Introduction
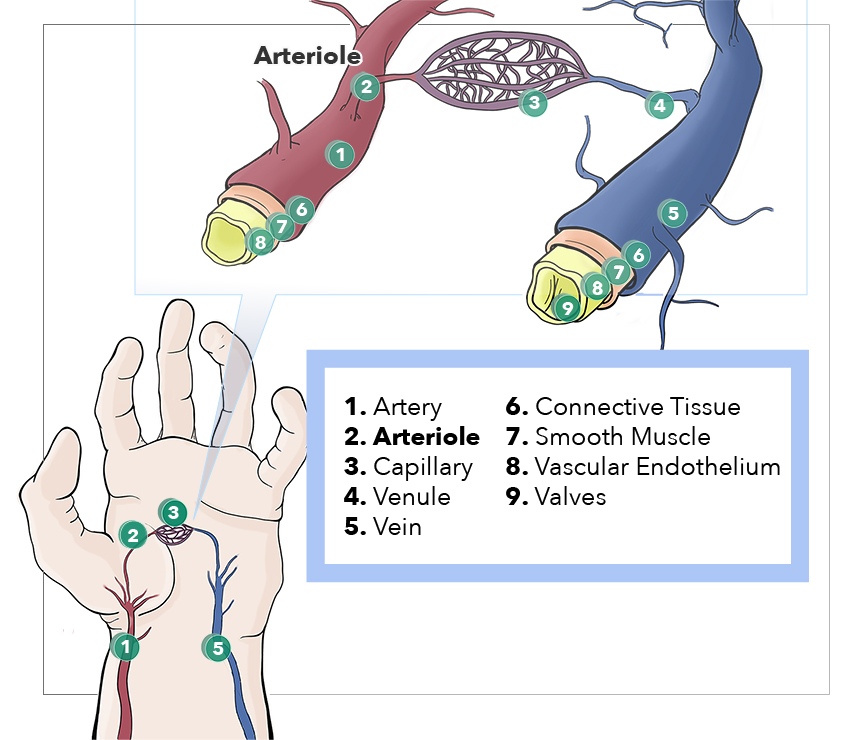
Oxygenated arterial blood circulates through the body via the vascular tree consisting of sequentially smaller arteries, arterioles, and capillary beds. Nutrients and waste exchange between the blood and body tissues occurs at the capillary bed. Venules serve as exit vessels in the capillary bed of various organs and unite to form veins, which return the blood to the heart. The arteriolar wall consists of three layers of cellular and extracellular components. The intima contains endothelial cells sitting on a basement membrane; the tunica media consists of internal elastic lamina and layers of smooth muscle; and the outer adventitia is made mainly of collagen, nerve endings, and fibroblasts. Arterioles contribute to maintaining mean arterial pressure and tissue perfusion as they are the important site of total peripheral resistance. By increasing or decreasing the diameter, they also play a principal role in regulating blood flow in an organ or tissue-specific manner.
Structure and Function
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Structure and Function
Arterioles are considered the primary resistance vessels as they distribute blood flow into capillary beds (see Image. Anatomy, Arteriores). Arterioles provide approximately 80% of the total resistance to blood flow through the body and are considered vital regulators of hemodynamics, contributing to the upstream pressure and the regional distribution of blood.[1][2] They have significantly variable diameters depending on the particular vascular bed (i.e., between different organs), and their diameter varies further when in a state of constriction or dilation. Therefore, size is not their main identifying feature, but rather the structural makeup of smooth muscle in their wall—the wall of arterioles composed of three structurally distinct layers: intima, media, and adventitia.
The Adventitia
The adventitial layer consists of fibroblasts, an extracellular matrix, and thick bundles of collagen fibers organized along the longitudinal axis of the blood vessel.[3] Evidence suggests that some of the adventitial fibroblasts may be considered stem mesenchymal progenitor cells.[4] Nonmyelinated free nerve endings are distributed in the adventitial layer approximately 5 µm from the outermost part of vascular smooth muscle.[5] Recent studies have shown that elastic fibers arranged longitudinally in the outer layer allow arterioles to elongate or recoil in expandable tissues such as skeletal muscle. Arterioles within these expandable tissues usually possess additional external elastic lamina, which is absent in non-expandable tissues like the brain.[6]
Historically, the function of the adventitial layer has been considered only as a structural support and scaffold for the anchored nerve endings. Recently, however, research into the role of adventitial fibroblasts has been dramatically expanded. Adventitial fibroblasts have a potential role in reactive oxygen species (ROS) production to modulate the activity of smooth muscle cells residing in tunica media, which, in turn, initiate vascular remodeling.[7] Furthermore, adventitial fibroblasts produce several growth factors and vasoactive compounds, which include transforming growth factor-beta, fibroblast growth factor, and endothelin-1. All of these compounds have an essential role in mediating cell proliferation and controlling vascular tone.[8] Adventitial fibroblasts also play a predominant role in vascular repair. In response to injury, fibroblasts can transform into myofibroblasts, which allows them to increase their contractile capabilities and for the synthesis of extracellular matrix (ECM) proteins, such as collagen.[9]
The Media
The medial layer of arterioles predominantly consists of smooth muscle cells and an internal elastic lamina. The internal elastic lamina primarily consists of degradation-resistant elastin molecules. The presence of an internal elastic lamina is restricted to the feeding arterioles of skeletal muscle, mesentery, and cerebrum. The enriched elastin content of the internal elastic lamina provides a potential recoil property to deal with pulsatile blood pressure when necessary. This function is apparent in conduit arteries and less evident in arterioles due to less pronounced pulsatile pressure. Besides, the internal elastic lamina in arterioles is not smooth/fully stretched, evident by the wavy appearance with evenly distributed ridges.[10] Thus, in physiological conditions, the internal elastic lamina contributes little to the viscoelastic property of the vessel wall. Under electron microscopy, the internal elastic lamina appears as a fenestrated solid sheet.[11][12] Studies have shown that fenestrations influence the transport of substances from the blood across the media and extravascular tissues.[13] Therefore, remodeling of the fenestrae may be tuned by vascular permeability requirements.[14] Besides, fenestrations allow direct contact between endothelial and medial smooth muscle cells. However, not all fenestrae possess these myoendothelial junctions.[14]
Vascular smooth muscle is the most abundant component of the media that is primarily delegated to control vascular diameter by the contraction-relaxation process. They are arranged circumferentially to the longitudinal axis of the vessel. Smooth muscle in feeding arterioles is arranged in one to two layers, which appear to wrap around the vascular circumference. Intercellular connections between medial smooth muscles are not continuous; instead, they are in the form of appositions, inter-digitations, intermediate junctions, and nexus junctions. These intercellular connections involve several junctional molecules such as integrins, cadherins, and connexins.[15][16]
Under physiological conditions, smooth muscles of the arteriole remain partially contracted to exert tone. Vascular smooth muscle can sense physical forces that primarily transmit to cells of the vascular wall through the adhesive contacts with the extracellular matrix (ECM) and one another. Additionally, it has previously been shown that the blockade of integrins or cadherins can inhibit myogenic vasoconstriction. Thus, intercellular connections and cell-extracellular matrix interactions are essential for myogenic phenomena and mechanotransduction.
The Intima
Traditionally the intima is considered a physical barrier between blood components and extravascular tissues. In arterioles, the intima predominantly consists of endothelial cells (ECs) and their basement membrane. Endothelial cells are longitudinally arranged with the direction of flow and have an overall thickness of 0.2 to 0.5 µm, except at the area of the cell nucleus. In feeding arterioles, ECs are approximately 100 µm in length and approximately 10 µm in width with a 10:1 ratio that reduces as downstream arterioles with a smaller diameter.[17]
Functionally, endothelial cells control vessel tone by synthesis and release of vasoactive factors that exert their potential effects on neighboring smooth muscle cells. The functional properties of endothelial cells interlink with their structure. ECs have an excellent capacity to sense and transduce mechanical forces and produce vasoactive compounds that can fine-tune the tone of adjacent smooth muscle cells for the appropriate regulation of arteriolar diameter.[2][18][19][20]
An additional functional characteristic of endothelial cells is their association with vascular permeability. Morphological changes of ECs from toxin and inflammation allow variations in microvascular permeability.[21][22] Furthermore, there have also been suggestions that regional functional heterogeneity is due to structural variations in endothelial cells from the retinal, mesenteric, renal, and pulmonary circulations.[17][23] Besides this phenotypic variation, some endothelial cells may possess multipotent characteristics evident by their transdifferentiation into smooth muscle cells. Endothelial progenitor cells (EPC) have stem-like properties that aid in vascular repair and neovascularization by endothelial-to-mesenchymal transdifferentiation to produce a smooth muscle phenotype. Transforming growth factor (TGF) beta, a cytokine produced by various cells, is considered the responsible stimulus for this endothelial-to-mesenchymal transdifferentiation.[24][25][26]
The basement membrane underneath ECs in arterioles is primarily composed of collagen type IV, laminin, heparan sulfate, and proteoglycans. Additional constituents are collagens type I, III, and V and fibronectin.[27] Primarily the basement functions as anchoring support to the endothelium. Previously the basement membrane has been considered an intrinsic part of the intima and produced by endothelial cells. However, recent results indicate close interaction with the media and intima for its proper formation and maintenance. For example, during vasculogenesis, the interaction between pericytes and endothelial cells is required to form and stabilize vascular tubes and endothelial basement membranes.[28]
Physiologic Role
The arterioles play a principal role in flow regulation and intravascular pressure. Arterioles are the site of the highest resistance across the vascular tree and thus act as the most significant contributor to total peripheral resistance and eventually mean arterial pressure. By maintaining total peripheral resistance, or mean arterial pressure, arterioles contribute to upstream perfusion pressure for all organs. Most commonly, arterioles branch into capillary beds and then collect into small venules and control the volume of blood distribution in a given capillary bed. They are the gatekeepers to the capillary network that supplies cells and tissues with oxygen and nutrients and removes waste products. They have a unique feature compared to other blood vessels defined by their active response to physical stimuli. They constrict and maintain a smaller diameter in response to high intravascular pressure (the myogenic response) and undergo a dilated state when flow increases (flow-included dilation). Furthermore, arterioles respond to changes in the chemical environment, including dilation in response to local hypoxia or secondary to various mediators during periods of high metabolic activity of the parenchyma. It has been evident that meaningful communication resides among the venules and adjacent arterioles, providing a feedback system to control arteriolar tone according to local metabolic needs.[29][30]
Embryology
The development of the cardiovascular system begins in the mesoderm as early as the third week of life, and fetal circulation begins through the vasculature system around the eighth week. Hematopoietic stem cells evolved into the vascular system to enable immune surveillance, supply oxygen and nutrients, and dispose of waste. Although most apparent near the early forming heart, vascular development begins in both the intra- and extraembryonic mesoderm. During their development, hematopoietic stem cells give rise to angioblasts, which eventually aggregate and line up to form tubes or sinuses. This process is called vasculogenesis. The term for establishing new vessels from already developed vasculature is angiogenesis. Angiogenesis is an essential mechanism of arteriole development as it is the terminal branch of the arterial side of the vascular tree.
There are three mechanisms of angiogenesis: sprouting, intussusception, and splitting [31]. Sprouting of new vessels occurs through a series of events where the first event is the signaling for new vessel growth. This process strictly depends on vascular endothelial growth factor (VEGF) and its receptor VEGFR2. It is a potent and endothelial-selective growth factor. In the next step, endothelial activation by nitric oxide (NO) converts the existing blood vessels to a dilated and hyperpermeable state. The activated endothelial cell then begins to synthesize enzymes that degrade the basement membrane (e.g., matrix metalloproteinases (MMPs). Endothelial cells migrate and proliferate through the digested matrix, and eventually, a lumen forms in the nascent blood vessels. Recruitment of supporting cells, pericytes, and smooth muscle cells stabilize the newly built vessel. At some point in the later events, blood begins to flow. Sprouting is a prolonged process and primarily depends on cell proliferation and bridging the vascular gaps.
In contrast, intussusception and splitting do not depend on cell proliferation. Intussusception begins with endothelial invagination outside the vascular wall or lumen, where the intraluminal protrusion of endothelial cells is the pre-representations of splitting [31]. Endothelial cell differentiation to the sites of active vascular growth may also occur in the adult by homing marrow-derived circulating precursors cells in a VEGF/VEGFR2-dependent manner.
Blood Supply and Lymphatics
All the large blood vessels depend on other tiny vessels called vasa vasorum for their blood perfusion as other organs of the body. They may arise directly from the central lumen of the artery, from its branches of the given artery, or from the nearby vessels. After their origin, they undergo further branching and supply the outer portion of tunica media and the adventitial layer. In contrast, the arteriole takes advantage of its smaller diameter. A short distance between the wall and inner luminal blood provides an opportunity for the direct diffusion of oxygen and nutrition to happen. Thus, arterioles do not have to depend on the vasa vasorum.[32]
Nerves
The innervation of arterioles mainly derives from the sympathetic autonomic nervous system. Sympathetic nerves form plexuses along the surfaces of the large arteries and run through the smaller arterioles as single, very tiny filaments and more exclusive branches distributed exclusively to the muscular tissue after penetrating the outer coat. The principal role of this sympathetic innervation is controlling the total peripheral resistance, thereby maintaining the upstream mean arterial pressure essential for organ perfusion. Sympathetic fibers are also extensively distributed in the precapillary sphincter and control localized flow by either contraction or relaxation.[33]
Muscles
Like other blood vessels, arterioles contain only smooth muscle cells in their middle coat, along with other connective tissues, including elastic tissue. Smooth muscle cells are the most abundant component of the tunica media of an arteriole.[34] They are spindle-shaped and transversally arranged to the long axis of the vessel, having a luminal side facing toward the internal elastic lamina and an abluminal side facing toward the adventitia. The primary function of these smooth muscle cells is to control arteriolar lumen diameter by their contraction or relaxation processes. Although they have the unique ability to detect and respond to mechanical forces under physiological conditions, arteriolar smooth muscle cells display a partial contraction to exert tone.[10][35]
Clinical Significance
Tubuloglomerular Feedback Mechanism in the Kidney
Afferent and efferent arterioles transport blood in and out of the glomerulus, respectively. Hypovolemia-induced reduction in glomerular filtration leads to decreased flow of NaCl to specialized epithelial cells (macula densa). This, in turn, sends signals to the adjacent afferent arteriole to vasodilate and increase glomerular filtration. Furthermore, the decreased delivery of NaCl also stimulates the renin secretion from specialized cells located in the wall of the afferent arteriole.[36]
Hypoxic Vasoconstriction in the Pulmonary Circulation
Pulmonary circulation is a low-pressure and high-compliance circuit where arterioles respond differently to hypoxia. Systemic arterioles dilate in response to hypoxia, whereas arterioles in the lungs constrict in hypoxia. This hypoxic pulmonary vasoconstriction is essential in directing blood away from under-ventilated parts of the lungs towards better-ventilated parts of the lungs.[37]
Coronary Circulation
Blood flow in the coronary circulation is not continuous as in other parts of the body. Vascular resistance in the transmural portion of the coronary circulation is significantly higher during ventricular contraction resulting in decreased flow during systole. And regulation of coronary microvascular resistance varies across different segments of the vasculature. Arterioles with less than 100 microns in diameter respond differently than larger arterioles at the level of autoregulation, myogenic control, and control by metabolic factors.[38]
Cerebral Circulation
Cerebral blood flow is mostly regulated by the arterial partial pressure of CO2 (pC02). As long as arterial pO2 is normal or above normal, cerebral blood flow is proportional to arterial pCO2. High pCO2 induces cerebral arteriolar vasodilation, thus increasing cerebral blood flow. This unique characteristic of cerebral arterioles allows therapeutic hyperventilation for lowering intracranial pressure (ICP) when indicated. Furthermore, the final step of blood flow regulation by the arterioles matches the focal demand of nearby tissue, which is mediated by microvascular pericytes.[39][40]
Skin
Arterioles most commonly branch into capillaries and are subsequently collected into small venules. In contrast, terminal arterioles of the skin are directly connected to the venue, bypassing the capillary bed. This bypassing or arteriovenous (A-V) shunt is particularly useful in body temperature regulation. Cutaneous A-V shunts receive sympathetic innervation. Sympathetic stimulation induces the constriction of arterioles resulting in a decrease in blood volume in the skin, thereby preventing heat loss. On the contrary, high skin temperature directly causes vasodilation, which increases heat loss.[41]
The function and structure of arterioles can be affected by several disease conditions, such as inflammation, atherosclerosis, and hypertensive vascular disease. Clinical manifestations depend on the involved organs that predominantly result from ischemia due to arterial insufficiency.
Vasculitis
Vasculitis refers to inflammation of the vessel wall, which may affect large vessels (elastic arteries), medium-sized (muscular arteries), or small (arterioles, venules, capillaries) combinations of these vessel types. Disease manifestations include nonspecific systemic symptoms of inflammation (e.g., fever, fatigue, weight loss, and myalgias) or organ-specific symptoms from ischemia due to luminal narrowing or thrombosis of the inflamed vessel. Vasculitis affecting arterioles solely or in combination with other vessels includes granulomatosis with polyangiitis, Churg-Strauss syndrome, microscopic polyangiitis, Henoch-Schönlein purpura, and cryoglobulinemic vasculitis.[42]
Arteriolosclerosis
Arteriosclerosis is a pathologic process involving the arteriole, eventually causing occlusion. Hyaline arteriolosclerosis and hyperplastic arteriolosclerosis are the two varieties of arteriolosclerosis. In hyaline arteriolosclerosis, increased protein deposition within the vascular wall occludes the arteriole lumen. Uncontrolled diabetes and hypertension are the two most common causes of this form of arteriosclerosis. Poorly controlled diabetes causes non-enzymatic glycosylation of the basement membrane that allows plasma proteins to leak into the vessel wall. In hypertensive patients, high luminal pressure pushes plasma proteins into the vessel wall.[43] Hyperplastic arteriolosclerosis is characterized by basement membrane duplication and smooth muscle cell hyperplasia as a reaction to a very rapid rise in blood pressure. This condition is common in the afferent and efferent arterioles of renal vasculature in patients with malignant hypertension.[44]
The Arteriolar Disease of Cerebral Circulation
The small cerebral penetrating arterioles are affected by diseases such as micro-atherosclerosis, lipohyalinosis, and microaneurysm (aka Charcot–Bouchard aneurysm). Atherothrombotic occlusion of the small, penetrating arterioles results in lacunar stroke, a subtype of ischemic stroke. In contrast, rupture of microaneurysm is the culprit of parenchymal hemorrhage or hemorrhagic stroke.
Diabetic Microangiopathy
This small-vessel disease commonly affects the arterioles, capillaries, and venules of diabetic patients. Diabetic microangiopathy is the main pathogenic contributor to diabetic complications of the eye (diabetic retinopathy), kidney (arteriolar nephrosclerosis), and lower extremities (gangrene). The structural hallmark of this disease is the thickening of the basement membrane, which leads to lumen occlusion, thus, tissue hypoxia and damage.
Media
(Click Image to Enlarge)
References
Christensen KL, Mulvany MJ. Location of resistance arteries. Journal of vascular research. 2001 Jan-Feb:38(1):1-12 [PubMed PMID: 11173989]
Level 3 (low-level) evidenceMeininger GA, Harris PD, Joshua IG. Distributions of microvascular pressure in skeletal muscle of one-kidney, one clip, two-kidney, one clip, and deoxycorticosterone-salt hypertensive rats. Hypertension (Dallas, Tex. : 1979). 1984 Jan-Feb:6(1):27-34 [PubMed PMID: 6693146]
Level 3 (low-level) evidenceSangiorgi S, Manelli A, Dell'Orbo C, Congiu T. A new method for the joint visualization of vascular structures and connective tissues: corrosion casting and 1 N NaOH maceration. Microscopy research and technique. 2006 Nov:69(11):919-23 [PubMed PMID: 16921528]
Level 3 (low-level) evidenceHoshino A, Chiba H, Nagai K, Ishii G, Ochiai A. Human vascular adventitial fibroblasts contain mesenchymal stem/progenitor cells. Biochemical and biophysical research communications. 2008 Apr 4:368(2):305-10. doi: 10.1016/j.bbrc.2008.01.090. Epub 2008 Jan 28 [PubMed PMID: 18230345]
Higuchi K,Hashizume H,Aizawa Y,Ushiki T, Scanning electron microscopic studies of the vascular smooth muscle cells and pericytes in the rat heart. Archives of histology and cytology. 2000 May; [PubMed PMID: 10885448]
Level 3 (low-level) evidenceLee RM. Morphology of cerebral arteries. Pharmacology & therapeutics. 1995 Apr:66(1):149-73 [PubMed PMID: 7630927]
Level 3 (low-level) evidenceHaurani MJ, Pagano PJ. Adventitial fibroblast reactive oxygen species as autacrine and paracrine mediators of remodeling: bellwether for vascular disease? Cardiovascular research. 2007 Sep 1:75(4):679-89 [PubMed PMID: 17689510]
Level 3 (low-level) evidenceDi Wang H, Rätsep MT, Chapman A, Boyd R. Adventitial fibroblasts in vascular structure and function: the role of oxidative stress and beyond. Canadian journal of physiology and pharmacology. 2010 Mar:88(3):177-86. doi: 10.1139/Y10-015. Epub [PubMed PMID: 20393583]
Level 3 (low-level) evidenceForte A,Della Corte A,De Feo M,Cerasuolo F,Cipollaro M, Role of myofibroblasts in vascular remodelling: focus on restenosis and aneurysm. Cardiovascular research. 2010 Dec 1; [PubMed PMID: 20621923]
Level 3 (low-level) evidenceSleek GE, Duling BR. Coordination of mural elements and myofilaments during arteriolar constriction. Circulation research. 1986 Dec:59(6):620-7 [PubMed PMID: 3815757]
Level 3 (low-level) evidenceArribas SM, Briones AM, Bellingham C, González MC, Salaices M, Liu K, Wang Y, Hinek A. Heightened aberrant deposition of hard-wearing elastin in conduit arteries of prehypertensive SHR is associated with increased stiffness and inward remodeling. American journal of physiology. Heart and circulatory physiology. 2008 Dec:295(6):H2299-307. doi: 10.1152/ajpheart.00155.2008. Epub 2008 Oct 10 [PubMed PMID: 18849339]
Level 3 (low-level) evidenceWong LC, Langille BL. Developmental remodeling of the internal elastic lamina of rabbit arteries: effect of blood flow. Circulation research. 1996 May:78(5):799-805 [PubMed PMID: 8620599]
Level 3 (low-level) evidenceGuo ZY, Yan ZQ, Bai L, Zhang ML, Jiang ZL. Flow shear stress affects macromolecular accumulation through modulation of internal elastic lamina fenestrae. Clinical biomechanics (Bristol, Avon). 2008:23 Suppl 1():S104-11 [PubMed PMID: 17923177]
Level 3 (low-level) evidenceSandow SL, Gzik DJ, Lee RM. Arterial internal elastic lamina holes: relationship to function? Journal of anatomy. 2009 Feb:214(2):258-66. doi: 10.1111/j.1469-7580.2008.01020.x. Epub [PubMed PMID: 19207987]
Level 3 (low-level) evidenceKrizmanich WJ, Lee RM. Correlation of vascular smooth muscle cell morphology observed by scanning electron microscopy with transmission electron microscopy. Experimental and molecular pathology. 1997:64(3):157-72 [PubMed PMID: 9439481]
Level 3 (low-level) evidenceSosa-Melgarejo JA, Berry CL, Robinson NA. Effects of hypertension on the intercellular contacts between smooth muscle cells in the rat thoracic aorta. Journal of hypertension. 1991 May:9(5):475-80 [PubMed PMID: 1649868]
Level 3 (low-level) evidenceYu PK,Yu Dy,Alder VA,Seydel U,Su En,Cringle SJ, Heterogeneous endothelial cell structure along the porcine retinal microvasculature. Experimental eye research. 1997 Sep; [PubMed PMID: 9299174]
Level 3 (low-level) evidenceSun D, Huang A, Kaley G. Mechanical compression elicits NO-dependent increases in coronary flow. American journal of physiology. Heart and circulatory physiology. 2004 Dec:287(6):H2454-60 [PubMed PMID: 15308477]
Level 3 (low-level) evidenceSun D, Huang A, Sharma S, Koller A, Kaley G. Endothelial microtubule disruption blocks flow-dependent dilation of arterioles. American journal of physiology. Heart and circulatory physiology. 2001 May:280(5):H2087-93 [PubMed PMID: 11299210]
Level 3 (low-level) evidenceBrum Cde A, Duarte ID, Webb RC, Leite R. Disruption of microtubular network attenuates histamine-induced dilation in rat mesenteric vessels. American journal of physiology. Cell physiology. 2005 Feb:288(2):C443-9 [PubMed PMID: 15483228]
Level 3 (low-level) evidenceThurston G, Baldwin AL, Wilson LM. Changes in endothelial actin cytoskeleton at leakage sites in the rat mesenteric microvasculature. The American journal of physiology. 1995 Jan:268(1 Pt 2):H316-29 [PubMed PMID: 7840278]
Level 3 (low-level) evidenceNag S, Robertson DM, Dinsdale HB. Intracerebral arteriolar permeability to lanthanum. The American journal of pathology. 1982 Jun:107(3):336-41 [PubMed PMID: 7081387]
Level 3 (low-level) evidenceOchoa CD, Wu S, Stevens T. New developments in lung endothelial heterogeneity: Von Willebrand factor, P-selectin, and the Weibel-Palade body. Seminars in thrombosis and hemostasis. 2010 Apr:36(3):301-8. doi: 10.1055/s-0030-1253452. Epub 2010 May 20 [PubMed PMID: 20490980]
Imamura H, Ohta T, Tsunetoshi K, Doi K, Nozaki K, Takagi Y, Kikuta K. Transdifferentiation of bone marrow-derived endothelial progenitor cells into the smooth muscle cell lineage mediated by tansforming growth factor-beta1. Atherosclerosis. 2010 Jul:211(1):114-21. doi: 10.1016/j.atherosclerosis.2010.02.040. Epub 2010 Mar 4 [PubMed PMID: 20303084]
Level 3 (low-level) evidenceMoonen JR,Krenning G,Brinker MG,Koerts JA,van Luyn MJ,Harmsen MC, Endothelial progenitor cells give rise to pro-angiogenic smooth muscle-like progeny. Cardiovascular research. 2010 Jun 1; [PubMed PMID: 20083576]
Frid MG, Kale VA, Stenmark KR. Mature vascular endothelium can give rise to smooth muscle cells via endothelial-mesenchymal transdifferentiation: in vitro analysis. Circulation research. 2002 Jun 14:90(11):1189-96 [PubMed PMID: 12065322]
Level 3 (low-level) evidenceDas A, Frank RN, Zhang NL, Turczyn TJ. Ultrastructural localization of extracellular matrix components in human retinal vessels and Bruch's membrane. Archives of ophthalmology (Chicago, Ill. : 1960). 1990 Mar:108(3):421-9 [PubMed PMID: 2310346]
Stratman AN, Malotte KM, Mahan RD, Davis MJ, Davis GE. Pericyte recruitment during vasculogenic tube assembly stimulates endothelial basement membrane matrix formation. Blood. 2009 Dec 3:114(24):5091-101. doi: 10.1182/blood-2009-05-222364. Epub 2009 Oct 12 [PubMed PMID: 19822899]
Level 3 (low-level) evidenceChaudhry R,Miao JH,Rehman A, Physiology, Cardiovascular 2020 Jan; [PubMed PMID: 29630249]
Satish M, Tadi P. Physiology, Vascular. StatPearls. 2024 Jan:(): [PubMed PMID: 31194409]
Zhan K, Bai L, Wang G, Zuo B, Xie L, Wang X. Different angiogenesis modes and endothelial responses in implanted porous biomaterials. Integrative biology : quantitative biosciences from nano to macro. 2018 Jul 16:10(7):406-418. doi: 10.1039/c8ib00061a. Epub [PubMed PMID: 29951652]
Gössl M, Rosol M, Malyar NM, Fitzpatrick LA, Beighley PE, Zamir M, Ritman EL. Functional anatomy and hemodynamic characteristics of vasa vasorum in the walls of porcine coronary arteries. The anatomical record. Part A, Discoveries in molecular, cellular, and evolutionary biology. 2003 Jun:272(2):526-37 [PubMed PMID: 12740947]
Level 3 (low-level) evidenceSheng Y, Zhu L. The crosstalk between autonomic nervous system and blood vessels. International journal of physiology, pathophysiology and pharmacology. 2018:10(1):17-28 [PubMed PMID: 29593847]
Pugsley MK, Tabrizchi R. The vascular system. An overview of structure and function. Journal of pharmacological and toxicological methods. 2000 Sep-Oct:44(2):333-40 [PubMed PMID: 11325577]
Level 3 (low-level) evidenceWaitkus-Edwards KR, Martinez-Lemus LA, Wu X, Trzeciakowski JP, Davis MJ, Davis GE, Meininger GA. alpha(4)beta(1) Integrin activation of L-type calcium channels in vascular smooth muscle causes arteriole vasoconstriction. Circulation research. 2002 Mar 8:90(4):473-80 [PubMed PMID: 11884378]
Level 3 (low-level) evidenceIto S,Abe K, Tubuloglomerular feedback. Japanese heart journal. 1996 Mar [PubMed PMID: 8676542]
Level 3 (low-level) evidenceLumb AB, Slinger P. Hypoxic pulmonary vasoconstriction: physiology and anesthetic implications. Anesthesiology. 2015 Apr:122(4):932-46. doi: 10.1097/ALN.0000000000000569. Epub [PubMed PMID: 25587641]
Level 3 (low-level) evidenceDeFily DV, Chilian WM. Coronary microcirculation: autoregulation and metabolic control. Basic research in cardiology. 1995 Mar-Apr:90(2):112-8 [PubMed PMID: 7646415]
Cipolla MJ. The Cerebral Circulation. 2009:(): [PubMed PMID: 21452434]
Paulson OB, Strandgaard S, Edvinsson L. Cerebral autoregulation. Cerebrovascular and brain metabolism reviews. 1990 Summer:2(2):161-92 [PubMed PMID: 2201348]
Curri SB. [Microvascular anatomy of the skin and its appendages]. Phlebologie. 1990 Jul-Oct:43(3):407-30 [PubMed PMID: 2290860]
Shavit E, Alavi A, Sibbald RG. Vasculitis-What Do We Have to Know? A Review of Literature. The international journal of lower extremity wounds. 2018 Dec:17(4):218-226. doi: 10.1177/1534734618804982. Epub 2018 Dec 3 [PubMed PMID: 30501545]
Balakrishnan M, Garcia-Tsao G, Deng Y, Ciarleglio M, Jain D. Hepatic arteriolosclerosis: a small-vessel complication of diabetes and hypertension. The American journal of surgical pathology. 2015 Jul:39(7):1000-9. doi: 10.1097/PAS.0000000000000419. Epub [PubMed PMID: 25786083]
Level 2 (mid-level) evidenceLüscher TF. [Hypertension and vascular diseases: molecular and cellular mechanisms]. Schweizerische medizinische Wochenschrift. 1995 Feb 18:125(7):270-82 [PubMed PMID: 7878405]