Introduction
Renal angiomyolipomas are the most prevalent benign renal tumor.[1] First described by Grawitz in 1900, renal angiomyolipomas are generally highly vascular tumors primarily composed of blood vessels, smooth muscles, and mature adipose tissues and are distinguished by perivascular epithelioid differentiation.[2] Although most of these tumors are often discovered incidentally during radiological imaging, symptomatic presentations such as flank pain, gross hematuria, or severe retroperitoneal hemorrhage may also exist.[3]
Angiomyolipomas are occasionally misidentified as hamartomas. However, while both are benign, the distinction lies in their nature—an angiomyolipoma is a genuine tumor, whereas a hamartoma constitutes a disorganized aggregation of normal local tissue and cells, often resulting from trauma, infection, infarction, obstruction, or hemorrhage. Notably, although Grawitz first described renal angiomyolipomas, they are distinct from Grawitz tumors, which are malignant renal cell carcinomas and hypernephromas.
Imaging plays a central role in the diagnosis and management of renal angiomyolipomas.[4] These tumors comprise variable amounts of 3 tissue elements—vascular, muscular, and adipose. These variations manifest in distinct pathological, radiological, and clinical features.[5]
The key diagnostic criterion of classic angiomyolipoma is identifying a significant amount of adipose tissue on radiological imaging.[6] Although considered benign, the tumors may extend into the surrounding perirenal fat or renal sinus, as well as nearby organs and lymphatics.[7] Rare reports also indicate isolated instances of tumor thrombi extending through the renal vein into the vena cava.[8][9]
Management of angiomyolipomas is determined by factors such as clinical symptoms, tumor size, number, growth pattern, and potential for malignancy.[2][5] For instance, epithelioid angiomyolipoma of the kidney, a rare subtype of angiomyolipoma, is regarded as potentially malignant.[10]
Etiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Etiology
Renal angiomyolipomas primarily occur as isolated "sporadic" cases (80%) but may also be associated with certain "hereditary" conditions (20%), such as tuberous sclerosis and pulmonary lymphangioleiomyomatosis.[3][5] Although the exact cause of sporadic cases remains unknown, some researchers postulate that it results from a spontaneous genetic mutation.[6]
Hereditary angiomyolipomas, associated with tuberous sclerosis complex or pulmonary lymphangioleiomyomatosis, result from a genetic mutation in the TSC1 or TSC2 genes.[5][11] These mutations affect the mammalian target of rapamycin (mTOR), which explains the response of these angiomyolipomas to mTOR inhibitors such as sirolimus and everolimus.[12]
In contrast to the more common sporadic type, hereditary angiomyolipomas, which are associated with tuberous sclerosis or lymphangioleiomyomatosis, are likely to be larger, begin at an earlier age, display multicentric and bilateral growth, have a faster growth rate, be prone to hemorrhage, contribute to chronic renal failure, and be more prone to malignant transformation.[13][14]
Causes of Renal Angiomyolipomas
Tuberous sclerosis: This is a rare, familial, autosomal dominant genetic disorder characterized by the development of benign tumors in various body parts, typically affecting the brain, eyes, heart, lungs, and kidneys. Tuberous sclerosis is also associated with adenoma sebaceum, epilepsy, and mental deficiencies. Although the overall incidence of tuberous sclerosis is estimated at about 1 per 10,000 population, it is believed to be significantly underdiagnosed. Tuberous sclerosis does not exhibit racial or sexual predilection. Over 50% of individuals with tuberous sclerosis will eventually develop renal angiomyolipomas.[5]
Tuberous sclerosis is usually diagnosed by the discovery of benign tumors in the brain, kidneys, heart, liver, and lungs. Initial symptoms often manifest as neurological, including seizures, mental deficiencies, autism, behavioral challenges (such as obsessive-compulsive disorder or overly aggressive tendencies), learning disabilities, and developmental delays. In addition, this condition is also associated with various skin manifestations. A definitive diagnosis can usually be made by genetic testing for the TSC1 or TSC2 genes.[15] Please see StatPearls' companion resource, "Tuberous Sclerosis," for further information.
Patients diagnosed with tuberous sclerosis typically present with larger renal angiomyolipomas (32% exceeding 3 cm), a higher likelihood of bilateral and multifocal lesions, and double the rate of tumors that increase in size over time compared to individuals without tuberous sclerosis who have renal angiomyolipomas.[16][17][18] In addition, the epithelioid variant of angiomyolipoma in these patients is more prone to malignant transformation.[19] Among women with tuberous sclerosis, 40% will exhibit some degree of pulmonary lymphangioleiomyomatosis, as described below.[16][20][21]
Pulmonary lymphangioleiomyomatosis: This is an exceedingly rare genetic disorder often associated with angiomyolipomas and occasionally with tuberous sclerosis. The overall incidence is about 5 cases per 1 million women.[22] This condition typically affects women of childbearing age, causing abnormal smooth muscle cell growth in the lungs.
Pulmonary lymphangioleiomyomatosis causes cyst formation in the lungs, which can affect pulmonary function, resulting in dyspnea. Approximately 45% to 60% of patients will develop angiomyolipomas in the kidneys, commonly occurring in multiple and bilateral patterns.[23][24][25] Pulmonary lymphangioleiomyomatosis should be suspected in younger women with interstitial lung disorders. Diagnosis of this condition can be confirmed through biopsy or elevated serum levels of vascular endothelial growth factor-D (VEGF-D).[26]
Patients newly diagnosed with lymphangioleiomyomatosis should undergo renal imaging to screen for angiomyolipomas. However, routine VEGF-D testing for lymphangioleiomyomatosis is not necessary or recommended for most female patients with angiomyolipomas due to its rarity. Nevertheless, it should be considered in younger women with multiple renal angiomyolipomas, particularly if they also present with pulmonary issues. Please see StatPearls' companion resource, "Lymphangioleiomyomatosis," for further information.[23]
Epidemiology
Renal angiomyolipomas are rare kidney tumors that account for only 0.3% to 3% of all renal neoplasms.[2][27] Their overall prevalence rate ranges from 0.13% to 2.2%, with 80% of cases being sporadic.[6][28][29][30] The remaining 20% are associated with pulmonary lymphangioleiomyomatosis or tuberous sclerosis complex.[28][31]
Renal angiomyolipomas tend to occur in females more than males, possibly due to estrogenic effects.[5] Additionally, the size of the tumors also tends to be larger in females than in males. Pregnant women experience an accelerated growth rate of angiomyolipomas and face a higher risk of hemorrhagic complications.[32][33][34][35] Furthermore, exogenous estrogen therapy appears to increase the risk of bleeding from angiomyolipomas.[36]
At least 50% to 75% of individuals diagnosed with tuberous sclerosis will develop renal angiomyolipomas.[5][37][38][39] In these cases, the incidence of renal angiomyolipomas increases with age, and the initial presentation of the tumors manifests at a younger age compared to those without tuberous sclerosis (12 versus 24 years).[37]
Pathophysiology
The exact cause of sporadic renal angiomyolipoma remains unknown.[5][28] All three tissue types appear to originate from pericytes (microcirculatory mural cells), as they express pericyte markers, except for the endothelial cell components that develop independently.[40][41]
A mutation in either the TSC1 or TSC2 gene causes tuberous sclerosis and hereditary renal angiomyolipomas.[2][11] These 2 genes encode the essential proteins hamartin and tuberin, respectively.[3][5] The interaction between these 2 proteins forms a tumor suppressor complex, which inhibits the mTOR pathway, crucial for cell growth, angiogenesis, and protein synthesis.[5][28]
Deficient or defective TSC1 or TSC2 gene activity leads to unchecked activation of the mTOR pathway, which results in the formation of angiomyolipomas.[5][16][42] This mechanism also elucidates the effectiveness of mTOR inhibitors such as sirolimus and everolimus in reducing tumor size and restraining their growth.
Angiomyolipomas are generally considered benign, with a very low likelihood of malignant transformation.[16] The vast majority (90%) of angiomyolipomas are asymptomatic and tend to remain stable in size. However, only about 9% may gradually increase in size over time.[43] Larger tumors (>6 cm) are more likely to enlarge, grow, hemorrhage, and develop symptoms.[43]
Histopathology
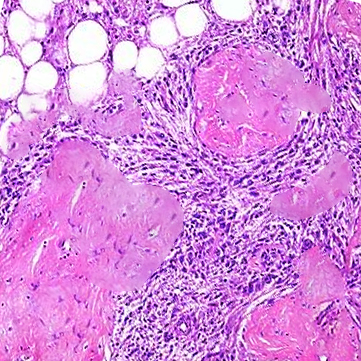
Renal angiomyolipomas can be histologically classified as either typical (triphasic) or atypical (monophasic or epithelioid).[3][44] Triphasic angiomyolipomas typically consist of 3 tumor components—blood vessels or vascular cells, smooth muscle cells, and mature adipocytes. Monophasic angiomyolipomas usually have a major component, while other elements are typically present only in minimal amounts (see Image. Histology, Renal Angiomyolipoma).[3][6][44]
Gross Pathology Descriptions
- The mean tumor size measures 6 cm, with a range varying from 0.5 to 25 cm.[3]
- A "large" tumor is considered larger than 6 cm, previously defined as 4 cm.
- Tumors exhibit a circumscribed morphology and lack encapsulation.[45]
- Cystic components within tumors are rare.[44]
- Tumors are usually unifocal and unilateral, although bilateral or multiple occurrences may suggest an underlying tuberous sclerosis.[44]
- Cut surface appearances vary, displaying gray-white (smooth muscle component), red (vascular component), or yellow (adipose component) colors. The characteristic golden-yellow appearance depends on fat content.[4]
- Invasion into the local perinephric fat, lymphatics, or renal sinus may or may not occur.[3]
- Occasionally, tumors may invade the renal vein, forming a tumor thrombus akin to malignant renal tumors, potentially extending intracardiacly and leading to pulmonary embolisms.[8][9]
- Epitheliod variant typically exhibits minimal fat and may demonstrate metastasis in up to a third of cases.[46]
Microscopic Pathology Descriptions
- The classic triphasic renal angiomyolipoma exhibits thick-walled, dysmorphic, disorganized, hyalinized blood vessels without elastic lamina.[47]
- The tumor comprises mature adipose tissue and myoid spindle cells.[45]
- The epithelioid variant typically features predominant polygonal cells with densely eosinophilic cytoplasm. Hemorrhage, necrosis, epithelioid cellular proliferation, and mitotic figures are commonly observed.[45][48]
- Renal angiomyolipomas with epithelial cysts display cysts lined by cuboidal or hobnail epithelial cells.[45]
Biomarkers
Renal angiomyolipomas are comprised of 3 cell types, all originating from pericytes.[5][40] These cell types include atypical smooth-muscle-like, adipocyte-like, and epithelioid cells.[5] Accordingly, they express pericyte markers.[40][45] Notably, the expressed pericyte markers include angiotensin II type 1 receptor, platelet-derived growth factor receptor-β desmin, calponin, α-smooth muscle actin, and VEGF receptor 2. However, the endothelial cell or adipocyte markers, including CD31 and S-100, are not expressed.[40]
Renal angiomyolipomas typically exhibit strong positive staining for estrogen, progesterone, and androgen receptors, partially explaining their increased incidence in females and the observed accelerated growth in patients undergoing hormonal therapy.[49]
Melanocytic markers for spindle cells, such as HMB-45 and Melan-A, can help differentiate angiomyolipomas from various sarcomas.[16][48]
Malignant angiomyolipomas are extremely rare. Risk factors include lesions without fat on imaging, pure epithelioid histology, size exceeding 7 cm, and signs of intralesional necrosis.[16][50][51]
History and Physical
Renal angiomyolipomas are mostly asymptomatic and typically diagnosed incidentally.[5] The advancements in imaging technology have significantly increased the likelihood of diagnosing more cases.[44]
Historically, up to 15% of patients have presented with spontaneous retroperitoneal hemorrhage, also known as Wunderlich syndrome, which can be severe and may result in shock in approximately one-third of cases.[5] Therefore, the primary clinical concern in patients newly diagnosed with renal angiomyolipoma is the risk of potentially life-threatening bleeding.[5] Although spontaneous retroperitoneal bleeding can also be caused by renal cancer or renal cystic disease, angiomyolipomas represent the most common etiology.
Possible clinical signs and symptoms include flank pain, palpable abdominal mass, urinary tract infection, hematuria, anemia, severe renal insufficiency, and shock.[5][6] Although classic renal angiomyolipomas generally have a benign prognosis, the epithelioid variant can potentially undergo malignant transformation.[16]
Renal angiomyolipomas associated with tuberous sclerosis complex or pulmonary lymphangioleiomyomatosis tend to be more aggressive. They typically manifest at an earlier age, are more likely to be bilateral and multifocal, and are larger at the time of initial diagnosis compared to the more common sporadic variety.[4][5][52]
Evaluation
Renal angiomyolipomas can manifest as either fat-rich, fat-poor, or fat-invisible masses.[53] Approximately 5% of angiomyolipomas belong to the fat-poor or fat-invisible variety.[54][55][56]
Computed tomography (CT) scanning is typically the initial imaging modality used, often performed for unrelated reasons, and may be adequate for diagnosis. If CT scanning is inconclusive or further characterization is needed, magnetic resonance imaging (MRI) is preferred as it is more sensitive in identifying fat-poor angiomyolipomas.[43][57] In addition, CT scans offer advantages such as widespread availability, lower cost, easier access, and rapid performance. However, for fat-invisible lesions, neither CT nor MRI may suffice, necessitating either a percutaneous biopsy or surgical intervention for accurate diagnosis.[43]
The cornerstone for identifying renal angiomyolipomas is diagnostic imaging. In most cases, a biopsy is not necessary for diagnosis, but it may be required to rule out malignancy in challenging or complex cases, such as fat-invisible renal mass lesions.
Imaging Modalities for Diagnosing Renal Angiomyolipomas
Various imaging techniques aid in the diagnosis of this condition, as outlined below.
Ultrasonography: A hyperechoic lesion accompanied by posterior acoustic shadowing is a typical feature of fat-rich angiomyolipomas.[6] Renal masses may present as isoechoic or hyperechoic, and up to 30% of smaller renal cell cancers can also exhibit hyperechogenicity.[5][6][58] The echogenicity of the lesion directly correlates with the total fat content.[31] Fat-poor angiomyolipomas have a mixed echogenicity.[4][5] Fat-invisible angiomyolipomas are homogeneously isoechoic.[6] Unfortunately, approximately one-third of renal cell carcinomas can exhibit similar features on ultrasound.[58] Consequently, the ultrasonographic findings in angiomyolipomas are frequently inconclusive, necessitating further investigations.[5][6] The overall diagnostic accuracy of ultrasound in the initial diagnosis of angiomyolipomas is approximately 78%.[59]
The combination of tumor size with ultrasonic image-based quantitative textural analysis improves the capability of ultrasonography to differentiate hyperechoic renal cell carcinomas from angiomyolipomas with increased accuracy.[60] Ultrasonography proves particularly valuable in monitoring the progression of known lesions, given its cost-effectiveness and minimal ionizing radiation exposure.[16][61]
If ultrasound reveals significant growth (>2.5 mm per year) or indicates a potentially serious change, it is advisable to proceed with a CT or MRI scan. Using the same imaging modality for tracking disease progression allows for more accurate comparisons over time. Therefore, a baseline ultrasound is advised for all newly diagnosed angiomyolipomas.[60]
Computed tomography: In CT scanning, the presence of a region of interest (ROI) exhibiting attenuations of −10 Hounsfield units (HU) or less is considered a dependable and reliable indicator of fatty tissue, which is typically prominent in most fat-rich angiomyolipomas.[5][6] However, CT scans may not depict a hypoattenuating area of less than −10 HU in fat-poor angiomyolipomas.[62] Thus, proper placement of the ROI is crucial for detecting the fatty tissue component of suspicious renal masses. Attenuation values will be higher if the ROI is positioned on the muscular or vascular component compared to when it is placed on fatty tissue.[5][6]
Fat-poor angiomyolipomas typically present as heterogeneously hyperattenuating or isoattenuating lesions. Conversely, fat-invisible angiomyolipomas demonstrate homogenously hyperattenuating characteristics due to the insufficient presence of adipose tissue, resulting in no density decrease. In such cases, an MRI is usually the next recommended imaging step. However, in selected cases where there is a high suspicion of active bleeding, a contrast-enhanced CT scan may be warranted to identify tortuous blood vessels prone to hemorrhage before considering therapeutic embolization.[5][6][63]
In rare instances, renal cell carcinomas may exhibit hyperattenuation on CT (HU < −20), similar to renal angiomyolipomas. However, homogeneous lesion enhancement and prolonged enhancement patterns observed on biphasic helical CT scans can aid in distinguishing fat-poor AML from renal cell carcinoma. Additionally, the presence of calcification within the fat also favors renal cell carcinoma over renal angiomyolipoma.[64]
The accuracy of detecting most renal angiomyolipomas is comparable between MRI and CT scans. However, MRI is deemed superior in detecting fat-poor lesions and is preferred in pediatric cases due to its avoidance of ionizing radiation. Children may require sedation during MRI scans due to extended study times.[5][6][31][65]
In a study involving 98 pathologically confirmed fat-poor angiomyolipomas, 23% were detectable on MRI but not on CT scans.[66] The classical approach for diagnosing this condition involves identifying the fatty component within the mass.[5] The most effective approach to locating the fatty area is comparing T1-weighted MRI images with and without selective fat suppression.[6]
Fat-rich angiomyolipomas typically exhibit T1 hypointensity (dark) with fat suppression and T1 and T2 hyperintensity (bright) without fat suppression. However, it is noteworthy that this T1 hyperintensity can also be observed in other conditions, such as renal cell carcinomas and hemorrhagic cysts, making it a non-specific or definitive diagnostic.[6][31]
On the other hand, fat-poor renal angiomyolipomas tend to appear heterogeneous or isointense to renal parenchyma. The primary diagnostic challenge lies in identifying a fat-invisible mass, which contains insufficient fat to demonstrate the hypointensity on fat-suppressed images necessary for diagnosis. Typically, these lesions appear homogeneously hypointense compared to the renal parenchyma due to their minimal fat content.[63][67]
Fat-invisible angiomyolipomas typically appear homogeneously hypointense on T2-weighted MRI images, whereas renal cell carcinomas, particularly the clear cell subtype, often present as hyperintense lesions. Notably, papillary renal cell carcinoma can also manifest as a hypointense mass on T2-weighted images.[5][66]
The application of MRI artifacts has been proposed for improved evaluation of fat content in renal tumors. One such artifact is the India ink artifact, which creates a black line at the boundary between fat and water. Applying this type of artifact can aid in diagnosing small tumors and fat-poor angiomyolipomas. Additionally, phase versus opposed-phase chemical shift MRI can help differentiate between fat-poor angiomyolipomas and renal cell carcinomas.[5][6]
Gadolinium contrast is generally unnecessary for diagnosis, but it can be useful in identifying internal aneurysms and assessing general vascularity. The reported overall sensitivity and specificity of MRI for diagnosing fat-poor angiomyolipomas are 83% and 90%, respectively.[57] In cases where MRI imaging fails to provide a definitive diagnosis, a biopsy may be required.
Percutaneous biopsy: Percutaneous renal biopsies are mainly indicated to differentiate malignant renal lesions from benign masses.[5] As most renal masses show characteristic signs of malignancy and benign lesions, such as angiomyolipomas, which are usually diagnosed reliably with imaging, routine percutaneous biopsies are not generally necessary or recommended.
Lesions that raise the suspicion of malignancy often exhibit necrosis, intralesional calcifications, or lymphadenopathy. In such cases, it is reasonable to proceed directly to surgical resection.[17] The primary objective of a percutaneous renal biopsy is to identify benign lesions that do not necessitate aggressive treatment, particularly in those rare equivocal cases where imaging modalities alone cannot provide a definitive diagnosis.[5][31][68]
A percutaneous renal biopsy is usually reserved for cases where it is challenging to distinguish between a renal cell carcinoma and a fat-invisible or low-fat angiomyolipoma, particularly when both MRI and CT scan images are inconclusive or indecisive.[5][69] Furthermore, a percutaneous renal biopsy may be necessary to differentiate between the papillary subtype of renal cell carcinoma and fat-invisible angiomyolipomas, especially if the lesion is small (<3 cm) and exhibits T2 hypointensity on MRI without any indication of fat within the tumor.[5][68] If there is a high possibility of bleeding or the mass is particularly large, opting for appropriate surgical intervention upfront might be the preferable course of action to mitigate hemorrhagic complications.[5][6] The risk of significant bleeding following a percutaneous biopsy in fat-invisible angiomyolipomas is no greater than for other renal masses.[70]
Renal nuclear scanning: Renal nuclear scanning with technetium-99m (Tc) mercapto-acetyl-triglycine (MAG3) renography is not generally used in the routine evaluation of angiomyolipomas and other renal masses. However, it evaluates relative renal function, which can be helpful before nephron-sparing surgery. Furthermore, it may help differentiate renal masses and pseudomasses in cases where other imaging modalities are ambiguous.
Evaluation Summary
- Although ultrasonography can be useful for detecting or monitoring a renal mass, it is not considered reliable enough for an initial diagnosis.
- CT scanning is generally the first line of approach in the initial evaluation of renal masses and is often sufficient for diagnostic information.
- In cases where CT scans yield inconclusive results, MRI proves more sensitive, particularly for identifying fat-poor angiomyolipomas.[43][57]
- For certain fat-invisible lesions, no imaging modality offers a definitive diagnosis, necessitating either a percutaneous biopsy or direct surgical intervention.[43]
- If none of these methods can confidently diagnose the mass lesion, it should be managed as a renal malignancy.
Treatment / Management
Indications for Treatment
Most angiomyolipoma cases are asymptomatic and usually do not require treatment, but therapeutic interventions are required in the minority of cases. In the past, the main indications for intervention included symptoms, suspicion of malignancy, presence of the tumor in women of childbearing age, and size larger than 4 cm.[5] However, recent studies challenge the historical 4 cm size threshold criterion, as only 30% of renal angiomyolipomas larger than 4 cm were symptomatic.[5]
Other studies revealed that using the 4 cm criterion as a predictor of hemorrhage has lower specificity than the tumoral aneurysmal diameter of larger than 5 mm. The degree and complexity of the lesion's vascularity (multiple, large, tortuous vessels, aneurysms) is a better general indicator of the need for an intervention to prevent significant bleeding.[31]
This evidence suggests that the vascularity of the angiomyolipoma and the size of any related aneurysms are ultimately more significant than the tumor size alone.[31] The average size of angiomyolipomas treated for bleeding is 7 cm, and recommendations for treatment based on tumor size range from 3 to 8 cm.[65] Using a size criteria larger than 6 cm avoids presumed overtreatment in 65% of patients and is a reasonable compromise.[71](B2)
Additional high-risk factors for predicting bleeding from angiomyolipomas include an exophytic growth pattern, clinical symptoms (flank pain, nausea, and hematuria), female gender, age 15 to 50, increased lesional vascularity, intralesional aneurysms larger than 5 mm in diameter, obesity, and the TSC2 genetic variant.[65][71][72][73][74][75](B2)
During pregnancy, angiomyolipomas pose heightened concerns due to elevated estrogen receptor levels, leading to potential enlargement and a relatively high rate of reported hemorrhage.[76][77][78] If an angiomyolipoma is found in a pregnant female, management of these lesions should align with renal tumor protocols, emphasizing individualized treatment.[79] Recommendations from the European Association of Urology and the Canadian Best Practice Report advocate for considering and discussing angiomyolipoma treatment options with reproductive-age women diagnosed with these lesions.[76][80][81](A1)
Currently, the European Association of Urology, the Canadian Urological Association, and the International Consensus Statement from the European Rare Kidney Disease Reference Network (ERKNET) Working Group for Autosomal Dominant Structural Kidney Disorders and the ERA Genes & Kidney Working Group concur that intervention should be considered in well-selected cases considering the following criteria:[13][65][80][81][82](A1)
- Female patients requiring ongoing hormonal therapy
- individuals in high-risk occupations, such as commercial airline pilots
- Intralesional aneurysms larger than 5 mm
- Large lesions exceeding 6 cm (The European Consensus Guidelines recommend prophylactic embolization starting at 4 cm diameter)
- Lesions that are enlarging more than 2.5 mm per year (>5 mm yearly is considered high risk)
- Presence of multiple lesions
- Tumors in women of childbearing age
- Limited access to emergency care or follow-up
- Poor patient compliance
- Symptomatic manifestations such as persistent pain
Possible interventions include medical treatment with mTOR inhibitors, thermal ablation methods such as radiofrequency ablation and cryotherapy, selective renal artery embolization, and surgical procedures, including total nephrectomy or partial nephron-sparing surgery.[5]
Medical treatment: Hereditary renal angiomyolipomas, linked with conditions such as tuberous sclerosis or pulmonary lymphangioleiomyomatosis, stem from mutations in the TSC1 or TSC2 genes, leading to uncontrolled activation of the mTOR pathway. This results in cell overgrowth and the formation of angiomyolipomas.[11][12]
Medications that inhibit the mTOR pathway can interrupt tumor progression and promote the reversion of the existing lesions.[5][12] Some recommendations suggested that all hereditary angiomyolipoma lesions 3 cm or larger be treated with mTOR inhibitor medications, even if asymptomatic.[1][5][83](A1)
The introduction of sirolimus marked the inception of mTOR inhibitor therapy used to treat hereditary angiomyolipomas.[84] Initial studies with sirolimus showed a greater volume reduction in fat-poor lesions than in fat-rich angiomyolipomas. The reduction in excessive tumoral vascularity and associated aneurysms were markedly reduced by 75% to 100%, particularly notable in areas of the tumor with heightened vascularity and low-fat content.[85](A1)
Everolimus, also an mTOR inhibitor, is approved by the US Food and Drug Administration (FDA) for treating hereditary angiomyolipomas in tuberous sclerosis complex cases.[5][16][28][83][86] Everolimus has been reported to reduce tumor size by an average of 55%.[87] The standard starting dosage of everolimus is 10 mg daily. Continued efficacy has been reported, even after 4 years of treatment.[88](A1)
After 6 months of standard everolimus therapy, patients with at least a 50% reduction may transition to a low daily dose of 5 mg everolimus. This adjustment aims to minimize adverse effects while effectively managing angiomyolipoma growth.[89] Patients who fail to meet this requirement remain on the standard 10 mg daily dosage of everolimus for an additional 6 months before transitioning to the lower dosage, regardless of their response.[89] This decision was based on research indicating that angiomyolipoma tumor size stabilizes six months after initiating everolimus therapy. This stabilization effect parallels those observed in patients with tuberous sclerosis and subependymal giant cell astrocytomas.[83][90][91](A1)
Intermittent everolimus therapy for controlling angiomyolipomas in tuberous sclerosis patients has been explored. Approximately 30% of participants in a study experienced angiomyolipoma stabilization upon everolimus withdrawal.[92] The remainder had to resume everolimus, and all responded with a significant reduction in angiomyolipoma tumor size, similar to the initial response.[92]
Using everolimus for sporadic angiomyolipomas poses challenges. A 2020 phase II multi-institutional prospective trial showed promising efficacy in patients with 4 to 6 cycles of monthly everolimus therapy based on response and tolerability.[83] Over 50% of patients experienced a tumor size reduction of 25% or more, but nearly 50% faced adverse effects or toxicities that required modification or discontinuation of therapy.[86] This compares favorably with the EXIST-2 trial findings of everolimus use in hereditary angiomyolipomas.[83](A1)
Medical therapy of angiomyolipomas is preferred over surgical management, whenever possible, to preserve renal function.[65][93] Although mTOR inhibitors are preferred, they carry risks of adverse effects, sometimes necessitating dose adjustment or discontinuation if the patient manifests severe adverse effects.[94] The most common adverse effects of these medications include amenorrhea, delayed wound healing, dyslipidemia, fatigue, hyperglycemia, hypophosphatemia, infections, lymphopenia, microcytic anemia, non-infectious pneumonitis, proteinuria, stomatitis, and thrombocytopenia.[14](A1)
Thermal ablative therapy: Thermal ablative therapy, including radiofrequency ablation and cryotherapy, has been recommended by the American Urological Association Guidelines for treating small renal masses (<3 cm).[82][95]
Specific selection depends on patient characteristics, tumor size, location, local experience, and expertise. Thermal ablative therapies, such as cryotherapy and radiofrequency ablation, offer the advantage of being performed simultaneously with percutaneous renal biopsies and effectiveness regardless of tumor characteristics. However, selective angiographic embolization, requiring a clear diagnosis before treatment, remains the preferred minimally invasive procedure for angiomyolipomas.[82]
- Cryotherapy: Cryotherapy has demonstrated success in a limited number of angiomyolipoma cases, yet routine recommendation lacks sufficient reported data.[31][95][96][97]
- Cryotherapy has some unique advantages, including minimal damage to surrounding renal tissue, a well-defined area of ablation, the ability to obtain a tissue biopsy during the same procedure, percutaneous accessibility, minimally invasive technology, and the delivery of definitive treatment to the mass regardless of whether it is eventually determined to be a benign or malignant lesion.
- Cryotherapy may be a reasonable option in institutions experienced with this modality. This is especially true when arterial embolization has failed or is not technically feasible. Cryotherapy is also suitable when minimal loss of renal function is desired or when diagnostic uncertainty exists. Additionally, it is an alternative to surgical resection for patients with preexisting renal failure or extensive bilateral disease.
- Participation in a clinical trial is advisable when contemplating renal cryoablation. While no data exist on combining angiographic embolization with renal cryotherapy, it could theoretically offer superior outcomes for larger tumors.
(B3)
- Radiofrequency ablation: Radiofrequency ablation of angiomyolipomas has shown minimal complications and good efficacy. This minimally invasive technique is ideal for small-sized asymptomatic tumors but can also target large masses.[5][31]
- Radiofrequency ablation targets the vascular and solid elements of the tumor mass without damaging normal renal tissue. Studies evaluating its efficacy for small renal angiomyolipomas have been conducted in relatively small series.[5]
- In a study involving 15 small renal angiomyolipomas, successful treatment resulted in the complete disappearance of tumoral enhancement on CT scan, with a low complication rate (13%) observed at a mean follow-up of 21 months.[98]
- In another study, radiofrequency ablation of a sporadic angiomyolipoma in a patient with a solitary kidney resulted in a reduction in tumor size from 4.5 cm to 2.9 cm. Also, no complications or tumor recurrence were observed at the 12-month follow-up.[99][100]
- A separate study used radiofrequency ablation to treat 4 large angiomyolipomas without any reported complications or hemorrhagic events. Over a 48-month follow-up period, the study demonstrated a significant decrease in the mean soft-tissue-to-total tumor ratio, although the total tumor volume did not significantly change.[100][101]
- CT scans usually guide the procedure, but ultrasonography is a possible alternative.[5]
- CT scan guidance becomes necessary for tumors that are not easily and safely targeted by ultrasonography. Numerous studies have shown that combining superselective renal artery embolization with radiofrequency ablation yields excellent success rates.[102] Further studies are needed to determine whether this approach offers superior long-term outcomes compared to alternative treatment.
(B3)
Selective renal artery embolization: In the past, the main treatment for angiomyolipoma was surgical excision because tumor malignancy could not be excluded.[3][16] Currently, selective renal artery embolization is considered the first-line treatment for angiomyolipoma, particularly in cases at risk of acute bleeding, active hemorrhaging, or hemodynamic instability.[5][14][31][80][82][103] (A1)
Selective renal artery embolization is the preferred procedural option, with surgery and thermal ablative therapies considered for cases with vascular malformations, uncontrollable symptoms, embolization failures, or diagnostic uncertainty. This approach boasts an impressive success rate of 93%, with an average reduction in tumor size of 38%.[5][14]
When the surgical option is indicated, preoperative embolization of the tumor may be considered to reduce the complications and the difficulty of the operation.[4][5][31] However, embolization is not a risk-free option and is associated with a relatively high rate of adverse effects (43%).[5] Most of these adverse effects are related to a post-embolization syndrome characterized by flank pain, fever, vomiting, nausea, and leukocytosis during the first 3 days after the procedure.[5][16]
These adverse effects are generally managed conservatively with standard supportive care. Prescribing antipyretics, antiemetics, and analgesia prophylactically could significantly minimize and control these symptoms.[5][16][80] Despite this, up to 30% of cases undergoing selective arterial embolization may require a second treatment primarily due to tumor revascularization.[14][81](A1)
Surgical excision: Surgical excision, whether total or partial nephrectomy, remains the definitive treatment option for completely eradicating the renal mass.[3] Every surgical operation should use a nephron-sparing approach whenever possible.[3][14][82] Preserving renal parenchyma is particularly crucial in patients with hereditary angiomyolipomas due to their elevated recurrence rate, accelerated growth, and multifocal disease pattern.[5] (B2)
Nephrectomy is usually indicated when the suspicion of malignancy is high, the renal mass is particularly large, or other treatment modalities cannot be performed.[5] However, in an emergency, a nephrectomy can be a lifesaving option.[5][11][104] Surgical excision offers the advantage of a higher cure rate and requires fewer follow-up procedures, resulting in reduced surveillance compared to arterial embolization.(B3)
Active Surveillance and Follow-Up
Active surveillance is generally used to monitor the progression of existing tumors and the emergence of new ones when treatment indications are absent, following the completion of diagnostic evaluations.[5][43] Given the low overall rate of spontaneous bleeding from angiomyolipomas (only 2%), surveillance is considered reasonable in most cases.[81] Currently, definitive guidelines do not exist for sporadic angiomyolipomas in terms of frequency or modality of imaging studies.[5][31] Decisions should be guided according to the individual clinical scenario and history.[31][105] (A1)
An annual repeat of imaging may be appropriate for small and solitary lesions.[6] For hereditary tumors (associated with tuberous sclerosis or lymphangioleiomyomatosis), multiple, bilateral, and larger lesions, biannual (twice annual) imaging is generally recommended at least initially, as some have been found to grow up to 1.5 cm a year.[43][106]
For hereditary angiomyolipomas, the International Tuberous Sclerosis Complex Consensus highly recommends using a yearly MRI to monitor the condition due to its higher sensitivity in detecting adipose tissue.[5] Most published protocols start with biannual imaging, which may be modified to annual screenings if there is little change.[43] In these cases, blood pressure and renal function should be checked at least annually.[5]
Ultrasound is usually adequate for monitoring, with a CT scan or MRI reserved for significant changes. Avoiding routine CT scanning to minimize radiation exposure is advisable for long-term follow-up.
Treatment Summary
- Most angiomyolipomas can be managed without treatment.
- Treatment considerations include current symptoms, size (>6 cm), growth rate (>2.5 mm per year), intratumoral aneurysms (>5 mm), high-risk scenarios, and potential lack of access to healthcare.
- The least invasive treatment modality should be used whenever possible, which minimizes complications and loss of renal function.
- Sirolimus and everolimus have demonstrated efficacy in controlling renal tumor growth and inducing shrinkage in hereditary angiomyolipomas. Their effectiveness in sporadic angiomyolipomas is less established, but early studies suggest it can be effective.
- Everolimus carries a high cost and notable adverse effects. Utilizing intermittent treatment and low-dose maintenance protocols can mitigate these challenges and minimize the negative aspects of everolimus administration.
- Thermal therapies such as radiofrequency ablation and cryotherapy offer simultaneous percutaneous biopsies and efficacy across benign and malignant lesions. Their consideration for smaller tumors (<3 cm) should align with local expertise and availability.
- Selective angiographic embolization is the preferred treatment for managing active bleeding and preventing high-risk angiomyolipomas. Despite its effectiveness, it comes with a high retreatment rate of 30% and necessitates a confirmed diagnosis before therapy.
- Surgery is the most definitive curative therapy but is reserved for situations that cannot be better managed with less invasive modalities or require urgent resolution. Nephron-sparing minimally invasive techniques are advocated as the preferred approach.
Differential Diagnosis
Recognizing the various types of renal angiomyolipomas, including fat-rich, fat-poor, and fat-invisible variants, is crucial.[31] Fat-rich angiomyolipomas are diagnosed easily on imaging due to the high amount of adipose tissue in the mass. However, accurately diagnosing renal angiomyolipomas with poor or no fat visualization presents a primary challenge.[5]
Before initiating treatment, it is crucial to rule out other potential differential diagnoses.[5] The possible differential diagnoses of renal angiomyolipoma include adrenal myelolipoma, oncocytoma, renal cell carcinoma, retroperitoneal liposarcoma, tumors metastatic to the kidney, and Wilms tumor.[107][108]
The widespread adoption of high-resolution imaging techniques has increased the detection of small renal masses. Often discovered incidentally, many of these small renal lesions are suspected of possible renal cell carcinomas and managed accordingly. However, approximately 22% of these masses are later determined benign after surgical excision, with renal angiomyolipomas comprising roughly half of these cases.[5]
Differentiating between renal cell carcinomas and angiomyolipomas can pose challenges in routine clinical practice.[62][109][110] A primary diagnostic challenge lies in differentiating fat-poor or fat-invisible angiomyolipomas from renal cell carcinomas.[5]
Clinical information that favors the diagnosis of angiomyolipoma includes female gender, younger age, and asymptomatic clinical manifestations.[5] Imaging results that raise suspicion of angiomyolipoma include the presence of multiple lesions, absence of calcifications, T2-weighted hypointensity on MRI, and hyperattenuation on CT scans.[5] However, it is noteworthy that exceptions exist, and no single clinical finding or imaging modality is definitive in all cases.[5][111][112][113]
Retroperitoneal liposarcomas may also appear similar to larger angiomyolipomas. However, as angiomyolipomas are renal parenchymal tumors, they typically present with an indentation or dimple at the site of origin. In contrast, liposarcoma tends to envelop and compress the entire kidney without a distinct originating marker.[114][115]
Although some angiomyolipomas can grow rapidly, the majority exhibit slow growth. Rapid enlargement (>5 mm per year) should raise suspicion of potential malignancy.[65][116][117][118] Failure of a lesion to respond to mTOR inhibitors is also considered suspicious.[65][119] Suspected malignant lesions, characterized by features such as tumoral necrosis or internal calcifications, regional lymphadenopathy, metastasis, or perinephric extension, typically warrant surgical intervention unless definitively determined to be benign by a percutaneous biopsy.[5][6][17][69]
Prognosis
Small angiomyolipomas and those lacking aneurysms usually carry a favorable prognosis due to a low risk of spontaneous bleeding, estimated at only 2%.[5][81] However, renal angiomyolipomas harboring aneurysms larger than 5 mm or exhibiting a size exceeding 6 cm present a considerable risk of spontaneous rupture and hemorrhage, constituting a life-threatening condition.[16][120] Additionally, the malignant potential of certain angiomyolipoma types serves as another crucial prognostic factor, as they may have a greater tendency for this transformation.[5]
Updated clinical guidelines are essential to assist physicians in determining optimal management strategies for these rare and atypical cases. Progress in treatments or potential cures will necessitate ongoing translational research in genomics, immunotherapy, and other emerging treatment modalities.[121]
Complications
Although renal angiomyolipoma is a rare benign neoplasm, it may still impose significant morbidity and mortality due to the tumor's unique vascular characteristics and the possible complications associated with treatment.[5]
The risk of bleeding associated with renal angiomyolipomas is a challenging factor.[5] Studies have consistently shown that large tumors (>6 cm) and lesions associated with tuberous sclerosis complex are considered more susceptible to bleeding than small sporadic lesions, highlighting the importance of prophylactic treatment.[16]
Another important complication associated with renal angiomyolipomas is the potential for renal impairment over time.[11]
Consultations
The management of renal angiomyolipoma and its complications requires a multidisciplinary team, which can include specialists from various fields such as general surgery, genetics, interventional radiology, nephrology, oncology, pathology, primary care, radiology, urology, and vascular surgery.[5]
Deterrence and Patient Education
Patients with renal angiomyolipoma should be informed about the generally benign nature of the disease.[5] Healthcare professionals should clarify that available medications and minimally invasive treatments can help halt the disease's progression and reduce the risk of spontaneous bleeding.[5] Healthcare providers must also advise patients about the importance of medication adherence and compliance, as well as the need for long-term surveillance.[16]
Any woman of childbearing age diagnosed with renal angiomyolipoma who plans to become pregnant should seek consultation to discuss the management of their condition before and during pregnancy. Pregnancy is considered a significant risk factor for disease progression and complications, as the tumor mass is likely to grow and may rupture during pregnancy.[5][11]
Pearls and Other Issues
- Many experts recommend that all patients with newly diagnosed angiomyolipomas be evaluated for tuberous sclerosis.
- Conversely, all patients with tuberous sclerosis should be screened for angiomyolipomas.
- Renal imaging for angiomyolipomas is advised for all patients with lymphangioleiomyomatosis.
- Asymptomatic patients with lesions smaller than 4 cm are unlikely to have tuberous sclerosis.
- Selective angiographic embolization is the preferred method for controlling active bleeding, although there is a 30% retreatment rate.
- Biopsy should be considered if the diagnosis cannot be conclusively made from CT and MRI scans alone.
- Therapy with the mTOR inhibitors, such as sirolimus and everolimus, should be considered for lesions 3 cm or larger.[122]
- Intermittent mTOR therapy and low-dose maintenance treatment protocols can help mitigate adverse effects and lower medication costs.
- Medical therapy is generally preferred over surgical procedures whenever possible.
- MRI imaging, being more sensitive to detecting fat than CT scans, is particularly valuable for identifying fat-poor lesions. Imaging fat-invisible lesions can pose challenges.
- Over 90% of all angiomyolipomas do not require treatment.
- Prophylactic surgical treatment is recommended for angiomyolipomas larger than 6 cm or those with intralesional aneurysms larger than 5 mm.
- The risk associated with percutaneous biopsy is not higher for patients with low-fat or invisible-fat angiomyolipomas compared to other renal tumors.
- Surgical intervention, employing minimally invasive ablative technologies and nephron-sparing techniques, should be pursued whenever feasible.
- Ultrasound is not sufficiently detailed to definitively diagnose angiomyolipomas but can track previously identified lesions.
- If all diagnostic measures have failed to confirm a reliable diagnosis, treating the lesion as malignant is advisable.
- In cases where a renal mass lesion cannot be definitively identified as an angiomyolipoma from imaging alone but is suspected to be one, a percutaneous biopsy is a reasonable option, particularly for high-risk patients, as it may help avoid potential surgical complications.[69]
- Women of childbearing age should consider prophylactic treatment for angiomyolipomas due to the elevated risk of complications during pregnancy.
Enhancing Healthcare Team Outcomes
Managing renal angiomyolipoma requires the collaboration of the entire interprofessional healthcare team. The primary healthcare provider may be the first to encounter the initial symptomatic presentation of the disease. Radiologists and interventional radiologists are crucial in confirming the diagnosis and monitoring disease progression through imaging. In some cases, a percutaneous renal biopsy may be necessary to confirm the diagnosis.
All medical efforts should prioritize preserving renal function while preventing disease progression and associated complications. Whenever feasible, medical therapy is preferred over nephron-sparing surgeries. Additionally, as detailed previously, other specialists may be consulted to provide expertise on the case.
The nursing staff are critical in patient counseling, surgical assistance, and coordination of interventions between specialties. They also play a vital role in notifying the relevant clinicians of any changes in patient clinical status and addressing patient inquiries. If medical therapy is included in the treatment plan, pharmacists can provide counseling on appropriate dosing and administration, conduct medication reconciliation, and communicate with the prescribing clinician regarding any medication-related concerns.
All interprofessional healthcare team members should meticulously document their activities and interventions in the patient's medical record so that all team members have access to the same up-to-date, accurate information. Open communication among healthcare team members is essential for discussing the case and implementing the best therapeutic options. This collaborative approach ensures optimal patient outcomes.
Media
References
Hatano T, Egawa S. Renal angiomyolipoma with tuberous sclerosis complex: How it differs from sporadic angiomyolipoma in both management and care. Asian journal of surgery. 2020 Oct:43(10):967-972. doi: 10.1016/j.asjsur.2019.12.008. Epub 2020 Jan 17 [PubMed PMID: 31959574]
Al-Thani H, El-Menyar A, Al-Sulaiti M, El-Mabrok J, Hajaji K, Elgohary H, Al-Malki A, Tabeb A. Clinical presentation, management, and outcome of patients with incidental renal angiomyolipoma in qatar. Oman medical journal. 2014 Nov:29(6):419-24. doi: 10.5001/omj.2014.112. Epub [PubMed PMID: 25584159]
Lane BR, Aydin H, Danforth TL, Zhou M, Remer EM, Novick AC, Campbell SC. Clinical correlates of renal angiomyolipoma subtypes in 209 patients: classic, fat poor, tuberous sclerosis associated and epithelioid. The Journal of urology. 2008 Sep:180(3):836-43. doi: 10.1016/j.juro.2008.05.041. Epub 2008 Jul 16 [PubMed PMID: 18635231]
Level 2 (mid-level) evidenceHalpenny D, Snow A, McNeill G, Torreggiani WC. The radiological diagnosis and treatment of renal angiomyolipoma-current status. Clinical radiology. 2010 Feb:65(2):99-108. doi: 10.1016/j.crad.2009.09.014. Epub 2009 Dec 3 [PubMed PMID: 20103431]
Vos N, Oyen R. Renal Angiomyolipoma: The Good, the Bad, and the Ugly. Journal of the Belgian Society of Radiology. 2018 Apr 20:102(1):41. doi: 10.5334/jbsr.1536. Epub 2018 Apr 20 [PubMed PMID: 30039053]
Jinzaki M, Silverman SG, Akita H, Nagashima Y, Mikami S, Oya M. Renal angiomyolipoma: a radiological classification and update on recent developments in diagnosis and management. Abdominal imaging. 2014 Jun:39(3):588-604. doi: 10.1007/s00261-014-0083-3. Epub [PubMed PMID: 24504542]
Minja EJ, Pellerin M, Saviano N, Chamberlain RS. Retroperitoneal extrarenal angiomyolipomas: an evidence-based approach to a rare clinical entity. Case reports in nephrology. 2012:2012():374107. doi: 10.1155/2012/374107. Epub 2012 Jul 12 [PubMed PMID: 24555133]
Level 3 (low-level) evidenceWang D, Liu M, Li Y, Xie M. Right renal angiomyolipoma with intracardiac extension and pulmonary embolism. Journal of clinical ultrasound : JCU. 2021 Jun:49(5):516-519. doi: 10.1002/jcu.22968. Epub 2020 Dec 15 [PubMed PMID: 33325037]
Bouaziz H, Ghalleb M, Tounsi N, Riahi N, Bouzaiene H, Ziadi J, Hassouna JB, Slimane M, Rahal K. A renal angiomyolipoma with a challenging presentation: a case report. Journal of medical case reports. 2021 Sep 27:15(1):477. doi: 10.1186/s13256-021-03073-0. Epub 2021 Sep 27 [PubMed PMID: 34565446]
Level 3 (low-level) evidenceZhan R, Li YQ, Chen CY, Hu HY, Zhang C. Primary kidney malignant epithelioid angiomyolipoma: Two cases report and review of literature. Medicine. 2018 Aug:97(32):e11805. doi: 10.1097/MD.0000000000011805. Epub [PubMed PMID: 30095646]
Level 3 (low-level) evidenceSeyam RM, Alkhudair WK, Kattan SA, Alotaibi MF, Alzahrani HM, Altaweel WM. The Risks of Renal Angiomyolipoma: Reviewing the Evidence. Journal of kidney cancer and VHL. 2017:4(4):13-25. doi: 10.15586/jkcvhl.2017.97. Epub 2017 Oct 16 [PubMed PMID: 29090118]
Wang W, Guo G, Shi G, Wei X, Hu Z, Li H, Zhang X, Ye D, Zhang Y. A Multi-Centric Study Assessing Safety and Efficacy of Everolimus in Adult Chinese Patients With Tuberous Sclerosis Complex Associated Renal Angiomyolipomas. Frontiers in oncology. 2022:12():871723. doi: 10.3389/fonc.2022.871723. Epub 2022 Jul 4 [PubMed PMID: 35860575]
Bhatt JR, Richard PO, Kim NS, Finelli A, Manickavachagam K, Legere L, Evans A, Pei Y, Sykes J, Jhaveri K, Jewett MAS. Natural History of Renal Angiomyolipoma (AML): Most Patients with Large AMLs }4cm Can Be Offered Active Surveillance as an Initial Management Strategy. European urology. 2016 Jul:70(1):85-90. doi: 10.1016/j.eururo.2016.01.048. Epub 2016 Feb 9 [PubMed PMID: 26873836]
Ariceta G, Buj MJ, Furlano M, Martínez V, Matamala A, Morales M, Robles NR, Sans L, Villacampa F, Torra R. Recommendations for the management of renal involvement in the tuberous sclerosis complex. Nefrologia. 2020 Mar-Apr:40(2):142-151. doi: 10.1016/j.nefro.2019.07.002. Epub 2019 Nov 10 [PubMed PMID: 31722796]
Zamora EA, Aeddula NR. Tuberous Sclerosis. StatPearls. 2024 Jan:(): [PubMed PMID: 30860727]
Flum AS, Hamoui N, Said MA, Yang XJ, Casalino DD, McGuire BB, Perry KT, Nadler RB. Update on the Diagnosis and Management of Renal Angiomyolipoma. The Journal of urology. 2016 Apr:195(4 Pt 1):834-46. doi: 10.1016/j.juro.2015.07.126. Epub 2015 Nov 21 [PubMed PMID: 26612197]
Sooriakumaran P, Gibbs P, Coughlin G, Attard V, Elmslie F, Kingswood C, Taylor J, Corbishley C, Patel U, Anderson C. Angiomyolipomata: challenges, solutions, and future prospects based on over 100 cases treated. BJU international. 2010 Jan:105(1):101-6. doi: 10.1111/j.1464-410X.2009.08649.x. Epub 2009 Jun 2 [PubMed PMID: 19493268]
Level 2 (mid-level) evidenceSeyam RM, Bissada NK, Kattan SA, Mokhtar AA, Aslam M, Fahmy WE, Mourad WA, Binmahfouz AA, Alzahrani HM, Hanash KA. Changing trends in presentation, diagnosis and management of renal angiomyolipoma: comparison of sporadic and tuberous sclerosis complex-associated forms. Urology. 2008 Nov:72(5):1077-82. doi: 10.1016/j.urology.2008.07.049. Epub 2008 Sep 21 [PubMed PMID: 18805573]
Level 2 (mid-level) evidenceAydin H, Magi-Galluzzi C, Lane BR, Sercia L, Lopez JI, Rini BI, Zhou M. Renal angiomyolipoma: clinicopathologic study of 194 cases with emphasis on the epithelioid histology and tuberous sclerosis association. The American journal of surgical pathology. 2009 Feb:33(2):289-97. doi: 10.1097/PAS.0b013e31817ed7a6. Epub [PubMed PMID: 18852677]
Level 3 (low-level) evidenceCuratolo P, Bombardieri R. Tuberous sclerosis. Handbook of clinical neurology. 2008:87():129-51. doi: 10.1016/S0072-9752(07)87009-6. Epub [PubMed PMID: 18809023]
Level 3 (low-level) evidenceFranz DN. Everolimus: an mTOR inhibitor for the treatment of tuberous sclerosis. Expert review of anticancer therapy. 2011 Aug:11(8):1181-92. doi: 10.1586/era.11.93. Epub [PubMed PMID: 21916571]
Level 3 (low-level) evidenceHarknett EC, Chang WY, Byrnes S, Johnson J, Lazor R, Cohen MM, Gray B, Geiling S, Telford H, Tattersfield AE, Hubbard RB, Johnson SR. Use of variability in national and regional data to estimate the prevalence of lymphangioleiomyomatosis. QJM : monthly journal of the Association of Physicians. 2011 Nov:104(11):971-9. doi: 10.1093/qjmed/hcr116. Epub 2011 Jul 15 [PubMed PMID: 21764810]
Khaddour K, Sankari A, Shayuk M. Lymphangioleiomyomatosis. StatPearls. 2024 Jan:(): [PubMed PMID: 30480964]
Ryu JH, Moss J, Beck GJ, Lee JC, Brown KK, Chapman JT, Finlay GA, Olson EJ, Ruoss SJ, Maurer JR, Raffin TA, Peavy HH, McCarthy K, Taveira-Dasilva A, McCormack FX, Avila NA, Decastro RM, Jacobs SS, Stylianou M, Fanburg BL, NHLBI LAM Registry Group. The NHLBI lymphangioleiomyomatosis registry: characteristics of 230 patients at enrollment. American journal of respiratory and critical care medicine. 2006 Jan 1:173(1):105-11 [PubMed PMID: 16210669]
Bernstein SM, Newell JD Jr, Adamczyk D, Mortenson RL, King TE Jr, Lynch DA. How common are renal angiomyolipomas in patients with pulmonary lymphangiomyomatosis? American journal of respiratory and critical care medicine. 1995 Dec:152(6 Pt 1):2138-43 [PubMed PMID: 8520787]
McCormack FX, Gupta N, Finlay GR, Young LR, Taveira-DaSilva AM, Glasgow CG, Steagall WK, Johnson SR, Sahn SA, Ryu JH, Strange C, Seyama K, Sullivan EJ, Kotloff RM, Downey GP, Chapman JT, Han MK, D'Armiento JM, Inoue Y, Henske EP, Bissler JJ, Colby TV, Kinder BW, Wikenheiser-Brokamp KA, Brown KK, Cordier JF, Meyer C, Cottin V, Brozek JL, Smith K, Wilson KC, Moss J, ATS/JRS Committee on Lymphangioleiomyomatosis. Official American Thoracic Society/Japanese Respiratory Society Clinical Practice Guidelines: Lymphangioleiomyomatosis Diagnosis and Management. American journal of respiratory and critical care medicine. 2016 Sep 15:194(6):748-61. doi: 10.1164/rccm.201607-1384ST. Epub [PubMed PMID: 27628078]
Level 1 (high-level) evidenceNelson CP, Sanda MG. Contemporary diagnosis and management of renal angiomyolipoma. The Journal of urology. 2002 Oct:168(4 Pt 1):1315-25 [PubMed PMID: 12352384]
Bissler JJ, Kingswood JC. Optimal treatment of tuberous sclerosis complex associated renal angiomyolipomata: a systematic review. Therapeutic advances in urology. 2016 Aug:8(4):279-290 [PubMed PMID: 27928430]
Level 3 (low-level) evidenceFujii Y, Ajima J, Oka K, Tosaka A, Takehara Y. Benign renal tumors detected among healthy adults by abdominal ultrasonography. European urology. 1995:27(2):124-7 [PubMed PMID: 7744154]
Rule AD, Sasiwimonphan K, Lieske JC, Keddis MT, Torres VE, Vrtiska TJ. Characteristics of renal cystic and solid lesions based on contrast-enhanced computed tomography of potential kidney donors. American journal of kidney diseases : the official journal of the National Kidney Foundation. 2012 May:59(5):611-8. doi: 10.1053/j.ajkd.2011.12.022. Epub 2012 Mar 6 [PubMed PMID: 22398108]
Level 2 (mid-level) evidenceKiefer RM, Stavropoulos SW. The Role of Interventional Radiology Techniques in the Management of Renal Angiomyolipomas. Current urology reports. 2017 May:18(5):36. doi: 10.1007/s11934-017-0687-6. Epub [PubMed PMID: 28299630]
Lewis EL, Palmer JM. Renal angiomyolipoma and massive retroperitoneal hemorrhage during pregnancy. The Western journal of medicine. 1985 Nov:143(5):675-6 [PubMed PMID: 4082575]
Level 3 (low-level) evidencePetrikovsky BM, Vintzileos AM, Cassidy SB, Egan JF. Tuberous sclerosis in pregnancy. American journal of perinatology. 1990 Apr:7(2):133-5 [PubMed PMID: 2184812]
Level 3 (low-level) evidenceRaft J, Lalot JM, Meistelman C, Longrois D. [Renal angiomyolipoma rupture during pregnancy]. Gynecologie, obstetrique & fertilite. 2006 Oct:34(10):917-9 [PubMed PMID: 16996772]
Level 3 (low-level) evidenceZapardiel I, Delafuente-Valero J, Bajo-Arenas JM. Renal angiomyolipoma during pregnancy: review of the literature. Gynecologic and obstetric investigation. 2011:72(4):217-9. doi: 10.1159/000329328. Epub 2011 Aug 27 [PubMed PMID: 21876331]
Level 3 (low-level) evidenceGould Rothberg BE, Grooms MC, Dharnidharka VR. Rapid growth of a kidney angiomyolipoma after initiation of oral contraceptive therapy. Obstetrics and gynecology. 2006 Sep:108(3 Pt 2):734-6 [PubMed PMID: 17018483]
Level 3 (low-level) evidenceKingswood JC, Belousova E, Benedik MP, Carter T, Cottin V, Curatolo P, Dahlin M, D' Amato L, d'Augères GB, de Vries PJ, Ferreira JC, Feucht M, Fladrowski C, Hertzberg C, Jozwiak S, Lawson JA, Macaya A, Marques R, Nabbout R, O'Callaghan F, Qin J, Sander V, Sauter M, Shah S, Takahashi Y, Touraine R, Youroukos S, Zonnenberg B, Jansen AC. Renal angiomyolipoma in patients with tuberous sclerosis complex: findings from the TuberOus SClerosis registry to increase disease Awareness. Nephrology, dialysis, transplantation : official publication of the European Dialysis and Transplant Association - European Renal Association. 2019 Mar 1:34(3):502-508. doi: 10.1093/ndt/gfy063. Epub [PubMed PMID: 29697822]
Level 2 (mid-level) evidenceBachour K, House AA, Andrade DM, Connolly M, Debicki DB, Desbiens R, Fantaneanu TA, Kyriakopoulos P, Lattouf JB, Suller-Marti A, Marques PT, Keezer MR. Adults with tuberous sclerosis complex: A distinct patient population. Epilepsia. 2022 Mar:63(3):663-671. doi: 10.1111/epi.17159. Epub 2021 Dec 29 [PubMed PMID: 34967000]
Ewalt DH, Sheffield E, Sparagana SP, Delgado MR, Roach ES. Renal lesion growth in children with tuberous sclerosis complex. The Journal of urology. 1998 Jul:160(1):141-5 [PubMed PMID: 9628635]
Siroky BJ, Yin H, Dixon BP, Reichert RJ, Hellmann AR, Ramkumar T, Tsuchihashi Z, Bunni M, Dillon J, Bell PD, Sampson JR, Bissler JJ. Evidence for pericyte origin of TSC-associated renal angiomyolipomas and implications for angiotensin receptor inhibition therapy. American journal of physiology. Renal physiology. 2014 Sep 1:307(5):F560-70. doi: 10.1152/ajprenal.00569.2013. Epub 2014 Jun 11 [PubMed PMID: 24920756]
Karbowniczek M, Yu J, Henske EP. Renal angiomyolipomas from patients with sporadic lymphangiomyomatosis contain both neoplastic and non-neoplastic vascular structures. The American journal of pathology. 2003 Feb:162(2):491-500 [PubMed PMID: 12547707]
von Ranke FM, Faria IM, Zanetti G, Hochhegger B, Souza AS Jr, Marchiori E. Imaging of tuberous sclerosis complex: a pictorial review. Radiologia brasileira. 2017 Jan-Feb:50(1):48-54. doi: 10.1590/0100-3984.2016.0020. Epub [PubMed PMID: 28298732]
Guo Y, Kapoor A, Cheon P, So AI, Lattouf JB, Jamal M. Canadian Urological Association best practice report: Diagnosis and management of sporadic angiomyolipomas. Canadian Urological Association journal = Journal de l'Association des urologues du Canada. 2020 Nov:14(11):E527-E536. doi: 10.5489/cuaj.6942. Epub [PubMed PMID: 33213697]
Jinzaki M, Silverman SG, Akita H, Mikami S, Oya M. Diagnosis of Renal Angiomyolipomas: Classic, Fat-Poor, and Epithelioid Types. Seminars in ultrasound, CT, and MR. 2017 Feb:38(1):37-46. doi: 10.1053/j.sult.2016.11.001. Epub 2016 Nov 5 [PubMed PMID: 28237279]
Martignoni G, Pea M, Rocca PC, Bonetti F. Renal pathology in the tuberous sclerosis complex. Pathology. 2003 Dec:35(6):505-12 [PubMed PMID: 14660101]
Brimo F, Robinson B, Guo C, Zhou M, Latour M, Epstein JI. Renal epithelioid angiomyolipoma with atypia: a series of 40 cases with emphasis on clinicopathologic prognostic indicators of malignancy. The American journal of surgical pathology. 2010 May:34(5):715-22. doi: 10.1097/PAS.0b013e3181d90370. Epub [PubMed PMID: 20410812]
Level 3 (low-level) evidenceTamboli P, Ro JY, Amin MB, Ligato S, Ayala AG. Benign tumors and tumor-like lesions of the adult kidney. Part II: Benign mesenchymal and mixed neoplasms, and tumor-like lesions. Advances in anatomic pathology. 2000 Jan:7(1):47-66 [PubMed PMID: 10640201]
Level 3 (low-level) evidenceEble JN, Amin MB, Young RH. Epithelioid angiomyolipoma of the kidney: a report of five cases with a prominent and diagnostically confusing epithelioid smooth muscle component. The American journal of surgical pathology. 1997 Oct:21(10):1123-30 [PubMed PMID: 9331283]
Level 3 (low-level) evidenceLogginidou H, Ao X, Russo I, Henske EP. Frequent estrogen and progesterone receptor immunoreactivity in renal angiomyolipomas from women with pulmonary lymphangioleiomyomatosis. Chest. 2000 Jan:117(1):25-30 [PubMed PMID: 10631194]
Christiano AP, Yang X, Gerber GS. Malignant transformation of renal angiomyolipoma. The Journal of urology. 1999 Jun:161(6):1900-1 [PubMed PMID: 10332463]
Level 3 (low-level) evidenceNese N, Martignoni G, Fletcher CD, Gupta R, Pan CC, Kim H, Ro JY, Hwang IS, Sato K, Bonetti F, Pea M, Amin MB, Hes O, Svec A, Kida M, Vankalakunti M, Berel D, Rogatko A, Gown AM, Amin MB. Pure epithelioid PEComas (so-called epithelioid angiomyolipoma) of the kidney: A clinicopathologic study of 41 cases: detailed assessment of morphology and risk stratification. The American journal of surgical pathology. 2011 Feb:35(2):161-76. doi: 10.1097/PAS.0b013e318206f2a9. Epub [PubMed PMID: 21263237]
Level 3 (low-level) evidenceLogue LG, Acker RE, Sienko AE. Best cases from the AFIP: angiomyolipomas in tuberous sclerosis. Radiographics : a review publication of the Radiological Society of North America, Inc. 2003 Jan-Feb:23(1):241-6 [PubMed PMID: 12533658]
Level 3 (low-level) evidencePark BK. Renal Angiomyolipoma: Radiologic Classification and Imaging Features According to the Amount of Fat. AJR. American journal of roentgenology. 2017 Oct:209(4):826-835. doi: 10.2214/AJR.17.17973. Epub 2017 Jul 20 [PubMed PMID: 28726505]
Schieda N, Kielar AZ, Al Dandan O, McInnes MD, Flood TA. Ten uncommon and unusual variants of renal angiomyolipoma (AML): radiologic-pathologic correlation. Clinical radiology. 2015 Feb:70(2):206-20. doi: 10.1016/j.crad.2014.10.001. Epub 2014 Nov 15 [PubMed PMID: 25468637]
Park SG, Park BK. New radiologic classification of renal angiomyolipoma: frequently asked questions. Clinical imaging. 2019 May-Jun:55():156-160. doi: 10.1016/j.clinimag.2019.01.025. Epub 2019 Feb 1 [PubMed PMID: 30825810]
Kim JK, Park SY, Shon JH, Cho KS. Angiomyolipoma with minimal fat: differentiation from renal cell carcinoma at biphasic helical CT. Radiology. 2004 Mar:230(3):677-84 [PubMed PMID: 14990834]
Level 2 (mid-level) evidenceWilson MP, Patel D, Murad MH, McInnes MDF, Katlariwala P, Low G. Diagnostic Performance of MRI in the Detection of Renal Lipid-Poor Angiomyolipomas: A Systematic Review and Meta-Analysis. Radiology. 2020 Sep:296(3):511-520. doi: 10.1148/radiol.2020192070. Epub 2020 Jun 30 [PubMed PMID: 32602827]
Level 1 (high-level) evidenceForman HP, Middleton WD, Melson GL, McClennan BL. Hyperechoic renal cell carcinomas: increase in detection at US. Radiology. 1993 Aug:188(2):431-4 [PubMed PMID: 8327692]
Ascenti G, Zimbaro G, Mazziotti S, Gaeta M, Settineri N, Scribano E. Usefulness of power Doppler and contrast-enhanced sonography in the differentiation of hyperechoic renal masses. Abdominal imaging. 2001 Nov-Dec:26(6):654-60 [PubMed PMID: 11907734]
Habibollahi P, Sultan LR, Bialo D, Nazif A, Faizi NA, Sehgal CM, Chauhan A. Hyperechoic Renal Masses: Differentiation of Angiomyolipomas from Renal Cell Carcinomas using Tumor Size and Ultrasound Radiomics. Ultrasound in medicine & biology. 2022 May:48(5):887-894. doi: 10.1016/j.ultrasmedbio.2022.01.011. Epub 2022 Feb 23 [PubMed PMID: 35219511]
Steiner MS, Goldman SM, Fishman EK, Marshall FF. The natural history of renal angiomyolipoma. The Journal of urology. 1993 Dec:150(6):1782-6 [PubMed PMID: 8230504]
Yang CW, Shen SH, Chang YH, Chung HJ, Wang JH, Lin AT, Chen KK. Are there useful CT features to differentiate renal cell carcinoma from lipid-poor renal angiomyolipoma? AJR. American journal of roentgenology. 2013 Nov:201(5):1017-28. doi: 10.2214/AJR.12.10204. Epub [PubMed PMID: 24147472]
Level 2 (mid-level) evidenceLi HM, Yeh LR, Lu K. Renal angiomyolipoma with coexistent hemorrhagic aneurysm formation and fatty thrombus in inferior vena cava: a rare presentation. Abdominal imaging. 2013 Feb:38(1):180-3. doi: 10.1007/s00261-012-9886-2. Epub [PubMed PMID: 22450723]
Level 3 (low-level) evidenceShin NY, Kim MJ, Chung JJ, Chung YE, Choi JY, Park YN. The differential imaging features of fat-containing tumors in the peritoneal cavity and retroperitoneum: the radiologic-pathologic correlation. Korean journal of radiology. 2010 May-Jun:11(3):333-45. doi: 10.3348/kjr.2010.11.3.333. Epub 2010 Apr 29 [PubMed PMID: 20461188]
Mekahli D, Müller RU, Marlais M, Wlodkowski T, Haeberle S, de Argumedo ML, Bergmann C, Breysem L, Fladrowski C, Henske EP, Janssens P, Jouret F, Kingswood JC, Lattouf JB, Lilien M, Maleux G, Rozenberg M, Siemer S, Devuyst O, Schaefer F, Kwiatkowski DJ, Rouvière O, Bissler J. Clinical practice recommendations for kidney involvement in tuberous sclerosis complex: a consensus statement by the ERKNet Working Group for Autosomal Dominant Structural Kidney Disorders and the ERA Genes & Kidney Working Group. Nature reviews. Nephrology. 2024 Mar 5:():. doi: 10.1038/s41581-024-00818-0. Epub 2024 Mar 5 [PubMed PMID: 38443710]
Level 3 (low-level) evidenceSong S, Park BK, Park JJ. New radiologic classification of renal angiomyolipomas. European journal of radiology. 2016 Oct:85(10):1835-1842. doi: 10.1016/j.ejrad.2016.08.012. Epub 2016 Aug 18 [PubMed PMID: 27666625]
Israel GM, Hindman N, Hecht E, Krinsky G. The use of opposed-phase chemical shift MRI in the diagnosis of renal angiomyolipomas. AJR. American journal of roentgenology. 2005 Jun:184(6):1868-72 [PubMed PMID: 15908544]
Level 2 (mid-level) evidenceSahni VA, Silverman SG. Biopsy of renal masses: when and why. Cancer imaging : the official publication of the International Cancer Imaging Society. 2009 Jul 6:9(1):44-55. doi: 10.1102/1470-7330.2009.0005. Epub 2009 Jul 6 [PubMed PMID: 19602467]
Silverman SG, Gan YU, Mortele KJ, Tuncali K, Cibas ES. Renal masses in the adult patient: the role of percutaneous biopsy. Radiology. 2006 Jul:240(1):6-22 [PubMed PMID: 16709793]
Lobo JM, Clements MB, Bitner DP, Mikula MD, Noona SW, Sultan MI, Cathro HP, Lambert DL, Schenkman NS, Krupski TL. Does renal mass biopsy influence multidisciplinary treatment recommendations? Scandinavian journal of urology. 2020 Feb:54(1):27-32. doi: 10.1080/21681805.2019.1703805. Epub 2019 Dec 23 [PubMed PMID: 31868063]
Ouzaid I, Autorino R, Fatica R, Herts BR, McLennan G, Remer EM, Haber GP. Active surveillance for renal angiomyolipoma: outcomes and factors predictive of delayed intervention. BJU international. 2014 Sep:114(3):412-7. doi: 10.1111/bju.12604. Epub 2014 Apr 16 [PubMed PMID: 24325283]
Level 2 (mid-level) evidenceKingswood JC, Belousova E, Benedik MP, Carter T, Cottin V, Curatolo P, Dahlin M, D'Amato L, Beaure d'Augères G, de Vries PJ, Ferreira JC, Feucht M, Fladrowski C, Hertzberg C, Jozwiak S, Lawson JA, Macaya A, Marques R, Nabbout R, O'Callaghan F, Qin J, Sander V, Shah S, Takahashi Y, Touraine R, Youroukos S, Zonnenberg B, Jansen AC, Sauter M. Renal Manifestations of Tuberous Sclerosis Complex: Key Findings From the Final Analysis of the TOSCA Study Focussing Mainly on Renal Angiomyolipomas. Frontiers in neurology. 2020:11():972. doi: 10.3389/fneur.2020.00972. Epub 2020 Sep 16 [PubMed PMID: 33041968]
Level 2 (mid-level) evidenceCockerell I, Guenin M, Heimdal K, Bjørnvold M, Selmer KK, Rouvière O. Prevalence of Renal Angiomyolipomas and Spontaneous Bleeding Related to Angiomyolipomas in Tuberous Sclerosis Complex Patients in France and Norway-a Questionnaire Study. Urology. 2017 Jun:104():70-76. doi: 10.1016/j.urology.2017.02.023. Epub 2017 Feb 21 [PubMed PMID: 28232177]
Yamakado K, Tanaka N, Nakagawa T, Kobayashi S, Yanagawa M, Takeda K. Renal angiomyolipoma: relationships between tumor size, aneurysm formation, and rupture. Radiology. 2002 Oct:225(1):78-82 [PubMed PMID: 12354988]
Rimon U, Duvdevani M, Garniek A, Golan G, Bensaid P, Ramon J, Morag B. Large renal angiomyolipomas: digital subtraction angiographic grading and presentation with bleeding. Clinical radiology. 2006 Jun:61(6):520-6 [PubMed PMID: 16713423]
Boorjian SA, Sheinin Y, Crispen PL, Lohse CM, Kwon ED, Leibovich BC. Hormone receptor expression in renal angiomyolipoma: clinicopathologic correlation. Urology. 2008 Oct:72(4):927-32. doi: 10.1016/j.urology.2008.01.067. Epub 2008 Mar 28 [PubMed PMID: 18374399]
Çetin C, Büyükkurt S, Demir C, Evrüke C. Renal angiomyolipoma during pregnancy: Case report and literature review. Turkish journal of obstetrics and gynecology. 2015 Jun:12(2):118-121. doi: 10.4274/tjod.32848. Epub 2015 Jun 15 [PubMed PMID: 28913054]
Level 3 (low-level) evidencePreece P, Mees B, Norris B, Christie M, Wagner T, Dundee P. Surgical management of haemorrhaging renal angiomyolipoma in pregnancy. International journal of surgery case reports. 2015:7C():89-92. doi: 10.1016/j.ijscr.2015.01.004. Epub 2015 Jan 8 [PubMed PMID: 25598402]
Level 3 (low-level) evidenceZhao Y, Yang Z, Xu W, Ji Z, Dong J. Management of renal tumors during pregnancy: case reports. BMC nephrology. 2021 Apr 9:22(1):127. doi: 10.1186/s12882-021-02318-w. Epub 2021 Apr 9 [PubMed PMID: 33836679]
Level 3 (low-level) evidenceMurray TE, Doyle F, Lee M. Transarterial Embolization of Angiomyolipoma: A Systematic Review. The Journal of urology. 2015 Sep:194(3):635-9. doi: 10.1016/j.juro.2015.04.081. Epub 2015 Apr 25 [PubMed PMID: 25916674]
Level 1 (high-level) evidenceFernández-Pello S, Hora M, Kuusk T, Tahbaz R, Dabestani S, Abu-Ghanem Y, Albiges L, Giles RH, Hofmann F, Kuczyk MA, Lam TB, Marconi L, Merseburger AS, Powles T, Staehler M, Volpe A, Ljungberg B, Bex A, Bensalah K. Management of Sporadic Renal Angiomyolipomas: A Systematic Review of Available Evidence to Guide Recommendations from the European Association of Urology Renal Cell Carcinoma Guidelines Panel. European urology oncology. 2020 Feb:3(1):57-72. doi: 10.1016/j.euo.2019.04.005. Epub 2019 Jun 4 [PubMed PMID: 31171501]
Level 1 (high-level) evidenceVaggers S, Rice P, Somani BK, Veeratterapillay R, Rai BP. Evidence-based protocol-led management of renal angiomyolipoma: A review of literature. Turkish journal of urology. 2021 Feb:47(Supp. 1):S9-S18. doi: 10.5152/tud.2020.20343. Epub 2020 Sep 21 [PubMed PMID: 32966208]
Bissler JJ, Kingswood JC, Radzikowska E, Zonnenberg BA, Frost M, Belousova E, Sauter M, Nonomura N, Brakemeier S, de Vries PJ, Whittemore VH, Chen D, Sahmoud T, Shah G, Lincy J, Lebwohl D, Budde K. Everolimus for angiomyolipoma associated with tuberous sclerosis complex or sporadic lymphangioleiomyomatosis (EXIST-2): a multicentre, randomised, double-blind, placebo-controlled trial. Lancet (London, England). 2013 Mar 9:381(9869):817-24 [PubMed PMID: 23312829]
Level 1 (high-level) evidencePeng ZF, Yang L, Wang TT, Han P, Liu ZH, Wei Q. Efficacy and safety of sirolimus for renal angiomyolipoma in patients with tuberous sclerosis complex or sporadic lymphangioleiomyomatosis: a systematic review. The Journal of urology. 2014 Nov:192(5):1424-30. doi: 10.1016/j.juro.2014.04.096. Epub 2014 May 9 [PubMed PMID: 24813310]
Level 1 (high-level) evidenceWatanabe EH, Coelho FMA, Filho HL, Balbo BEP, Neves PDMM, Franzin FM, Yamauchi FI, Onuchic LF. The effect of sirolimus on angiomyolipoma is determined by decrease of fat-poor compartments and includes striking reduction of vascular structures. Scientific reports. 2021 Apr 19:11(1):8493. doi: 10.1038/s41598-021-87930-4. Epub 2021 Apr 19 [PubMed PMID: 33875750]
Geynisman DM, Kadow BT, Shuch BM, Boorjian SA, Matin SF, Rampersaud E, Milestone BN, Plimack ER, Zibelman MR, Kutikov A, Smaldone MC, Chen DY, Viterbo R, Joshi S, Greenberg RE, Malizzia L, McGowan T, Ross EA, Uzzo RG. Sporadic Angiomyolipomas Growth Kinetics While on Everolimus: Results of a Phase II Trial. The Journal of urology. 2020 Sep:204(3):531-537. doi: 10.1097/JU.0000000000001065. Epub 2020 Apr 6 [PubMed PMID: 32250730]
Wang W, Guo H, Shi B, Sun H, Li H, Zhang Y, Cai Y. CT characteristics predict the response to everolimus or sirolimus of renal angiomyolipomas in patients with tuberous sclerosis complex. International urology and nephrology. 2019 Apr:51(4):671-676. doi: 10.1007/s11255-019-02093-6. Epub 2019 Feb 11 [PubMed PMID: 30756282]
Bissler JJ, Kingswood JC, Radzikowska E, Zonnenberg BA, Belousova E, Frost MD, Sauter M, Brakemeier S, de Vries PJ, Berkowitz N, Voi M, Peyrard S, Budde K. Everolimus long-term use in patients with tuberous sclerosis complex: Four-year update of the EXIST-2 study. PloS one. 2017:12(8):e0180939. doi: 10.1371/journal.pone.0180939. Epub 2017 Aug 9 [PubMed PMID: 28792952]
Luo C, Ye WR, Zu XB, Chen MF, Qi L, Li YL, Cai Y. Low-Dose Everolimus Maintenance Therapy for Renal Angiomyolipoma Associated With Tuberous Sclerosis Complex. Frontiers in medicine. 2021:8():744050. doi: 10.3389/fmed.2021.744050. Epub 2021 Nov 24 [PubMed PMID: 34901059]
Level 2 (mid-level) evidenceBissler JJ, Budde K, Sauter M, Franz DN, Zonnenberg BA, Frost MD, Belousova E, Berkowitz N, Ridolfi A, Christopher Kingswood J. Effect of everolimus on renal function in patients with tuberous sclerosis complex: evidence from EXIST-1 and EXIST-2. Nephrology, dialysis, transplantation : official publication of the European Dialysis and Transplant Association - European Renal Association. 2019 Jun 1:34(6):1000-1008. doi: 10.1093/ndt/gfy132. Epub [PubMed PMID: 30053159]
Trelinska J, Dachowska I, Baranska D, Stawiski K, Kotulska K, Fendler W, Jozwiak S, Mlynarski W. Maintenance therapy with everolimus for subependymal giant cell astrocytoma in patients with tuberous sclerosis (the EMINENTS study). Pediatric blood & cancer. 2017 Jun:64(6):. doi: 10.1002/pbc.26347. Epub 2016 Nov 15 [PubMed PMID: 27860334]
Hatano T, Inaba H, Endo K, Egawa S. Intermittent everolimus administration for renal angiomyolipoma associated with tuberous sclerosis complex. International journal of urology : official journal of the Japanese Urological Association. 2017 Nov:24(11):780-785. doi: 10.1111/iju.13428. Epub 2017 Sep 14 [PubMed PMID: 28905429]
Sasongko TH, Kademane K, Chai Soon Hou S, Jocelyn TXY, Zabidi-Hussin Z. Rapamycin and rapalogs for tuberous sclerosis complex. The Cochrane database of systematic reviews. 2023 Jul 11:7(7):CD011272. doi: 10.1002/14651858.CD011272.pub3. Epub 2023 Jul 11 [PubMed PMID: 37432030]
Level 1 (high-level) evidenceLee L, Ito T, Jensen RT. Everolimus in the treatment of neuroendocrine tumors: efficacy, side-effects, resistance, and factors affecting its place in the treatment sequence. Expert opinion on pharmacotherapy. 2018 Jun:19(8):909-928. doi: 10.1080/14656566.2018.1476492. Epub 2018 May 24 [PubMed PMID: 29757017]
Level 3 (low-level) evidenceAbdelsalam ME, Ahrar K. Ablation of Small Renal Masses. Techniques in vascular and interventional radiology. 2020 Jun:23(2):100674. doi: 10.1016/j.tvir.2020.100674. Epub 2020 May 25 [PubMed PMID: 32591194]
Leflore A, McKenna PH, Schwartz BF. Laparoscopic cryotherapy for pediatric angiomyolipoma: the first report. Journal of endourology. 2007 Mar:21(3):321-4; discussion 324 [PubMed PMID: 17444779]
Level 3 (low-level) evidenceJohnson SC, Graham S, D'Agostino H, Elmajian DA, Shingleton WB. Percutaneous renal cryoablation of angiomyolipomas in patients with solitary kidneys. Urology. 2009 Dec:74(6):1246-9. doi: 10.1016/j.urology.2008.09.005. Epub 2009 Oct 7 [PubMed PMID: 19815260]
Level 3 (low-level) evidenceCastle SM, Gorbatiy V, Ekwenna O, Young E, Leveillee RJ. Radiofrequency ablation (RFA) therapy for renal angiomyolipoma (AML): an alternative to angio-embolization and nephron-sparing surgery. BJU international. 2012 Feb:109(3):384-7. doi: 10.1111/j.1464-410X.2011.10376.x. Epub 2011 Dec 16 [PubMed PMID: 22176671]
Prevoo W, van den Bosch MA, Horenblas S. Radiofrequency ablation for treatment of sporadic angiomyolipoma. Urology. 2008 Jul:72(1):188-91. doi: 10.1016/j.urology.2008.02.059. Epub 2008 May 2 [PubMed PMID: 18455777]
Level 3 (low-level) evidenceStamatiou KN, Moschouris H, Marmaridou K, Kiltenis M, Kladis-Kalentzis K, Malagari K. Combination of Superselective Arterial Embolization and Radiofrequency Ablation for the Treatment of a Giant Renal Angiomyolipoma Complicated with Caval Thrombus. Case reports in oncological medicine. 2016:2016():8087232. doi: 10.1155/2016/8087232. Epub 2016 May 11 [PubMed PMID: 27293932]
Level 3 (low-level) evidenceGregory SM, Anderson CJ, Patel U. Radiofrequency ablation of large renal angiomyolipoma: median-term follow-up. Cardiovascular and interventional radiology. 2013 Jun:36(3):682-9. doi: 10.1007/s00270-012-0483-0. Epub 2013 Jan 25 [PubMed PMID: 23354958]
Liu F, Yuan H, Li X, Tang J, Tian X, Ji K. A new management strategy for renal angiomyolipomas: superselective arterial embolization in combination with radiofrequency ablation. Annals of translational medicine. 2019 Dec:7(23):766. doi: 10.21037/atm.2019.11.63. Epub [PubMed PMID: 32042782]
Nozadze G, Larsen SB, Heerwagen S, Juhl Jensen R, Lönn L, Røder MA. Selective arterial embolization of renal angiomyolipomas: A 10-year experience. BJUI compass. 2022 Jan:3(1):86-92. doi: 10.1002/bco2.107. Epub 2021 Aug 31 [PubMed PMID: 35475158]
Urciuoli P, D'Orazi V, Livadoti G, Foresi E, Panunzi A, Anichini S, Cialini M, Wlderk A, Cirelli C, Colangelo M, Mongardini M, Custureri F. Treatment of renal angiomyolipoma: surgery versus angioembolization. Il Giornale di chirurgia. 2013 Nov-Dec:34(11-12):326-31 [PubMed PMID: 24342162]
Level 3 (low-level) evidenceZeid M, Sayedin H, Nabi N, Abdelrahman M, Jacob PT, Alhadi B, Giri S. Active Surveillance for Renal Angiomyolipoma Less Than 4 Centimeters: A Systematic Review of Cohort Studies. Cureus. 2022 Feb:14(2):e22678. doi: 10.7759/cureus.22678. Epub 2022 Feb 28 [PubMed PMID: 35371642]
Level 1 (high-level) evidenceMues AC, Palacios JM, Haramis G, Casazza C, Badani K, Gupta M, McKiernan J, Benson MC, Landman J. Contemporary experience in the management of angiomyolipoma. Journal of endourology. 2010 Nov:24(11):1883-6. doi: 10.1089/end.2010.0223. Epub 2010 Oct 4 [PubMed PMID: 20919915]
Schieda N, Davenport MS, Pedrosa I, Shinagare A, Chandarana H, Curci N, Doshi A, Israel G, Remer E, Wang J, Silverman SG. Renal and adrenal masses containing fat at MRI: Proposed nomenclature by the society of abdominal radiology disease-focused panel on renal cell carcinoma. Journal of magnetic resonance imaging : JMRI. 2019 Apr:49(4):917-926. doi: 10.1002/jmri.26542. Epub 2019 Jan 28 [PubMed PMID: 30693607]
Hélénon O, Merran S, Paraf F, Melki P, Correas JM, Chrétien Y, Moreau JF. Unusual fat-containing tumors of the kidney: a diagnostic dilemma. Radiographics : a review publication of the Radiological Society of North America, Inc. 1997 Jan-Feb:17(1):129-44 [PubMed PMID: 9017804]
Potretzke AM, Potretzke TA, Bauman TM, Knight BA, Park AM, Mobley JM, Figenshau RS, Siegel CL. Computed Tomography and Magnetic Resonance Findings of Fat-Poor Angiomyolipomas. Journal of endourology. 2017 Feb:31(2):119-128. doi: 10.1089/end.2016.0219. Epub 2017 Jan 3 [PubMed PMID: 27897036]
Jeon HG, Lee SR, Kim KH, Oh YT, Cho NH, Rha KH, Yang SC, Han WK. Benign lesions after partial nephrectomy for presumed renal cell carcinoma in masses 4 cm or less: prevalence and predictors in Korean patients. Urology. 2010 Sep:76(3):574-9. doi: 10.1016/j.urology.2009.11.082. Epub 2010 Mar 29 [PubMed PMID: 20303148]
Level 2 (mid-level) evidenceKrishna S, Murray CA, McInnes MD, Chatelain R, Siddaiah M, Al-Dandan O, Narayanasamy S, Schieda N. CT imaging of solid renal masses: pitfalls and solutions. Clinical radiology. 2017 Sep:72(9):708-721. doi: 10.1016/j.crad.2017.05.003. Epub 2017 Jun 4 [PubMed PMID: 28592361]
Kang SK, Huang WC, Pandharipande PV, Chandarana H. Solid renal masses: what the numbers tell us. AJR. American journal of roentgenology. 2014 Jun:202(6):1196-206. doi: 10.2214/AJR.14.12502. Epub [PubMed PMID: 24848816]
Dahl DM, Simeone JF, Iliopoulos O, Saylor PJ, Wu CL. Case 36-2018: A 29-Year-Old Man with an Incidentally Discovered Renal Mass. The New England journal of medicine. 2018 Nov 22:379(21):2064-2072. doi: 10.1056/NEJMcpc1802832. Epub [PubMed PMID: 30462933]
Level 3 (low-level) evidenceWang LJ, Wong YC, Chen CJ, See LC. Computerized tomography characteristics that differentiate angiomyolipomas from liposarcomas in the perinephric space. The Journal of urology. 2002 Feb:167(2 Pt 1):490-3 [PubMed PMID: 11792904]
Level 2 (mid-level) evidenceClark PE, Novick AC. Exophytic noninvasive growth pattern of renal angiomyolipomas: implications for nephron sparing surgery. The Journal of urology. 2001 Feb:165(2):513-4 [PubMed PMID: 11176411]
Level 3 (low-level) evidencePatel U, Simpson E, Kingswood JC, Saggar-Malik AK. Tuberose sclerosis complex: analysis of growth rates aids differentiation of renal cell carcinoma from atypical or minimal-fat-containing angiomyolipoma. Clinical radiology. 2005 Jun:60(6):665-73; discussion 663-4 [PubMed PMID: 16038693]
Avila NA, Dwyer AJ, Moss J. Active Surveillance of Nonfatty Renal Masses in Patients With Lymphangioleiomyomatosis: Use of CT Features and Patterns of Growth to Differentiate Angiomyolipoma From Renal Cancer. AJR. American journal of roentgenology. 2017 Sep:209(3):611-619. doi: 10.2214/AJR.16.17530. Epub 2017 Jul 5 [PubMed PMID: 28678574]
Yeoh ZW, Navaratnam V, Bhatt R, McCafferty I, Hubbard RB, Johnson SR. Natural history of angiomyolipoma in lymphangioleiomyomatosis: implications for screening and surveillance. Orphanet journal of rare diseases. 2014 Oct 3:9():151. doi: 10.1186/s13023-014-0151-3. Epub 2014 Oct 3 [PubMed PMID: 25277108]
Battelli C, Cho DC. mTOR inhibitors in renal cell carcinoma. Therapy (London, England : 2004). 2011 Jul:8(4):359-367 [PubMed PMID: 21894244]
Wang C, Li X, Peng L, Gou X, Fan J. An update on recent developments in rupture of renal angiomyolipoma. Medicine. 2018 Apr:97(16):e0497. doi: 10.1097/MD.0000000000010497. Epub [PubMed PMID: 29668633]
Restrepo JCÁ, Millan DAC, Sabogal CAR, Bernal AFP, Donoso WD. New Trends and Evidence for the Management of Renal Angiomyolipoma: A Comprehensive Narrative Review of the Literature. Journal of kidney cancer and VHL. 2022:9(1):33-41. doi: 10.15586/jkcvhl.v9i1.177. Epub 2022 Jan 21 [PubMed PMID: 35096516]
Level 3 (low-level) evidenceMacKeigan JP, Krueger DA. Differentiating the mTOR inhibitors everolimus and sirolimus in the treatment of tuberous sclerosis complex. Neuro-oncology. 2015 Dec:17(12):1550-9. doi: 10.1093/neuonc/nov152. Epub 2015 Aug 19 [PubMed PMID: 26289591]