Anatomy, Thorax, Heart Anomalous Left Anterior Descending (LAD) Artery
Anatomy, Thorax, Heart Anomalous Left Anterior Descending (LAD) Artery
Introduction
Coronary anomalies of the left anterior descending artery (LAD) are very rare, with a frequency of less than 1% in the general population.[1] Most patients with LAD anomalies are asymptomatic, and therefore most are discovered incidentally. However, some anomalies of the LAD put patients at risk for myocardial dysfunction, myocardial infarction, ventricular arrhythmias, and even sometimes produce a steal phenomenon. These potentially life-threatening coronary artery anomalies need to be identified and treated promptly to lower the risk of morbidity and mortality. Traditionally, the percutaneous transcatheter arteriography (PCA) was the recommended diagnostic test of choice. However, now with advancement in technology, the electrocardiographically (ECG)–gated multi-detector row computed tomography (CT) is the preferred method of diagnosis an anomalous LAD.[2] The CT scan allows for the accurate and noninvasive depiction of coronary artery anomalies of origin, course, and termination. Treatment modalities vary by the type of LAD anomaly present. Some require surgical intervention or transcatheter intervention, while others require only medical therapy or observation. This article will describe in detail the normal structure and function of the LAD, embryologic anomalies of the LAD, surgical considerations, and clinical significance of anomalous LAD.
Structure and Function
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Structure and Function
Normal Anatomy of the LAD
The normal origin of the LAD is at the bifurcation of the distal left main coronary artery (LMCA). From there, it travels around the left side of the pulmonary artery (PA) and passes into the epicardial fat obliquely in the direction of the apex of the heart in the anterior interventricular sulcus. The LAD is made up of a proximal segment, middle segment, and distal segment. The proximal segment contains the first branch of the septal perforator. The middle segment is bordered by the first septal branch and represents the halfway point between the septal branch and the ventricular apex. The distal segment is from the halfway point to the apex. The length of the LAD usually measures approximately 10 cm to 13 cm long. Branches of the LAD are called diagonal branches and septal perforator branches. There are usually one or two large diagonal branches that may arise and descend diagonally across the anterior surface of the LV. The septal perforator branches supply the anterior two-thirds of the basal interventricular septum and the entire septum at the mid and apical levels.[2]
Anomalies of Origin
The most common coronary artery anomalies of the LAD are congenital abnormalities of the origin. One of the most dangerous anomalies of origin of the LAD is when the LAD originates off the pulmonary artery, which is a very rare anomaly that can lead to myocardial injury and potential death.[3] Patients tend to experience symptoms early in life. Approximately 90% of untreated infants die in the 1st year of life, and only a few patients survive to adulthood.[4] Another potentially fatal LAD anomaly is when the LAD is the only coronary artery to arise with a single ostium from the aortic trunk, called a single coronary artery.[5] Population studies reveal a frequency of single LAD to be less than 0.0024 to 0.044%. [6] These patients are at increased risk of sudden cardiac death (SCD), especially if the vessel takes an inter-atrial path, or passes between the aorta and pulmonary artery. Another anomaly of origin of the LAD is when the LAD originates from the RCA; this correlates with the tetralogy of Fallot. The LAD can also originate from the right sinus of Valsalva and has been known to correlate with double outlet right ventricle, tetralogy of Fallot, and transposition complexes. Sometimes these occur in the congenital absence of the LMCA. The absence of the LMCA is associated with aortic valve disease and left dominance. When the LMCA is atretic, a fibrous connection may be seen between the LAD, left circumflex (LCx) arterial junction, and the left sinus of Valsalva. The LAD and the LCx are found on rare occasions to branch from the same origin and bifurcate at the same time; this is called multiple Ostia. Patients with multiple Ostia coronary anomalies are a high risk of coronary artery disease in the affected vessels.[7] While there can be multiple Ostia in some patients, rarely there can also be congenital ostial stenosis or atresia.[8] This condition can be a life-threatening congenital anomaly when it involves the LAD unless there is sufficient collateral supply to the LAD cardiac muscle domain.
Anomalies of Course
There are five different possible paths by which the coronary arteries can connect with the opposite coronary cusps. These paths are called pre-pulmonic, petro-aortic, inter-atrial, trans-septal, and retro-cardiac. The LAD is more commonly associated with the transeptal path; however, it can also be involved with all the other paths. The trans-septal path is when the coronary artery has a subpulmonic course as the artery travels anteriorly and inferiorly through the interventricular septum and takes an intramyocardial course, giving off septal branches and finally emerging at its normal epicardial position.[9]
Sometimes arteries are duplicated. The LAD has been noted in the literature to be a duplicate in 0.13% to 1% of the general population. It is noteworthy that sometimes one of the duplicated LAD arteries may arise from the RCA and take a pre-pulmonic, septal or inter-arterial course. Dual LAD is associated with congenital heart disease, most commonly Tetralogy of Fallot and transposition of the great vessels. One case report by Belostotsky et al. depicts a dual LAD from the LMCA and the other from the right aortic sinus of Valsalva.[10] This anomaly is not intrinsically hemodynamically significant, but its presence may complicate surgical intervention when aortocoronary bypass or other coronary artery surgery is performed.
Angiographic classification of dual LADs was a proposal by Spinaldo-Franco et al. According to Spinaldo-Franco et al. there are four types of dual LADs. Type I dual LAD has a short and long arm, both of which travels in the anterior interventricular sulcus (AIVS). The shorter LAD usually branches into all the major proximal septal perforators. The longer LAD travels on the left-ventricular side of the AIVS and then reenters the distal AIVS to reach the apex. Type II dual LAD has a similar short LAD as seen in Type I. The long arm, however, travels on the right ventricular side before reentering the AIVS to supply the apex. Type III also has a short arm similar to Type I, but the long arm descends intramyocardially into the septum. Type IV the long arm arises from the RCA.[11] Manchanda et al. described another variant termed a type V dual LAD where the shorter LAD originates independently, starting from the left coronary sinus, and the longer LAD originates from the right sinus separate from the right coronary artery and undergoes an intramyocardial course before reaching the distal interventricular groove.[12]
Anomalies Termination
Coronary fistulas occur about 0.3% to 0.87% in patients who undergo cardiac catheterization. In 60% of cases, these fistulae drain into the right heart chambers.[13] Coronary fistulas are communications between a coronary artery and either the cardiac chamber, coronary sinus, pulmonary artery, or superior vena cava (SVC). Fistulas that connect to the cardiac chamber are called coronary cameral fistulas. The LAD has been noted to form all of these fistula types.[14][15][16] Coronary fistulas correlate with an increased risk of bacterial endocarditis. There have also been rare cases of communication between the LAD and the systemic circulation through an anomalous coronary artery formation. Systemic communications also correlate with the presence of congenital heart disease.
Muscles
The heart muscle, particularly the anterolateral wall, can be affected in the presence of LAD anomalies. Depending on the specific anomaly, the heart muscle may be thin or thick.
Physiologic Variants
Normal Origin Variants
The origin of the LAD is a trifurcation of the LMCA into a LAD, LCx, and ramus intermedius instead of the regular bifurcation consisting of the LCx and LAD.[17]
Normal Course Variants
Usually, the coronary arteries travel through the epicardial fat, but in rare cases, a coronary artery may travel into the heart muscle; this is called a myocardial bridge. The mid LAD is the most commonly involved coronary artery. The myocardial bridge usually involves the proximal segment, however other segments can be affected. The length of the myocardial bridging is variable, but it typically ranges from 10 mm to 50 mm. Most patients with myocardial bridging are asymptomatic, and no abnormalities are observable during functional stress testing. However, occasionally when the muscle contracts, the blood vessels that travel underneath become occluded, and symptoms of the acute coronary syndrome can present.[18]
Normal Termination Variants
A "wraparound LAD" is a term coined to describe a normal variant course of the LAD when it wraps around the cardiac apex and supplies a part of the inferior apical wall.[19] In most cases, the posterior descending artery (PDA) supplies this region of the heart. There is some controversy in the literature regarding whether the wraparound LAD should be deemed a normal variant or anomalous. However, the prevalence of the wraparound LAD suggests it should be considered a normal variant. The presence of a wraparound LAD correlates with larger infarct sizes and a high incidence of heart failure.[20]
Clinical Significance
Coronary artery anomalies seldom present in clinical practice; however, when present, they can be associated with a very high risk for mortality and morbidity. In a study by Eckart et al. on 126 patients, 61% of sudden cardiac deaths were due to coronary artery anomalies.[1] Coronary anomalies of the LAD have been noted to be associated with myocardial ischemia/infarction, increased risk of atherosclerosis, aortic valve disease, increased risk of bacterial endocarditis, increased incidence of cardiomyopathy, volume overload, heart failure, and increased risk of complications during cardiac surgery.[8] It is essential to thoroughly evaluate patients with anomalies of the LAD, including a thorough physical exam and mapping of the coronary arteries. Patients with high-risk coronary anomalies may need a referral to interventional cardiology and/or cardiothoracic surgery for further evaluation.
Media
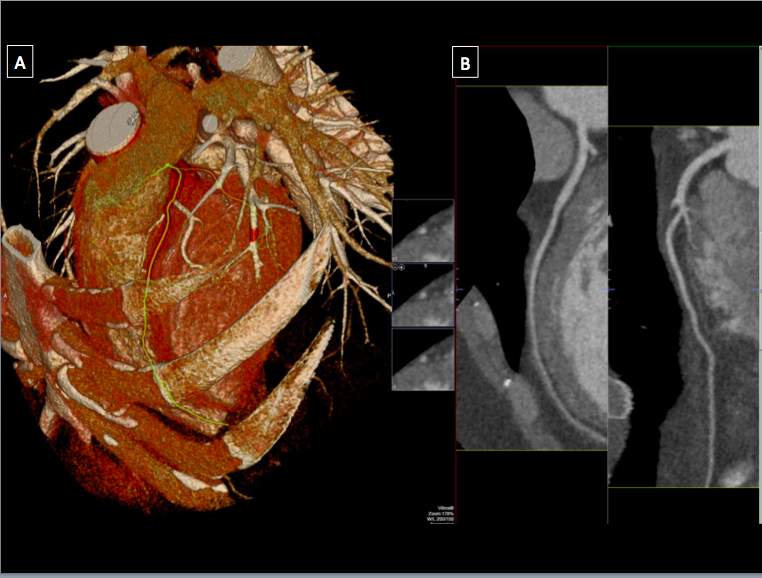
(Click Image to Enlarge)
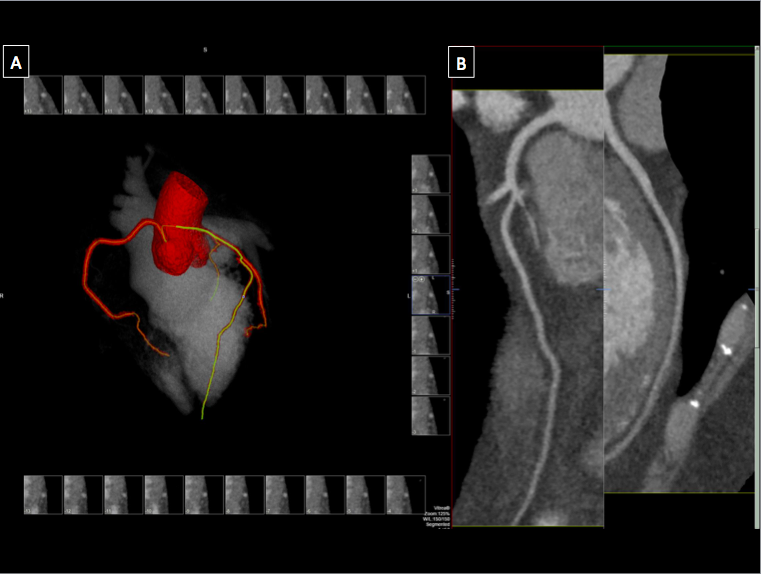
(Click Image to Enlarge)
References
Eckart RE, Scoville SL, Campbell CL, Shry EA, Stajduhar KC, Potter RN, Pearse LA, Virmani R. Sudden death in young adults: a 25-year review of autopsies in military recruits. Annals of internal medicine. 2004 Dec 7:141(11):829-34 [PubMed PMID: 15583223]
Level 2 (mid-level) evidenceYoung PM, Gerber TC, Williamson EE, Julsrud PR, Herfkens RJ. Cardiac imaging: Part 2, normal, variant, and anomalous configurations of the coronary vasculature. AJR. American journal of roentgenology. 2011 Oct:197(4):816-26. doi: 10.2214/AJR.10.7249. Epub [PubMed PMID: 21940568]
Vohra A, Narula H. Dual left anterior descending artery with anomalous origin of long LAD from pulmonary artery - rare coronary anomaly detected on computed tomography coronary angiography. The Indian journal of radiology & imaging. 2016 Apr-Jun:26(2):201-5. doi: 10.4103/0971-3026.184423. Epub [PubMed PMID: 27413266]
Wesselhoeft H, Fawcett JS, Johnson AL. Anomalous origin of the left coronary artery from the pulmonary trunk. Its clinical spectrum, pathology, and pathophysiology, based on a review of 140 cases with seven further cases. Circulation. 1968 Aug:38(2):403-25 [PubMed PMID: 5666852]
Level 3 (low-level) evidenceSurhonne PS, Bogle SR, Bhairappa S, Gupta AK. Percutaneous coronary intervention in a case of anomalous single coronary artery. BMJ case reports. 2016 May 17:2016():. doi: 10.1136/bcr-2016-215651. Epub 2016 May 17 [PubMed PMID: 27190134]
Level 3 (low-level) evidenceDesmet W, Vanhaecke J, Vrolix M, Van de Werf F, Piessens J, Willems J, de Geest H. Isolated single coronary artery: a review of 50,000 consecutive coronary angiographies. European heart journal. 1992 Dec:13(12):1637-40 [PubMed PMID: 1289093]
Level 2 (mid-level) evidenceAl-Mohaissen M, Heilbron B, Leipsic J, Ignaszewski A. Anomalous origin of the entire coronary system by three separate ostia within the right coronary sinus--a rarely observed coronary anomaly. The Canadian journal of cardiology. 2010 Jun-Jul:26(6):206-8 [PubMed PMID: 20548983]
Level 3 (low-level) evidenceAngelini P. Coronary artery anomalies: an entity in search of an identity. Circulation. 2007 Mar 13:115(10):1296-305 [PubMed PMID: 17353457]
Kastellanos S, Aznaouridis K, Vlachopoulos C, Tsiamis E, Oikonomou E, Tousoulis D. Overview of coronary artery variants, aberrations and anomalies. World journal of cardiology. 2018 Oct 26:10(10):127-140. doi: 10.4330/wjc.v10.i10.127. Epub [PubMed PMID: 30386490]
Level 3 (low-level) evidenceBelostotsky V, Veljanovska L, Hristov N, Mitrev Z. Dual left anterior descending artery distribution. Interactive cardiovascular and thoracic surgery. 2010 Apr:10(4):648-9. doi: 10.1510/icvts.2009.221820. Epub 2010 Jan 15 [PubMed PMID: 20080532]
Level 3 (low-level) evidenceSpindola-Franco H, Grose R, Solomon N. Dual left anterior descending coronary artery: angiographic description of important variants and surgical implications. American heart journal. 1983 Mar:105(3):445-55 [PubMed PMID: 6829406]
Manchanda A, Qureshi A, Brofferio A, Go D, Shirani J. Novel variant of dual left anterior descending coronary artery. Journal of cardiovascular computed tomography. 2010 Mar-Apr:4(2):139-41. doi: 10.1016/j.jcct.2009.12.007. Epub 2010 Jan 14 [PubMed PMID: 20430346]
Level 3 (low-level) evidenceDadkhah-Tirani H, Salari A, Shafighnia S, Hosseini SF, Naghdipoor M. Coronary artery to pulmonary artery fistula. The American journal of case reports. 2013:14():486-8. doi: 10.12659/AJCR.889416. Epub 2013 Nov 18 [PubMed PMID: 24298301]
Level 3 (low-level) evidenceBarsoum EA, Saiful FB, Asti D, Morcus R, Khoueiry G, Lafferty J, McCord DA. Rare case of coronary to pulmonary vein fistula with coronary steal phenomenon. World journal of cardiology. 2014 Jul 26:6(7):682-4. doi: 10.4330/wjc.v6.i7.682. Epub [PubMed PMID: 25068029]
Level 3 (low-level) evidenceRusso FD, Ahmadian HR, Slim AM. Anomalous left anterior descending artery to coronary sinus fistula with associated localized ischemia: A clinical dilemma. The American journal of case reports. 2014:15():107-10. doi: 10.12659/AJCR.890002. Epub 2014 Mar 10 [PubMed PMID: 24644528]
Level 3 (low-level) evidenceCakar MA, Tatli E. Coronary-cameral fistula with angina pectoris. Case reports in medicine. 2010:2010():362532. doi: 10.1155/2010/362532. Epub 2010 Dec 6 [PubMed PMID: 21209744]
Level 3 (low-level) evidenceFuruichi S, Sangiorgi GM, Palloshi A, Godino C, Airoldi F, Montorfano M, Chieffo A, Michev I, Carlino M, Colombo A. Drug-eluting stent implantation in coronary trifurcation lesions. The Journal of invasive cardiology. 2007 Apr:19(4):157-62 [PubMed PMID: 17404400]
Morales AR, Romanelli R, Tate LG, Boucek RJ, de Marchena E. Intramural left anterior descending coronary artery: significance of the depth of the muscular tunnel. Human pathology. 1993 Jul:24(7):693-701 [PubMed PMID: 8319950]
Ilia R, Rosenshtein G, Weinstein J, Cafri C, Abu-Ful A, Gueron M. Left anterior descending artery length in left and right coronary artery dominance. Coronary artery disease. 2001 Feb:12(1):77-8 [PubMed PMID: 11211170]
Level 2 (mid-level) evidenceKobayashi N, Maehara A, Mintz GS, Wolff SD, Généreux P, Xu K, Mehran R, Gibson CM, Brener SJ, Stone GW. Usefulness of the Left Anterior Descending Artery Wrapping Around the Left Ventricular Apex to Predict Adverse Clinical Outcomes in Patients With Anterior Wall ST-Segment Elevation Myocardial Infarction (an INFUSE-AMI Substudy). The American journal of cardiology. 2015 May 15:115(10):1389-95. doi: 10.1016/j.amjcard.2015.02.034. Epub 2015 Feb 19 [PubMed PMID: 25770973]
Level 1 (high-level) evidence