Introduction
Alopecia is the absence or loss of hair in an area where it is expected to be present. This condition can be localized or diffuse, temporary or permanent, and affect both sexes and all age groups. Recognized as a sign or symptom arising from heterogeneous etiologies, alopecia is broadly classified as nonscarring (the most prevalent) and scarring (cicatricial). Patients may present with significant distress, diminishing their quality of life. A detailed history, physical examination, and focused evaluation are necessary to determine the underlying cause, which will guide the decision-making process for the most appropriate management for the best outcomes. Of note, the nomenclature in the literature typically uses nonscarring rather than noncicatricial and varies between scarring and cicatricial. This review uses nonscarring and scarring for simplicity, except when cicatricial is part of the named disorder.
Etiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Etiology
The etiology of alopecia varies according to the type and subtype of alopecia. Some of the causative mechanisms are given below:
Nonscarring Alopecias
The hair follicles are preserved in nonscarring alopecia; therefore, hair loss is potentially reversible, and hair regrowth is possible. Nonscarring alopecias include androgenetic alopecia, alopecia areata, telogen effluvium, anagen effluvium, traction alopecia, trichotillomania, and alopecia syphilitica, among others.
Androgenetic alopecia
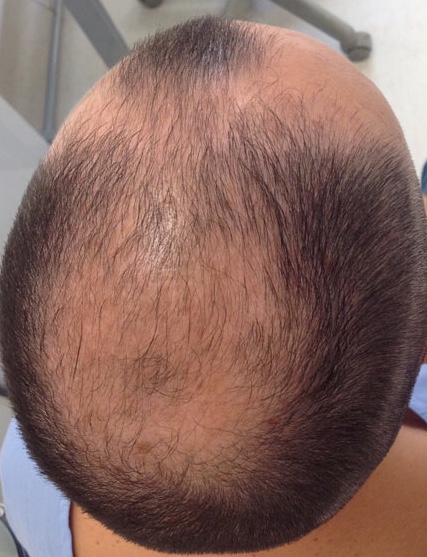
Androgenetic alopecia is referred to as androgenic alopecia, hereditary alopecia, male balding, female-pattern alopecia, and female-pattern hair loss, to name a few. Due to an improved understanding of the condition, newer nomenclature is moving toward male- and female-pattern hair loss.[1] Pattern hair loss is usually slowly progressive and is the most common type of alopecia in men, women, and adolescents. Men tend to lose hair in the vertex, bitemporal, and midfrontal scalp (see Image. Male-Pattern Androgenetic Alopecia), whereas women tend to lose hair throughout the central scalp, with minimal involvement of the frontal hairline.[1][2][3][4][5]
Alopecia areata
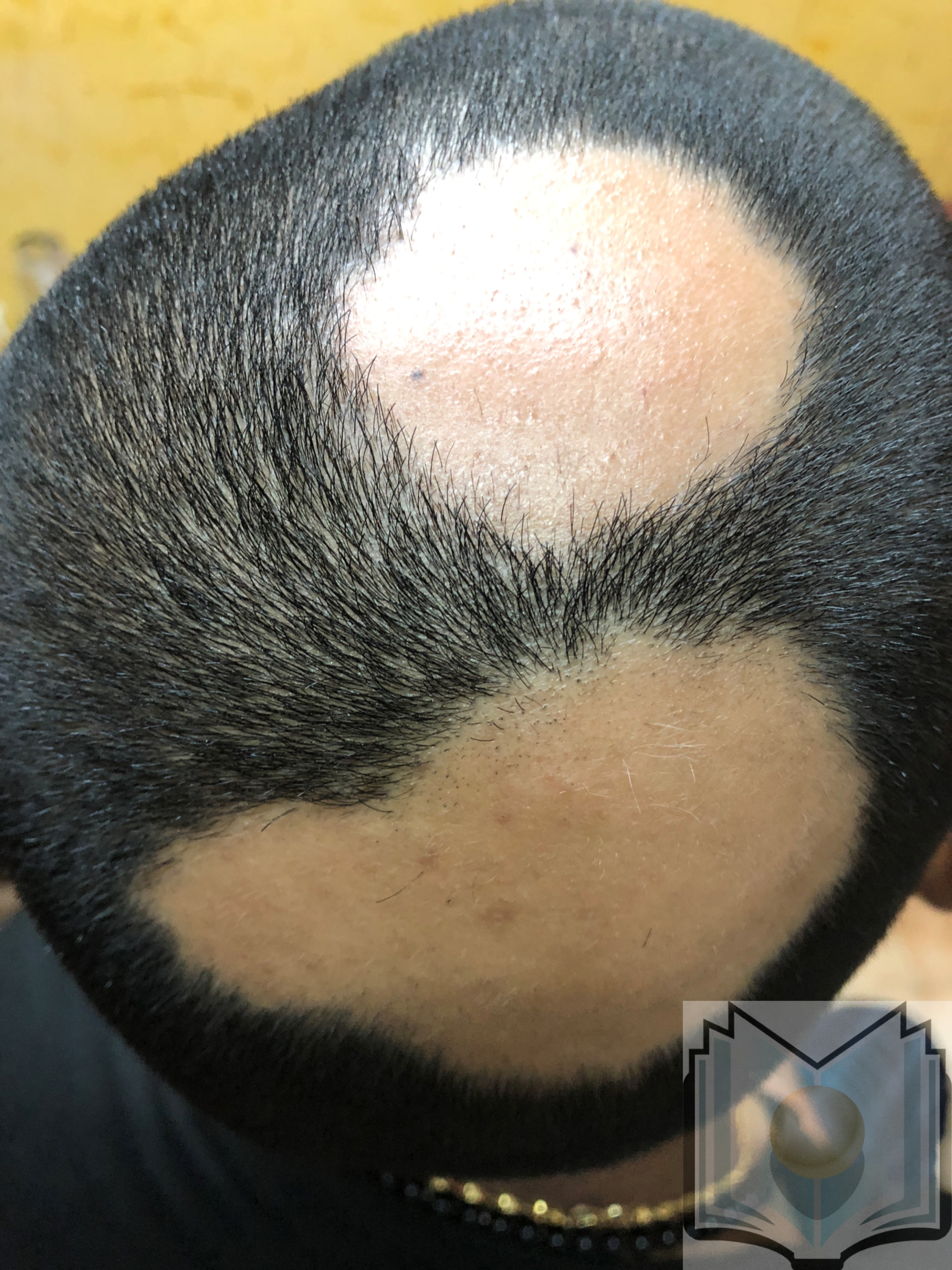
Alopecia areata is a chronic immune-mediated disease commonly presenting as acute onset patchy hair loss on the scalp, affecting both sexes and children, adolescents, and adults (see Image. Alopecia Areata). Rarely do patients present with total scalp hair loss (alopecia totalis) or hair over the entire body (alopecia universalis). Patients may experience a single episode or a pattern of remission and recurrence.[3][4][5][6]
Telogen effluvium
Telogen effluvium is a noninflammatory acute or chronic alopecia usually affecting the entire scalp in both sexes of children, adolescents, and adults. Disruption in the transition from the anagen phase to the telogen phase may be due to psychological stress, chronic illness, pregnancy or postpartum, malnutrition, severe infection, endocrine disorders, metabolic disturbances, surgery, and medications, such as anticonvulsants, antidepressants, anticoagulants, oral contraceptive pills, and retinoids.[4][5] Hair loss occurs approximately 3 months after a triggering event, is usually self-limiting, and lasts for about 6 months.[7]
Scarring Alopecias
The hair follicles are irreversibly destroyed in scarring alopecia, leading to permanent hair loss. Scarring alopecias are classified as primary and secondary. The primary cicatricial alopecia classes are lymphocytic, neutrophilic, and mixed.[8] Lymphocytic primary cicatricial alopecias include frontal fibrosing alopecia (FFA), lichen planopilaris (LPP), central centrifugal cicatricial alopecia, discoid lupus erythematosus (the most common type of chronic cutaneous lupus erythematosus), pseudopelade of Brocq, alopecia mucinosa, and keratosis follicularis spinulosa decalvans. Neutrophilic primary cicatricial alopecias are dissecting cellulitis and folliculitis decalvans, and mixed primary cicatricial alopecias are acne keloidalis nuchae, acne necrotica, and erosive pustular dermatosis of the scalp. The 2 most common scarring alopecias are FFA and LPP.
FFA
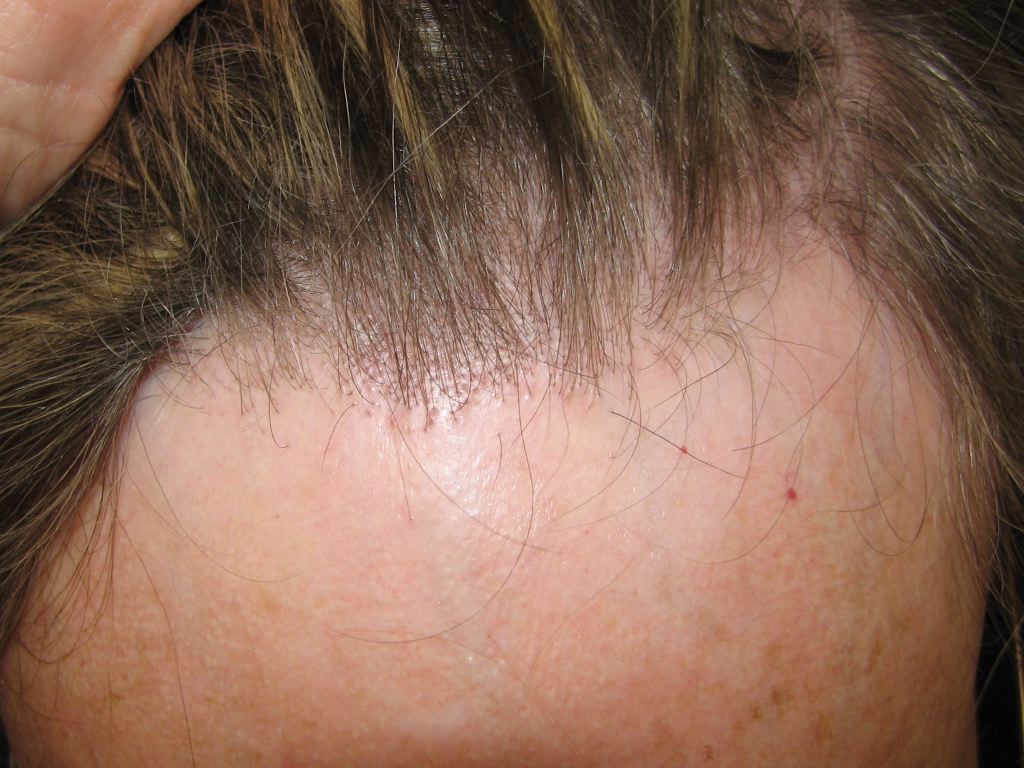
FFA is characterized by a slowly progressive symmetric band of frontal hairline alopecia and primarily affects postmenopausal women (see Image. FFA). About 25% of patients report itching and pain in the affected area; otherwise, the condition is asymptomatic. FFA is often considered a variant of LPP based on histological similarities.[9][10]
LPP
LPP most commonly affects women and initially presents with perifollicular erythema and follicular hyperkeratosis, evolving to single or multifocal patches of irregular alopecia on the scalp vertex. Hair loss is usually limited to the scalp but may occasionally affect body hair. Patients may report itching, burning, or tenderness in the affected area(s).[10]
Causes of secondary cicatricial alopecia include localized scleroderma, pemphigoid, neoplasm, radiation therapy, chemical or physical trauma, and bacterial or fungal infections.[11]
Epidemiology
A 2019 multicenter retrospective study was performed to analyze "the frequency of the types of alopecia in patients consulting at specialist hair clinics and to assess for global variations."[12]
Over 3000 diagnoses of alopecia were included, of which 73% were nonscarring, and 27% were scarring.
Nonscarring Alopecia Frequency
- Androgenetic alopecia: 37.7%
- Alopecia areata: 18.2%
- Telogen effluvium: 11.3%
Scarring Alopecia Frequency
- Frontal fibrosing alopecia: 10.8%
- Lichen planopilaris: 7.6%
- Folliculitis decalvans: 2.8%
The frequency of the other alopecia types listed is less than 2%. The article also provides "differences in the frequency of hair disorders" for gender, median age range by gender, and continent.[12]
Pathophysiology
Vellus hair is short, thin, and present on most of the body. Terminal hair is long, thick, and present on the scalp, eyelashes, eyebrows, beards, axillae, and pubic regions. Both types have a stationary upper segment and a dynamic lower segment, with the lower segment going through the follicular cycle of anagen (growing, 85%-90%), catagen (involuting, <5%), and telogen (resting, 5%-10%) phases.[2][4]
The mean duration of the anagen phase is 2 to 7 years. The hair follicle then enters the catagen phase, the shortest phase of the cycle, with an approximate 2- to 3-week duration, then telogen, which lasts 2 to 4 months. The duration of anagen determines hair length.[2][3]
Nonscarring Alopecias
Androgenetic alopecia
Androgenetic alopecia (male- and female-pattern hair loss) develops after puberty, with variable progression and severity. Although the androgen-dependent nature of male pattern hair loss is well established, this is not the case with females. Both conditions are due to altered hair follicle cycling and miniaturization, leading to the transformation of terminal hair follicles to vellus hair follicles, resulting in shorter, finer hair shafts. Additionally, the duration of the anagen phase shortens from years to only weeks or months. A strong genetic predisposition is clear, but no causative gene has been identified, and epigenetic modifications may alter the genetic susceptibility.[13]
Male-pattern hair loss
Male-pattern hair loss is ascribed to genetic variants of the androgen receptor, particularly the androgen receptor gene (AR) at the AR⁄ectodysplasin A2 receptor gene (EDA2R) locus.[14] Another susceptible locus is on chromosome 20p11, for which no candidate gene has been identified.[14] Dihydrotestosterone (DHT), a testosterone metabolite synthesized through the activity of 5-alpha reductase, plays a key role in hair loss in androgen-sensitive areas. Androgen sensitivity of hair follicles varies depending on the scalp region, with male hair loss typically occurring in the vertex, bitemporal, and midfrontal scalp. Of note, occipital hair follicles are androgen-independent.
Female-pattern hair loss
Female-pattern hair loss shows no involvement of the locus on chromosome 20p11, but the X-chromosomal AR⁄EDA2R locus may be involved in the pathogenesis of early-onset female-pattern hair loss.[14] The age-related increase in female-pattern hair loss, with the highest rates in postmenopausal women, suggests a protective role of estrogen.[15] Evidence is conflicting regarding whether estrogen inhibits or stimulates the hair follicle.[13]
Alopecia areata
Alopecia areata is a chronic immune-mediated disease characterized by acute or chronic hair loss, with the anagen phase of the hair growth cycle significantly shortened during acute flares. Although the exact pathophysiology is not fully understood, the collapse of the immune privilege (IP) that usually protects the hair follicle bulb plays an important role.[16] Interferon-gamma and substance P are potent inducers of the collapse secondary to increasing proinflammatory factors while decreasing IP guardians.[17] Interleukin (IL)-2 and IL-15 have also been implicated as important mediators capable of inducing a topical immune reaction in alopecia areata.[6]
Genome-wide association studies provide evidence for susceptibility loci on chromosomes 6, 10, 16, and 18. Furthermore, the major loci on 16 and 18 coincide with loci for psoriasis seen in other studies.[18]
Telogen effluvium
Telogen effluvium occurs if a significant number of anagen hair is triggered to prematurely stop growing and enter the catagen phase, then the telogen phase. Approximately 2 to 3 months after the triggering event, the patient’s hair sheds excessively. If the trigger is not repetitive, the alopecia will resolve as the new anagen hair grows over 3 to 6 months.[7] See the Etiology section for examples of triggers.
Scarring Alopecias
FFA and LPP
FFA and LPP are characterized by inflamed hair follicles surrounding an area of permanent scarring alopecia. The central pathophysiological process in FFA and LPP is inflammation-induced, irreversible damage to epithelial stem cells in the hair follicle bulge. Massive inflammation can attack the proximal hair bulb in alopecia areata without destroying the hair follicle; however, in FAA and LPP, relatively discrete infiltrates around the bulge are sufficient to damage hair follicles irreversibly. Sclerotic fibrous tracts replace the destroyed hair follicles, with no follicles in the catagen or telogen phases.[11][19][20]
Histopathology
The histopathologic features of alopecia vary according to the subtype of alopecia. Interpretation of the histopathological findings of primary scarring and nonscarring alopecias may prove daunting. This is especially true if the biopsy specimen is inadequate, and the clinical history and pattern of the alopecia are not known.[21] Hence, proper history, physical examination, and adequate sampling techniques are necessary for diagnosis.
Scarring Alopecia
In the case of scarring alopecia due to chronic cutaneous discoid lupus, histopathological features include follicular hyperkeratosis and a vacuolar-interface folliculitis at the level of the infundibulum. The interfollicular epidermis may at times also be involved, in association with a perivascular and periappendageal superficial and deep lymphoid cell infiltrate with plasma cells. Late stages are characterized by concentric lamellar perifollicular fibroplasia and by basement membrane zone thickening that is highlighted by Periodic Acid Schiff staining.
Lichen planopilaris
In the case of lichen planopilaris, histopathologically, the features of LPP and its variants are similar, irrespective of clinical presentation. In early lesions, there is a vacuolar interface change with a moderately dense perifollicular lichenoid lymphocytic cell infiltrate at the level of the infundibulum and isthmus.
Nonscarring Alopecia
In nonscarring alopecia, the most common alopecia is androgenetic alopecia. Histopathologically, the use of transverse sections is the most valuable method to reach a diagnosis, as all the hair follicles can be visualized. Although the total number of hair follicles is unchanged, there is progressive miniaturization, with a variation in the size of the hair follicles and increased vellus hairs.
The terminal (T) to vellus (V) ratio for a normal scalp is T: V=7:1. A ratio of T: V=3:1 or less is considered to be diagnostic. However, the intermediate hairs that have a hair shaft diameter in between the terminal and vellus hair follicles, and are currently classified as terminal in follicular counts. Other findings include an increased number of telogen hairs, decreased numbers of terminal hair follicles in the subcutaneous fat, variation of shaft diameter, and increased numbers of fibrous tracts. A mild peri-infundibular lymphocytic cell infiltrate and perifollicular collagen deposition are present in 40% of cases.
History and Physical
A thorough history will likely include the following:
- Whether there is visible hairlessness (alopecia) or an increased amount of hair falling out daily (effluvium)
- Age at onset of hair loss
- Time course
- Progression
- Distribution, such as focal (patchy), patterned, or diffuse
- Accompanying symptoms
- Gynecological and obstetrics history for females
- Dietary history
- Family history of hair loss
- Recent or current medications
Recent environmental exposures and surgical history may also be part of a relevant history.
A complete skin evaluation, including the scalp, face, and nails, should be conducted. When examining the scalp, position the patient in a chair rather than on an exam table, and use good lighting and possibly magnification to ensure proper inspection.[22] Assess for erythema, papules, scaling, pustules, crusting, and nail discoloration and pitting. Dermoscopy is essential for visualizing epidermal and subepidermal structures undetectable by the naked eye.
The hair-pull test may help evaluate patients with excessive hair shedding. Grasp a group of approximately 40 to 60 hairs, apply firm but gentle traction away from the scalp, and repeat in 3 to 4 areas. Traditionally, more than 10% of hairs (4 to 6) pulled from the scalp is a positive test. A more recently recommended cutoff for a positive test is more than 3 hairs per 60 hairs removed.[4][23]
Extracted hair can be examined with light microscopy to determine the phase (anagen or telogen) and whether the hair is broken or dystrophic. The roots in each growth phase can be counted, with more than 20% in the telogen phase indicating increased hair shedding.[3]
Nonscarring Alopecias
Androgenetic alopecia
Androgenetic alopecia (male and female pattern hair loss) in males usually presents as a receding hairline and hair miniaturization on the vertex, bitemporal, and midfrontal scalp. Females typically present with prominent central scalp and minimal frontal hairline involvement. The hair-pull test is usually negative, and dermoscopy will likely show an increased number of thin and vellus hairs, heterogenous hair shaft thickness, perifollicular pigmentation, and a variable number of yellow dots (sebaceous glands) without scarring.[1][5]
Alopecia areata
Alopecia areata commonly presents with patchy hair loss on the scalp (see Image. Alopecia Areata Hair Loss); less commonly, the conditions presents with total scalp hair loss (alopecia totalis) or complete loss of all body hair (alopecia universalis). The skin within the alopecic area(s) is smooth and intact without scarring or signs of inflammation. The hair-pull test is performed at the border of a patchy lesion and the contralateral clinically unaffected side. A positive test indicates active disease, and a negative test indicates the disease is stable or resolving. A positive pull test on the clinically unaffected side may indicate disease progression toward alopecia totalis. Dermoscopy will likely show vellus hairs (associated with remission or long-standing disease), broken hairs, black dots, and yellow dots. Exclamation mark hairs, usually seen in the periphery of active lesions, are pathognomonic for alopecia areata. Nail pitting and, in severe cases, trachyonychia (rough, longitudinally ridged nails) may be seen.[5][6]
The Severity of Alopecia Tool or SALT score can help guide therapeutic decision-making and monitor the therapeutic response in patients with alopecia areata.[24][25] The scalp area is divided into 4 quadrants, with each quadrant representing a percentage of the total scalp area: right side (18%), left side (18%), top (40%), and back (24%). The percentage of hair loss is visually estimated in each quadrant and then summated to determine the SALT score, with the 5 subgroups of hair involvement as follows:
- S0: No hair loss
- S1: Less than 25% hair loss
- S2: 25% to 49% hair loss
- S3: 50% to 74% hair loss
- S4: 75% to 99% hair loss
- S5: 100% hair loss [25]
Telogen effluvium
Telogen effluvium typically presents with hair loss over the entire scalp. The presence of inflammation, scaling, pustules, or scarring suggests other diagnoses. A positive hair-pull test performed on multiple scalp sites strongly suggests active shedding associated with telogen effluvium. Examining the hair shafts with light microscopy can confirm the loose hairs are telogen hairs; dermoscopy may help distinguish chronic telogen effluvium (hair loss for more than 6 months) from female-pattern hair loss, with the latter demonstrating more variability in hair diameter.[7][26] Additionally, the wash test or modified wash test may aid in assessing disease severity.[7][27]
Scarring Alopecias
Vano-Galvan et al and Tolkachjov et al have proposed FFA diagnostic criteria.[28][29] The diagnosis requires 2 major criteria or 1 major criterion and 2 minor criteria.
Major criteria:
- Cicatricial alopecia of the frontal, temporal, or frontotemporal scalp on examination, in the absence of follicular keratotic papules on the body
- Diffuse bilateral eyebrow alopecia
Minor criteria:
- Typical trichoscopic features (perifollicular erythema, follicular hyperkeratosis, or both)
- Histopathologic features of cicatricial alopecia in the pattern of FFA and LPP on biopsy
- Involvement (hair loss or perifollicular erythema) of additional FFA sites (occipital area, facial hair, sideburns, or body hair)
- Noninflammatory facial papules
LPP characteristically presents as multiple, pruritic, patchy alopecic areas on the scalp vertex, with follicular hyperkeratosis and perifollicular erythema along the periphery of the scarring lesions. The hair-pull test may demonstrate anagen hair with thick root sheaths. Scalp dermoscopy may show peripilar casts (concentrically arranged scale encircling an emerging hair shaft), perifollicular erythema and scaling, and follicular keratotic plugs.[11][30]
In 2010, an LPP activity index was introduced by Chiang et al to assess clinical activity.[31] The index consists of symptoms (pain, pruritus, and burning), signs (scalp erythema, perifollicular erythema, and peripilar desquamation), the hair-pull test (presence of anagen hairs), and progression (the patient’s perception of clinical worsening). After assigning a score for each, an equation is used to calculate a final activity score. Although it may not always provide an accurate disease activity estimate, it can be used in conjunction with other assessment tools.[10][30]
Evaluation
Laboratory testing is indicated when information gathered during the history and physical exam suggests an underlying comorbidity or the cause of alopecia is unclear. Generally, tests may include a complete blood count, complete metabolic panel, iron studies (serum ferritin, serum iron, and total iron binding capacity), thyroid-stimulating hormone (TSH), and vitamin D (25[OH]D). Ferritin and vitamin D are essential for a normal hair cycle. [22]
Scalp biopsy offers additional information in nonscarring alopecias and is essential in scarring alopecias. Generally, 1 or 2 4-mm punch biopsies are taken in the direction of the hair shaft down to the subcutaneous fat, allowing for horizontal or vertical sectioning. Horizontal sections have the advantage of facilitating the assessment of many follicles for hair density, anagen-to-telogen hair ratios, T-to-V hair ratios, and the extent of inflammatory infiltrate when present.[22]
Nonscarring Alopecias
Androgenetic alopecia
Androgenetic alopecia is most commonly diagnosed clinically. A complete blood count, complete metabolic panel, iron studies, TSH, and 25(OH)D may be helpful. Laboratory testing is recommended for females presenting with signs or symptoms of hyperandrogenism, such as hirsutism, menstrual irregularities, acanthosis nigricans, and adult acne, including free or total testosterone with or without dehydroepiandrosterone sulfate (DHEAS).[32] A scalp biopsy may be needed when a coexisting scalp condition is suspected. Histologic examination is likely to show terminal hair miniaturization, decreased hair shaft thickness, increased percentage of telogen hairs, and fewer total number of hairs per unit area.[32][33]
Alopecia areata
Alopecia areata is usually diagnosed based on the history and physical (see History and Physical section for details). When clinically uncertain, additional evaluation may include serology for other autoimmune disorders or infectious diseases (eg, syphilis), a fungal culture, and a scalp biopsy.[6] A single biopsy usually suffices and should be taken from the edge of the lesion, avoiding sites susceptible to androgenetic alopecia when possible.[24] Histologic examination during acute disease likely shows dense lymphocytic infiltration of anagen hair bulbs and dermal papillae.
Telogen effluvium
Telogen effluvium is typically diagnosed clinically. If needed, additional investigations may include a complete blood count, iron studies (serum ferritin, serum iron, and total iron binding capacity), TSH, T3, and T4 because 2 common conditions associated with telogen effluvium are iron deficiency anemia and thyroid disorders.[34] A serum zinc level and antinuclear antibody titer may also be indicated.[35] Scalp biopsy for acute telogen effluvium (shedding lasting for less than 6 months) will show an increase in telogen follicles. Normal counts are 6% to 13%; more than 15% suggests telogen effluvium, and more than 25% is definitive. A scalp biopsy can also help to rule out female pattern hair loss and alopecia areata.[34] Multiple biopsies increase the diagnostic accuracy of chronic telogen effluvium, with an anagen-to-telogen ratio of 8:1 compared with 14:1 with normal scalp biopsies.[27]
Scarring Alopecias
FFA and LPP share many histological features; however, differences have been found, which support FFA as a specific type of lymphocytic cicatricial alopecia rather than an LPP variant.[36] Histopathology is the gold standard for diagnosis but not ideal for monitoring because biopsies are invasive.[37]
FFA
FFA lesions are characterized by a dense lichenoid lymphocytic infiltrate around the infundibulum and isthmus of the hair follicle. An absence of sebaceous glands is an early finding. Additional features may include vacuolar degeneration of the basal layer of the follicular epithelium, keratinocyte necrosis, apoptosis in the outer sheath, fibrous tracts, and elastin fiber loss.[9][11][37]
There are no specific laboratory tests for FFA or LPP. Basic serology, thyroid function tests, antinuclear antibodies, and sex hormone levels may be ordered, but findings are usually normal. Currently, there is no consensus on whether or not to screen all patients with FFA or LPP.[37][38][39]
LLP
As with FFA, LPP lesions are characterized by a lymphocytic infiltrate around the infundibulum and isthmus of the hair follicle. A lichenoid pattern may be present; the epithelium-stroma junction will likely show necrotic keratinocytes, and the lower portion of the hair follicle may demonstrate inflammatory cells. Sebaceous glands are usually totally destroyed, and destroyed hair follicles are replaced by connective tissue or sclerotic fibrous tracts.[11][40]
Treatment / Management
Management varies according to the type of alopecia.
Nonscarring Alopecias
Androgenetic alopecia
Androgenetic alopecia treatment and management is aimed at stopping hair loss, preventing or slowing further hair thinning and loss, and promoting the resumption of hair growth. Topical minoxidil and oral finasteride are the first-line treatments for males, and topical minoxidil is the first-line treatment for females. Numerous alternatives exist, including other pharmacologic treatments, platelet-rich plasma, low-level laser therapy, microneedling adjunctive therapy, and autologous hair transplantation. Treating comorbidities such as hyperandrogenism and nutritional deficiency may help maintain hair growth.[1][3][5]
Alopecia areata
Alopecia areata therapies "aim to immunosuppress or immunomodulate the activity of the disease."[6] The Alopecia Areata Consensus of Experts (ACE) study: Results of an international expert opinion on treatments for alopecia areata was published in 2020.[41] Topical corticosteroids are recommended as a first-line therapy for treating limited patchy alopecia areata and adjunctive therapy in more severe forms. Intralesional corticosteroid injections are also a first-line recommendation for limited patchy alopecia areata, alone or combined with topical corticosteroids. The preferred corticosteroid is triamcinolone acetonide. Oral corticosteroids can be used as first-line treatment for moderate-to-severe disease (SALT score >30%) in adults, alone or combined with topical corticosteroids.[41](B3)
Other therapeutic options include topical sensitization with diphenylcyclopropenone (DPCP) or anthralin and methotrexate with or without oral corticosteroids. [6] Also, the U. Food and Drug Administration recently approved the JAK inhibitor baricitinib to treat adult patients with severe alopecia areata. Pediatric patients with a few alopecic patches can be managed by watchful waiting because up to 50% demonstrate spontaneous regrowth within 6 to 12 months of onset. Topical corticosteroids can be used in patients younger than 10, intralesional steroid injections for those older than 10, and systemic corticosteroids for alopecia areata refractory to other local treatments.[5] Nonsteroidal treatments include topical immunotherapy with DPCP or anthralin and methotrexate as an adjunctive treatment for severe disease in patients older than 12 when standard therapies have failed.[6](B3)
Telogen effluvium
Telogen effluvium is entirely reversible, with trigger removal and treating the underlying disorder if one is present. An essential aspect of the management is counseling the patient about the condition. Inform them that hair shedding takes 3 to 6 months to cease; therefore, regrowth can be seen 3 to 6 months after removing the trigger, but cosmetically significant regrowth may take 12 to 18 months.
Scarring Alopecias
Due to their rarity, limited evidence exists on the efficacy of the therapeutic options for primary cicatricial alopecias.
FFA and LPP
The aims of treating FFA and LPP are to stop or delay hair loss and progression in the scarring alopecic area(s) and provide symptom relief. There are no specific treatment guidelines for FFA and LPP treatment and management; however, class III or IV topical corticosteroids (with or without intralesional triamcinolone acetonide injections) may be considered. Additional pharmacotherapeutic treatment options are suggested, and psychological support and camouflage techniques are recommended.[10][42][43](B3)
Psychosocial support should be considered when treating and managing all patients with alopecia.
Differential Diagnosis
Various subtypes of alopecia are confused with each other and require detailed examination through dermoscopy and biopsy. Some of the differentials include:
- Androgenetic alopecia: Alopecia areata, telogen effluvium, traction alopecia, trichotillomania, FFA, LPP, and central centrifugal cicatricial alopecia [1]
- Alopecia areata: Tinea capitis, trichotillomania, and temporal triangular alopecia for patchy alopecia areata and female-pattern hair loss, telogen effluvium, and drug-induced alopecia for diffuse forms of alopecia areata [6]
- Telogen effluvium: Androgenetic alopecia and diffuse forms of alopecia areata [44]
- FFA and LPP: Chronic cutaneous lupus erythematosus, central centrifugal cicatricial alopecia, and folliculitis decalvans [45]
Prognosis
The prognosis of alopecia also varies according to the clinical subtype and comorbidities of the patient.
Androgenetic Alopecia: Androgenetic alopecia typically progresses over time, leading to diminished coverage of the affected areas. Progression to complete baldness may occur in males but rarely in females. Early recognition and treatment can aid in halting progression and preserving as much hair as possible.
Alopecia Breata: Alopecia areata has an unpredictable prognosis, often including relapses and remissions, as part of a chronic course. The following factors may indicate a worse prognosis: onset in childhood, long duration, extensive involvement, nail involvement, comorbid autoimmune disease, and positive family history.[3]
Telogen Effluvium: The prognosis of telogen effluvium depends on the ability to remove or treat the triggering factor. The course is self-limited if the underlying etiology resolves spontaneously or can be eliminated.
FFA: FFA prognosis is difficult to predict. Factors associated with a poor prognosis include facial papules, eyelashes body hair loss, and diffuse involvement of the frontotemporal recess. Factors associated with a less severe course include younger age at onset, mild involvement of eyebrows, and a clinical pattern of pseudo fringe.[46][47]
LPP: LPP has an unpredictable clinical course; it may stabilize or continue to progress slowly.
Complications
Although alopecia has few physically harmful effects, it can lead to negative psychological consequences, such as anxiety and depression, in both sexes and all age groups.[5][48] Females are more likely to experience adverse psychosocial consequences than males, including decreased social contact and lower self-esteem.[15][49] Also, children and adolescents with alopecia areata are vulnerable to social isolation and bullying by their peers.[50][51]
Deterrence and Patient Education
There is a dearth of information regarding the impact of patient education on alopecia. In a 2018 population-based study, half of the participants were provided knowledge about androgenetic alopecia; treatment willingness was then compared between the group that received patient education and the one that did not. Those with accurate knowledge about androgenetic alopecia showed a 1.7-fold higher tendency to receive treatment.[52]
Whether these data can be extrapolated to other types of alopecia is unknown. A 2022 pre-test/post-test study to "investigate the efficacy of an online social media video library for improving patient knowledge of hair loss disorders" showed that video-based educational content can effectively educate patients about alopecia. The study included both sexes of all ages with androgenetic alopecia, alopecia areata, telogen effluvium, FFA, and LPP.[53]
Enhancing Healthcare Team Outcomes
Patients with alopecia often initially present to a primary care clinician. A referral may be warranted depending on the type, severity, and patient impact. For example, if scarring alopecia is suspected, a referral to a dermatologist for a biopsy is necessary. A multidisciplinary and interprofessional team of providers will include any of the following: primary care providers, dermatologists, dermatopathologists, psychiatrists, psychologists, pharmacists, and nurses.
An accurate diagnosis can optimize clinical management for patients and help reduce the psychosocial burden. Specialty nurses can help educate patients about expected outcomes and encourage treatment compliance. The clinical pharmacist can help streamline medications to prevent interactions and adverse effects and offer patients medication counseling. Nurses often help coordinate activities and interventions among various clinicians and can provide counsel to patients. A collaborative interprofessional team of clinicians, psychotherapists, nurses, and pharmacists can best treat patients with alopecia, optimizing clinical outcomes while minimizing adverse events.
Ideally, the interprofessional team will work to build long-term relationships with the patient and their family to consistently provide pharmacological and adjunct therapy, patient education, psychological and emotional support, ongoing monitoring, and social support.
Media
(Click Image to Enlarge)
(Click Image to Enlarge)
(Click Image to Enlarge)
(Click Image to Enlarge)
References
Tamashunas NL, Bergfeld WF. Male and female pattern hair loss: Treatable and worth treating. Cleveland Clinic journal of medicine. 2021 Mar 1:88(3):173-182. doi: 10.3949/ccjm.88a.20014. Epub 2021 Mar 1 [PubMed PMID: 33648970]
Bernárdez C, Molina-Ruiz AM, Requena L. Histologic features of alopecias-part I: nonscarring alopecias. Actas dermo-sifiliograficas. 2015 Apr:106(3):158-67. doi: 10.1016/j.ad.2014.07.006. Epub 2014 Oct 25 [PubMed PMID: 25444580]
Wolff H, Fischer TW, Blume-Peytavi U. The Diagnosis and Treatment of Hair and Scalp Diseases. Deutsches Arzteblatt international. 2016 May 27:113(21):377-86. doi: 10.3238/arztebl.2016.0377. Epub [PubMed PMID: 27504707]
Phillips TG, Slomiany WP, Allison R. Hair Loss: Common Causes and Treatment. American family physician. 2017 Sep 15:96(6):371-378 [PubMed PMID: 28925637]
Xu L, Liu KX, Senna MM. A Practical Approach to the Diagnosis and Management of Hair Loss in Children and Adolescents. Frontiers in medicine. 2017:4():112. doi: 10.3389/fmed.2017.00112. Epub 2017 Jul 24 [PubMed PMID: 28791288]
Lintzeri DA, Constantinou A, Hillmann K, Ghoreschi K, Vogt A, Blume-Peytavi U. Alopecia areata - Current understanding and management. Journal der Deutschen Dermatologischen Gesellschaft = Journal of the German Society of Dermatology : JDDG. 2022 Jan:20(1):59-90. doi: 10.1111/ddg.14689. Epub [PubMed PMID: 35040577]
Level 3 (low-level) evidenceMalkud S. Telogen Effluvium: A Review. Journal of clinical and diagnostic research : JCDR. 2015 Sep:9(9):WE01-3. doi: 10.7860/JCDR/2015/15219.6492. Epub 2015 Sep 1 [PubMed PMID: 26500992]
Olsen EA, Bergfeld WF, Cotsarelis G, Price VH, Shapiro J, Sinclair R, Solomon A, Sperling L, Stenn K, Whiting DA, Bernardo O, Bettencourt M, Bolduc C, Callendar V, Elston D, Hickman J, Ioffreda M, King L, Linzon C, McMichael A, Miller J, Mulinari F, Trancik R, Workshop on Cicatricial Alopecia. Summary of North American Hair Research Society (NAHRS)-sponsored Workshop on Cicatricial Alopecia, Duke University Medical Center, February 10 and 11, 2001. Journal of the American Academy of Dermatology. 2003 Jan:48(1):103-10 [PubMed PMID: 12522378]
Porriño-Bustamante ML, Fernández-Pugnaire MA, Arias-Santiago S. Frontal Fibrosing Alopecia: A Review. Journal of clinical medicine. 2021 Apr 21:10(9):. doi: 10.3390/jcm10091805. Epub 2021 Apr 21 [PubMed PMID: 33919069]
Fechine COC, Valente NYS, Romiti R. Lichen planopilaris and frontal fibrosing alopecia: review and update of diagnostic and therapeutic features. Anais brasileiros de dermatologia. 2022 May-Jun:97(3):348-357. doi: 10.1016/j.abd.2021.08.008. Epub 2022 Apr 2 [PubMed PMID: 35379508]
Fanti PA, Baraldi C, Misciali C, Piraccini BM. Cicatricial alopecia. Giornale italiano di dermatologia e venereologia : organo ufficiale, Societa italiana di dermatologia e sifilografia. 2018 Apr:153(2):230-242. doi: 10.23736/S0392-0488.18.05889-3. Epub 2018 Jan 24 [PubMed PMID: 29368842]
Vañó-Galván S, Saceda-Corralo D, Blume-Peytavi U, Cucchía J, Dlova NC, Gavazzoni Dias MFR, Grimalt R, Guzmán-Sánchez D, Harries M, Ho A, Holmes S, Larrondo J, Mosam A, Oliveira-Soares R, Pinto GM, Piraccini BM, Pirmez R, De la Rosa Carrillo D, Rudnicka L, Shapiro J, Sinclair R, Tosti A, Trüeb RM, Vogt A, Miteva M. Frequency of the Types of Alopecia at Twenty-Two Specialist Hair Clinics: A Multicenter Study. Skin appendage disorders. 2019 Aug:5(5):309-315. doi: 10.1159/000496708. Epub 2019 Apr 2 [PubMed PMID: 31559256]
Level 2 (mid-level) evidenceYip L, Rufaut N, Sinclair R. Role of genetics and sex steroid hormones in male androgenetic alopecia and female pattern hair loss: an update of what we now know. The Australasian journal of dermatology. 2011 May:52(2):81-8. doi: 10.1111/j.1440-0960.2011.00745.x. Epub 2011 Mar 29 [PubMed PMID: 21605090]
Redler S, Brockschmidt FF, Tazi-Ahnini R, Drichel D, Birch MP, Dobson K, Giehl KA, Herms S, Refke M, Kluck N, Kruse R, Lutz G, Wolff H, Böhm M, Becker T, Nöthen MM, Messenger AG, Betz RC. Investigation of the male pattern baldness major genetic susceptibility loci AR/EDA2R and 20p11 in female pattern hair loss. The British journal of dermatology. 2012 Jun:166(6):1314-8. doi: 10.1111/j.1365-2133.2012.10877.x. Epub [PubMed PMID: 22309448]
Level 2 (mid-level) evidenceVujovic A, Del Marmol V. The female pattern hair loss: review of etiopathogenesis and diagnosis. BioMed research international. 2014:2014():767628. doi: 10.1155/2014/767628. Epub 2014 Apr 9 [PubMed PMID: 24812631]
Paus R, Bulfone-Paus S, Bertolini M. Hair Follicle Immune Privilege Revisited: The Key to Alopecia Areata Management. The journal of investigative dermatology. Symposium proceedings. 2018 Jan:19(1):S12-S17. doi: 10.1016/j.jisp.2017.10.014. Epub [PubMed PMID: 29273098]
Bertolini M, McElwee K, Gilhar A, Bulfone-Paus S, Paus R. Hair follicle immune privilege and its collapse in alopecia areata. Experimental dermatology. 2020 Aug:29(8):703-725. doi: 10.1111/exd.14155. Epub [PubMed PMID: 32682334]
Martinez-Mir A, Zlotogorski A, Gordon D, Petukhova L, Mo J, Gilliam TC, Londono D, Haynes C, Ott J, Hordinsky M, Nanova K, Norris D, Price V, Duvic M, Christiano AM. Genomewide scan for linkage reveals evidence of several susceptibility loci for alopecia areata. American journal of human genetics. 2007 Feb:80(2):316-28 [PubMed PMID: 17236136]
Harries MJ, Jimenez F, Izeta A, Hardman J, Panicker SP, Poblet E, Paus R. Lichen Planopilaris and Frontal Fibrosing Alopecia as Model Epithelial Stem Cell Diseases. Trends in molecular medicine. 2018 May:24(5):435-448. doi: 10.1016/j.molmed.2018.03.007. Epub 2018 Apr 13 [PubMed PMID: 29661566]
Tavakolpour S, Mahmoudi H, Abedini R, Kamyab Hesari K, Kiani A, Daneshpazhooh M. Frontal fibrosing alopecia: An update on the hypothesis of pathogenesis and treatment. International journal of women's dermatology. 2019 Jun:5(2):116-123. doi: 10.1016/j.ijwd.2018.11.003. Epub 2019 Jan 23 [PubMed PMID: 30997385]
Stefanato CM. Histopathology of alopecia: a clinicopathological approach to diagnosis. Histopathology. 2010 Jan:56(1):24-38. doi: 10.1111/j.1365-2559.2009.03439.x. Epub [PubMed PMID: 20055903]
Jackson AJ, Price VH. How to diagnose hair loss. Dermatologic clinics. 2013 Jan:31(1):21-8. doi: 10.1016/j.det.2012.08.007. Epub 2012 Sep 29 [PubMed PMID: 23159173]
McDonald KA, Shelley AJ, Colantonio S, Beecker J. Hair pull test: Evidence-based update and revision of guidelines. Journal of the American Academy of Dermatology. 2017 Mar:76(3):472-477. doi: 10.1016/j.jaad.2016.10.002. Epub 2016 Dec 20 [PubMed PMID: 28010890]
Meah N, Wall D, York K, Bhoyrul B, Bokhari L, Asz-Sigall D, Bergfeld WF, Betz RC, Blume-Peytavi U, Callender V, Chitreddy V, Combalia A, Cotsarelis G, Craiglow B, Donovan J, Eisman S, Farrant P, Green J, Grimalt R, Harries M, Hordinsky M, Irvine AD, Itami S, Jolliffe V, King B, Lee WS, McMichael A, Messenger A, Mirmirani P, Olsen E, Orlow SJ, Piraccini BM, Rakowska A, Reygagne P, Roberts JL, Rudnicka L, Shapiro J, Sharma P, Tosti A, Vogt A, Wade M, Yip L, Zlotogorski A, Sinclair RD. The Alopecia Areata Consensus of Experts (ACE) study part II: Results of an international expert opinion on diagnosis and laboratory evaluation for alopecia areata. Journal of the American Academy of Dermatology. 2021 Jun:84(6):1594-1601. doi: 10.1016/j.jaad.2020.09.028. Epub 2020 Sep 12 [PubMed PMID: 32926985]
Level 3 (low-level) evidenceOlsen EA, Hordinsky MK, Price VH, Roberts JL, Shapiro J, Canfield D, Duvic M, King LE Jr, McMichael AJ, Randall VA, Turner ML, Sperling L, Whiting DA, Norris D, National Alopecia Areata Foundation. Alopecia areata investigational assessment guidelines--Part II. National Alopecia Areata Foundation. Journal of the American Academy of Dermatology. 2004 Sep:51(3):440-7 [PubMed PMID: 15337988]
Level 1 (high-level) evidenceMiteva M, Tosti A. Hair and scalp dermatoscopy. Journal of the American Academy of Dermatology. 2012 Nov:67(5):1040-8. doi: 10.1016/j.jaad.2012.02.013. Epub 2012 Mar 8 [PubMed PMID: 22405573]
Asghar F, Shamim N, Farooque U, Sheikh H, Aqeel R. Telogen Effluvium: A Review of the Literature. Cureus. 2020 May 27:12(5):e8320. doi: 10.7759/cureus.8320. Epub 2020 May 27 [PubMed PMID: 32607303]
Vañó-Galván S, Saceda-Corralo D, Moreno-Arrones ÓM, Camacho-Martinez FM. Updated diagnostic criteria for frontal fibrosing alopecia. Journal of the American Academy of Dermatology. 2018 Jan:78(1):e21-e22. doi: 10.1016/j.jaad.2017.08.062. Epub [PubMed PMID: 29241802]
Tolkachjov SN, Chaudhry HM, Imhof RL, Camilleri MJ, Torgerson RR. Reply to: "Updated diagnostic criteria for frontal fibrosing alopecia". Journal of the American Academy of Dermatology. 2018 Jan:78(1):e23-e24. doi: 10.1016/j.jaad.2017.09.027. Epub [PubMed PMID: 29241803]
Naeini FF, Saber M, Faghihi G. Lichen planopilaris: A review of evaluation methods. Indian journal of dermatology, venereology and leprology. 2021 May-Jun:87(3):442-445. doi: 10.25259/IJDVL_775_18. Epub [PubMed PMID: 33871194]
Chiang C, Sah D, Cho BK, Ochoa BE, Price VH. Hydroxychloroquine and lichen planopilaris: efficacy and introduction of Lichen Planopilaris Activity Index scoring system. Journal of the American Academy of Dermatology. 2010 Mar:62(3):387-92. doi: 10.1016/j.jaad.2009.08.054. Epub 2010 Jan 12 [PubMed PMID: 20061052]
Level 2 (mid-level) evidenceOlsen EA, Messenger AG, Shapiro J, Bergfeld WF, Hordinsky MK, Roberts JL, Stough D, Washenik K, Whiting DA. Evaluation and treatment of male and female pattern hair loss. Journal of the American Academy of Dermatology. 2005 Feb:52(2):301-11 [PubMed PMID: 15692478]
Blume-Peytavi U, Blumeyer A, Tosti A, Finner A, Marmol V, Trakatelli M, Reygagne P, Messenger A, European Consensus Group. S1 guideline for diagnostic evaluation in androgenetic alopecia in men, women and adolescents. The British journal of dermatology. 2011 Jan:164(1):5-15. doi: 10.1111/j.1365-2133.2010.10011.x. Epub 2010 Dec 8 [PubMed PMID: 20795997]
Level 3 (low-level) evidenceShrivastava SB. Diffuse hair loss in an adult female: approach to diagnosis and management. Indian journal of dermatology, venereology and leprology. 2009 Jan-Feb:75(1):20-7; quiz 27-8 [PubMed PMID: 19172026]
Sinclair R. Diffuse hair loss. International journal of dermatology. 1999 May:38 Suppl 1():8-18 [PubMed PMID: 10369535]
Poblet E, Jiménez F, Pascual A, Piqué E. Frontal fibrosing alopecia versus lichen planopilaris: a clinicopathological study. International journal of dermatology. 2006 Apr:45(4):375-80 [PubMed PMID: 16650161]
Lis-Święty A, Brzezińska-Wcisło L. Frontal fibrosing alopecia: a disease that remains enigmatic. Postepy dermatologii i alergologii. 2020 Aug:37(4):482-489. doi: 10.5114/ada.2020.98241. Epub 2020 Sep 2 [PubMed PMID: 32994767]
Moreno-Ramírez D, Ferrándiz L, Camacho FM. [Diagnostic and therapeutic assessment of frontal fibrosing alopecia]. Actas dermo-sifiliograficas. 2007 Nov:98(9):594-602 [PubMed PMID: 17961448]
Iorizzo M, Tosti A. Frontal Fibrosing Alopecia: An Update on Pathogenesis, Diagnosis, and Treatment. American journal of clinical dermatology. 2019 Jun:20(3):379-390. doi: 10.1007/s40257-019-00424-y. Epub [PubMed PMID: 30659454]
Sperling LC, Cowper SE. The histopathology of primary cicatricial alopecia. Seminars in cutaneous medicine and surgery. 2006 Mar:25(1):41-50 [PubMed PMID: 16616302]
Meah N, Wall D, York K, Bhoyrul B, Bokhari L, Sigall DA, Bergfeld WF, Betz RC, Blume-Peytavi U, Callender V, Chitreddy V, Combalia A, Cotsarelis G, Craiglow B, Donovan J, Eisman S, Farrant P, Green J, Grimalt R, Harries M, Hordinsky M, Irvine AD, Itami S, Jolliffe V, King B, Lee WS, McMichael A, Messenger A, Mirmirani P, Olsen E, Orlow SJ, Piraccini BM, Rakowska A, Reygagne P, Roberts JL, Rudnicka L, Shapiro J, Sharma P, Tosti A, Vogt A, Wade M, Yip L, Zlotogorski A, Sinclair R. The Alopecia Areata Consensus of Experts (ACE) study: Results of an international expert opinion on treatments for alopecia areata. Journal of the American Academy of Dermatology. 2020 Jul:83(1):123-130. doi: 10.1016/j.jaad.2020.03.004. Epub 2020 Mar 9 [PubMed PMID: 32165196]
Level 3 (low-level) evidenceKanti V, Röwert-Huber J, Vogt A, Blume-Peytavi U. Cicatricial alopecia. Journal der Deutschen Dermatologischen Gesellschaft = Journal of the German Society of Dermatology : JDDG. 2018 Apr:16(4):435-461. doi: 10.1111/ddg.13498. Epub [PubMed PMID: 29645394]
Cardoso CO, Tolentino S, Gratieri T, Cunha-Filho M, Lopez RFV, Gelfuso GM. Topical Treatment for Scarring and Non-Scarring Alopecia: An Overview of the Current Evidence. Clinical, cosmetic and investigational dermatology. 2021:14():485-499. doi: 10.2147/CCID.S284435. Epub 2021 May 12 [PubMed PMID: 34012282]
Level 3 (low-level) evidenceWerner B, Mulinari-Brenner F. Clinical and histological challenge in the differential diagnosis of diffuse alopecia: female androgenetic alopecia, telogen effluvium and alopecia areata--part II. Anais brasileiros de dermatologia. 2012 Nov-Dec:87(6):884-90 [PubMed PMID: 23197208]
Cummins DM, Chaudhry IH, Harries M. Scarring Alopecias: Pathology and an Update on Digital Developments. Biomedicines. 2021 Nov 24:9(12):. doi: 10.3390/biomedicines9121755. Epub 2021 Nov 24 [PubMed PMID: 34944572]
Moreno-Arrones OM, Saceda-Corralo D, Fonda-Pascual P, Rodrigues-Barata AR, Buendía-Castaño D, Alegre-Sánchez A, Pindado-Ortega C, Molins M, Perosanz D, Segurado-Miravalles G, Jaén P, Vañó-Galván S. Frontal fibrosing alopecia: clinical and prognostic classification. Journal of the European Academy of Dermatology and Venereology : JEADV. 2017 Oct:31(10):1739-1745. doi: 10.1111/jdv.14287. Epub 2017 May 15 [PubMed PMID: 28426907]
Strazzulla LC, Avila L, Li X, Lo Sicco K, Shapiro J. Prognosis, treatment, and disease outcomes in frontal fibrosing alopecia: A retrospective review of 92 cases. Journal of the American Academy of Dermatology. 2018 Jan:78(1):203-205. doi: 10.1016/j.jaad.2017.07.035. Epub [PubMed PMID: 29241787]
Level 2 (mid-level) evidenceHunt N, McHale S. The psychological impact of alopecia. BMJ (Clinical research ed.). 2005 Oct 22:331(7522):951-3 [PubMed PMID: 16239692]
Dinh QQ, Sinclair R. Female pattern hair loss: current treatment concepts. Clinical interventions in aging. 2007:2(2):189-99 [PubMed PMID: 18044135]
Bilgiç Ö, Bilgiç A, Bahalı K, Bahali AG, Gürkan A, Yılmaz S. Psychiatric symptomatology and health-related quality of life in children and adolescents with alopecia areata. Journal of the European Academy of Dermatology and Venereology : JEADV. 2014 Nov:28(11):1463-8. doi: 10.1111/jdv.12315. Epub 2013 Nov 16 [PubMed PMID: 24237476]
Level 2 (mid-level) evidenceChristensen T, Yang JS, Castelo-Soccio L. Bullying and Quality of Life in Pediatric Alopecia Areata. Skin appendage disorders. 2017 Aug:3(3):115-118. doi: 10.1159/000466704. Epub 2017 Mar 24 [PubMed PMID: 28879186]
Level 2 (mid-level) evidenceKim BK, Lee S, Jun M, Chung HC, Oh SS, Lee WS. Perception of Hair Loss and Education Increases the Treatment Willingness in Patients With Androgenetic Alopecica: A Population-Based Study. Annals of dermatology. 2018 Aug:30(4):402-408. doi: 10.5021/ad.2018.30.4.402. Epub 2018 Jun 27 [PubMed PMID: 30065579]
Klein EJ, Karim M, Sukhdeo K, Svigos K, Yin L, Peterson E, Gutierrez D, Shapiro J, Lo Sicco K. Improving knowledge of hair loss disorders: Assessing the efficacy of a dermatologist-developed social media video library for patient education. JAAD international. 2022 Dec:9():122-124. doi: 10.1016/j.jdin.2022.08.022. Epub 2022 Sep 7 [PubMed PMID: 36248208]