Introduction
A myocardium that is dysfunctional due to ischemia but potentially salvageable upon revascularization is viable. Viable myocardium can be ‘stunned’ or ‘hibernating.’ This is believed to be due to the adaptive response of the myocardium to the ischemic milieu with the downregulation of its metabolism and contractility. Myocardial viability assessment is essential in selecting patients with heart failure with reduced ejection fraction (HFrEF) for revascularization. Coronary artery disease is the most common cause of HFrEF (LVEF <40%) worldwide and is termed ischemic cardiomyopathy.[1]
The blood supply of the epicardium, myocardium, and endocardium is by epicardial coronary arteries, and atherosclerotic coronary artery disease is the most common cause of myocardial ischemia, which leads to myocardial necrosis and ischemic cardiomyopathy in the long run.
Repetitive ischemia lasting for a lesser duration causes a temporary loss of contraction with a potential for recovery in the function of viable myocardium in days to weeks, an entity called ‘myocardial stunning.’[2] Chronic stunning is the phenomenon where repetitive episodes of reversible ischemia before myocardial function normalizes lead to chronic myocardial dysfunction.
Myocardial hibernation is an adaptation to chronic ischemia with only a little cell death in which myocardial segments supplied by the coronary artery with fixed stenosis remain noncontractile due to the downregulation of cellular metabolism but retain their potential to recover partially or entirely upon revascularization.[3] It is a state of chronic left ventricular systolic dysfunction resulting from the adaptation of ischemic myocardium to critically reduced perfusion, thus establishing a new balance between reduced flow and function.[3]
Etiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Etiology
Myocardial Hibernation due to Ischemic Cardiomyopathy
Chronic reversible ischemia due to obstructive coronary artery disease leads to a reduction in resting blood flow and subsequent downregulation in metabolism and contractility. This myocardium is viable and recovers partially or fully upon revascularization.
Myocardial Stunning due to Acute Coronary Syndrome
An acute coronary syndrome due to plaque rupture leads to an ischemic cascade downstream and subsequent impairment in LV contractility. The contractile dysfunction persists despite early revascularization and before irreversible myocardial injury sets. Duration of transient post-ischemic dysfunction or stunning is the function of the duration of ischemia. It is believed to be caused by acute ischemia followed by reperfusion injury.
Myocardial Stunning Post-Cardiac Surgery
On-pump cardiac surgical procedures like CABG involve the institution of cardiopulmonary bypass machines, aortic cross-clamping, and cardioplegia, which lead to an interruption in coronary artery blood flow. This leads to ischemia, initially followed by reperfusion injury once the heart is restarted, resulting in reversible post-ischemic systolic dysfunction.[4] This may require using vasopressors and inotropes, mechanical circulatory assist devices like ECMO, thus prolonging the hospital stay.
Left Atrial Stunning Post Cardioversion
Conversion of atrial fibrillation or flutter to sinus rhythm by either electrical or pharmacological cardioversion leads to transient left atrial and left atrial appendage contractile dysfunction lasting about 4 to 6 weeks. A stunned atrium and appendage can be a nidus for thrombus formation; hence, patients should be on anticoagulation for at least four weeks.
Myocardial Stunning in Resuscitated Cardiac Arrest
Myocardial dysfunction may result after the return of spontaneous circulation following a cardiac arrest. It manifests as global myocardial hypokinesis with a severe reduction in ejection fraction, reduction in cardiac output, and hypotension.[5] Studies have shown that myocardial blood flow is not reduced during the period of dysfunction, and myocardial recovery occurs in about 72 hours in survivors.[6]
Epidemiology
In developed countries, ischemic cardiomyopathy due to atherosclerotic coronary artery disease accounts for about 70% of HFrEF patients. Studies have shown that 50% of patients with coronary artery disease and heart failure with reduced ejection have a significant volume of viable myocardium.[7]
The overall mortality in patients with HFrEF due to ischemic cardiomyopathy is 39%, which is lower in patients with viable myocardium than with non-viable myocardium, ie, 37% vs. 51%.[8]
Pathophysiology
Atherosclerosis leads to lipid-rich plaque accumulation in the coronary artery's intima.[9] Plaques with thin fibrous caps, increased macrophage activity, large necrotic lipid-rich cores, and superficial microcalcifications are considered vulnerable as they easily get eroded by exogenous shear stresses leading to plaque fissuring, rupture, destabilization, and exposing the underlying thrombogenic and proinflammatory necrotic core. This leads to the initiation of the coagulation cascade and superimposed thrombosis leading to downstream myocardial ischemia, fatal myocardial infarction, and sudden cardiac death.[10]
Myocardial ischemia follows a temporal sequence of events called an ischemic cascade, a function of the duration of ischemia. It begins with a reduction in perfusion, leading to flow disparity and, thus, impairment of the Na/K ATPase pump, followed by diastolic dysfunction, systolic dysfunction, electrocardiographic changes, and angina. The extent of myocardial ischemia is the function of the duration of obstruction and the area of myocardium subtended by the diseased vessel.
Partial occlusion for more than 5 hours causes patchy infarction, whereas complete occlusion for more than 20 minutes causes irreversible myocyte death, termed transmural infarction. The cascade of irreversible myocardial injury begins from the endocardium and then progresses to the myocardium. If reperfusion is established before the onset of irreversible damage after myocardial infarction, stunning develops, and the time required for recovery depends on the duration of ischemia.
In chronic stunning, oxygen-free radicals released when interrupted blood flow damages the myocytes and cause myocardial stunning. The sequential evolution of the extent of myocardial injury from ischemia depends on the duration of ischemia, residual blood flow from collateral circulation, location of the occlusion, and myocardial oxygen consumption.[2]
Ischemic pre- and post-conditioning are the adaptive responses believed to reduce infarct size. Brief reversible ischemic episodes followed by reperfusion preceding the major ischemic event are cardioprotective as it is believed to reduce the extent of myocardial necrosis. This is called ischemic preconditioning; preconditioned myocytes can tolerate ischemia better than naive myocytes.[11]
Myocardial post-conditioning is when intermittent episodes of ischemia and reperfusion during reperfusion therapy are believed to be cardioprotective by reducing reperfusion injury and infarct size. Complex endogenous second messenger systems like Protein Kinase C, inducible Nitric oxide (iNOS), COX-2, and mitochondrial K channels are believed to be active.[2]
Studies suggest hibernation and stress-induced ischemia without angina are common in patients with HFrEF, and revascularization may improve resting blood flow and left ventricular contractility.[12] Thus, both chronic stunning and hibernation present with reversible systolic dysfunction, but the resting blood flow is normal in myocardial stunning, whereas it is critically reduced in hibernation.[13]
The resting myocardium uses beta-oxidation of fatty acids as a metabolic fuel to produce high-energy phosphates like ATP to power myocardial contraction. However, the ischemic myocardium switches its energy metabolism from fatty acids to anaerobic glycolysis. Hence, glucose uptake is upregulated in ischemic and viable myocardium, and this phenomenon is utilized to assess myocardial viability in 18F-fluorodeoxyglucose (F-FDG) positron emission tomography (PET).[2]
History and Physical
History: Patients with left ventricular dysfunction may be asymptomatic or present with exertional fatigue, palpitation, angina, paroxysmal nocturnal dyspnea, or orthopnea. Sudden cardiac arrest or sudden cardiac death may be the initial presentation.
Physical: General physical examination varies with the stage of the disease and may reveal respiratory distress, cyanosis, elevation in JVP, bilateral pitting pedal edema, or congestive hepatomegaly. The precordial examination may reveal a displaced apex beat or pan systolic murmur of secondary mitral regurgitation.
Evaluation
The physiology of the ischemic cascade is fundamental for a stress test and myocardial viability assessment, as different imaging modalities utilize different stages of the cascade. The following imaging modalities are commonly used in the evaluation of myocardial viability.
Dobutamine Stress Echocardiography
Dobutamine stress echocardiography is used to assess the myocardial viability in patients with resting systolic dysfunction before revascularization. It is based on the principle that viable myocardium will undergo augmentation in contractility in response to beta-adrenergic stimulation, whereas non-viable myocardium will not. Dobutamine is infused at a rate of 2.5 to 10 micrograms/kg/min.
Improvement in contractility at low dose followed by diminished wall motion at high dose (10 to 40 micrograms/kg/min) is termed a biphasic response and suggests viable myocardium. A biphasic response is predictive of recovery in myocardial contractility after revascularization.[14] This phenomenon is explained by an increase in myocardial flow and contractility at low doses but significant myocardial oxygen demand and supply mismatch during the high dose of dobutamine infusion.
Radionuclide Techniques
Radiotracers used in the viability study are the analogs of our endogenous second messenger system and metabolites used in cell signaling and metabolism. Quantitative analysis of these radiotracers' uptake thus yields indirect evidence of intact cellular function and the cell membrane and subcellular organelles' integrity. These tracers have unstable nuclei and electrons, which emit characteristic radiation after being taken up by the myocardial cells, which are converted to photons and imaged by a camera system.
The degree of uptake is proportional to myocardial blood flow. The strength of the emitted radiation correlates with the degree of uptake and, thus, the extent of viability. The infarcted myocardium does not take up tracers; hence, no radiation is emitted by this portion of the myocardium. The images show perfusion defects in that area. Radiotracer perfusion study thus aims to study the flow disparity component of the ischemic cascade, which is the early stage. The areas with normal perfusion are viable, but those with perfusion defects may be infarcted or hibernating.
Metabolic tracers like 18F-FDG, as well as delayed imaging with Thallium-201, help differentiate this. The following imaging protocols are commonly used for viability assessment:
- Thallium-201 Single Photon Emission Computed Tomography (SPECT) Study: Thallium-201 is a potassium analog, and its uptake via Na/K ATPase is evidence of intact cell membrane and, thus, cellular viability. Thallium-201 has a high first-pass myocardial extraction of about 85% after the intravenous injection. The initial myocardial uptake after intravenous injection is a function of blood flow, and hence uptake will be high in normal myocardium and low in ischemic myocardium. Thallium has a unique property of late redistribution from the myocardium to the blood pool, which depends on its concentration difference between the myocardium and blood pool, and not on blood flow. Since the concentration difference between the blood pool and ischemic myocardium is low, washout is low, retaining more TI-201 activity, while washout is high in normal myocardium. This phenomenon of differential washout is utilized in the viability study. The rest or stress images are taken 15 to 20 minutes after injection, and then redistribution images are obtained after 3 to 4 hours. The presence of perfusion defect in the initial images followed by improvement in redistribution images is the hallmark of myocardial viability. The specificity of this study can be further improved by enhancing redistribution by reinjecting the tracer during the redistribution stage or taking late distribution images at 24 to 48 hours later, thus allowing more time for redistribution and improving positive predictive value for improvement in LV function.[15]
- Technetium 99m Sestamibi SPECT: Tc-99m Sestamibi is a cation that diffuses across the cell membrane and then into the mitochondria, and positive uptake is suggestive of viable myocardial cells. The protocols used are rest-stress and rest protocol with nitrate enhancement to promote uptake.[16] Normal uptake is consistent with viable myocardium, moderate uptake is consistent with a mixture of infarct and viable tissue, and absent uptake is consistent with an infarct.
- 18F-Fluorodeoxyglucose (FDG) PET Scan: 18F-FDG is an analog of glucose tagged with a radiotracer, taken up by mitochondria, phosphorylated in the stage of glycolysis, and gets trapped in this state without further metabolism. In hibernating myocardium, GLUT receptors are overexpressed; hence, the myocardium is FDG avid. In ischemic myocardium, beta-oxidation of fatty acids is suppressed by hypoxia, so cells switch to anaerobic glycolysis for energy metabolism. Viable tissue characteristically shows flow-metabolism mismatch. The first component tests for the perfusion using PET radiotracers like rubidium-82, 13-N ammonia, and hibernating myocardium exhibit perfusion defects. The metabolic component of the test consists of keeping the patient in a fasting state for 6 hours, followed by a glucose load, insulin infusion, and finally, radiotracer F-FDG. The uptake in hibernating and normal myocardium is thus facilitated by insulin, and an image with uniform uptake is shown in a PET scanner.[17] The absence of uptake is consistent with an infarct.
Contrast-Enhanced Cardiac Magnetic Resonance (CMR)
The initial method of viability assessment using CMR is quantifying end-diastolic wall thickness (EDWT). A myocardial thickness of less than 6 mm is likely a 'scar' and less likely to improve after revascularization. One of the fundamental histologic changes in necrotic myocardium is extracellular volume expansion and death of myocytes. This change enables the high uptake and retention of high-density gadolinium-based contrast agents in the cell as well as extracellular space, termed late gadolinium enhancement (LGE) and forms the basis for a viability study with contrast-enhanced CMR.[17]
The LGE starts from the endocardium (subendocardial infarct) and then progresses transmurally (transmural infarct) in the segments supplied by the coronary artery. The degree of LGE correlates with the extent of viability and potential benefit from revascularization. The myocardial segment with >50% LGE is non-viable and will not improve after revascularization, <25% is viable and will benefit from revascularization, and 25 to 50% LGE needs to be further investigated for inotropic contractile reserve by a dobutamine infusion and assessment of wall motion in echocardiography or CMR.[18][19]
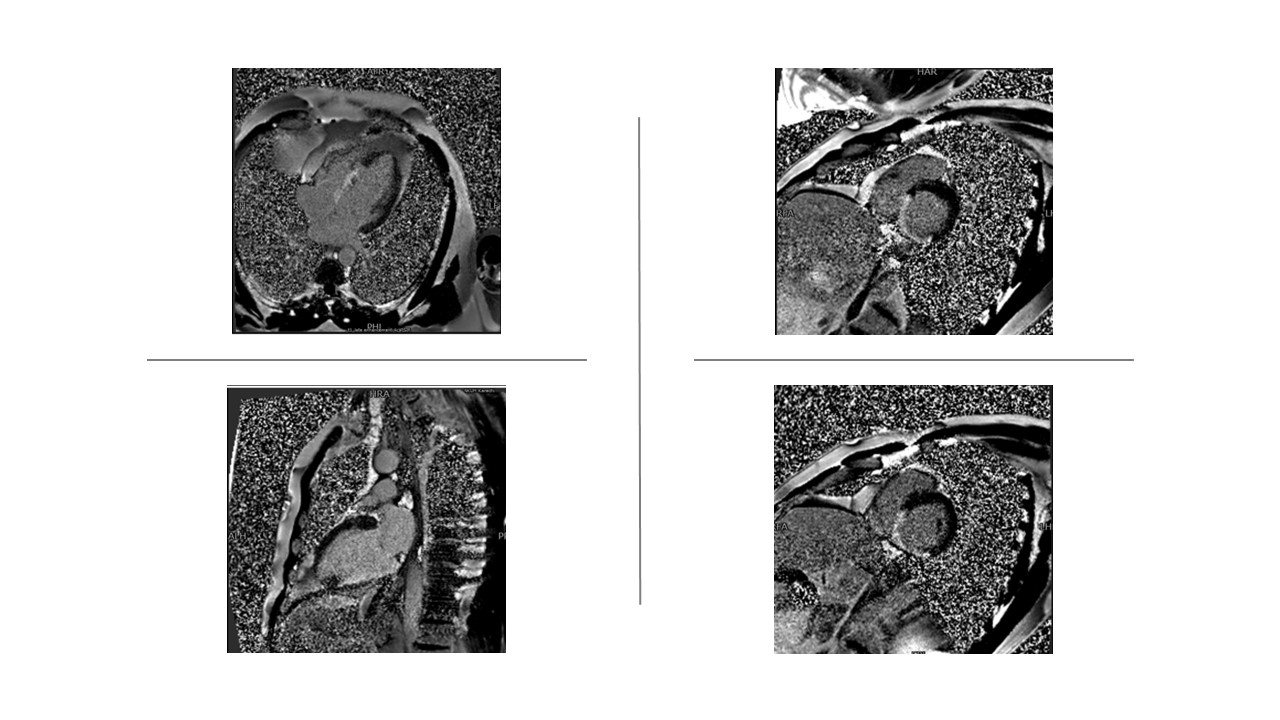
Figure 1 shows a transmural LGE in the right coronary artery consistent with transmural infarction and lack of viability. Dobutamine stress CMR can be used to assess viability. A dysfunctional but viable myocardium will exhibit an increased systolic thickening of over 2 mm on low-dose dobutamine stress.
Treatment / Management
Identifying patients with ischemic cardiomyopathy who would benefit from revascularization in addition to guideline-directed medical therapy (GDMT) is important. All patients with HFrEF should be on optimal medical therapy, irrespective of the presence or absence of viable myocardium.
Medical Therapy
Beta-Blockers: Selected beta-blockers carvedilol, metoprolol, and bisoprolol in maximum tolerated doses have proven mortality and morbidity benefit in HFrEF patients. (ACC/AHA Class IA).
ACEIs/ARBs/Angiotensin-Neprlisyn inhibitors (ARNI): RAAS inhibitors (particularly Enalapril) and ARNI (Sacubitril-Valsartan) also have a mortality benefit (ACC/AHA Class IA). ACEI and ARNIs should not be used together, and a washout period of at least 36 hours should be given before transitioning from ACEI to ARNI. (ACC/AHA Class IA )
Mineralocorticoid Receptor Antagonists (MRA): Spironolactone and eplerenone are proven to reduce mortality and morbidity and are recommended with LOR Class IA by ACC/AHA for NYHA Class II-IV heart failure and LVEF <35%. (ACC/AHA Class IA)
Hydralazine and isosorbide dinitrate: The combination reduces mortality and morbidity and is used predominantly in African American patients with NYHA Class III-IV HFrEF despite receiving beta-blockers and ACEI (Class IA). These drugs can be used as alternative preload and afterload-reducing agents in patients who cannot be on ACEI/ARBs/ARNI due to hypersensitivity, hypotension, and renal failure.
Sodium-Glucose Cotransporter 2 (SGLT2i) Inhibitors: SGLT2i (empagliflozin and dapagliflozin) are shown to reduce heart failure-related hospitalization and cardiovascular mortality in patients with symptomatic HFrEF irrespective of the presence of type 2 diabetes (ACC/AHA Class I).[20]
Coronary Artery Revascularization
CABG surgery is done for the left main disease, triple vessel disease, complex coronary artery disease, and patients with CAD and reduced left ventricular ejection fraction. Percutaneous coronary intervention with balloon angioplasty and stent placement is done for uncomplicated single or two-vessel disease.
Device-Based Therapies for Cardiomyopathy-Related Arrhythmias
An implantable cardioverter defibrillator (ICD) is used in patients with end-stage cardiomyopathy with non-viable myocardium as a prevention against sudden cardiac arrest from malignant ventricular arrhythmias. Cardiac resynchronization therapy (CRT) synchronizes LV and RV contraction in HFrEF, prevents heart failure progression, and improves symptoms.
ICD is indicated for the primary prevention of sudden cardiac death in patients with LVEF <35%, NYHA class II/III heart failure who have been on maximally tolerated doses of beta blocker, ACEI/ARNI, and MRA for 90 days, and with survival for more than a year. Patients with LBBB with a QRS duration of 150 msec or more and NYHA Class II, III symptoms on GDMT benefit from its upgrade to a CRT device.
Differential Diagnosis
Stress-induced cardiomyopathy: Any form of myocardial stress, like sepsis, and emotional and physical trauma, can lead to catecholaminergic surge, microvascular spasm, and myocardial dysfunction. This is characterized by reversible distal myocardial hypo-contractility with basal hyper-contractility in echocardiography. Its presentation is similar to acute coronary syndrome, and it is a diagnosis of exclusion. Obstructive coronary artery disease needs to be ruled out by cardiac catheterization.
LV Systolic dysfunction due to myocarditis: Myocarditis due to viruses like Coxsackie and Parvovirus B19 can lead to transient reversible myocardial dysfunction.
Tachycardia-induced cardiomyopathy: Any tachycardia like atrial fibrillation with a rapid ventricular response, supraventricular tachycardia, and atrial flutter with rapid ventricular response can cause reversible myocardial systolic dysfunction. Myocardial function recovers after the rate is controlled.
Peripartum cardiomyopathy: Peripartum cardiomyopathy is defined by the development of congestive heart failure in the last month of pregnancy or within five weeks of puerperium. It is thought to be due to the hemodynamic stress of pregnancy on the heart. LV systolic function often improves with time.
Pertinent Studies and Ongoing Trials
Extensive landmark studies assessing the mortality benefits of revascularization in a viable myocardium are not available, and studies are ongoing. Small- and moderate-sized studies showed no mortality benefits with revascularization and medical therapy versus medical therapy only in patients with viable myocardium. But these studies were statistically underpowered and had many confounding factors. In a sub-analysis of the STICH (Surgical Treatment for Ischemic Heart Failure) trial, 601 patients underwent a viability study before revascularization; about half received optimal medical therapy with CABG, and half received optimal medical therapy only. The arm with CABG and medical therapy had lower mortality, but it was statistically insignificant.[2]
The STICH trial additionally showed that the increase in ejection fraction was more in patients with viable myocardium. Still, this increase was similar in both CABG with medical therapy and medical therapy-only groups. PARR-2 (FDG-PET assisted management of patients with severe left ventricular dysfunction and suspected coronary disease: a randomized, controlled trial) study showed PET viability imaging did not demonstrate survival benefits with revascularization compared to medical therapy.[21]
The most important benefit mechanism from revascularization may be an improvement in ejection fraction and prevention of future coronary events, as the patients who benefitted most in the STICH trial were those with multivessel disease and severely reduced ejection fraction.[22]
The benefits of CABG can be extrapolated from survival benefits to improving quality of life and exercise tolerance in patients with ischemic cardiomyopathy.[23] The revascularized myocardium is electrically stable and less likely to be a source of malignant ventricular arrhythmias.
Prognosis
It is essential to diagnose hibernating myocardium in patients with ischemic cardiomyopathy because symptoms of chronic LV dysfunction may not always be from necrotic and scarred myocardium from previous MIs, but rather from reversible ischemia. Hibernating myocardium may be the predominant etiology of chronic heart failure in patients with a history of known or suspected coronary artery disease, and the degree of cardiac dysfunction may not be solely accounted for by previous MIs.
The prognosis of ischemic cardiomyopathy with scarred myocardium due to previous MIs is dismal, and ventricular assist devices and cardiac transplant may be the only alternatives ultimately.[8] However, the prognosis of patients with hibernating myocardium presenting as chronic heart failure may be better with revascularization. ACC/AHA recommends myocardial viability testing in chronic ischemic cardiomyopathy with Class IIa level of recommendation (LOR).
Complications
Untreated myocardial dysfunction may lead to:
- End-stage heart failure
- Multi-organ failure
- Heart failure hospitalization
- Arrhythmia hospitalization
- Sudden cardiac death
- Need for device-based therapy/left ventricular assist devices.
- Need for cardiac transplantation
Deterrence and Patient Education
The importance of compliance with medical therapy should be discussed with patients with HFrEF as these medications are proven to halt the progression of cardiomyopathy, improve ejection fraction and have mortality and morbidity benefits. Regular follow-up to ensure optimal fluid-volume status, electrolytes, and renal function is important as these medications frequently cause renal injury and hyperkalemia. Revascularization is likely to improve the patient's heart function if viability is present.
Enhancing Healthcare Team Outcomes
Managing patients with chronic heart failure requires close multidisciplinary collaborative efforts of emergency physicians, hospitalists, general cardiologists, advanced cardiovascular imaging specialists, nuclear cardiologists, radiologists, interventional cardiologists, cardiothoracic surgeons, and nurses.
Acutely decompensated patients generally present in the emergency room and are initially seen by emergency physicians and get admitted to floors under a hospitalist or in CCU under a general cardiologist, where the patients are started on appropriate medications, including diuretics, inotropes, and vasopressors. Advanced cardiovascular imaging physicians, radiologists, and nuclear cardiologists are responsible for interpreting cardiac imaging and viability study.
If substantial viability is present, an interventional cardiologist or a cardiothoracic surgeon should be consulted for PCI or CABG. A general cardiologist follows the patient in the clinic. When medical therapy is used, the pharmacist should provide input regarding the drugs chosen, verify dosing, perm medication reconciliation, and can counsel the patients on their medications.
Nurses are essential in medicating patients, monitoring telemetry, vitals, fluid, and volume status while inpatient, counseling about dietary and lifestyle modification, following them up in heart failure clinics, and monitoring them during cardiac rehabilitation sessions. Nurses must be empowered to bring any concerns to the attention of the clinicians, and their suggestions must be received as those of a colleague. This type of open communication model is crucial for interprofessional care to succeed.
Media
(Click Image to Enlarge)
References
Tsao CW, Aday AW, Almarzooq ZI, Alonso A, Beaton AZ, Bittencourt MS, Boehme AK, Buxton AE, Carson AP, Commodore-Mensah Y, Elkind MSV, Evenson KR, Eze-Nliam C, Ferguson JF, Generoso G, Ho JE, Kalani R, Khan SS, Kissela BM, Knutson KL, Levine DA, Lewis TT, Liu J, Loop MS, Ma J, Mussolino ME, Navaneethan SD, Perak AM, Poudel R, Rezk-Hanna M, Roth GA, Schroeder EB, Shah SH, Thacker EL, VanWagner LB, Virani SS, Voecks JH, Wang NY, Yaffe K, Martin SS. Heart Disease and Stroke Statistics-2022 Update: A Report From the American Heart Association. Circulation. 2022 Feb 22:145(8):e153-e639. doi: 10.1161/CIR.0000000000001052. Epub 2022 Jan 26 [PubMed PMID: 35078371]
Kloner RA, Jennings RB. Consequences of brief ischemia: stunning, preconditioning, and their clinical implications: part 2. Circulation. 2001 Dec 18:104(25):3158-67 [PubMed PMID: 11748117]
Level 3 (low-level) evidenceRahimtoola SH. The hibernating myocardium. American heart journal. 1989 Jan:117(1):211-21 [PubMed PMID: 2783527]
Leung JM. Clinical evidence of myocardial stunning in patients undergoing CABG surgery. Journal of cardiac surgery. 1993 Mar:8(2 Suppl):220-3 [PubMed PMID: 8461506]
Kern KB, Hilwig RW, Rhee KH, Berg RA. Myocardial dysfunction after resuscitation from cardiac arrest: an example of global myocardial stunning. Journal of the American College of Cardiology. 1996 Jul:28(1):232-40 [PubMed PMID: 8752819]
Level 3 (low-level) evidenceLaurent I, Monchi M, Chiche JD, Joly LM, Spaulding C, Bourgeois B, Cariou A, Rozenberg A, Carli P, Weber S, Dhainaut JF. Reversible myocardial dysfunction in survivors of out-of-hospital cardiac arrest. Journal of the American College of Cardiology. 2002 Dec 18:40(12):2110-6 [PubMed PMID: 12505221]
Di Carli MF, Davidson M, Little R, Khanna S, Mody FV, Brunken RC, Czernin J, Rokhsar S, Stevenson LW, Laks H. Value of metabolic imaging with positron emission tomography for evaluating prognosis in patients with coronary artery disease and left ventricular dysfunction. The American journal of cardiology. 1994 Mar 15:73(8):527-33 [PubMed PMID: 8147295]
Level 2 (mid-level) evidencePerera D, Clayton T, O'Kane PD, Greenwood JP, Weerackody R, Ryan M, Morgan HP, Dodd M, Evans R, Canter R, Arnold S, Dixon LJ, Edwards RJ, De Silva K, Spratt JC, Conway D, Cotton J, McEntegart M, Chiribiri A, Saramago P, Gershlick A, Shah AM, Clark AL, Petrie MC, REVIVED-BCIS2 Investigators. Percutaneous Revascularization for Ischemic Left Ventricular Dysfunction. The New England journal of medicine. 2022 Oct 13:387(15):1351-1360. doi: 10.1056/NEJMoa2206606. Epub 2022 Aug 27 [PubMed PMID: 36027563]
Nepal S, Bishop MA. Spontaneous Coronary Artery Dissection. StatPearls. 2023 Jan:(): [PubMed PMID: 35881748]
Virmani R, Kolodgie FD, Burke AP, Farb A, Schwartz SM. Lessons from sudden coronary death: a comprehensive morphological classification scheme for atherosclerotic lesions. Arteriosclerosis, thrombosis, and vascular biology. 2000 May:20(5):1262-75 [PubMed PMID: 10807742]
Cohen MV, Downey JM. Signalling pathways and mechanisms of protection in pre- and postconditioning: historical perspective and lessons for the future. British journal of pharmacology. 2015 Apr:172(8):1913-32. doi: 10.1111/bph.12903. Epub 2014 Nov 24 [PubMed PMID: 25205071]
Level 3 (low-level) evidencePatel MR, White RD, Abbara S, Bluemke DA, Herfkens RJ, Picard M, Shaw LJ, Silver M, Stillman AE, Udelson J, American College of Radiology Appropriateness Criteria Committee, American College of Cardiology Foundation Appropriate Use Criteria Task Force. 2013 ACCF/ACR/ASE/ASNC/SCCT/SCMR appropriate utilization of cardiovascular imaging in heart failure: a joint report of the American College of Radiology Appropriateness Criteria Committee and the American College of Cardiology Foundation Appropriate Use Criteria Task Force. Journal of the American College of Cardiology. 2013 May 28:61(21):2207-31. doi: 10.1016/j.jacc.2013.02.005. Epub 2013 Apr 23 [PubMed PMID: 23500216]
Panza JA, Chrzanowski L, Bonow RO. Myocardial Viability Assessment Before Surgical Revascularization in Ischemic Cardiomyopathy: JACC Review Topic of the Week. Journal of the American College of Cardiology. 2021 Sep 7:78(10):1068-1077. doi: 10.1016/j.jacc.2021.07.004. Epub [PubMed PMID: 34474740]
Camici PG, Prasad SK, Rimoldi OE. Stunning, hibernation, and assessment of myocardial viability. Circulation. 2008 Jan 1:117(1):103-14. doi: 10.1161/CIRCULATIONAHA.107.702993. Epub [PubMed PMID: 18172050]
Henzlova MJ, Duvall WL, Einstein AJ, Travin MI, Verberne HJ. ASNC imaging guidelines for SPECT nuclear cardiology procedures: Stress, protocols, and tracers. Journal of nuclear cardiology : official publication of the American Society of Nuclear Cardiology. 2016 Jun:23(3):606-39. doi: 10.1007/s12350-015-0387-x. Epub [PubMed PMID: 26914678]
Bisi G, Sciagrà R, Santoro GM, Fazzini PF. Rest technetium-99m sestamibi tomography in combination with short-term administration of nitrates: feasibility and reliability for prediction of postrevascularization outcome of asynergic territories. Journal of the American College of Cardiology. 1994 Nov 1:24(5):1282-9 [PubMed PMID: 7930251]
Level 1 (high-level) evidenceGarcia MJ, Kwong RY, Scherrer-Crosbie M, Taub CC, Blankstein R, Lima J, Bonow RO, Eshtehardi P, Bois JP, American Heart Association Council on Cardiovascular Radiology and Intervention and Council on Clinical Cardiology. State of the Art: Imaging for Myocardial Viability: A Scientific Statement From the American Heart Association. Circulation. Cardiovascular imaging. 2020 Jul:13(7):e000053. doi: 10.1161/HCI.0000000000000053. Epub 2020 Jul 13 [PubMed PMID: 32833510]
Kim RJ, Wu E, Rafael A, Chen EL, Parker MA, Simonetti O, Klocke FJ, Bonow RO, Judd RM. The use of contrast-enhanced magnetic resonance imaging to identify reversible myocardial dysfunction. The New England journal of medicine. 2000 Nov 16:343(20):1445-53 [PubMed PMID: 11078769]
Baer FM, Theissen P, Crnac J, Schmidt M, Deutsch HJ, Sechtem U, Schicha H, Erdmann E. Head to head comparison of dobutamine-transoesophageal echocardiography and dobutamine-magnetic resonance imaging for the prediction of left ventricular functional recovery in patients with chronic coronary artery disease. European heart journal. 2000 Jun:21(12):981-91 [PubMed PMID: 10901510]
Zelniker TA, Braunwald E. Mechanisms of Cardiorenal Effects of Sodium-Glucose Cotransporter 2 Inhibitors: JACC State-of-the-Art Review. Journal of the American College of Cardiology. 2020 Feb 4:75(4):422-434. doi: 10.1016/j.jacc.2019.11.031. Epub [PubMed PMID: 32000955]
Beanlands RS, Nichol G, Huszti E, Humen D, Racine N, Freeman M, Gulenchyn KY, Garrard L, deKemp R, Guo A, Ruddy TD, Benard F, Lamy A, Iwanochko RM, PARR-2 Investigators. F-18-fluorodeoxyglucose positron emission tomography imaging-assisted management of patients with severe left ventricular dysfunction and suspected coronary disease: a randomized, controlled trial (PARR-2). Journal of the American College of Cardiology. 2007 Nov 13:50(20):2002-12 [PubMed PMID: 17996568]
Level 1 (high-level) evidencePanza JA, Velazquez EJ, She L, Smith PK, Nicolau JC, Favaloro RR, Gradinac S, Chrzanowski L, Prabhakaran D, Howlett JG, Jasinski M, Hill JA, Szwed H, Larbalestier R, Desvigne-Nickens P, Jones RH, Lee KL, Rouleau JL. Extent of coronary and myocardial disease and benefit from surgical revascularization in ischemic LV dysfunction [Corrected]. Journal of the American College of Cardiology. 2014 Aug 12:64(6):553-61. doi: 10.1016/j.jacc.2014.04.064. Epub [PubMed PMID: 25104523]
Level 1 (high-level) evidenceMark DB, Knight JD, Velazquez EJ, Wasilewski J, Howlett JG, Smith PK, Spertus JA, Rajda M, Yadav R, Hamman BL, Malinowski M, Naik A, Rankin G, Harding TM, Drew LA, Desvigne-Nickens P, Anstrom KJ. Quality-of-life outcomes with coronary artery bypass graft surgery in ischemic left ventricular dysfunction: a randomized trial. Annals of internal medicine. 2014 Sep 16:161(6):392-9. doi: 10.7326/M13-1380. Epub [PubMed PMID: 25222386]
Level 1 (high-level) evidence