex that project to the cochlear nucleus in guinea pigs. Journal of the Association for Research in Otolaryngology : JARO. 2006 Jun:7(2):95-109 [PubMed PMID: 16557424]
Level 3 (low-level) evidenceCoomes Peterson D, Schofield BR. Projections from auditory cortex contact ascending pathways that originate in the superior olive and inferior colliculus. Hearing research. 2007 Oct:232(1-2):67-77 [PubMed PMID: 17643879]
Level 3 (low-level) evidenceWeinberger NM. Associative representational plasticity in the auditory cortex: a synthesis of two disciplines. Learning & memory (Cold Spring Harbor, N.Y.). 2007 Jan-Feb:14(1-2):1-16 [PubMed PMID: 17202426]
Level 3 (low-level) evidenceSuga N. Role of corticofugal feedback in hearing. Journal of comparative physiology. A, Neuroethology, sensory, neural, and behavioral physiology. 2008 Feb:194(2):169-83. doi: 10.1007/s00359-007-0274-2. Epub 2008 Jan 29 [PubMed PMID: 18228080]
Level 3 (low-level) evidenceForbes CE, Grafman J. The role of the human prefrontal cortex in social cognition and moral judgment. Annual review of neuroscience. 2010:33():299-324. doi: 10.1146/annurev-neuro-060909-153230. Epub [PubMed PMID: 20350167]
Level 3 (low-level) evidenceLitovsky R. Development of the auditory system. Handbook of clinical neurology. 2015:129():55-72. doi: 10.1016/B978-0-444-62630-1.00003-2. Epub [PubMed PMID: 25726262]
Level 3 (low-level) evidenceMei X, Atturo F, Wadin K, Larsson S, Agrawal S, Ladak HM, Li H, Rask-Andersen H. Human inner ear blood supply revisited: the Uppsala collection of temporal bone-an international resource of education and collaboration. Upsala journal of medical sciences. 2018 Sep:123(3):131-142. doi: 10.1080/03009734.2018.1492654. Epub 2018 Sep 11 [PubMed PMID: 30204028]
Pan WR, le Roux CM, Levy SM, Briggs CA. Lymphatic drainage of the external ear. Head & neck. 2011 Jan:33(1):60-4. doi: 10.1002/hed.21395. Epub [PubMed PMID: 20848416]
Lim DJ, Hussl B. Macromolecular transport by the middle ear and its lymphatic system. Acta oto-laryngologica. 1975 Jul-Aug:80(1-2):19-31 [PubMed PMID: 1166775]
Level 3 (low-level) evidenceSalt AN, Hirose K. Communication pathways to and from the inner ear and their contributions to drug delivery. Hearing research. 2018 May:362():25-37. doi: 10.1016/j.heares.2017.12.010. Epub 2017 Dec 19 [PubMed PMID: 29277248]
Greywoode JD, Pribitkin EA, Krein H. Management of auricular hematoma and the cauliflower ear. Facial plastic surgery : FPS. 2010 Dec:26(6):451-5. doi: 10.1055/s-0030-1267719. Epub 2010 Nov 17 [PubMed PMID: 21086231]
Fireman P. Otitis media and eustachian tube dysfunction: connection to allergic rhinitis. The Journal of allergy and clinical immunology. 1997 Feb:99(2):S787-97 [PubMed PMID: 9042072]
Briggs RJ, Fabinyi G, Kaye AH. Current management of acoustic neuromas: review of surgical approaches and outcomes. Journal of clinical neuroscience : official journal of the Neurosurgical Society of Australasia. 2000 Nov:7(6):521-6 [PubMed PMID: 11029233]
Level 2 (mid-level) evidenceGreene J, Al-Dhahir MA. Acoustic Neuroma. StatPearls. 2023 Jan:(): [PubMed PMID: 29262098]
Møller AR. The role of neural plasticity in tinnitus. Progress in brain research. 2007:166():37-45 [PubMed PMID: 17956769]
Level 3 (low-level) evidenceCrocetti A, Forti S, Del Bo L. Neurofeedback for subjective tinnitus patients. Auris, nasus, larynx. 2011 Dec:38(6):735-8. doi: 10.1016/j.anl.2011.02.003. Epub 2011 May 17 [PubMed PMID: 21592701]
Weise C, Heinecke K, Rief W. Biofeedback-based behavioral treatment for chronic tinnitus: results of a randomized controlled trial. Journal of consulting and clinical psychology. 2008 Dec:76(6):1046-57. doi: 10.1037/a0013811. Epub [PubMed PMID: 19045972]
Level 1 (high-level) evidenceHandscomb L. Use of bedside sound generators by patients with tinnitus-related sleeping difficulty: which sounds are preferred and why? Acta oto-laryngologica. Supplementum. 2006 Dec:(556):59-63 [PubMed PMID: 17114145]
Norrix LW, Velenovsky D. Clinicians' Guide to Obtaining a Valid Auditory Brainstem Response to Determine Hearing Status: Signal, Noise, and Cross-Checks. American journal of audiology. 2018 Mar 8:27(1):25-36. doi: 10.1044/2017_AJA-17-0074. Epub [PubMed PMID: 29392291]