 Fertility Sparing Management in Uterine Fibroids
Fertility Sparing Management in Uterine Fibroids
Introduction
Uterine leiomyoma (fibroids) are the most common benign gynecologic tumors, occurring in up to 70% of women by menopause.[1] While many fibroids are asymptomatic and are only discovered incidentally, 25 to 30 % of women experience a spectrum of symptoms that increase morbidity and adversely affect their quality of life.[2]
The most common symptoms include abnormal uterine bleeding (AUB), heavy menstrual bleeding (HMB), pelvic pain and pressure, anemia, and bladder and/or bowel dysfunction. Importantly, the presence of one or multiple fibroids may affect fertility, as the distortion of the uterus can prevent successful implantation and/or continued survival of an intrauterine pregnancy.
The presence of fibroids can introduce obstetrical complications, such as recurrent pregnancy loss (RPL), preterm labor (PTL), abnormal placentation, increased rates of cesarean section, and postpartum hemorrhage. With the current trend of increased childbearing age, fertility-sparing management of uterine fibroids is critical. In this review, we discuss the clinical presentation of fibroids in women of reproductive age and the medical and surgical management options available for women with fibroids who wish to become pregnant in the future.
Etiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Etiology
Fibroids develop as inappropriate yet benign growths of monoclonal uterine cells, specifically smooth muscle cells and fibroblasts.[3] Though pathogenesis typically occurs in the myometrium, fibroids can extend superficially from the uterine serosa from the pelvis into the lower abdomen and, on occasion, into the upper abdomen as well. Alternatively. they can arise from the myometrium and delve into the uterine cavity.
Epidemiology
Uterine fibroids may present at any age and in any community. However, prevalence varies significantly amongst different age groups and ethnicities. The prevalence of fibroids in women of reproductive age is estimated to be 20 to 40%.[4] In women presenting with infertility, 5 to 10% are determined to have uterine fibroids, and also they are found to be the sole factor affecting fertility in 2 to 4% of these women.[5][6]
The incidence of fibroids increases with increasing age until menopause. The cumulative incidence of fibroids has been documented to be as high as 80% in black women and approximately 70% in white women. The incidence is higher among women in their fifth and sixth decade of life. Premenopausal women demonstrated a higher fibroid risk than post-menopausal women, likely due to the role of hormones in stimulating fibroid growth.
Studies have consistently shown a 3-fold increased risk of fibroids among black women compared to white women when controlled for other accepted risk factors. In addition, black women tend to develop uterine fibroids at a younger age, present with larger uteri at the time of diagnosis. They are more likely to suffer from blood-loss anemia compared to white women. This may be related to the fact that black women generally tend to have had earlier menarche compared to white women.[7] Racial disparities observed are likely due largely to social determinants of health, including but not limited to access to care, job security, and adequate health insurance seen in the black community.
Other risk factors for fibroids include early menarche, increased interval since last birth, obesity, diet, family history, and hypertension.[8] Notably, one study revealed that women with hypertension have a fivefold increased risk of developing fibroids compared to those without hypertension.[9]
The risk of fibroids decreases with an increasing number of term pregnancies, such that women having > 5 pregnancies have a quarter of the risk of women who had none.[10] The use of hormonal contraceptives and/or depot medroxyprogesterone acetate (DMPA) for any length of time has also been shown to be protective against fibroids.
Pathophysiology
The exact initiating factor of fibroid formation remains unclear, though prevailing theories highlight reproductive hormones, inherent abnormalities of the myometrium, and predisposing genetic variations as contributing factors.[11] Epidemiological studies also underscore the role of estrogen and progesterone since an increased incidence of fibroids has been found in premenopausal women, have undergone early menarche or DES exposure, or have an elevated BMI. Perhaps the most striking evidence supporting this relationship is the tendency of fibroids to grow during early pregnancy and regress during the postpartum period or with the use of hormone-suppressing GnRH analogs. Molecular studies help clarify this link. Myometrial cells with high levels of estrogen and progesterone receptors have been shown to stimulate the growth of adjacent fibroid stem cells in a paracrine fashion.[12]
Other research models using leiomyoma xenografts were used to explore this molecular relationship have identified both estrogen and progesterone as necessary and interdependent for the growth of abnormal myocytes.[13] In addition, somatic mutations (i.e., MED12, HMGA1, and HMGA2) and inherited mutations (fumarate hydratase gene FH, somatic chromosomal rearrangements) have been identified as discernible genetic components that contribute to this fibroid pathogenesis.[14]
Histopathology
Fibroids are composed of monoclonal smooth muscle and fibroblast cells that have transformed at the microscopic level to abnormal cells.[15] These cells feature upregulated expression of progesterone receptors (predominantly the PR-a isoform) and estrogen receptors (mainly ER-a).[13][16]
With the endocrinological influence of sex steroid hormones, these cells may grow into clinically detectable fibroids. Histological samples of fibroids demonstrate a significant extracellular matrix (ECM) composed of collagen, proteoglycan, and fibronectin adjacent to the disordered myometrium cells.[17][18] Fibroids are characteristically encapsulated by a thin layer of areolar tissue and compressed muscle fibers.
History and Physical
The diversity in size, location, and volume of fibroids is reflected in the diversity of their clinical presentation, which ranges from asymptomatic to intensely debilitating. While most patients with fibroids are asymptomatic, approximately 25-30% of patients present with severe symptoms.[3] Moreover, the size and nature of fibroids are often dynamic, as they may involute, degenerate, become fibrotic, calcify, or even hemorrhage over time, and thus symptoms may evolve over time.
When fibroids present symptomatically, the most common complaints include AUB, pelvic pain/pressure, increased urinary frequency, constipation, and infertility.[19] Fibroids that arise from the submucosa are more likely to disrupt the contraction of endometrial vasculature and, as a result, may cause extensive bleeding, even when small.[20] Alternatively, large bulky fibroids that extend from the myometrium and/or serosal surface into the peritoneum are more likely to cause symptoms secondary to mass effect.[21] For example, large anterior fibroids tend to manifest with pelvic pressure and bladder symptoms, while posterior fibroids can lead to constipation.
Fibroids’ adverse effects on fertility are largely dependent on their location and ability to distort normal uterine and adnexal anatomy, thereby preventing gamete transport, embryo implantation, and/or the maintenance of early pregnancy.[22] There is also significant evidence suggesting that fibroids alter endometrial receptivity, impair blood flow, and lead to chronic inflammation that creates a hostile environment for successful intrauterine pregnancy.[23][22]
On a bimanual exam, the examiner may palpate an enlarged, bulky, irregularly shaped uterus. If the fibroids are predominantly posterior, one may appreciate fullness in the posterior cul-de-sac with an anteriorly displaced cervix. An irregular contour or increased uterine size may not be appreciated in patients with small intramural or submucosal fibroids. Whether or not physical findings are significant, a patient with the symptoms described above is sufficient to warrant further evaluation.
Evaluation
The relationship of the leiomyoma with the uterine cavity is important for understanding symptoms and treatment options. Myomas are classified using the International Federation of Gynecology and Obstetrics (FIGO) classification system, which predominantly categorizes based on location (Figure 1).[24][25] There are three main categories: submucosal (FIGO 0, 1, 2), intramural (FIGO 3, 4, 5), subserosal (FIGO 6, 7), and other (e.g., cervical, parasitic) (FIGO 8).
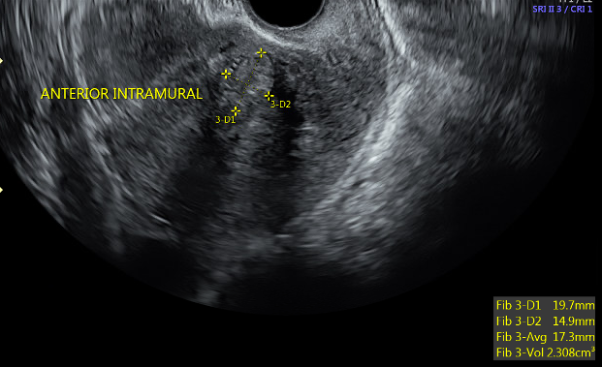
Transvaginal ultrasonography (TVUS) imaging remains the best tool for initial evaluation when fibroids are suspected.[26] With uterine size less than ten weeks gestation, transvaginal ultrasound has 95 to 100 percent sensitivity in detecting fibroids.[27] On TVUS, fibroids are typically described as well-circumscribed, concentric, hypoechoic masses with acoustic shadowing (figure 2).[28]
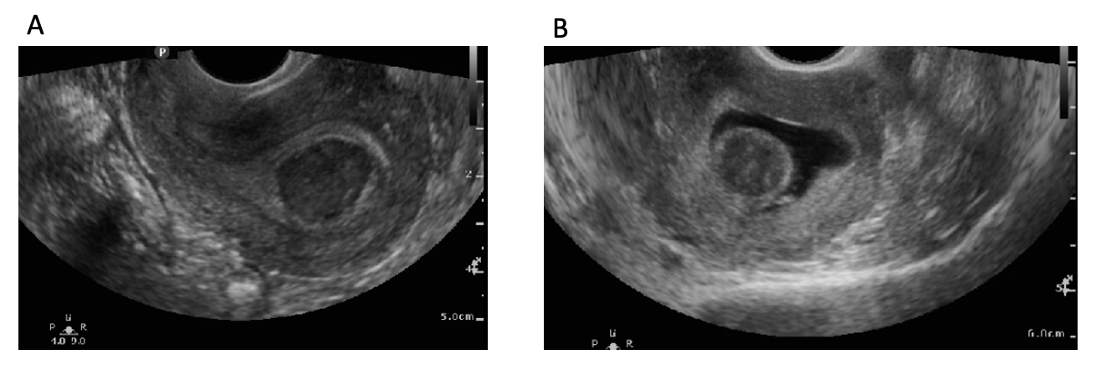
Saline infusion sonohysterography (SIS) is also useful in evaluating the distinction between Type 1-3 fibroids as it provides a clearer picture of extension into the endometrial cavity (figure 3).[26] Suppose the distinction between Type 2 and Type 3 fibroids cannot be made on SIS. In that case, diagnostic hysteroscopy can be a useful tool to do so as it can characterize the extent of protrusion under direct visualization. However, hysteroscopy is less accurate in determining the size of the fibroid compared to ultrasound and SIS.[29]
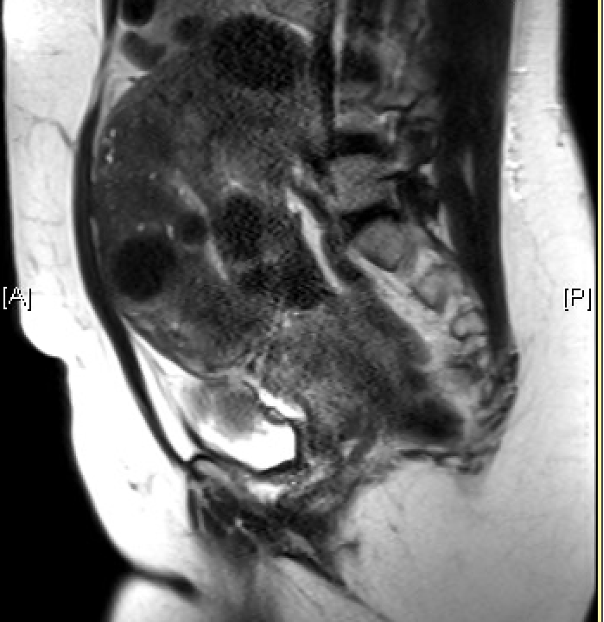
Magnetic resonance imaging (MRI) is the most effective imaging to detect the size and location of fibroids; however, given the cost of this modality, it is not routinely used and is instead useful for surgical planning since it can help identify vasculature, degenerating fibroids, location, and depth into the myometrium (figure 4).[2][30]
Treatment / Management
Medical Management
NSAIDs
NSAIDs are commonly used as first-line agents for the use of fibroid-related dysmenorrhea given their affordability, limited adverse effects, and availability.[31] Typically, ibuprofen (600 to 1800 mg daily) or naproxen (550 to 1100 mg daily) is given and advised to be taken 1 to 2 days before the onset of menstruation and through the duration of menstruation.[32] A Cochrane review showed that NSAIDs reduce heavy menstrual bleeding (HMB) compared to placebo. However, they are less effective compared to tranexamic acid, danazol, or LNG-IUD.[32](A1)
Combined Oral Contraceptives
Combined oral contraceptives work by preventing endometrial proliferation and have shown some benefits in improving fibroid-related heavy menstrual bleeding. However, there is no evidence to support their use for the relief of bulk symptoms.[33][31] In a randomized trial comparing the use of the levonorgestrel intrauterine device (LNG-IUD) and a low-dose combined oral contraceptive (COCs), COCs showed a decrease in menstrual blood loss. However, the LNG-IUD group had significantly less blood loss.[34] The use of progestin-only oral contraceptives is a reasonable option for patients with fibroid-associated heavy menstrual bleeding and contraindications to estrogen use. However, limited direct evidence supporting their benefit exists.(A1)
Levonorgestrel Intrauterine Device
Levonorgestrel IUDs (LNG-IUD) are currently FDA approved for the treatment of HMB, including fibroid-related uterine bleeding.[31] The LNG-IUD works via a local progesterone effect on the uterine cavity that induces endometrial decidualization, glandular atrophy, and desensitizing endometrial estrogen receptors rendering them less responsive.[35][36] The device has been clinically FDA-approved for five years, but recent studies demonstrate effectiveness for up to 7 years.[37] Around 20% of patients experience amenorrhea in the first year after insertion, which is the most common reason for removal.[38] Multiple studies have demonstrated that despite reducing fibroid-related menorrhagia, the LNG-IUD had no significant reduction in fibroid volume among premenopausal women.[39][34] Expulsion rates of IUDs seem to depend on fibroid size, with one study demonstrating a 15.4% expulsion rate in women with fibroids >3 cm compared to a rate of 6.3% in those with fibroids <3 cm, regardless of fibroid type.(A1)
Gonadotropin-Releasing Hormone (GnRH) Agonists
Gonadotropin-Releasing Hormone (GnRH) agonists are peptide analogs of the GnRH molecule that act as agonists at the GnRH receptor in the pituitary gland. Their initial activation at the GnRH receptor causes a flare effect, temporarily increasing serum gonadotropins and downstream sex hormone concentrations. Prolonged activation of the GnRH receptor, typically following 2-4 weeks of continuous therapy, leads to involution or downregulation of the receptor, thereby suppressing pituitary function leading to hypoestrogenism.[40] GnRH agonists have a diverse array of clinical applications. Regarding symptomatic fibroid management, they are used for short-term treatment of AUB, typically inducing a state of amenorrhea, and help improve symptoms of uterine enlargement. Given their side effect profile with long-term use, GnRH agonists are meant to be utilized as a bridge therapy prior to surgical or interventional procedures or natural menopause.[2] Leuprolide acetate, the most studied and most frequently administered GnRH agonist for fibroid therapy, is given as an intramuscular depot injection, either as a monthly injection of 3.75 mg for up to three months or an 11.25 mg 3-month formulation as a single dose.
GnRH agonists are often used to decrease uterine volume prior to surgical therapy, which may ultimately allow for a more minimally invasive surgical approach. In a recent Cochrane review, GnRH agonists were shown to reduce uterine volume by 175ml and fibroid volume by 155 ml compared to placebo controls. The same review demonstrated GnRH agonist benefit in increasing preoperative hemoglobin by an average of 0.88 g/dL, decreasing intraoperative blood loss, and decreasing postoperative complications.[41](A1)
With prolonged therapy, there is a risk of chemically induced menopause leading to vasomotor symptoms, bone loss, vaginal atrophy/dryness, and unfavorable changes to lipid profile.[2] If therapy is to exceed six months, it is important to start hormone add-back therapy with estrogen and progesterone, which can prolong acceptable treatment to an additional six months (for a total of 12 months of therapy).[42]
Gonadotropin-Releasing Hormone (GnRH) Antagonists
Elagolix and relugolix are oral, non-peptide, GnRH antagonists that function by reversible, dose-dependent suppression of GnRH, thereby suppressing gonadotropins and ovarian sex hormones and leading to a hypoestrogenic state.[43] These medications have both been recently FDA-approved (elagolix in 2020 and relugolix in 2021) for the use of HMB related to fibroids. An advantage of these medications is they are formulated with hormone-add back therapy. Elagolix comes co-packaged with two capsules; one containing elagolix 300 mg plus estradiol 1 mg plus norethindrone 0.5 mg taken in the morning and the other capsule containing elagolix 300mg alone taken at night.[44] Relugolix is also a combined formulation consisting of relugolix 40 mg plus estradiol 1mg plus norethindrone acetate 0.5 mg; however, it is only a single daily dose.[45] Both treatments are FDA approved for 24 months of use.
Regarding efficacy, elagolix has been shown to be efficacious in reducing fibroid-related bleeding. In two recent identical double-blinded, randomized, placebo-controlled, 6-month phase III trials (trials UF1 and UF2), 68.5% of 206 women in UF-1 and 76.5% of 189 women in UF-2 receiving elagolix + add back therapy achieved the primary endpoint of reduced menstrual blood loss to less than 80 mL during the final month of treatment and at least a 50% reduction in menstrual blood loss compared to baseline. In those who received elagolix alone (without add-back therapy), the same primary endpoint was met in 84.1% of 104 women in UF-1 and 77% of 95 women in UF-2.[43]
In a subsequent extension phase III trial, when therapy was given for an additional six months (total of 12 months of treatment), more than half of the participants demonstrated amenorrhea showing a significant improvement in quality of life.[44] The most common side effects of elagolix were hot flushes (7%) and headaches (5.5%). With prolonged therapy, the bone mineral density of the lumbar spine was reduced by 1.5%; however, the reduction was improved to only 0.6% when measured one year following completion of treatment.
Following the elagolix trials, the protocol was replicated for two international, randomized, double-blind placebo-controlled, 24-week, phase 3 trials for the study of relugolix. The relugolix trials demonstrated a reduction of menstrual blood loss in 71 to 80 % of participants compared to placebo and resulted in a significant decrease in pain, anemia, bulk-related symptoms, and uterine volume.[45] Moreover, relugolix was shown to preserve bone mineral density. The most common adverse effects of relugolix combination therapy were hot flashes and hypertension.
Selective Progesterone Receptor Modulators (SPRMs)
SPRMs are a class of synthetic steroids that bind to progesterone receptors and have tissue-specific antagonist effects of varying degrees.[46] Specifically, Ulipristal Acetate (UPA) binds to the pituitary gland, endometrium, and uterine fibroids to elicit actions that have been beneficial in managing uterine fibroids. At the level of the pituitary, UPA inhibits ovulation and induces amenorrhea. At the level of the endometrium, UPA prevents endometrial proliferation and decreases uterine bleeding. UPA also binds progesterone receptors that are abundant within the fibroids themselves to decrease cellular proliferation and increase apoptosis, ultimately reducing the size of the fibroids. Of note, SPRMs lead to histologic changes in the endometrium, referred to as progesterone receptor modulator-associated endometrial changes (PAECs), which are reversible and are not associated with malignancy.[46][47] (A1)
Clinical efficacy in the reduction of AUB-L and fibroid volume has been demonstrated in the European-based PEARL trials.[48][49] UPA is currently not approved in the United States for the treatment of fibroids. UPA use outside the United States is limited due to the rare side effect of severe liver injury.(A1)
Aromatase Inhibitors
Letrozole is a reversible non-steroidal competitive aromatase inhibitor that works by inhibiting the conversion of androgens into estrogens. Studies have demonstrated that uterine fibroid tissue over-expresses aromatase. Thus, aromatase inhibitors reduce the local production of estrogen from the fibroid itself, thereby preventing further fibroid proliferation and assisting in fibroid volume reduction.[50][51] Letrozole can be administered orally 2.5 to 5 mg/day for 12 weeks. One trial of 70 participants demonstrated a reduction in fibroid volume by 46% after 12 weeks of treatment; however, there was no significant difference compared to GnRH agonists.[52] Another study demonstrated a decreased operative time and intraoperative blood loss when letrozole was used preoperatively for 12 weeks.[53] Letrozole is not currently FDA approved for symptomatic fibroids; however, it has been used off-label for this purpose. Most reported adverse effects are hot flashes and bone mineral density loss with prolonged use.[52](A1)
Surgical Management
Myomectomy is the surgical removal of uterine fibroids via hysteroscopic, laparoscopic, or abdominal surgery. Common indications for myomectomy include AUB with or without a history of symptomatic anemia, bulk-related symptoms such as urinary urgency, frequency, hydronephrosis, constipation, infertility, prevention of obstetrical complications, and in some cases, for the evaluation of potential malignancy. Establishing the type (location), size, and quantity of fibroids using the modalities described above is a crucial step in a patient’s evaluation that guides the optimal excision strategy.
Hysteroscopic Myomectomy
Hysteroscopic myomectomy (HSC-M) is a minimally invasive approach to fibroid removal with high success rates and low risk for complications in the appropriately selected patient (figure 5). The feasibility of performing a successful HSC-M is largely dependent on the type and size of the existing fibroids. HSC-M has shown to be most successful in resecting Type 0 and Type I fibroids.[54] Complete resection rates of Type 0 and Type I fibroids range from 96 to 97% and 86 to 90%, respectively.[54]
HSC-M can be used for type 2 fibroids; however, this often involves repeat procedures due to the lower complete resection rate of 61 to 83%. The type of fibroid also correlates to operative time and the amount of distention fluid absorbed during the procedure. The deeper the penetration of the fibroid into the myometrium, the greater the volume of fluid absorbed (type 0: 450 ml, type I: 957 ml, type II: 1682ml).[55] Patients with fibroids >3 cm and/or with two or more fibroids present have the highest risk of needing a repeat procedure.[56] The overall rate of incomplete resection is approximately 5 to 17%.[57]
HSC-M has a 94.4% success rate, defined as improved quality of life (QoL) scores and resolution of symptoms.[58][59] One of the biggest advantages of HSC-M is the benefits that come with a minimally invasive approach, such as minimal recovery time, minimal need for opioids, minimal postoperative restrictions, sooner return to daily activity, and decreased perioperative morbidity.[60] Patients typically report a major improvement in symptom severity and quality of life within 6-12 weeks of surgery.[60] (B2)
Regarding fertility, a 2009 study of infertile women with submucosal fibroids by Pritts et al. found that women who underwent submucosal myomectomy were twice as likely to become pregnant than women who did not undergo myomectomy (RR 2.03, CI 1.08–3.82).[61] Another study investigating conception rates up to 3 years after hysteroscopic myomectomy found a 49% conception rate after resection of type 0 fibroids and 33% after resection of type I fibroids.[62](A1)
HSC-M is overall a very safe procedure with a complication rate of <1%.[63] The most common complication with hysteroscopy is uterine perforation at a rate of 0.76%, which most frequently occurs during uterine entry. This is also the most common reason for excessive bleeding during the procedure. Other complications of HSC-M are associated with excess fluid absorption of the distention medium, which can occur during complicated resections with large or multiple fibroids that prolong the procedure.[64](B2)
Laparoscopic Myomectomy
Laparoscopic myomectomy (LSC-M) is a minimally invasive surgical approach to remove symptomatic fibroids of type 2 through 8. Location, size, and the number of fibroids are all important determinants for the success of the surgery and reducing recurrence.
LSC-M provides an 80% success rate in the relief of symptoms, similar to abdominal myomectomy. However, it comes with significantly fewer complications.[65] Furthermore, LSC-M shares the benefits of a minimally invasive approach providing patients with shorter recovery time and fewer complications. One study looking specifically at the short-term quality of life post-LSC-M demonstrated a median 20-day shorter time to return to work and a 1-week shorter time to return to normal activity than abdominal myomectomy.[60] (B2)
In another study looking at outcomes immediately post-op, patients who underwent laparoscopic myomectomy had less post-op pain at the 6-hour and 48-hour mark, a 50% lower chance of perioperative fever, and shorter hospitalization compared to those who underwent abdominal myomectomy.[66](A1)
Risk of complication during LSC-M is increased in patients with fibroids >5 cm, >3 total fibroids, and in those with intramural and intraligamentous fibroids.[67] Complications include hemorrhage, fever/infection, bowel or bladder injury, conversion to laparotomy, and adhesive disease. A large multicenter study looking at over 2000 cases demonstrated a total complication rate of 11.1% (n=225), of which 9.1% were minor complications and 2.02% were major complications. The most common minor complications were fever of unexplained origin (5.1%) and urinary tract infections (3.4%). Among the major complications were hemorrhage (0.7%), hematoma (0.48%), bowel injury (0.04%), and post-op kidney failure (0.04%).[67] (B2)
Conversion to laparotomy is overall a rare event. In a study looking at over 700 intended laparoscopic myomectomies, only seven had to be converted to an abdominal open approach.[68] Risk factors for conversion to abdominal myomectomy include the size of fibroids (5 cm on ultrasound), intramural type fibroids, anterior fibroids, and preoperative use of GnRH agonists, and elevated BMI.[69] Long-term complications of surgery include adhesive disease (36-66%) and recurrence of fibroids.[70][71][72](A1)
It is worth mentioning the use of robot-assisted laparoscopic myomectomy (RLSC-M), which has grown in popularity amongst many minimally invasive gynecologic surgeons in recent years. Preference for the use of either technique is based on the surgeon’s skill level in using the conventional vs. robotic approach in handling complex cases. Surgeons tend to prefer RLSC-M in cases that involve ≥10 fibroids ≤ 7 cm or in patients with obesity due to improved visibility.[73] The most significant difference between the two techniques is conversion to abdominal myomectomy. In a systematic review and meta-analysis looking at over 2000 patients, laparoscopic myomectomy was associated with a 4.5 times increased risk of conversion to abdominal myomectomy compared to robot-assisted laparoscopic myomectomy.[74] (B2)
Regarding outcomes and complications, a meta-analysis of eight retrospective trials comparing the two techniques demonstrated no significant difference in complications, operative time, length of hospital stays, and postoperative fertility.[75] Given these subtle differences and the increased costs associated with robotic-assisted laparoscopy, there is no significant indication for choosing it over LSC-M. With both approaches, the failure rate remains comparably low.[76](A1)
Abdominal Myomectomy
Abdominal myomectomy (ABD-M) is a major surgical procedure typically reserved for complex cases where a minimally invasive approach is contraindicated or in failed intended laparoscopy cases. Similar to LSC-M, an abdominal myomectomy is best used for symptomatic intramural and subserosal fibroids.
In a retrospective cohort study looking at 30 patients who underwent ABD-M, overall symptoms improved in 90% of patients, and 79% of patients were satisfied with their choice of the procedure.[65] With regards to fertility outcomes, a Cochrane review demonstrated no difference between LSC-M and ABD-M.[77] When looking at the difference in operative time, Barakat et al. demonstrated that ABD-M is associated with a shorter operative time of 126 minutes, compared to 155 and 181 minutes with LSC-M and RLSC-M, respectively.[78] Another benefit is the lower risk of recurrence with ABD-M eight years post-procedure (63.4% vs. 76.2%) compared with LSC-M.[79] This is likely due to the ability to identify and remove sub-centimeter-sized fibroids more easily with an ABD-M approach. (A1)
Despite the similar outcomes in symptom reduction, fertility, and benefits in operative time, ABD-M is associated with significant morbidity. Blood loss is significantly higher with ABD-M compared to its minimally invasive alternatives, with an average of 200 mL blood loss during ABD-M compared to 150 ml and 100 ml in LSC-M and RLSC-M, respectively.[78] A systematic review and meta-analysis performed by Chen et al. demonstrated that ABD-M caused a longer duration of post-op ileus and increased length of hospital stay.[80] A Cochrane review concluded that pain was increased at the 6-hour and 48-hour mark after an ABD-M compared to LSC-M. This review also found a 50% increased risk of postoperative fever in patients who received ABD-M compared to LSC-M.[66] Due to the higher rate of surgical complications compared to its minimally invasive counterparts, patient selection for ABD-M and preoperative optimization are paramount.(A1)
Myomectomy for Improving Fertility
Many studies have demonstrated a clear relationship between uterine fibroids and infertility; however surgical management of fibroids for the sole purpose of improving fertility (i.e., in otherwise asymptomatic patients) is less clear cut and largely depends on the location of the fibroids.
Fibroids that distort the uterine cavity, i.e., submucosal fibroids (FIGO Type 0-2), strongly influence reproductive outcomes.[81] In one systematic review and meta-analysis of 23 randomized controlled trials evaluating pregnancy outcomes of infertile women with and without SM fibroids, women with SM fibroids had a significantly reduced rate of implantation (RR=0.283), clinical pregnancy (RR=0.363), and ongoing pregnancy/live birth rate (RR=0.318). Furthermore, women with SM fibroids have increased rates of spontaneous abortion.[82][61] Evidence suggests that cavity-distorting myomas should be surgically removed to improve reproductive outcomes even if the patient is otherwise asymptomatic.[81] A prospective study of 181 infertile women with a solitary fibroid <4 cm compared pregnancy outcomes in those who underwent myomectomy vs. those who underwent expectant management. In patients with submucosal fibroids (n=52), those who underwent submucosal myomectomy had a clinical pregnancy rate of 43% compared to 27% in the expectant management group after one year of enrollment (p < .05).[83] At this time, there is insufficient evidence to suggest that HSC-M for submucosal fibroids reduces miscarriage rates.[81](A1)
The negative impact of intramural (IM) fibroids on fertility outcomes has been demonstrated in multiple studies. However, limited data suggest whether or not the removal of the intramural fibroids improves these outcomes. In general, women with intramural fibroids have a lower implantation rate, clinical pregnancy, and ongoing pregnancy/live birth.[82][61] One of the largest prospective cohort studies investigating the success of in vitro fertilization (IVF) in women with and without fibroids showed that in women with intramural fibroids ≤ 5 cm (mean size 2.3 cm), the clinical pregnancy rate per embryo transfer was 23.3% compared to 34.1% in women without fibroids.[84] (A1)
An RCT of 181 women with a solitary fibroid < 4 cm and at least one year of infertility showed that myomectomy was not associated with an improved clinical pregnancy rate in women with intramural fibroids (n=23, 56.5%, NS).[83] On the other hand, a prospective, nonrandomized cohort study of 318 women with unexplained infertility or recurrent pregnancy loss showed that women who underwent LSC-M had higher birth rates than those with fibroids who did not undergo LSC-M (42% [44/106] vs. 11% [12/106], P <.001, respectively).[85] In summary, myomectomy for intramural fibroids does not necessarily guarantee the resolution of infertility. However, there is fair evidence to support that it does not impair reproductive outcomes. If a patient with large intramural fibroids has failed multiple IVF cycles, myomectomy should be strongly considered. Ultimately, the decision to undergo myomectomy for intramural fibroids should result from a shared decision-making process between the patient and her surgeon. (B2)
Studies looking at subserosal (SS) fibroids suggest they do not play a role in infertility and thus do not need to be removed to improve reproductive outcomes.[82][61](A1)
Alternative Procedural Treatment Modalities
Uterine Artery Embolization (UAE)
Uterine artery embolization (UAE) is a percutaneous transcatheter embolization technique that occludes the cervicovaginal branch of both uterine arteries to deprive fibroids of their blood supply leading to degeneration [76] (figure 6). Ideal candidates for this approach are patients with heavy menstrual bleeding (HMB) caused by intramural fibroids who are either resistant to or not ideal candidates for surgery.
In a study looking at ten-year follow-up after UAE, 62% of patients reported their HMB had resolved at two years, which increased to 83% at five years post-procedure. Overall, 78% of patients reported they were very satisfied with the treatment at the ten-year mark.[86] UAE is associated with decreased risk for transfusion, shorter hospital stays, and quicker return to baseline functioning.[87][88](A1)
Although the UAE can provide relief for many patients and avoid surgery-associated risks, studies have shown that as many as 19-38% of patients undergo reintervention for persisting symptoms.[89] Over five years, the rate of re-intervention requiring hysterectomy was 28%.[90] Major complications occur about 1 to 12% of the time and include rehospitalization, ovarian failure, pulmonary embolism, and unplanned hysterectomy.[91] (A1)
Minor complications include fever, pain, nausea, pelvic infection, postembolization syndrome, and vaginal discharge. There is conflicting data regarding reproductive outcomes after UAE compared to myomectomy. Overall, UAE is not the ideal treatment option for patients who desire future fertility due to a potential post-procedure decline in ovarian reserve and increased risk of pregnancy-related complications. Regarding pregnancy-related complications, UAE for uterine fibroids is associated with an increase of miscarriage (35.2% versus 16.5%; OR, 2.8; 95% CI, 2.0–3.8), cesarean delivery (66% versus 48.5%; OR, 2.1; 95% CI, 1.4–2.9), and postpartum hemorrhage (13.9% versus 2.5%; OR, 6.4; 95% CI, 3.5–11.7) compared to expectant management.[92](A1)
Magnetic Resonance–guided Focused Ultrasound Surgery (MRgFUS)
Magnetic resonance-guided focused ultrasound surgery is a form of therapy that involves continuous waves of ultrasound energy over a 3 to the 5-hour period under magnetic resonance guidance. Eventually, the goal is to heat the entire fibroid to >57 degrees C for at least 1 second to induce coagulative necrosis.[76][93] Fibroids that possess a high T2-weighted signal intensity are more likely to be responsive to the treatment.[94] MRgFUS is a minimally invasive approach with very minimal recovery time, low radiation exposure and provides symptomatic treatment serving as a great option for patients who are opposed to surgery.
A study performed by Funaki et al. looking at 91 women over 24 months who underwent MRgFUS demonstrated a mean reduction in the volume of 36.5% at six months and 39.5% at 24 months.[95] Another study looking at 12-month follow-up showed an 86%, 93%, and 88% at 3, 6, and 12 months, respectively.[96] In patients with primarily intramural myomas >3cm and uteri size < 24 weeks of gestation, MRgFUS decreased the percentage of patients reporting excessive bleeding from 69 to 31% and relieving pressure symptoms in 49% of patients.[97] There is limited research regarding reproductive outcomes after MRgFUS, with most data coming from a few case reports and small case series. Existing data is reassuring, with one case series describing 54 pregnancies in 51 women after MRgFUS.[98] More studies are needed to determine whether MRgFUS should be recommended for patients seeking future fertility.(A1)
The overall complication rate with MRgFUS is low at about 13.1% in a study that looked at 150 women who underwent the procedure. However, only one of these was a major complication (deep vein thrombosis).[96] In the same study, 7.4% of patients required treatment for fibroid-related symptoms at one year following the procedure. Other side effects of the therapy involve skin burns, nerve damage, necrosis of non-targeted tissue, bone heating, and venous thrombosis due to prolonged immobility.[76][97][99] (B2)
Ultrasound-guided Radiofrequency Ablation (Laparoscopic RFA)
Radiofrequency ablation (RFA) is a therapy that involves applying energy via ultrasound which utilizes direct thermal conduction to induce coagulative necrosis in tissue.[100] The technique now includes using laparoscopic guidance using a laparoscopic transducer allowing direct placement of the radiofrequency ablation tip directly into the fibroid.
In a prospective international trial observing a cohort of 135 women with fibroids and HMB, menstrual blood loss decreased from baseline levels by 38.3 months, and fibroid volume decreased by 45.1% at the 1-year mark. Furthermore, symptom severity scores improved significantly along with the quality of life, and 94% of women reported satisfaction with the procedure.[101] Another study of 31 women had improved symptom severity scores by 82% and a mean reduction of uterine volume from 194.4 cm to 113.2 cm at one year.[102] (B2)
There is very limited data on fertility and pregnancy after RFA. There is documentation of 15 pregnancies in 13 patients yielding 13 live births after RFA for fibroids.[100] In addition, a case series of 30 pregnancies by Berman et al. demonstrated 26 full-term live birth and four pregnancy losses.[103] Current trials are beginning to include more patients desiring fertility, given that RFA causes minimal myometrial damage. However, no definitive conclusions can be drawn about pregnancy outcomes and pregnancy-related risks.(B2)
A meta-analysis regarding the rate of surgical reintervention after RFA was 4.2%, 8.2%, and 11.5% at 1, 2, and 3 years, respectively.[104] The Halt trial looked at the 3-year outcome of RFA in 104 patients and reported a reintervention rate of 11% (14 patients) which included 11 hysterectomies, two laparoscopic myomectomies, and 1 UAE.[105] The incidence of complications and adverse effects remains inconsistent across studies.(A1)
Differential Diagnosis
The differential diagnosis for uterine fibroids includes conditions that lead to a similar symptom profile of uterine enlargement (e.g., bulk symptoms), HMB, infertility, and/or dysmenorrhea. The main diagnoses to consider when evaluating a patient for these symptoms are pregnancy, adenomyosis, benign or malignant uterine neoplasms, endometrial carcinoma, and uterine leiomyosarcoma.
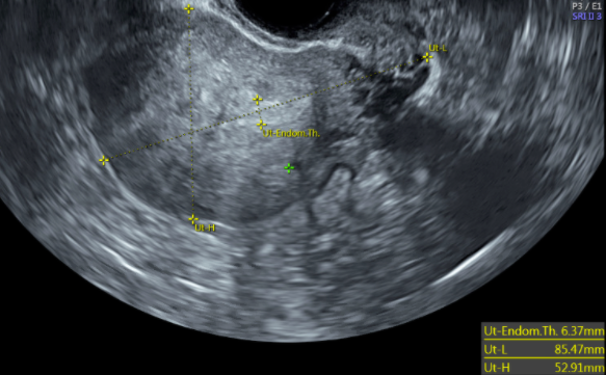
Adenomyosis often presents with an enlarged uterus, HMB, and dysmenorrhea. However, there are a few factors that can help in discerning between the two. Typically, the uterus is described to be smooth, globular, and boggy. However, this can be difficult to distinguish from fibroids. One of the main differentiating tools is transvaginal ultrasonography.[26] Features that support adenomyosis on ultrasound are asymmetric myometrial thickness, heterogeneous myometrium, heterogenous cysts, and sub endometrial echogenic linear striations (figure 7).[106][107]
A fibroid is typically well-circumscribed, whereas adenomyomas are more ill-defined. Adenomyomas are also more difficult to excise than fibroids. Thus suspicion of an adenomyoma might not be discovered until excision is attempted.[108] Ultimately, the gold standard for diagnosis is pathology, and an official diagnosis cannot be made solely with imaging.
Uterine leiomyosarcoma is a malignant neoplasm that is important to consider in the differential for uterine fibroids. They commonly present as fibroids with abnormal vaginal bleeding and a rapidly enlarging uterus. It is important to consider this differential in any post-menopausal women with rapidly enlarging uterine fibroids, as a correct preoperative diagnosis occurs less than 50% of the time. This should prompt further evaluation with hysterectomy without morcellation because the only way to exclude uterine leiomyosarcoma is through excision and pathologic examination.[109]
Endometrial carcinoma is another important differential to rule out when evaluating a patient with uterine bleeding, pain, and enlarging uterus.[110] On transvaginal ultrasound, endometrial carcinoma is typically described as irregular endometrial margins, a polypoid intrauterine mass, intrauterine fluid collection, and in more advanced disease, frank myometrial invasion from the endometrium. The use of color doppler to distinguish benign lesions from malignant is still controversial, and ultimately the diagnosis can only be made by pathologic examination.[111]
Prognosis
The overall prognosis of patients depends on the size and number of fibroids and the treatment modality they choose. Comparing Options For Management: Patient-centered results for Uterine Fibroids (COMPARE-UF), a prospective nationwide registry that looked at the short-term interval follow-up after different routes of myomectomy in 1206 women, showed that regardless of the type of myomectomy, all had improvement in short-term health-related quality of life and symptom severity score improvement. Return to usual activity averaged at 0 days in HSC-M patients compared to 21 and 28 days for laparoscopic and abdominal myomectomy, respectively.[60]
Some notable risk factors for fibroid recurrence include age less than 35.5 years, two or fewer fibroids at time of surgery, size of the uterus less than 13 weeks of gestation, and no childbearing after surgery.[112] At eight years post-operation, ABD-M has a 63.4% risk of fibroid recurrence compared to 76.2% after LSC-M.[79] Reintervention rates range from 19 to 38% after UAE and as high as 54% after MRgFUS.[89][58]
For those patients who become pregnant following myomectomy, delivery modality and timing of delivery recommendations are dependent on factors as the type of fibroid removed, number and location of incisions on the uterus, and if hysterotomy was made. The main concern in pregnancy in patients with a non-low transverse scar on the uterus, especially those with a mid-vertical or trans-fundal uterine scar, is the risk of uterine rupture, which is as high as 10%. Although published data is limited, the risk of uterine rupture after myomectomy is reportedly closer to 1%, similar to the risk of a trial of labor after cesarean section with successful vaginal delivery being achieved in about 90% of patients.[113]
According to the Society of Maternal-Fetal Medicine (SMFM), patients with a prior myomectomy with entry into the uterine cavity should undergo a scheduled cesarean delivery between 37-38 weeks gestation. If extensive trans-fundal scarring is suggested from the myomectomy operative report, delivery should be considered between 36 and 37 weeks gestation.[114]
Complications
Every medical intervention has a side effect profile, and every IR or surgical intervention has its associated risks, as described in detail above. Counseling patients on the adverse effects of medications such as altered bleeding profile and medication-induced menopause are worth mentioning to patients when they are deciding on treatment modality. Informing patients about the risks of bleeding, infection, injury to nearby organs, scarring, and failed treatment are all important to mention when consenting a patient for surgery or alternative procedure. Although the overall risk of hysterectomy for uncontrolled intraoperative bleeding is fewer than 1 out of 100 women, it is especially important to mention this risk to patients, particularly in women with multiple large fibroids.
Deterrence and Patient Education
When a patient presents for management of symptomatic fibroids, the discussion of management options should not only be guided by treatments most likely to improve their symptomatology but also by the patient’s desire for future fertility and the overall health status that may preclude them at higher risk of complication with one treatment strategy over another. As described in detail above, treatment strategies include medical therapy, surgical intervention, and procedures with interventional radiology (IR). Expectant management is an option for those who are asymptomatic and do not desire intervention. For patients not seeking immediate fertility, treatment strategies typically follow a “step-up approach,” which begins with minimally invasive or medical therapy before moving on to excisional surgery.[3]
Alternatively, in patients desiring immediate fertility and expectant management is not recommended due to location and size of the fibroid, surgical intervention is typically recommended as the initial treatment modality, as this is typically the most efficient way to remove the space-occupying mass. Nonetheless, once goals of care are reviewed, the treatment strategy should be individualized for each patient through shared decision-making. Risks, benefits, and alternatives to each treatment modality should be discussed at length to ensure the patient is making an educated decision and is providing informed consent for the next steps in their care.
Enhancing Healthcare Team Outcomes
Uterine fibroids are the most common tumor of the reproductive tract, and their presence has a significant impact on health care delivery and costs worldwide. All obstetricians and gynecologists are trained in the management of fibroids. However, skills and expertise, especially within surgical management, can vary significantly across providers. Depending on the patients’ range of symptoms and goals of care, management strategies are typically decided upon through shared decision-making between the patient and the provider. Symptomatic patients desiring pregnancy should be made aware of their fertility potential before selecting a management option that may significantly delay their time to conception. Gynecologists should consider consulting a fertility specialist to assist in counseling, especially in patients >35 years old.
Multiple other health professionals are needed to coordinate care to improve patient safety, satisfaction, and overall outcomes. For example, when medical management of symptomatic fibroids is adopted, care coordination between the physician, nursing staff, and pharmacists helps to ensure the patient understands expectations for treatment, how to self-administer the medication, potential side effects, and when to follow up to assess the medication’s effect. When a patient prefers a more aggressive form of therapy, but either does not want surgery or is not an ideal surgical candidate due to medical comorbidities, calling on colleagues in Interventional Radiology to assist in treatment may be the best option for specific patients. Follow-up visits to assess symptom improvement should be a shared responsibility between the interventional radiologist and gynecologist.
Lastly, when surgical management is deemed most appropriate for a specific patient, a massive effort of patient care coordination between multiple providers is required to ensure safe transitions between the preoperative, intra-operative, and postoperative phases of care. For some patients, preoperative medical clearance from their Primary Care Provider may be indicated before surgery. For example, patients with chronic blood loss anemia from heavy menstrual bleeding may benefit from hematology consultation for preoperative optimization.
All potential medical issues must be communicated to the anesthesiologist involved prior to surgery. Communication between the office and surgical center or hospital operating room is of utmost importance to ensure the appropriate equipment and staffing is available to perform the surgery. Additionally, many health systems have adopted Early Recovery After Surgery (ERAS) protocols to decrease the length of hospital stays and improve patient outcomes. If an ERAS protocol is utilized, it is crucial for the outpatient nurses to relay the preoperative preparation steps to the patient. The surgeon should communicate the need for such a protocol to inpatient nurses to ensure that the remainder of the protocol is carried out. As such, a coordinated effort amongst health professionals at all levels is necessary to provide the optimum level of patient care and promote patient safety.
Media
(Click Image to Enlarge)
(Click Image to Enlarge)
(Click Image to Enlarge)
(Click Image to Enlarge)
(Click Image to Enlarge)
References
Stewart EA, Cookson CL, Gandolfo RA, Schulze-Rath R. Epidemiology of uterine fibroids: a systematic review. BJOG : an international journal of obstetrics and gynaecology. 2017 Sep:124(10):1501-1512. doi: 10.1111/1471-0528.14640. Epub 2017 May 13 [PubMed PMID: 28296146]
Level 1 (high-level) evidenceAmerican College of Obstetricians and Gynecologists’ Committee on Practice Bulletins–Gynecology., Management of Symptomatic Uterine Leiomyomas: ACOG Practice Bulletin, Number 228. Obstetrics and gynecology. 2021 Jun 1 [PubMed PMID: 34011888]
Giuliani E,As-Sanie S,Marsh EE, Epidemiology and management of uterine fibroids. International journal of gynaecology and obstetrics: the official organ of the International Federation of Gynaecology and Obstetrics. 2020 Apr [PubMed PMID: 31960950]
Wallach EE,Vlahos NF, Uterine myomas: an overview of development, clinical features, and management. Obstetrics and gynecology. 2004 Aug; [PubMed PMID: 15292018]
Level 3 (low-level) evidenceGuo XC,Segars JH, The impact and management of fibroids for fertility: an evidence-based approach. Obstetrics and gynecology clinics of North America. 2012 Dec [PubMed PMID: 23182558]
Buttram VC Jr,Reiter RC, Uterine leiomyomata: etiology, symptomatology, and management. Fertility and sterility. 1981 Oct [PubMed PMID: 7026295]
Wise LA,Palmer JR,Harlow BL,Spiegelman D,Stewart EA,Adams-Campbell LL,Rosenberg L, Reproductive factors, hormonal contraception, and risk of uterine leiomyomata in African-American women: a prospective study. American journal of epidemiology. 2004 Jan 15 [PubMed PMID: 14718211]
Level 2 (mid-level) evidencePavone D,Clemenza S,Sorbi F,Fambrini M,Petraglia F, Epidemiology and Risk Factors of Uterine Fibroids. Best practice & research. Clinical obstetrics & gynaecology. 2018 Jan [PubMed PMID: 29054502]
Takeda T,Sakata M,Isobe A,Miyake A,Nishimoto F,Ota Y,Kamiura S,Kimura T, Relationship between metabolic syndrome and uterine leiomyomas: a case-control study. Gynecologic and obstetric investigation. 2008 [PubMed PMID: 18230910]
Level 2 (mid-level) evidenceRoss RK,Pike MC,Vessey MP,Bull D,Yeates D,Casagrande JT, Risk factors for uterine fibroids: reduced risk associated with oral contraceptives. British medical journal (Clinical research ed.). 1986 Aug 9 [PubMed PMID: 3730804]
Flake GP,Andersen J,Dixon D, Etiology and pathogenesis of uterine leiomyomas: a review. Environmental health perspectives. 2003 Jun [PubMed PMID: 12826476]
Level 3 (low-level) evidenceBulun SE, Uterine fibroids. The New England journal of medicine. 2013 Oct 3; [PubMed PMID: 24088094]
Level 3 (low-level) evidenceIshikawa H,Ishi K,Serna VA,Kakazu R,Bulun SE,Kurita T, Progesterone is essential for maintenance and growth of uterine leiomyoma. Endocrinology. 2010 Jun [PubMed PMID: 20375184]
Level 3 (low-level) evidenceBertsch E,Qiang W,Zhang Q,Espona-Fiedler M,Druschitz S,Liu Y,Mittal K,Kong B,Kurita T,Wei JJ, MED12 and HMGA2 mutations: two independent genetic events in uterine leiomyoma and leiomyosarcoma. Modern pathology : an official journal of the United States and Canadian Academy of Pathology, Inc. 2014 Aug [PubMed PMID: 24390224]
Level 2 (mid-level) evidenceStewart EA, Uterine fibroids. Lancet (London, England). 2001 Jan 27 [PubMed PMID: 11214143]
Brandon DD,Erickson TE,Keenan EJ,Strawn EY,Novy MJ,Burry KA,Warner C,Clinton GM, Estrogen receptor gene expression in human uterine leiomyomata. The Journal of clinical endocrinology and metabolism. 1995 Jun [PubMed PMID: 7775635]
Islam MS,Ciavattini A,Petraglia F,Castellucci M,Ciarmela P, Extracellular matrix in uterine leiomyoma pathogenesis: a potential target for future therapeutics. Human reproduction update. 2018 Jan 1; [PubMed PMID: 29186429]
Stewart EA,Friedman AJ,Peck K,Nowak RA, Relative overexpression of collagen type I and collagen type III messenger ribonucleic acids by uterine leiomyomas during the proliferative phase of the menstrual cycle. The Journal of clinical endocrinology and metabolism. 1994 Sep [PubMed PMID: 8077380]
Stewart EA, Clinical practice. Uterine fibroids. The New England journal of medicine. 2015 Apr 23 [PubMed PMID: 25901428]
Nishino M,Togashi K,Nakai A,Hayakawa K,Kanao S,Iwasaku K,Fujii S, Uterine contractions evaluated on cine MR imaging in patients with uterine leiomyomas. European journal of radiology. 2005 Jan [PubMed PMID: 15607866]
Borah BJ,Nicholson WK,Bradley L,Stewart EA, The impact of uterine leiomyomas: a national survey of affected women. American journal of obstetrics and gynecology. 2013 Oct; [PubMed PMID: 23891629]
Level 2 (mid-level) evidencePractice Committee of American Society for Reproductive Medicine in collaboration with Society of Reproductive Surgeons., Myomas and reproductive function. Fertility and sterility. 2008 Nov [PubMed PMID: 19007608]
Cook H,Ezzati M,Segars JH,McCarthy K, The impact of uterine leiomyomas on reproductive outcomes. Minerva ginecologica. 2010 Jun; [PubMed PMID: 20595947]
Munro MG, Critchley HO, Broder MS, Fraser IS, FIGO Working Group on Menstrual Disorders. FIGO classification system (PALM-COEIN) for causes of abnormal uterine bleeding in nongravid women of reproductive age. International journal of gynaecology and obstetrics: the official organ of the International Federation of Gynaecology and Obstetrics. 2011 Apr:113(1):3-13. doi: 10.1016/j.ijgo.2010.11.011. Epub 2011 Feb 22 [PubMed PMID: 21345435]
Munro MG,Critchley HOD,Fraser IS,FIGO Menstrual Disorders Committee., The two FIGO systems for normal and abnormal uterine bleeding symptoms and classification of causes of abnormal uterine bleeding in the reproductive years: 2018 revisions. International journal of gynaecology and obstetrics: the official organ of the International Federation of Gynaecology and Obstetrics. 2018 Dec; [PubMed PMID: 30198563]
Committee on Practice Bulletins—Gynecology. Practice bulletin no. 128: diagnosis of abnormal uterine bleeding in reproductive-aged women. Obstetrics and gynecology. 2012 Jul:120(1):197-206. doi: 10.1097/AOG.0b013e318262e320. Epub [PubMed PMID: 22914421]
Dueholm M,Lundorf E,Hansen ES,Ledertoug S,Olesen F, Accuracy of magnetic resonance imaging and transvaginal ultrasonography in the diagnosis, mapping, and measurement of uterine myomas. American journal of obstetrics and gynecology. 2002 Mar; [PubMed PMID: 11904599]
Woźniak A,Woźniak S, Ultrasonography of uterine leiomyomas. Przeglad menopauzalny = Menopause review. 2017 Dec; [PubMed PMID: 29483851]
Cicinelli E,Romano F,Anastasio PS,Blasi N,Parisi C,Galantino P, Transabdominal sonohysterography, transvaginal sonography, and hysteroscopy in the evaluation of submucous myomas. Obstetrics and gynecology. 1995 Jan [PubMed PMID: 7800322]
Level 1 (high-level) evidenceOmary RA,Vasireddy S,Chrisman HB,Ryu RK,Pereles FS,Carr JC,Resnick SA,Nemcek AA Jr,Vogelzang RL, The effect of pelvic MR imaging on the diagnosis and treatment of women with presumed symptomatic uterine fibroids. Journal of vascular and interventional radiology : JVIR. 2002 Nov; [PubMed PMID: 12427815]
Sohn GS,Cho S,Kim YM,Cho CH,Kim MR,Lee SR,Working Group of Society of Uterine Leiomyoma., Current medical treatment of uterine fibroids. Obstetrics & gynecology science. 2018 Mar [PubMed PMID: 29564309]
Bofill Rodriguez M, Lethaby A, Farquhar C. Non-steroidal anti-inflammatory drugs for heavy menstrual bleeding. The Cochrane database of systematic reviews. 2019 Sep 19:9(9):CD000400. doi: 10.1002/14651858.CD000400.pub4. Epub 2019 Sep 19 [PubMed PMID: 31535715]
Level 1 (high-level) evidenceLethaby A,Wise MR,Weterings MA,Bofill Rodriguez M,Brown J, Combined hormonal contraceptives for heavy menstrual bleeding. The Cochrane database of systematic reviews. 2019 Feb 11; [PubMed PMID: 30742315]
Level 1 (high-level) evidenceSayed GH,Zakherah MS,El-Nashar SA,Shaaban MM, A randomized clinical trial of a levonorgestrel-releasing intrauterine system and a low-dose combined oral contraceptive for fibroid-related menorrhagia. International journal of gynaecology and obstetrics: the official organ of the International Federation of Gynaecology and Obstetrics. 2011 Feb [PubMed PMID: 21092958]
Level 1 (high-level) evidenceBahamondes L, Fernandes A, Monteiro I, Bahamondes MV. Long-acting reversible contraceptive (LARCs) methods. Best practice & research. Clinical obstetrics & gynaecology. 2020 Jul:66():28-40. doi: 10.1016/j.bpobgyn.2019.12.002. Epub 2019 Dec 20 [PubMed PMID: 32014434]
Kokolina VF,Iakunina LN,Dubovaia TN, [The system of regulation of blood coagulation in girls with juvenile uterine hemorrhage]. Pediatriia. 1990 [PubMed PMID: 2399071]
Wu JP,Pickle S, Extended use of the intrauterine device: a literature review and recommendations for clinical practice. Contraception. 2014 Jun [PubMed PMID: 24679478]
Sergison JE,Maldonado LY,Gao X,Hubacher D, Levonorgestrel intrauterine system associated amenorrhea: a systematic review and metaanalysis. American journal of obstetrics and gynecology. 2019 May [PubMed PMID: 30527945]
Level 1 (high-level) evidenceJiang W,Shen Q,Chen M,Wang Y,Zhou Q,Zhu X,Zhu X, Levonorgestrel-releasing intrauterine system use in premenopausal women with symptomatic uterine leiomyoma: a systematic review. Steroids. 2014 Aug; [PubMed PMID: 24832215]
Level 1 (high-level) evidenceNewton CL,Riekert C,Millar RP, Gonadotropin-releasing hormone analog therapeutics. Minerva ginecologica. 2018 Oct [PubMed PMID: 30264955]
Lethaby A,Puscasiu L,Vollenhoven B, Preoperative medical therapy before surgery for uterine fibroids. The Cochrane database of systematic reviews. 2017 Nov 15 [PubMed PMID: 29139105]
Level 1 (high-level) evidenceBradley LD, Gueye NA. The medical management of abnormal uterine bleeding in reproductive-aged women. American journal of obstetrics and gynecology. 2016 Jan:214(1):31-44. doi: 10.1016/j.ajog.2015.07.044. Epub 2015 Aug 5 [PubMed PMID: 26254516]
Schlaff WD,Ackerman RT,Al-Hendy A,Archer DF,Barnhart KT,Bradley LD,Carr BR,Feinberg EC,Hurtado SM,Kim J,Liu R,Mabey RG Jr,Owens CD,Poindexter A,Puscheck EE,Rodriguez-Ginorio H,Simon JA,Soliman AM,Stewart EA,Watts NB,Muneyyirci-Delale O, Elagolix for Heavy Menstrual Bleeding in Women with Uterine Fibroids. The New England journal of medicine. 2020 Jan 23 [PubMed PMID: 31971678]
Simon JA,Al-Hendy A,Archer DF,Barnhart KT,Bradley LD,Carr BR,Dayspring T,Feinberg EC,Gillispie V,Hurtado S,Kim J,Liu R,Owens CD,Muneyyirci-Delale O,Wang A,Watts NB,Schlaff WD, Elagolix Treatment for Up to 12 Months in Women With Heavy Menstrual Bleeding and Uterine Leiomyomas. Obstetrics and gynecology. 2020 Jun [PubMed PMID: 32459423]
Al-Hendy A,Lukes AS,Poindexter AN 3rd,Venturella R,Villarroel C,Critchley HOD,Li Y,McKain L,Arjona Ferreira JC,Langenberg AGM,Wagman RB,Stewart EA, Treatment of Uterine Fibroid Symptoms with Relugolix Combination Therapy. The New England journal of medicine. 2021 Feb 18 [PubMed PMID: 33596357]
Ali M,Al-Hendy A, Selective progesterone receptor modulators for fertility preservation in women with symptomatic uterine fibroids. Biology of reproduction. 2017 Sep 1 [PubMed PMID: 29025038]
Murji A,Whitaker L,Chow TL,Sobel ML, Selective progesterone receptor modulators (SPRMs) for uterine fibroids. The Cochrane database of systematic reviews. 2017 Apr 26 [PubMed PMID: 28444736]
Level 1 (high-level) evidenceDonnez J,Tatarchuk TF,Bouchard P,Puscasiu L,Zakharenko NF,Ivanova T,Ugocsai G,Mara M,Jilla MP,Bestel E,Terrill P,Osterloh I,Loumaye E,PEARL I Study Group., Ulipristal acetate versus placebo for fibroid treatment before surgery. The New England journal of medicine. 2012 Feb 2 [PubMed PMID: 22296075]
Level 1 (high-level) evidenceDonnez J,Vázquez F,Tomaszewski J,Nouri K,Bouchard P,Fauser BC,Barlow DH,Palacios S,Donnez O,Bestel E,Osterloh I,Loumaye E,PEARL III and PEARL III Extension Study Group., Long-term treatment of uterine fibroids with ulipristal acetate ☆. Fertility and sterility. 2014 Jun; [PubMed PMID: 24630081]
Level 1 (high-level) evidenceGurates B,Parmaksiz C,Kilic G,Celik H,Kumru S,Simsek M, Treatment of symptomatic uterine leiomyoma with letrozole. Reproductive biomedicine online. 2008 Oct [PubMed PMID: 18854113]
Level 3 (low-level) evidenceBulun SE,Imir G,Utsunomiya H,Thung S,Gurates B,Tamura M,Lin Z, Aromatase in endometriosis and uterine leiomyomata. The Journal of steroid biochemistry and molecular biology. 2005 May [PubMed PMID: 16024248]
Song H,Lu D,Navaratnam K,Shi G, Aromatase inhibitors for uterine fibroids. The Cochrane database of systematic reviews. 2013 Oct 23 [PubMed PMID: 24151065]
Level 1 (high-level) evidenceLeone Roberti Maggiore U,Scala C,Venturini PL,Ferrero S, Preoperative treatment with letrozole in patients undergoing laparoscopic myomectomy of large uterine myomas: a prospective non-randomized study. European journal of obstetrics, gynecology, and reproductive biology. 2014 Oct [PubMed PMID: 25150954]
Level 1 (high-level) evidenceWamsteker K,Emanuel MH,de Kruif JH, Transcervical hysteroscopic resection of submucous fibroids for abnormal uterine bleeding: results regarding the degree of intramural extension. Obstetrics and gynecology. 1993 Nov; [PubMed PMID: 8414318]
Emanuel MH,Hart A,Wamsteker K,Lammes F, An analysis of fluid loss during transcervical resection of submucous myomas. Fertility and sterility. 1997 Nov; [PubMed PMID: 9389820]
Hart R,Molnár BG,Magos A, Long term follow up of hysteroscopic myomectomy assessed by survival analysis. British journal of obstetrics and gynaecology. 1999 Jul [PubMed PMID: 10428527]
Van Dongen H,Emanuel MH,Smeets MJ,Trimbos B,Jansen FW, Follow-up after incomplete hysteroscopic removal of uterine fibroids. Acta obstetricia et gynecologica Scandinavica. 2006 [PubMed PMID: 17260223]
Sandberg EM,Tummers FHMP,Cohen SL,van den Haak L,Dekkers OM,Jansen FW, Reintervention risk and quality of life outcomes after uterine-sparing interventions for fibroids: a systematic review and meta-analysis. Fertility and sterility. 2018 Apr [PubMed PMID: 29653718]
Level 2 (mid-level) evidencePolena V,Mergui JL,Perrot N,Poncelet C,Barranger E,Uzan S, Long-term results of hysteroscopic myomectomy in 235 patients. European journal of obstetrics, gynecology, and reproductive biology. 2007 Feb [PubMed PMID: 16530319]
Level 2 (mid-level) evidenceLaughlin-Tommaso SK,Lu D,Thomas L,Diamond MP,Wallace K,Wegienka G,Vines AI,Anchan RM,Wang T,Maxwell GL,Jacoby V,Marsh EE,Spies JB,Nicholson WK,Stewart EA,Myers ER, Short-term quality of life after myomectomy for uterine fibroids from the COMPARE-UF Fibroid Registry. American journal of obstetrics and gynecology. 2020 Apr [PubMed PMID: 31678093]
Level 2 (mid-level) evidencePritts EA, Parker WH, Olive DL. Fibroids and infertility: an updated systematic review of the evidence. Fertility and sterility. 2009 Apr:91(4):1215-23. doi: 10.1016/j.fertnstert.2008.01.051. Epub 2008 Mar 12 [PubMed PMID: 18339376]
Level 1 (high-level) evidenceVercellini P,Zàina B,Yaylayan L,Pisacreta A,De Giorgi O,Crosignani PG, Hysteroscopic myomectomy: long-term effects on menstrual pattern and fertility. Obstetrics and gynecology. 1999 Sep; [PubMed PMID: 10472856]
Jansen FW,Vredevoogd CB,van Ulzen K,Hermans J,Trimbos JB,Trimbos-Kemper TC, Complications of hysteroscopy: a prospective, multicenter study. Obstetrics and gynecology. 2000 Aug [PubMed PMID: 10908775]
Level 2 (mid-level) evidenceThe Use of Hysteroscopy for the Diagnosis and Treatment of Intrauterine Pathology: ACOG Committee Opinion, Number 800. Obstetrics and gynecology. 2020 Mar [PubMed PMID: 32080054]
Level 3 (low-level) evidenceBroder MS,Goodwin S,Chen G,Tang LJ,Costantino MM,Nguyen MH,Yegul TN,Erberich H, Comparison of long-term outcomes of myomectomy and uterine artery embolization. Obstetrics and gynecology. 2002 Nov [PubMed PMID: 12423842]
Level 2 (mid-level) evidenceBhave Chittawar P,Franik S,Pouwer AW,Farquhar C, Minimally invasive surgical techniques versus open myomectomy for uterine fibroids. The Cochrane database of systematic reviews. 2014 Oct 21 [PubMed PMID: 25331441]
Level 1 (high-level) evidenceSizzi O,Rossetti A,Malzoni M,Minelli L,La Grotta F,Soranna L,Panunzi S,Spagnolo R,Imperato F,Landi S,Fiaccamento A,Stola E, Italian multicenter study on complications of laparoscopic myomectomy. Journal of minimally invasive gynecology. 2007 Jul-Aug [PubMed PMID: 17630163]
Level 2 (mid-level) evidenceSandberg EM,Cohen SL,Jansen FW,Einarsson JI, Analysis of Risk Factors for Intraoperative Conversion of Laparoscopic Myomectomy. Journal of minimally invasive gynecology. 2016 Mar-Apr [PubMed PMID: 26546180]
Dubuisson JB,Fauconnier A,Fourchotte V,Babaki-Fard K,Coste J,Chapron C, Laparoscopic myomectomy: predicting the risk of conversion to an open procedure. Human reproduction (Oxford, England). 2001 Aug [PubMed PMID: 11473973]
Dubuisson JB,Fauconnier A,Chapron C,Kreiker G,Nörgaard C, Second look after laparoscopic myomectomy. Human reproduction (Oxford, England). 1998 Aug; [PubMed PMID: 9756277]
Mais V,Ajossa S,Piras B,Guerriero S,Marongiu D,Melis GB, Prevention of de-novo adhesion formation after laparoscopic myomectomy: a randomized trial to evaluate the effectiveness of an oxidized regenerated cellulose absorbable barrier. Human reproduction (Oxford, England). 1995 Dec [PubMed PMID: 8822429]
Level 1 (high-level) evidenceHasson HM,Rotman C,Rana N,Sistos F,Dmowski WP, Laparoscopic myomectomy. Obstetrics and gynecology. 1992 Nov [PubMed PMID: 1407934]
Vargas MV,Moawad GN,Sievers C,Opoku-Anane J,Marfori CQ,Tyan P,Robinson JK, Feasibility, Safety, and Prediction of Complications for Minimally Invasive Myomectomy in Women With Large and Numerous Myomata. Journal of minimally invasive gynecology. 2017 Feb [PubMed PMID: 27939896]
Level 2 (mid-level) evidenceGkegkes ID,Iatrakis G,Iavazzo PE,Bakalianou K,Iavazzo C, Robotic Management of Fibroids: Discussion of Use, Criteria and Advantages. Acta medica (Hradec Kralove). 2020 [PubMed PMID: 32771070]
Iavazzo C,Mamais I,Gkegkes ID, Robotic assisted vs laparoscopic and/or open myomectomy: systematic review and meta-analysis of the clinical evidence. Archives of gynecology and obstetrics. 2016 Jul; [PubMed PMID: 26969650]
Level 1 (high-level) evidenceGingold JA,Gueye NA,Falcone T, Minimally Invasive Approaches to Myoma Management. Journal of minimally invasive gynecology. 2018 Feb [PubMed PMID: 28734973]
Metwally M,Cheong YC,Horne AW, Surgical treatment of fibroids for subfertility. The Cochrane database of systematic reviews. 2012 Nov 14 [PubMed PMID: 23152222]
Level 1 (high-level) evidenceBarakat EE,Bedaiwy MA,Zimberg S,Nutter B,Nosseir M,Falcone T, Robotic-assisted, laparoscopic, and abdominal myomectomy: a comparison of surgical outcomes. Obstetrics and gynecology. 2011 Feb [PubMed PMID: 21252737]
Level 2 (mid-level) evidenceKotani Y,Tobiume T,Fujishima R,Shigeta M,Takaya H,Nakai H,Suzuki A,Tsuji I,Mandai M,Matsumura N, Recurrence of uterine myoma after myomectomy: Open myomectomy versus laparoscopic myomectomy. The journal of obstetrics and gynaecology research. 2018 Feb; [PubMed PMID: 29227004]
Chen R,Su Z,Yang L,Xin L,Yuan X,Wang Y, The effects and costs of laparoscopic versus abdominal myomectomy in patients with uterine fibroids: a systematic review and meta-analysis. BMC surgery. 2020 Mar 20; [PubMed PMID: 32192462]
Level 1 (high-level) evidencePractice Committee of the American Society for Reproductive Medicine. Electronic address: ASRM@asrm.org.,Practice Committee of the American Society for Reproductive Medicine., Removal of myomas in asymptomatic patients to improve fertility and/or reduce miscarriage rate: a guideline. Fertility and sterility. 2017 Sep [PubMed PMID: 28865538]
Desai P,Patel P, Fibroids, infertility and laparoscopic myomectomy. Journal of gynecological endoscopy and surgery. 2011 Jan [PubMed PMID: 22442534]
Level 2 (mid-level) evidenceCasini ML,Rossi F,Agostini R,Unfer V, Effects of the position of fibroids on fertility. Gynecological endocrinology : the official journal of the International Society of Gynecological Endocrinology. 2006 Feb [PubMed PMID: 16603437]
Level 2 (mid-level) evidenceHart R,Khalaf Y,Yeong CT,Seed P,Taylor A,Braude P, A prospective controlled study of the effect of intramural uterine fibroids on the outcome of assisted conception. Human reproduction (Oxford, England). 2001 Nov; [PubMed PMID: 11679530]
Level 1 (high-level) evidenceBulletti C,De Ziegler D,Polli V,Flamigni C, The role of leiomyomas in infertility. The Journal of the American Association of Gynecologic Laparoscopists. 1999 Nov; [PubMed PMID: 10548702]
de Bruijn AM, Ankum WM, Reekers JA, Birnie E, van der Kooij SM, Volkers NA, Hehenkamp WJ. Uterine artery embolization vs hysterectomy in the treatment of symptomatic uterine fibroids: 10-year outcomes from the randomized EMMY trial. American journal of obstetrics and gynecology. 2016 Dec:215(6):745.e1-745.e12. doi: 10.1016/j.ajog.2016.06.051. Epub 2016 Jul 5 [PubMed PMID: 27393268]
Level 1 (high-level) evidenceGupta JK, Sinha A, Lumsden MA, Hickey M. Uterine artery embolization for symptomatic uterine fibroids. The Cochrane database of systematic reviews. 2014 Dec 26:(12):CD005073. doi: 10.1002/14651858.CD005073.pub4. Epub 2014 Dec 26 [PubMed PMID: 25541260]
Level 1 (high-level) evidenceMara M,Maskova J,Fucikova Z,Kuzel D,Belsan T,Sosna O, Midterm clinical and first reproductive results of a randomized controlled trial comparing uterine fibroid embolization and myomectomy. Cardiovascular and interventional radiology. 2008 Jan-Feb [PubMed PMID: 17943348]
Level 1 (high-level) evidenceHartmann KE,Fonnesbeck C,Surawicz T,Krishnaswami S,Andrews JC,Wilson JE,Velez-Edwards D,Kugley S,Sathe NA, Management of Uterine Fibroids. 2017 Dec [PubMed PMID: 30789683]
van der Kooij SM,Hehenkamp WJ,Volkers NA,Birnie E,Ankum WM,Reekers JA, Uterine artery embolization vs hysterectomy in the treatment of symptomatic uterine fibroids: 5-year outcome from the randomized EMMY trial. American journal of obstetrics and gynecology. 2010 Aug [PubMed PMID: 20579960]
Level 1 (high-level) evidenceCarrillo TC, Uterine Artery Embolization in the Management of Symptomatic Uterine Fibroids: An Overview of Complications and Follow-up. Seminars in interventional radiology. 2008 Dec [PubMed PMID: 21326579]
Level 3 (low-level) evidenceHomer H,Saridogan E, Uterine artery embolization for fibroids is associated with an increased risk of miscarriage. Fertility and sterility. 2010 Jun [PubMed PMID: 19361799]
Level 1 (high-level) evidenceHesley GK,Gorny KR,Woodrum DA, MR-guided focused ultrasound for the treatment of uterine fibroids. Cardiovascular and interventional radiology. 2013 Feb [PubMed PMID: 22453202]
Funaki K,Fukunishi H,Funaki T,Sawada K,Kaji Y,Maruo T, Magnetic resonance-guided focused ultrasound surgery for uterine fibroids: relationship between the therapeutic effects and signal intensity of preexisting T2-weighted magnetic resonance images. American journal of obstetrics and gynecology. 2007 Feb [PubMed PMID: 17306674]
Funaki K,Fukunishi H,Sawada K, Clinical outcomes of magnetic resonance-guided focused ultrasound surgery for uterine myomas: 24-month follow-up. Ultrasound in obstetrics & gynecology : the official journal of the International Society of Ultrasound in Obstetrics and Gynecology. 2009 Nov [PubMed PMID: 19852041]
Level 2 (mid-level) evidenceGorny KR,Woodrum DA,Brown DL,Henrichsen TL,Weaver AL,Amrami KK,Hangiandreou NJ,Edmonson HA,Bouwsma EV,Stewart EA,Gostout BS,Ehman DA,Hesley GK, Magnetic resonance-guided focused ultrasound of uterine leiomyomas: review of a 12-month outcome of 130 clinical patients. Journal of vascular and interventional radiology : JVIR. 2011 Jun [PubMed PMID: 21482137]
Level 2 (mid-level) evidenceHesley GK,Felmlee JP,Gebhart JB,Dunagan KT,Gorny KR,Kesler JB,Brandt KR,Glantz JN,Gostout BS, Noninvasive treatment of uterine fibroids: early Mayo Clinic experience with magnetic resonance imaging-guided focused ultrasound. Mayo Clinic proceedings. 2006 Jul [PubMed PMID: 16835973]
Clark NA,Mumford SL,Segars JH, Reproductive impact of MRI-guided focused ultrasound surgery for fibroids: a systematic review of the evidence. Current opinion in obstetrics & gynecology. 2014 Jun [PubMed PMID: 24751998]
Level 1 (high-level) evidenceStewart EA, Rabinovici J, Tempany CM, Inbar Y, Regan L, Gostout B, Hesley G, Kim HS, Hengst S, Gedroyc WM. Clinical outcomes of focused ultrasound surgery for the treatment of uterine fibroids. Fertility and sterility. 2006 Jan:85(1):22-9 [PubMed PMID: 16412721]
Level 2 (mid-level) evidenceLee BB,Yu SP, Radiofrequency Ablation of Uterine Fibroids: a Review. Current obstetrics and gynecology reports. 2016; [PubMed PMID: 27917310]
Chudnoff SG,Berman JM,Levine DJ,Harris M,Guido RS,Banks E, Outpatient procedure for the treatment and relief of symptomatic uterine myomas. Obstetrics and gynecology. 2013 May; [PubMed PMID: 23635746]
Garza Leal JG,Hernandez Leon I,Castillo Saenz L,Lee BB, Laparoscopic ultrasound-guided radiofrequency volumetric thermal ablation of symptomatic uterine leiomyomas: feasibility study using the Halt 2000 Ablation System. Journal of minimally invasive gynecology. 2011 May-Jun; [PubMed PMID: 21545960]
Level 2 (mid-level) evidenceBerman JM,Shashoua A,Olson C,Brucker S,Thiel JA,Bhagavath B, Case Series of Reproductive Outcomes after Laparoscopic Radiofrequency Ablation of Symptomatic Myomas. Journal of minimally invasive gynecology. 2020 Mar - Apr; [PubMed PMID: 31238151]
Level 2 (mid-level) evidenceBradley LD,Pasic RP,Miller LE, Clinical Performance of Radiofrequency Ablation for Treatment of Uterine Fibroids: Systematic Review and Meta-Analysis of Prospective Studies. Journal of laparoendoscopic [PubMed PMID: 31702440]
Level 1 (high-level) evidenceBerman JM,Guido RS,Garza Leal JG,Pemueller RR,Whaley FS,Chudnoff SG,Halt Study Group., Three-year outcome of the Halt trial: a prospective analysis of radiofrequency volumetric thermal ablation of myomas. Journal of minimally invasive gynecology. 2014 Sep-Oct; [PubMed PMID: 24613404]
Kepkep K,Tuncay YA,Göynümer G,Tutal E, Transvaginal sonography in the diagnosis of adenomyosis: which findings are most accurate? Ultrasound in obstetrics [PubMed PMID: 17659649]
Level 2 (mid-level) evidenceAtri M,Reinhold C,Mehio AR,Chapman WB,Bret PM, Adenomyosis: US features with histologic correlation in an in-vitro study. Radiology. 2000 Jun; [PubMed PMID: 10831700]
Level 2 (mid-level) evidenceVan den Bosch T,Van Schoubroeck D, Ultrasound diagnosis of endometriosis and adenomyosis: State of the art. Best practice [PubMed PMID: 29506961]
Mallmann P, Uterine Sarcoma - Difficult to Diagnose, Hard to Treat. Oncology research and treatment. 2018; [PubMed PMID: 30326471]
Dave KS,Chauhan A,Bhansali R,Arora R,Purohit S, Uterine carcinosarcomas: 8-year single center experience of 25 cases. Indian journal of medical and paediatric oncology : official journal of Indian Society of Medical [PubMed PMID: 22557781]
Level 3 (low-level) evidenceFaria SC,Devine CE,Rao B,Sagebiel T,Bhosale P, Imaging and Staging of Endometrial Cancer. Seminars in ultrasound, CT, and MR. 2019 Aug; [PubMed PMID: 31375169]
Yoo EH,Lee PI,Huh CY,Kim DH,Lee BS,Lee JK,Kim D, Predictors of leiomyoma recurrence after laparoscopic myomectomy. Journal of minimally invasive gynecology. 2007 Nov-Dec; [PubMed PMID: 17980328]
Level 2 (mid-level) evidenceOdejinmi F,Strong S,Sideris M,Mallick R, Caesarean section in women following an abdominal myomectomy: a choice or a need? Facts, views [PubMed PMID: 32696025]
ACOG Practice Bulletin No. 205: Vaginal Birth After Cesarean Delivery. Obstetrics and gynecology. 2019 Feb; [PubMed PMID: 30681543]

![FIGO Leiomyoma Subclassification System, 2018 [30609040]](/pictures/getimagecontent//18370)