Introduction
Myopia, colloquially referred to as shortsightedness or nearsightedness, is one of the most common global eye diseases. It occurs when the eye's optical power is too high for the corresponding axial length of the eye, resulting in images of distant objects being focused in front of the retina. Myopia can be corrected by wearing glasses, contact lenses, or refractive surgery. However, pathologic myopia (PM) refers to a subset of ocular conditions associated with high myopia. The exact diagnostic criteria of PM have not been established. Some have defined PM based on an absolute cutoff value of refractive power (-6 to -8 D) or axial length (26.5 mm), whereas others have based it on myopic maculopathy.[1][2]
Etiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Etiology
Myopia is attributable to a complex interaction between genetics and the environment. Familial clustering in myopia is evident in twin studies and high sibling risk ratios.[3] Several genetic variants have been associated with the development of myopia. For the syndromic myopias, which include Marfans and Sticklers syndrome, the implicated genes are involved in the extracellular matrix of the sclera. Gene studies have identified 25 different genetic loci in association with nonsyndromic myopia. The pathogenicity of genetic mutations is difficult to identify due to variable gene expression and interactions.[4]
There is some evidence to suggest that spending more time outdoors is protective against the development of myopia.[5] Likewise, a shift towards urbanization and higher education is thought to be a significant driver of the increasing prevalence of myopia among schoolchildren.[6] The predominating theory is that the increased time spent accommodating during near reading promotes signals for eye growth. Animal studies have also found the placement of both positive and negative lenses that defocus images on the retina can promote or inhibit eye growth.[7][8]
Epidemiology
The prevalence of myopia varies according to ethnicity and geography. The prevalence of myopia is highest in regions of East Asia (47%) and Southeast Asia (39%) as compared to Central Europe (27%) and Central Africa (7%).[9] There are also observed differences in the prevalence of myopia between urban and rural areas, which is attributed to the socioeconomic and educational systems in place.[8] The prevalence of pathologic myopia is reportedly from 0.9% to 3.1%.[10]
Pathophysiology
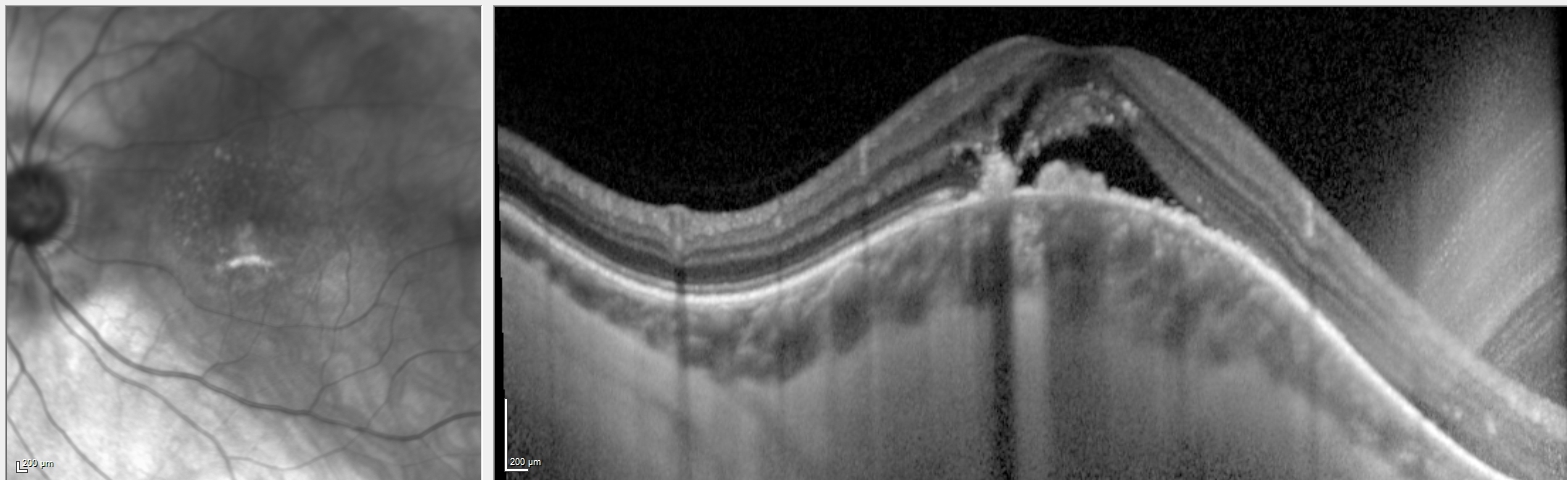
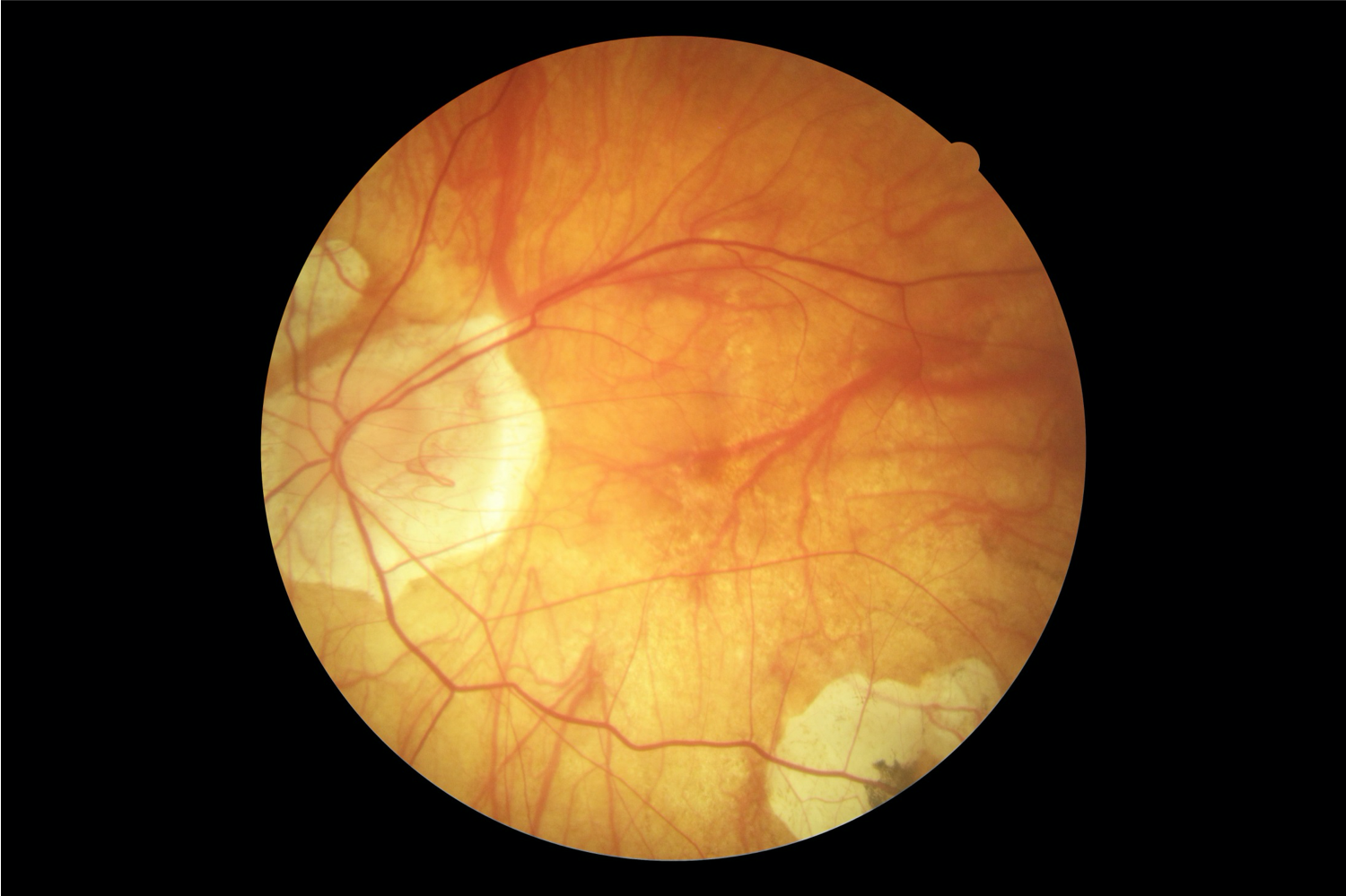
Myopia develops in the early to middle childhood and continues to early adulthood. Most children are born hyperopic. Then, closer to the age of 5 to 8, they experience a shift in their refraction through emmetropisation. After this process, the cornea stabilizes but increases in axial length, leading to progressive myopia.[11] In addition to longer axial lengths, individuals with pathological myopia typically manifest different ocular shapes with irregular protrusions, termed staphylomas. Excessive axial elongation stretches and thins the retina, choroid, and sclera, which results in various pathological findings (see Image. Grade 3 Pathological Myopia With Diffuse Chorioretinal Atrophy).
History and Physical
A complete history should focus on specific ocular and systemic associations of high myopia. A review of systems focused on joint, hearing, and heart problems should be conducted to screen for an underlying connective tissue disorder. Family history and previous ocular surgery should also be elicited, paying attention to previous retinal detachments, corrective lens wear, or refractive surgery. New complaints of metamorphopsia, scotoma, flashes, and onset of new floaters suggest retinal pathology.
Physical examination should begin with measuring visual acuity and intraocular pressure and assessing pupillary reaction. Next, a dilated fundal examination should be performed on the macula, optic disc, and peripheral retina. Finally, an assessment of visual fields can be requested if glaucoma is suspected.
In 2015, a specialist panel of researchers developed a universal classification system for pathological myopia.
| Category 0 | No macular lesions |
| Category 1 |
Tessellated fundus - Visible choroidal vessels |
| Category 2 |
Diffuse chorioretinal atrophy - Yellowish-white appearance to the posterior pole |
| Category 3 |
Patchy chorioretinal atrophy Discrete atrophic areas in the macula and peripapillary region |
| Category 4 |
Macular atrophy Atrophy in the foveal region |
|
Plus Lesions
|
Lacquer cracks Choroidal neovascularisation Fuchs spots |
Proposed Classification of Myopic Maculopathy. Submitted by Meta-analysis for Pathologic Myopia (META-PM) Study Group.[2]
Findings
- Staphyloma is an outpouching of the wall of the eye. Posterior staphyloma is unique to pathological myopia. Posterior staphylomas associated with pathological myopia have been classified into ten types based on shape and location.[12]
- Lacquer cracks are irregular yellow linear lesions in the macula. They represent mechanical linear breaks in Bruch’s membrane and are potential foci for choroidal neovascularisation. Newly formed lacquer cracks may be associated with bleeding.
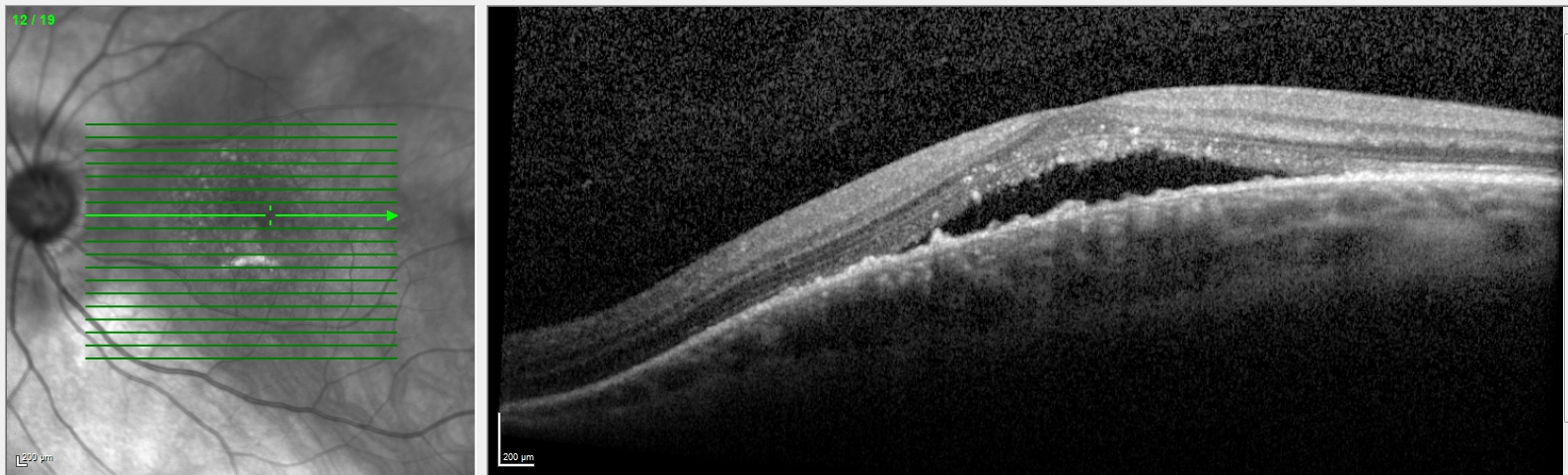
- Myopic choroidal neovascular membrane (CNV) is a common cause of visual impairment in patients with pathological myopia. The incidence of myopic CNV is estimated to be 10%, but it is even higher (35%) if the fellow eye was previously affected. Myopic CNV typically appears as a small, gray subretinal lesion beneath or near the fovea, and it may be associated with subretinal fluid or hemorrhage (see Image. Dome-Shaped Maculopathy With Subretinal Fluid). Fuchs spots are pigmented gray scars corresponding to regressed myopic CNV sites.
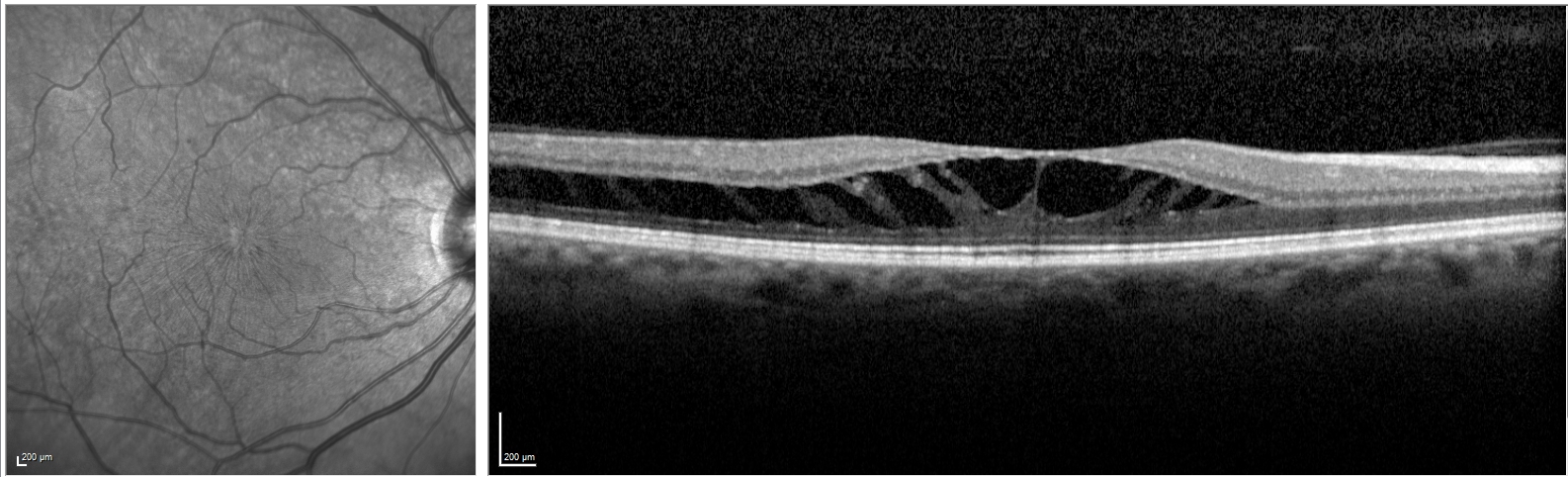
- Myopic tractional maculopathy results from vitreoretinal traction with manifestations that include retinoschisis, foveal retinal detachment, and lamellar or full-thickness macular hole.
- A dome-shaped macula is an inward bulge around the foveal region associated with scleral thickening. It is present in 20% of eyes with high myopia.[13]
- Structural anomalies and atrophy around the optic nerve fibers and optic disc pits can result in visual field loss.[14]
- Peripheral retinal holes or breaks
Evaluation
Spectral-domain optical coherence tomography (OCT) is noninvasive, quick, and widely available. It is suitable for diagnosing and monitoring retinal pigment epithelium atrophy, myopic CNV, and myopic tractional maculopathy. OCT provides a regular measurement of retinal and choroidal thickness and subretinal or intraretinal fluid. It also highlights the presence of macular holes, retinoschisis, or detachments. Dome-shaped maculopathy is best visualized on radial or vertical OCT slices (see Image. Dome-Shaped Maculopathy on Vertical Optical Coherence Tomography Scan).
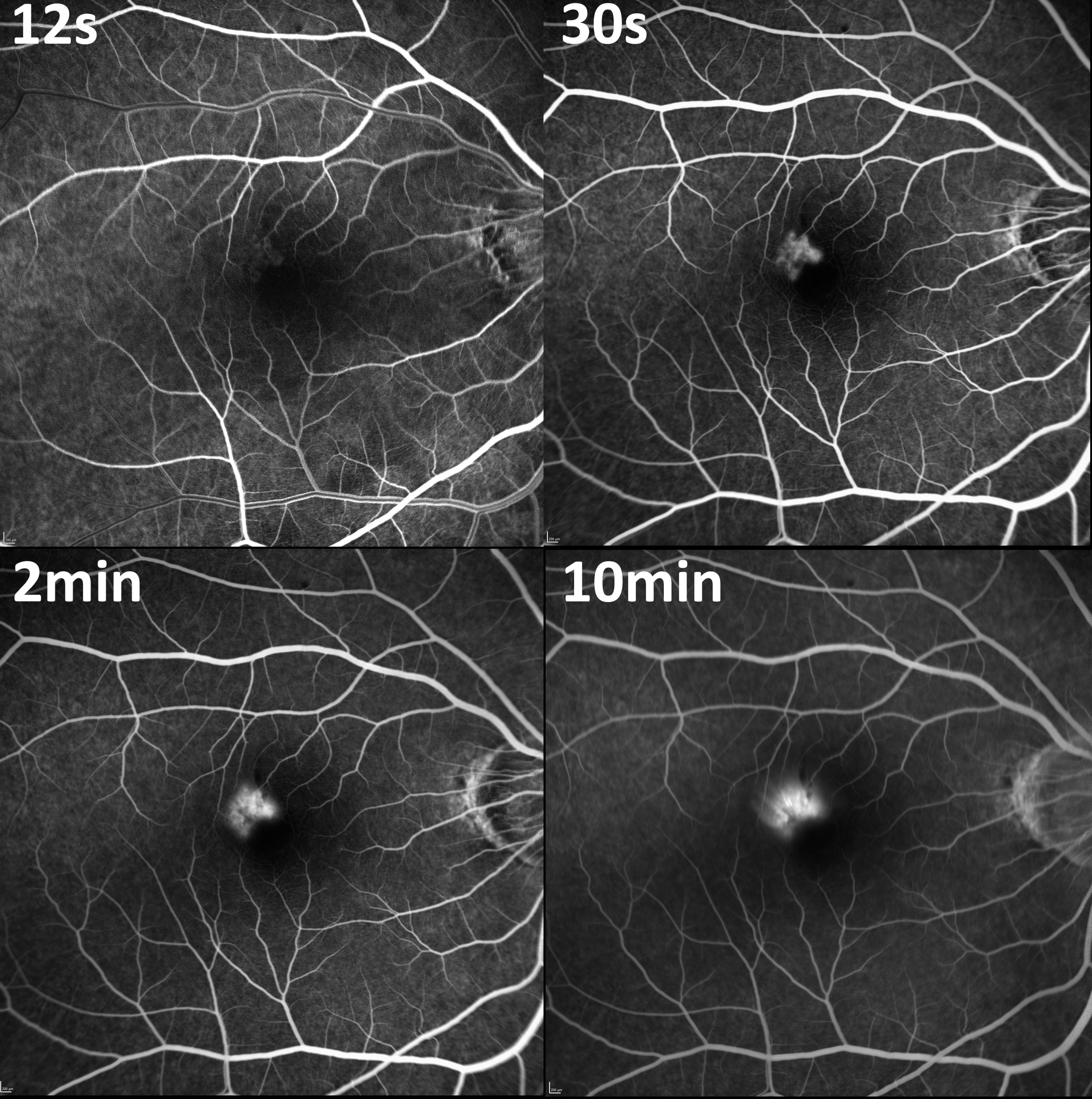
Fluorescein angiography provides detailed information about the type and activity of myopic CNV and helps plan treatment. Myopic CNV resembles the appearance of type 2 or classic CNV on fluorescein angiography, demonstrating early hyperfluorescence that increases in size and intensity through to the late phase. It can help differentiate between hemorrhage from a myopic CNV versus a lacquer crack, with the latter displaying blocked fluorescence instead of increasing hyperfluorescence (see Image. Fluorescein Angiogram Demonstrating Myopic Choroidal Neovascular Membrane).
Other tests, such as OCT angiography, ultrasonography, magnetic resonance imaging, and Indocyanine Green Angiography, may be useful for evaluating the shape and perfusion of the retina and choroid. However, these tests are rarely utilized in a clinical setting.[15][16][17]
Treatment / Management
Simple myopia can be satisfactorily corrected using refractive lenses (contact lenses, spectacles) or refractive surgery. Incentives to encourage outdoor play among children may help prevent myopia progression. Other methods include specialized contact lenses and low-dose topical atropine.[18] However, no proven therapy can reverse or halt the progressive retinal atrophy associated with pathological myopia.
The mainstay treatment of myopic CNV is anti-vascular endothelial growth factors (anti-VEGF) intravitreal injections. Large, randomized control trials have demonstrated superior visual and anatomical outcomes with anti-VEGF therapy compared to photodynamic therapy or control.[19][20] Fewer treatments with anti-VEGF therapy are needed for disease control compared with other subtypes of CNV (age-related macular degeneration).[19](A1)
The management of myopic tractional maculopathy remains debatable and depends on specific OCT findings.[21] Some practitioners favor a conservative approach as some cases remain stable or resolve following the release of retinal traction. However, those with pre-existing extensive macular retinoschisis may be more likely to progress and require intervention.[22] The surgical approaches consist of pars plana vitrectomy +/- internal limiting membrane peel versus a macular buckle procedure (see Image. Myopic Macular Schisis).(B2)
Differential Diagnosis
The differential diagnoses for degenerative myopia include the following:
- Age-related macular degeneration
- Choroidal rupture or angioid streaks may be mistaken for lacquer cracks
- Retinal pigment epithelial changes from previous central serous chorioretinopathy
- Ocular histoplasmosis
- Pattern dystrophies (eg, reticular dystrophy, Doyne honeycomb retinal dystrophy)
Other associated systemic disorders include:
Pertinent Studies and Ongoing Trials
The following trials demonstrated that anti-VEGF therapy was effective for the treatment of myopic CNV, with good visual and anatomic benefits achieved with a limited number of injections:
RADIANCE
RADIANCE is a multicenter, randomized controlled study of ranibizumab in patients with secondary to pathologic myopia (n=277). It compared intravitreal ranibizumab 0.5 mg to verteporfin photodynamic therapy to treat myopic CNV. The ranibizumab treatment group experienced a superior visual gain (14 letters vs 9 letters) at 12 months. Patients received a median of 2 treatments during the 12-month study period as guided by disease activity.[19]
MYRROR
MYRROR is a multicenter, randomized control trial of intravitreal aflibercept injection in patients with myopic CNV (n=122). It compared intravitreal aflibercept 0.5 mg versus placebo injection to treat myopic CNV. Additional injections were administered if there were signs of CNV activity during monthly visits. At week 24, the treatment group experienced superior visual gains (+12 letters vs -2 letters). Patients in the treatment group received a median of 2 injections in weeks 0–8 and 0 injections in weeks 8 to 32.[20]
Treatment Planning
The treatment regimen for a myopic choroidal neovascular membrane typically consists of intravitreal anti-VEGF injections. Often, 1 or 2 treatments spaced approximately four weeks apart are given, followed by monitoring and further treatment as needed if signs of choroidal neovascular activity such as subretinal fluid, hemorrhage, or leakage. Typical anti-VEGF agents and dosing is as follows:
- Bevacuzimab (1.25 mg/0.05 ml)
- Aflibercept (2 mg/0.05 ml)
- Ranibizumab (0.5 mg/0.05 ml)
Prognosis
Pathologic myopia is a significant cause of vision loss worldwide. The prevalence of visual impairment secondary to pathological myopia reaches as high as 0.5% in European studies and 1.4% in Asian studies.[10] Long-term follow-up studies of patients diagnosed with myopic CNV report a mean decline in visual acuity, even despite adequate disease control on anti-VEGF therapy.[25]
Complications
Patients with pathological myopia may experience a marked drop in visual acuity secondary to a subretinal hemorrhage or myopic choroidal neovascular membrane. Despite prompt therapy, they can develop scarring and atrophy in the macula that results in a permanent central scotoma. They also face an increased risk of early cataract formation, glaucoma, and retinal detachments, which can also lead to visual impairment.[26]
Deterrence and Patient Education
Patients should be advised to attend their regular eye appointments to ensure their disease remains optimally controlled by the available treatments. Any sudden changes to vision, including metamorphopsia, scotoma, flashes, floaters, or blurry vision, should prompt an urgent assessment by an eye care professional. Patients should be advised to check each eye with an Amsler grid regularly.
Enhancing Healthcare Team Outcomes
Despite the lack of formal guidelines, a collaborative care approach towards myopia prevention, detection, and management is required. Given the rising prevalence of myopia, which develops from childhood, public health needs to advocate for policies that encourage outdoor activity. Screening for myopia in schools or other forums requires a team of individuals, such as trained technicians or nurses, and a system in place for referral to an eye care provider for evaluation. Ophthalmologists and optometrists manage treatment decisions such as the initiation of pharmacological or optical therapy to halt myopic progression.
Additionally, optometrists and ophthalmologists play an important role in screening for pathological myopia and its associated complications. Collaboration between glaucoma, vitreoretinal, and medical retinal specialists ensures patients receive specialized care when necessary. Occupational therapists and optometrists specializing in low vision play an important role in visual rehabilitation, helping individuals maintain an independent living and maximize their vision when it is limited due to their disease.
Media
(Click Image to Enlarge)
(Click Image to Enlarge)
(Click Image to Enlarge)
References
Fredrick DR. Myopia. BMJ (Clinical research ed.). 2002 May 18:324(7347):1195-9 [PubMed PMID: 12016188]
Level 3 (low-level) evidenceOhno-Matsui K, Kawasaki R, Jonas JB, Cheung CM, Saw SM, Verhoeven VJ, Klaver CC, Moriyama M, Shinohara K, Kawasaki Y, Yamazaki M, Meuer S, Ishibashi T, Yasuda M, Yamashita H, Sugano A, Wang JJ, Mitchell P, Wong TY, META-analysis for Pathologic Myopia (META-PM) Study Group. International photographic classification and grading system for myopic maculopathy. American journal of ophthalmology. 2015 May:159(5):877-83.e7. doi: 10.1016/j.ajo.2015.01.022. Epub 2015 Jan 26 [PubMed PMID: 25634530]
Level 2 (mid-level) evidenceGuggenheim JA, Kirov G, Hodson SA. The heritability of high myopia: a reanalysis of Goldschmidt's data. Journal of medical genetics. 2000 Mar:37(3):227-31 [PubMed PMID: 10777361]
Level 3 (low-level) evidenceCai XB, Shen SR, Chen DF, Zhang Q, Jin ZB. An overview of myopia genetics. Experimental eye research. 2019 Nov:188():107778. doi: 10.1016/j.exer.2019.107778. Epub 2019 Aug 28 [PubMed PMID: 31472110]
Level 3 (low-level) evidenceRose KA, Morgan IG, Ip J, Kifley A, Huynh S, Smith W, Mitchell P. Outdoor activity reduces the prevalence of myopia in children. Ophthalmology. 2008 Aug:115(8):1279-85. doi: 10.1016/j.ophtha.2007.12.019. Epub 2008 Feb 21 [PubMed PMID: 18294691]
Level 2 (mid-level) evidenceMorgan I, Rose K. How genetic is school myopia? Progress in retinal and eye research. 2005 Jan:24(1):1-38 [PubMed PMID: 15555525]
Wallman J, Winawer J. Homeostasis of eye growth and the question of myopia. Neuron. 2004 Aug 19:43(4):447-68 [PubMed PMID: 15312645]
Level 3 (low-level) evidenceMorgan IG, Ohno-Matsui K, Saw SM. Myopia. Lancet (London, England). 2012 May 5:379(9827):1739-48. doi: 10.1016/S0140-6736(12)60272-4. Epub [PubMed PMID: 22559900]
Holden BA, Fricke TR, Wilson DA, Jong M, Naidoo KS, Sankaridurg P, Wong TY, Naduvilath TJ, Resnikoff S. Global Prevalence of Myopia and High Myopia and Temporal Trends from 2000 through 2050. Ophthalmology. 2016 May:123(5):1036-42. doi: 10.1016/j.ophtha.2016.01.006. Epub 2016 Feb 11 [PubMed PMID: 26875007]
Wong TY, Ferreira A, Hughes R, Carter G, Mitchell P. Epidemiology and disease burden of pathologic myopia and myopic choroidal neovascularization: an evidence-based systematic review. American journal of ophthalmology. 2014 Jan:157(1):9-25.e12. doi: 10.1016/j.ajo.2013.08.010. Epub 2013 Oct 5 [PubMed PMID: 24099276]
Level 1 (high-level) evidenceGordon RA, Donzis PB. Refractive development of the human eye. Archives of ophthalmology (Chicago, Ill. : 1960). 1985 Jun:103(6):785-9 [PubMed PMID: 4004614]
Curtin BJ. The posterior staphyloma of pathologic myopia. Transactions of the American Ophthalmological Society. 1977:75():67-86 [PubMed PMID: 613534]
Liang IC, Shimada N, Tanaka Y, Nagaoka N, Moriyama M, Yoshida T, Ohno-Matsui K. Comparison of Clinical Features in Highly Myopic Eyes with and without a Dome-Shaped Macula. Ophthalmology. 2015 Aug:122(8):1591-600. doi: 10.1016/j.ophtha.2015.04.012. Epub 2015 May 16 [PubMed PMID: 25986898]
Ohno-Matsui K, Shimada N, Yasuzumi K, Hayashi K, Yoshida T, Kojima A, Moriyama M, Tokoro T. Long-term development of significant visual field defects in highly myopic eyes. American journal of ophthalmology. 2011 Aug:152(2):256-265.e1. doi: 10.1016/j.ajo.2011.01.052. Epub 2011 Jun 12 [PubMed PMID: 21664594]
Level 3 (low-level) evidenceVaghefi E, Kauv K, Pan W, Squirrell D. Application of Arterial Spin Labelling in Detecting Retinal Ischemia. Case reports in ophthalmology. 2017 Sep-Dec:8(3):545-557. doi: 10.1159/000485316. Epub 2017 Dec 14 [PubMed PMID: 29422857]
Level 3 (low-level) evidenceAl-Sheikh M, Phasukkijwatana N, Dolz-Marco R, Rahimi M, Iafe NA, Freund KB, Sadda SR, Sarraf D. Quantitative OCT Angiography of the Retinal Microvasculature and the Choriocapillaris in Myopic Eyes. Investigative ophthalmology & visual science. 2017 Apr 1:58(4):2063-2069. doi: 10.1167/iovs.16-21289. Epub [PubMed PMID: 28388703]
Moriyama M, Ohno-Matsui K, Hayashi K, Shimada N, Yoshida T, Tokoro T, Morita I. Topographic analyses of shape of eyes with pathologic myopia by high-resolution three-dimensional magnetic resonance imaging. Ophthalmology. 2011 Aug:118(8):1626-37. doi: 10.1016/j.ophtha.2011.01.018. Epub 2011 Apr 29 [PubMed PMID: 21529960]
Baird PN, Saw SM, Lanca C, Guggenheim JA, Smith Iii EL, Zhou X, Matsui KO, Wu PC, Sankaridurg P, Chia A, Rosman M, Lamoureux EL, Man R, He M. Myopia. Nature reviews. Disease primers. 2020 Dec 17:6(1):99. doi: 10.1038/s41572-020-00231-4. Epub 2020 Dec 17 [PubMed PMID: 33328468]
Wolf S, Balciuniene VJ, Laganovska G, Menchini U, Ohno-Matsui K, Sharma T, Wong TY, Silva R, Pilz S, Gekkieva M, RADIANCE Study Group. RADIANCE: a randomized controlled study of ranibizumab in patients with choroidal neovascularization secondary to pathologic myopia. Ophthalmology. 2014 Mar:121(3):682-92.e2. doi: 10.1016/j.ophtha.2013.10.023. Epub 2013 Dec 8 [PubMed PMID: 24326106]
Level 1 (high-level) evidenceIkuno Y, Ohno-Matsui K, Wong TY, Korobelnik JF, Vitti R, Li T, Stemper B, Asmus F, Zeitz O, Ishibashi T, MYRROR Investigators. Intravitreal Aflibercept Injection in Patients with Myopic Choroidal Neovascularization: The MYRROR Study. Ophthalmology. 2015 Jun:122(6):1220-7. doi: 10.1016/j.ophtha.2015.01.025. Epub 2015 Mar 4 [PubMed PMID: 25745875]
Level 1 (high-level) evidenceFrisina R, Gius I, Palmieri M, Finzi A, Tozzi L, Parolini B. Myopic Traction Maculopathy: Diagnostic and Management Strategies. Clinical ophthalmology (Auckland, N.Z.). 2020:14():3699-3708. doi: 10.2147/OPTH.S237483. Epub 2020 Nov 2 [PubMed PMID: 33173268]
Shimada N, Tanaka Y, Tokoro T, Ohno-Matsui K. Natural course of myopic traction maculopathy and factors associated with progression or resolution. American journal of ophthalmology. 2013 Nov:156(5):948-957.e1. doi: 10.1016/j.ajo.2013.06.031. Epub 2013 Aug 20 [PubMed PMID: 23972301]
Level 2 (mid-level) evidenceMrugacz M, Rydzanicz M, Frajdenberg A, Podfigurna-Musielak M, Gajecka M. [Myopia in systemic disorders]. Klinika oczna. 2009:111(1-3):84-8 [PubMed PMID: 19517854]
Marr JE, Halliwell-Ewen J, Fisher B, Soler L, Ainsworth JR. Associations of high myopia in childhood. Eye (London, England). 2001 Feb:15(Pt 1):70-4 [PubMed PMID: 11318301]
Level 2 (mid-level) evidenceCheung CMG, Arnold JJ, Holz FG, Park KH, Lai TYY, Larsen M, Mitchell P, Ohno-Matsui K, Chen SJ, Wolf S, Wong TY. Myopic Choroidal Neovascularization: Review, Guidance, and Consensus Statement on Management. Ophthalmology. 2017 Nov:124(11):1690-1711. doi: 10.1016/j.ophtha.2017.04.028. Epub 2017 Jun 24 [PubMed PMID: 28655539]
Level 3 (low-level) evidenceMitchell P, Hourihan F, Sandbach J, Wang JJ. The relationship between glaucoma and myopia: the Blue Mountains Eye Study. Ophthalmology. 1999 Oct:106(10):2010-5 [PubMed PMID: 10519600]
Level 2 (mid-level) evidence