Introduction
Blepharospasm is a disease that results in an increased rate of bilateral eyelid closure, mainly attributed to the involuntary contraction of the orbicularis oculi muscles. Blepharospasm is a type of dystonia. Dystonia falls under the classification of movement disorders and is characterized by either sustained or intermittent contraction of a muscle. This leads to abnormal repetitive movements or postures, which tend to have a certain pattern and may be twisting or tremulous. In most dystonias, voluntary action typically leads to exacerbation of dystonia due to overactivation of muscles.[1]
Dystonia can potentially affect any part of the body and can present at a wide range of ages.[2]
Dystonia can be classified according to its distribution across the body:
- Focal dystonia refers to dystonia that affects only one isolated region of the body.
- Segmental dystonia refers to dystonia that affects 2 or more contiguous regions of the body.
- Multifocal dystonia refers to dystonia which affects 2 or more non-contiguous regions.
- Hemidystonia refers to dystonia, which affects half of the body.
- Generalized dystonia refers to dystonia, which affects the trunk along with 3 other sites.
Dystonia can have a static or progressive course of the disease. Furthermore, the variability of symptoms can be classified according to how often they occur:[1]
- Persistent dystonia refers to dystonia that persists to the same extent throughout the day.
- Action-specific dystonia refers to dystonia that only occurs when performing a certain activity.
- Diurnal fluctuation refers to dystonia, which fluctuates throughout the day with a circadian variation in severity.
- Paroxysmal dystonia refers to sudden episodes of dystonia typically induced by a trigger.
Examples of focal dystonia include blepharospasm, oromandibular dystonia, writer's cramp, spasmodic dysphonia, and torticollis.
Blepharospasm is a focal dystonia characterized by the simultaneous contraction of agonist and antagonist muscles, resulting in involuntary eyelid closure; the first report of patients with blepharospasm comes from a description of 10 patients, made by Henri Meige in 1910: these patients had involuntary eyelid closure in association with contraction of the muscles of the jaw. In his paper, Meige named this phenomenon "Convulsions de la Face" (convulsions of the face).[3]
This article will review the etiology, epidemiology, history, evaluation, and management of blepharospasm.
Etiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Etiology
The etiology of blepharospasm is not well understood. Some gene mutations have been implicated in the development of the disease: 20% to 30 % of cases have a positive family history of benign essential blepharospasm (BEB), and some genes have been associated with a greater susceptibility for developing the disease.[4] Other factors thought to be implicated in the pathogenesis of the disease are neurotransmitter dysregulations, structural damage, and previous underlying eye disorders.[5][6]
Non-inherited Risk Factors
Many environmental risk factors are associated with an increased risk of developing benign essential blepharospasm. These include high levels of urbanization and those working 'white-collar' jobs associated with a stressful lifestyle.
There are other factors that are loosely associated with an increased risk of developing blepharospasm, such as reading, watching television, and computer screen use.[7] It is believed that eye strain while watching television, reading, or using the computer may lead to aggravation of blepharospasm.[8]
Furthermore, patients suffering from psychiatric conditions such as obsessive-compulsive disorder, depression, and anxiety appear to have an increased risk of developing blepharospasm.[9]
It has been shown by Conte et al. that 40% to 60% of patients present with ophthalmic symptoms such as burning, dryness, or grittiness, which precedes the development of blepharospasm. Furthermore, there is an association between diseases of the anterior segment, such as keratoconjunctivitis/blepharitis, and an increased risk of developing blepharospasm, although this is more correctly termed "secondary blepharospasm" as the condition improves when the underlying conditions have been appropriately treated.[10][11][10]
Inherited Risk Factors
Genetic contribution to the disease is suggested by the finding of multiple affected individuals within families. In such cases, the inheritance pattern appears to be autosomal dominant with reduced penetrance. GNAL, CIZ1, TOR1A, DRD5, and REEP4 are genes reported to play a role in the development of BEB.[12][13][14]
GNAL:
The GNAL gene encodes for the G alpha subunit of the G protein receptor. It is found in the olfactory epithelium and helps to mediate odorant signaling.[14] It is also expressed in striatal neurons found in the basal ganglia. Mouse models have shown that it has a role in the coupling of adenyl cyclase in response to dopamine and adenosine via the Drd1 and Adora2a receptors.[15]
Mutations in the GNAL gene have been shown to be associated with autosomal dominant dystonia-25, and it is thought that mutations in GNAL leading to abnormalities in the function of DRD1/Adora2a contribute to the development of dystonia.[16]
CIZ1:
The CIZ1 gene encodes for the CKDN1A-interacting zinc finger protein 1.[17] This protein interacts with CIP1 to regulate the distribution of CIP1. CIZ1 has been found to be associated with autosomal dominant cervical dystonia.[13]
TOR1A:
The TOR1A gene encodes for torsin-1A, otherwise known as DYT1. This is an adenosine triphosphatase that aids with a wide range of cellular activities.[18] TOR1A has high levels of expression in melanized neurons found in the pars compacta (substantia nigra), dentate gyrus, cerebellum, and stratum pyramidale of CA3 (hippocampus).[19] It is thought that a mutation in the TOR1A gene may lead to changes in interactions involving TOR1A in the nuclear envelope.[20] Therefore, it is thought that mutations in TOR1A contribute to dystonia through defects in the structure and function of the nuclear membrane.
TOR1A mutations are associated with torsion dystonia and are inherited in an autosomal dominant pattern.[21]
DRD5:
DRD5 encodes for the dopamine receptor D1B. DRD5 functions to increase adenylate cyclase activity, leading to the accumulation of intracellular cAMP.[22] Polymorphisms in the D5 receptor gene have been shown to be associated with blepharospasm.[14][23]
Epidemiology
The estimated prevalence of benign essential blepharospasm worldwide is 20 to 133 cases per million and varies with geographic area.[24] Blepharospasm is less common than cervical dystonia in the United States and Europe. However, blepharospasm is more common in Italy and Japan than cervical dystonia.[6]
Gender
There is a preponderance of blepharospasm in women.[6] It is thought that one of the contributing factors to an increased risk of blepharospasm in women is menopause.[25] The ratio of blepharospasm in women to men is 2.3:1.[8][26] Women may also present with a higher symptom frequency and severity.[27]
Age
Blepharospasm peaks between the ages of 50 to 70.[6]
Pathophysiology
Blepharospasm is a disease that results in an increased rate of bilateral eyelid closure, mainly attributed to the involuntary contraction of the orbicularis oculi muscles.[28] The exact pathophysiology of the disease remains unknown. Many pathophysiological mechanisms have been suggested. Pathophysiologic mechanisms involved can be grouped into several categories: genetic, environmental, functional, and structural, which are not mutually exclusive. In fact, within a single subject, many of these factors must be present in order for the disease to develop. This is supported by what is called the two-hit hypothesis, which proposes that a predisposing factor and an environmental trigger must be present concomitantly for the disease to develop.[5][29]
The role of environmental factors in BEB is supported by studies showing the association of underlying eye disease and the antecedent of a traumatic stressor as triggers of the disease.[30][31]
Structural and functional mechanisms are thought to occur together as the condition is thought to be due to a structural defect that leads to neurotransmitter dysregulation.
Blink Reflex
Patients with BEB have an increased blinking rate and an increased response in the R2 phase of the trigeminal blinking reflex (TBR). TBR is a physiologic response that consists of two phases: phase R1 begins with corneal stimulation and consists of an afferent pathway that travels through the ophthalmic branch of the trigeminal nerve to reach the trigeminal nucleus in the brainstem. This phase is unilateral. After reaching the trigeminal nucleus, signals are sent to the facial nucleus bilaterally, which results in the R2 phase of the TBR.
The R2 phase is the efferent phase of the reflex, and it is carried by the zygomatic, buccal, and temporal branches of the facial nerve to reach the muscles of facial expression and result in their contraction bilaterally. As previously mentioned, patients with BEB exhibit a more intense R2 phase of the TBR as compared to healthy subjects.[32][33] Similarly, as there is a reflex that results in the stimulation of orbicularis oculi muscles, there is also a reflex for inhibition of the levator palpebrae superioris(LPS). This inhibition reflex is also composed of two phases: SP1 and SP2, the latter being more intense and of longer duration than the former. In healthy subjects, inhibition of LPS occurs synchronically with orbicularis oculi muscle action.[31] Whether or not a pathological response of the inhibitory reflex of LPS plays a role in the pathophysiology of BEB is not yet known.
Specific changes in brain structures have been reported by many authors. In one study performed by Etgen et al., it was found that patients with BEB exhibit gray matter increase bilaterally at the level of the putamen independent of the duration of the blepharospasm and a gray matter decrease in parietal lobes that appear to correlate with BEB duration. This finding is also supported by the fact that there is an increased glucose metabolism in the thalamus, as was evidenced by Suzuki et al.[34][35][34]
Functional magnetic resonance imaging has also been used to evaluate patients with BEB. When compared with healthy subjects, it was found that the anterior visual cortex, the thalamus, anterior cingulate cortex, primary motor cortex, and superior cerebellum exhibit a greater activation with voluntary and involuntary blinking.[36] Dopamine also appears to play a role in the pathogenesis of the disease. It was found in one study that these patients appear to have a decreased binding of dopamine to D2 receptors at the striatum when compared with healthy subjects.[37]
Anterior Segment Disease
As there is an association between prior anterior segment diseases of the eye and an increased risk of developing blepharospasm, it is thought that in people who are predisposed to blepharospasm, because of either genetic abnormalities or abnormalities in neurological signaling, there is a failure to regulate the blinking reflex leading to the development of trigeminal hyperexcitability and involuntary eyelid closure.[38]
History and Physical
BEB is an underdiagnosed entity. A study performed in Japan reported that 60% of patients see at least five physicians before they receive a definitive diagnosis. One-third of patients are diagnosed within the first year of symptom onset, one-third in a period between one and five years, and in one-third of patients, it takes more than five years before they are diagnosed with BEB.[39]
Precipitating Factors
Lee et al. have shown that 42.6% of patients experienced some sort of stressful life event before the onset of symptoms. This included major life events such as divorce (19.8%), problems at work (13.9%), illness of a family member (2%), death of a parent (1%), moving house (1%), head trauma (1%), traffic accident (1%) and cerebral infarction (1%). In a study in Tokyo that involved 1,116 participants, 9.7% reported symptoms following surgery: the most common surgery performed was cataract surgery.[30][39]
Motor Manifestations
The spasm associated with blepharospasm tends to be synchronous and bilateral, affecting the orbicularis oculi muscle.
Symptom severity is highly variable, and it can range from mild symptoms with increased blinking rate to functional blindness due to persistent muscle contraction. In many cases, the course of the disease is progressive, initially presenting with contractions limited to the orbicularis oculi muscles and later extending to the musculature of the lower face and neck, which is known as Meige syndrome.[33]
Apraxia of Eyelid Opening
Some patients suffering from blepharospasm may also present with apraxia of eyelid opening (AEO). This presents with an inability to reopen the eyes in the absence of orbicularis oculi muscle spasm. This occurs as a result of a spasm of the pretarsal orbicularis oculi muscle, which acts against the opening of the eyelid.[40]
Non-motor Manifestations
It is important to keep in mind that depression and anxiety are commonly present in patients with BEB. These occur either before the disease or as a consequence of it. This should be sought during evaluation and psychiatric consultation considered.[8]
Half of the patients with BEB have accompanying ocular symptoms, with most reporting eye dryness and photophobia.[41]
Relieving Factors
There are particular things some patients can do to help relieve their blepharospasm. This includes resting (35.6%), concentrating on work (12.9%), and various other things such as singing, talking, eating, sleep, and exercise, which were all reported at a rate of 2%. However, 31.7% of patients could not identify a relieving factor.[30]
Aggravating Factors
Things that have been shown to aggravate the symptoms of blepharospasm include fatigue (55.4%), stressful events (46.5%), watching television (27.7%), bright lights (18.9%), dry eye symptoms (14.9%), feeling sick (10.9%) or reading a book (8.9%).
Evaluation
The diagnosis of blepharospasm is mainly based on clinical assessment.
Clinical Assessment
A clinical history determining the nature of the eyelid spasm is highly important. Patients suffering from blepharospasm will complain of bilateral, synchronous, stereotyped spasms of the orbicularis oculi muscle. These spasms may present with brief and repetitive blinking or persistent closure of the eyes, leading to functional impairment. There is a diagnostic algorithm that is based on the presence of bilateral, synchronous, and stereotyped movements of the orbicularis oculi muscle, the presence of a sensory trick, or increased blinking. This diagnostic algorithm has been shown to have a sensitivity of 93% and a specificity of 90% in differentiating BEB from other similar conditions.[42][43][44]
Furthermore, as mentioned before, patients suffering from blepharospasm may also suffer from related non-motor manifestations such as psychiatric disorders, sleep disorders, sensory symptoms, and cognitive disturbances.
Sensory symptoms such as a sensation of dry eye or photophobia have been identified to be associated with a specificity of 94% and a sensitivity of 77% in patients suffering from BEB.[45]
Psychiatric disorders, such as anxiety and depression, are associated with BEB. However, other related conditions are also associated with higher rates of anxiety and depression.[28]
Severity Assessment
In order to assess the severity of the disease, many scales have been developed. These include The Jankovic Rating Scale (JRC), The Blepharospasm Severity Scale (BSS), The Blepharospasm Disability Scale (BDS), and The Blepharospasm Disability Index (BDI).[33]
The most widely used scale is the Jankovic Rating Scale. This is commonly used during the initial assessment and to monitor treatment response during patient follow-up. This scale assesses severity and frequency separately, giving a score of 0 to 4 for each one of these, as shown in Table 1.[46]
There also exist scales for severity grading of focal dystonia not specific for blepharospasm, but that are commonly used by some physicians when assessing patients with BEB. These include The Global Dystonia Rating Scale, The Burke-Fahn-Marsden Dystonia Rating Scale, and The Unified Dystonia Rating Scale.[42] Direct comparison of these grading scales has not been performed, and which one to use is determined by physician preference.
Neurophysiology
Electromyography
Dystonia presents with the contraction of antagonistic muscles. Electromyography studies can be used to identify such abnormalities. However, in practice, this is not always used.
Blink Reflex
Assessment of the blink reflex is useful in the evaluation of BEB. The tapping of the forehead (glabella reflex) may be done to induce reflexive blinking. By repeatedly tapping the forehead, it is possible to assess the patient's ability to inhibit the glabella reflex. This is known as Myerson's maneuver.[28] Another way to induce reflexive blinking is the use of a startling stimulus, such as visual or auditory stimuli.
In practice, the most frequently used method of inducing the blink reflex is electrical stimulation of the supraorbital nerve while performing an EMG on the orbicularis oculi muscle.[28] As mentioned earlier, the trigemino-facial blink reflex consists of an early ipsilateral R1 component and a late bilateral R2 component. The R1 component is facilitated by the pons, whereas the R2 component is dependent on both the pons and the lateral medulla.
The blink reflex can be assessed by using the paired shock technique. Both of the supraorbital nerves are stimulated with an impulse of identical intensity.[47] The first stimulus used induces a change in the excitability of the reflex circuits (conditioning), whereas the second stimulus (test), which is delivered at varying intervals (100 to 1000ms), is used as a probe stimulus. The size of the induced response is measured, and the test stimulus is compared to the conditioning stimulus. This is done for varying stimulus intervals. A normal patient will present with no R2 response during small intervals (100 to 200 ms), whereas a patient suffering from blepharospasm may have an R2 response with small intervals (100 to 200 ms).[28]
This test does not have high specificity for dystonia but can be used to rule out psychogenic dystonia, which will present with normal features.
Treatment / Management
The exact cause of benign essential blepharospasm is not well understood, and, as such, there is no definitive cure for BEB. However, there is a range of treatment options available.
Photochromatic Modulation
It has been shown that patients suffering from blepharospasm are able to tolerate light intensities similar to those tolerated by patients without blepharospasm. However, when particular wavelengths were blocked out using lens tints, the patients suffering from blepharospasm were no longer able to tolerate similar light intensities. This implies that photophobia in blepharospasm is dependent on both light intensity and wavelength.
Multiple studies have shown symptomatic improvement in patients treated with photochromatic modulation.[48] In particular, the FL-41 lens tint has been shown to reduce symptoms of photophobia in patients suffering from blepharospasm.[49][50](B2)
Botulinum Toxin injections
Botulinum toxins:
There are seven serotypes of the botulinum toxin (A, B, C, D, E, F, G), but only serotypes A and B are available for use.[51] In the United States, the only botulinum toxin products approved for use in blepharospasm are abobotulinumtoxinA, onabotulinumtoxinA, and incobotulinumtoxinA.[52] Studies have shown that abobotulinumtoxinA may have a longer duration of action compared to incobotulinumtoxinA.[53](B2)
Mode of action:
Botulinum toxin injection is the standard treatment for patients with BEB. It exerts its effect by inhibiting acetylcholine release at the neuromuscular junction, which leads to decreased muscle contraction.[54]
Technique:
Approximately 1.25-5 units of botulinum toxin are used per injection site.[53] However, with repeated injections, increasingly larger doses may be needed due to the formation of antibodies against the botulinum toxin in a few patients, along with the worsening of the underlying condition. The injection sites that are typically used include the lateral lower and upper eyelid margins, the lateral canthi, and the medial upper eyelid.[55] The toxin is typically injected into the procerus, corrugator, and orbicularis oculi muscles. The dose of botulinum toxin should not exceed more than 200 units (of onabotulinumtoxinA) within 30 days.[53]
It usually takes 48 hours from injection to clinical response. However, the response to treatment is highly variable. Nevertheless, the reduction of abnormal movements is seen in all subjects. Treatment response and duration of the effect appear to be dose-related, and the latter may be as long as 170 days.[56] On average, most patients become significantly symptomatic about 90 days after injection.
Botulinum toxin injections are recommended every three to four months. Some authors report a more intense effect seen with the first round of injections and decreasing efficacy with subsequent doses.[48](B3)
Potential reasons for a poor response to botulinum toxin injections include the development of antibodies against the botulinum toxin, poor injection technique, and the presence of apraxia of eyelid opening.[51] Antibodies against the botulinum toxin are more likely to develop if there is a short interval between injections, with the use of 'booster' injections, increasingly larger doses over time, and an early onset of botulinum toxin injection therapy.[57](B2)
Oral Pharmacotherapy
Oral pharmacotherapy for BEB has failed to show persistent relief from symptoms and tends to vary in effectiveness between patients.[48](B3)
This includes medications such as benzodiazepines, anticholinergics (benztropine and trihexyphenidyl), levodopa, baclofen, VMAT2 inhibitors (tetrabenazine), lithium, valproate, methylphenidate, zolpidem amongst others. All of these have a limited role but might be considered for refractory disease before taking patients to the operating room for a more invasive strategy and as an adjunct to botulinum toxin injections. Oral pharmacotherapy is also associated with potentially undesirable side effects, which are not associated with botulinum toxin injection, along with the potential risk of developing dependence on benzodiazepines.[33][48][58](B3)
Surgical Management
Surgical intervention is indicated in patients who fail to show a response to medical therapy and have persisting symptoms. Surgical management is performed with the intent to improve functionality and reduce the frequency of spasms.
In patients whose disease is refractory to standard measures, neurectomies, myomectomies, and deep brain stimulation (DBS) represent appropriate treatment alternatives. About half of patients treated with myomectomies or neurectomies will require botulinum toxin injection five years after surgery, but at lower doses than previously needed. Evidence supporting the use of DBS for BEB is not robust and should be considered a last resort.
Surgical myectomy:
Surgical myectomy involves the resection of the protractor muscles (corrugator supercili, orbicularis oculi, procerus, and depressor supercili). Gillum and Anderson described the surgical technique for full myectomy, which succeeds in controlling blepharospasm as there is an 88% improvement in patients with blepharospasm but may be associated with postoperative lymphedema, lagophthalmos, and facial anesthesia. Currently, modified and partial myectomy is the preferred technique. This is performed in addition to correcting the effects of blepharospasm, which include brow ptosis, ptosis, dermatochalasis, and lateral canthal dystopia.[48][59][60](B2)
Differential Diagnosis
There are many conditions that may present in a somewhat similar fashion to BEB. It is important to differentiate between these conditions and recognize their differences.
Apraxia of Eyelid Opening (AEO)
Most cases of AEO present in the context of extrapyramidal disorders. However, there are reports of AEO as an isolated finding in healthy individuals.
AEO shares many demographic aspects with BEB: its incidence is greater in the sixth decade of life, and it also has a female predominance (2:1). It tends to be bilateral, and most patients exhibit sensory tricks. Anxiety and depression are also common before and after diagnosis. In patients with isolated AEO, instead of contraction of the orbicularis oculi muscle, frontalis muscle contraction is seen in an effort to open the eyes.
In some cases, it is not possible to differentiate between BEB and AEO since many patients with BEB also have AEO, and some that initially present as AEO later develop blepharospasm, usually after 1.5 years. AEO is also treated with botulinum toxin injections, and surgery is reserved for refractory cases.[61]
Hemifacial Spasm
Hemifacial spasm has the same female-to-male distribution (2 to 1), and the age at onset is between 40 to 59 years. The estimated prevalence of hemifacial spasm is 11 per 100,000 people, 7.4 per 100,000 in men, and 14.5 per 100,000 in women.[62]
It is characterized by involuntary muscle contractions, but in this case, limited to one side of the face. These muscle contractions are typically intermittent, clonic, or tonic in nature. The muscle is relaxed in between the spasm episodes. It is aggravated by voluntary movement of the face, fatigue, stress, or a change in head position. Patients may complain of hemifacial spasms persisting in their sleep.[63] (In contrast, blepharospasm is not seen in sleep). Hemifacial spasms may be relieved by the consumption of alcohol, relaxation, or touching of the affected areas.[64] Hemifacial spasms may be associated with low-pitched tinnitus, which is thought to occur due to contraction of the stapedius muscle.[65]
The most common underlying cause of hemifacial spasm is compression by an ectatic vessel, typically the vertebral artery or the anterior/posterior cerebellar artery.[63]
Following botulinum toxin injection, 85 to 90% of patients experience improvement in their symptoms. Patients refractory to botulinum toxin injection may benefit from vascular decompression.[62]
Post Facial Palsy Synkinesis
Nerve regeneration occurs after an episode of facial nerve palsy. If there is aberrant muscle reinnervation, synkinesis between the lower and upper facial muscles following facial palsy may occur. This generally tends to occur six months after an episode of Bell palsy.[66] It does not tend to present with spontaneous spasms.[67] The prevalence of synkinesis following an episode of facial nerve palsy ranges from 8.9 to 51 percent.[68] Furthermore, postparalytic synkinesis has been shown to be associated with infectious and idiopathic causes of facial nerve palsy and patients who have had partial paralysis without the need for reanimation surgery.[68]
A patient suffering from post-facial palsy synkinesis will present with abnormal involuntary facial movements. There are different patterns of synkinesis, such as oral-ocular, which presents with non-intended eye closure on voluntary movement of the muscles of the mouth. Ocular-oral synkinesis occurs when voluntary eye movements lead to non-intended movements of the muscles of the mouth.[69]
Psychogenic facial spasm:
Studies have shown that the mean age of psychogenic spasms is 34.6 years of age.[70]
A patient suffering from psychogenic facial spasm will present with a non-patterned, variable facial spasm.[66] The facial spasm is distractable such that asking the patient to perform a task will lead to a reduction in symptoms. It also tends to be bilateral and asynchronous. It is worsened by anxiety and stress.[63]
Patients suffering from psychogenic facial spasms may be differentiated from dystonic conditions by the presence of particular clinical signs. Psychogenic facial spasm tends to involve the muscles of the lower face (downward deviation of the angle of the mouth). Furthermore, the 'brow-lift sign' may be seen, which consists of the frontalis and orbicularis oculi muscles contracting, leading to a raising of the eyebrow during occlusion of the eye.
Facial Motor Tics
Facial motor tics tend to be short-lived, brief, and stereotyped in nature. The motor tic may resemble a normal coordinated voluntary movement. A patient with facial motor tics may have a sudden urge or premonitory feeling to perform the movement. As such, the patient may be able to suppress the movement with effort.[63]
These motor tics may be associated with motor tics of the limbs, along with other features of Tourettes syndrome.
Facial Myokymia
A myokymia is an undulating, rippling movement that affects individual muscle fascicles, which may affect the upper or lower eyelid. This is associated with sleep deprivation, excessive caffeine consumption, and excessive fatigue.[66] An EMG performed on a patient suffering from facial myokymia will show brief, sudden bursts of doublets, triplets, or multiples originating from repetitively firing motor unit potentials. Facial myokymia is usually self-limiting but may need botulinum toxin injections in some cases. In most patients, one injection tends to cure the condition.
Tardive Dyskinesia
Tardive dyskinesia presents with repetitive, involuntary movements around the orofacial region. There may be smacking of the lips, protraction of the tongue, or excessive blinking of the eyes. These movements tend to be irregular and asynchronous and involve muscles that are not innervated by the facial nerve, such as the masseter muscle or the external muscles of the tongue. Tardive dyskinesia is a potential complication of long-term neuroleptic use.[66]
Prognosis
The life expectancy of patients with isolated BEB appears not to be affected by the disease. However, it poses a tremendous impact on the quality of life. In a series of 238 patients with the diagnosis of BEB or Meige syndrome reported by Castelbuono and Miller, it was found that 27(11.3%) patients experienced spontaneous remission and good quality of life. However, this is not the case for the majority of patients who experience a constant fear of the disease worsening, reaching a point where they need increasing doses of botulinum toxin to achieve symptom improvement or worsen to a level where the response to the botulinum toxin is inadequate. There is also a sense of insecurity and embarrassment when performing social tasks, which, in turn, results in social withdrawal.[71][72][73]
Complications
Botulinum toxin injection can lead to a wide range of complications, such as ptosis, diplopia, photophobia, ecchymosis, epiphora, and blurred vision, among others. The likelihood of complications of botulinum toxin injection reduces with repeated injection episodes.[51]
The most frequently reported side effect is ptosis. Ptosis following botulinum toxin injection may occur due to the migration of the botulinum toxin into the levator palpebrae superioris muscle, leading to ptosis.[53] The incidence of ptosis following botulinum toxin injection to treat blepharospasm has been shown to vary between studies (5.88% to 20%), and it is thought that this variance exists due to differences in injection technique and the amount of botulinum toxin injected. Methods that have been shown to reduce the likelihood of ptosis following botulinum toxin injection include staying away from the eyelid center when injecting the toxin. A weakness of the orbicularis oculi muscle may result in lagophthalmos. This can result in dry eye and exposure keratitis.
As many patients with blepharospasm have a variable degree of apraxia of eyelid opening, most patients need injections into the pretarsal orbicularis oculi muscle. Therefore, we always remind patients to apply lubricating ointment into the eyes for the first two to three weeks after injections and to increase the use of topical lubrication to the eyes for that period. Botulinum toxin injection has been shown to cause reduced lacrimal drainage as a result of a lower blink rate and laxity of the lower eyelid due to paralysis of the lacrimal portion of the orbicularis oculi muscle.[55]
Enhancing Healthcare Team Outcomes
Interprofessional management of patients with BEB is recommended, Neurology consultation is the basis of treatment for the disease, particularly a movement disorder specialist. Ophthalmology consultation should be considered early in the disease, in order to address underlying eye disease, psychiatry consultation is also part of the evaluation of these patients, once diagnosed patients should be evaluated by a psychiatrist to rule out depression and anxiety disorders, and follow up visits must continue.[72][41] [Level 5]
Media
(Click Image to Enlarge)
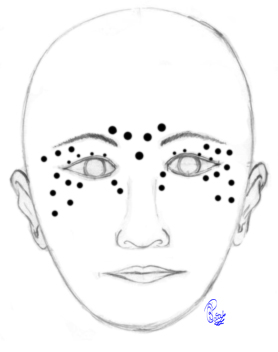
Botulinum Toxin Injections for Blepharospasm. Botulinum toxin injections may be administered at various sites, as depicted in the image. The dosage varies depending on the severity of the blepharospasm and apraxia of eyelid opening at each specific point. Pretarsal injections are administered to specifically counteract apraxia of eyelid opening. Injections just lateral to the lateral nasal wall aim to alleviate the squeezing of the nasalis muscle, which is observed in some patients. Injections into the corrugator and procerus muscles reduce the downward movement of the brow, consequently aiding eyelid control. Injections just below the brows provide a chemical lift to the brows, thereby improving the ability to open the eyelids. Caution is warranted to inject a minimal amount over the zygomaticus major and minor muscles to prevent the appearance of lower facial weakness following injections.
Contributed by BCK Patel, MD, FRCS
<p>Contributed by BCK Patel, MD, FRCS</p>
(Click Image to Enlarge)
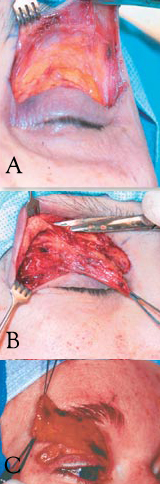
Limited Myectomy for Blepharospasm. The upper eyelid orbital and preseptal orbicularis muscle are dissected from the skin through an eyelid crease incision. The degree to which the pretarsal muscle is removed is determined by the extent of apraxia of eyelid opening (A). Care is exercised to safeguard the skin through meticulous dissection (B). The image illustrates en bloc removal of the orbicularis muscle (C).
Contributed by BCK Patel, MD, FRCS
References
Albanese A, Bhatia K, Bressman SB, Delong MR, Fahn S, Fung VS, Hallett M, Jankovic J, Jinnah HA, Klein C, Lang AE, Mink JW, Teller JK. Phenomenology and classification of dystonia: a consensus update. Movement disorders : official journal of the Movement Disorder Society. 2013 Jun 15:28(7):863-73. doi: 10.1002/mds.25475. Epub 2013 May 6 [PubMed PMID: 23649720]
Level 3 (low-level) evidenceJinnah HA, Factor SA. Diagnosis and treatment of dystonia. Neurologic clinics. 2015 Feb:33(1):77-100. doi: 10.1016/j.ncl.2014.09.002. Epub [PubMed PMID: 25432724]
Gautam P, Bhatia MS, Kaur J, Rathi A. Meige's syndrome. Industrial psychiatry journal. 2016 Jul-Dec:25(2):232-233. doi: 10.4103/0972-6748.207853. Epub [PubMed PMID: 28659707]
Dong H, Luo Y, Fan S, Yin B, Weng C, Peng B. Screening Gene Mutations in Chinese Patients With Benign Essential Blepharospasm. Frontiers in neurology. 2019:10():1387. doi: 10.3389/fneur.2019.01387. Epub 2020 Jan 23 [PubMed PMID: 32038460]
Hallett M, Evinger C, Jankovic J, Stacy M, BEBRF International Workshop. Update on blepharospasm: report from the BEBRF International Workshop. Neurology. 2008 Oct 14:71(16):1275-82. doi: 10.1212/01.wnl.0000327601.46315.85. Epub [PubMed PMID: 18852443]
Level 3 (low-level) evidenceDefazio G, Hallett M, Jinnah HA, Conte A, Berardelli A. Blepharospasm 40 years later. Movement disorders : official journal of the Movement Disorder Society. 2017 Apr:32(4):498-509. doi: 10.1002/mds.26934. Epub 2017 Feb 10 [PubMed PMID: 28186662]
Bali J, Navin N, Thakur BR. Computer vision syndrome: a study of the knowledge, attitudes and practices in Indian ophthalmologists. Indian journal of ophthalmology. 2007 Jul-Aug:55(4):289-94 [PubMed PMID: 17595478]
Level 2 (mid-level) evidenceSun Y, Tsai PJ, Chu CL, Huang WC, Bee YS. Epidemiology of benign essential blepharospasm: A nationwide population-based retrospective study in Taiwan. PloS one. 2018:13(12):e0209558. doi: 10.1371/journal.pone.0209558. Epub 2018 Dec 26 [PubMed PMID: 30586395]
Level 2 (mid-level) evidenceKuyper DJ, Parra V, Aerts S, Okun MS, Kluger BM. Nonmotor manifestations of dystonia: a systematic review. Movement disorders : official journal of the Movement Disorder Society. 2011 Jun:26(7):1206-17. doi: 10.1002/mds.23709. Epub 2011 Apr 11 [PubMed PMID: 21484874]
Level 1 (high-level) evidenceConte A, Berardelli I, Ferrazzano G, Pasquini M, Berardelli A, Fabbrini G. Non-motor symptoms in patients with adult-onset focal dystonia: Sensory and psychiatric disturbances. Parkinsonism & related disorders. 2016 Jan:22 Suppl 1():S111-4. doi: 10.1016/j.parkreldis.2015.09.001. Epub 2015 Sep 3 [PubMed PMID: 26360238]
Defazio G, Berardelli A, Abbruzzese G, Lepore V, Coviello V, Acquistapace D, Capus L, Carella F, De Berardinis MT, Galardi G, Girlanda P, Maurri S, Albanese A, Bertolasi L, Liguori R, Rossi A, Santoro L, Tognoni G, Livrea P. Possible risk factors for primary adult onset dystonia: a case-control investigation by the Italian Movement Disorders Study Group. Journal of neurology, neurosurgery, and psychiatry. 1998 Jan:64(1):25-32 [PubMed PMID: 9436723]
Level 2 (mid-level) evidenceHammer M, Abravanel A, Peckham E, Mahloogi A, Majounie E, Hallett M, Singleton A. Blepharospasm: A genetic screening study in 132 patients. Parkinsonism & related disorders. 2019 Jul:64():315-318. doi: 10.1016/j.parkreldis.2019.04.003. Epub 2019 Apr 2 [PubMed PMID: 30956059]
Xiao J, Uitti RJ, Zhao Y, Vemula SR, Perlmutter JS, Wszolek ZK, Maraganore DM, Auburger G, Leube B, Lehnhoff K, LeDoux MS. Mutations in CIZ1 cause adult onset primary cervical dystonia. Annals of neurology. 2012 Apr:71(4):458-69. doi: 10.1002/ana.23547. Epub 2012 Mar 23 [PubMed PMID: 22447717]
Misbahuddin A, Placzek MR, Chaudhuri KR, Wood NW, Bhatia KP, Warner TT. A polymorphism in the dopamine receptor DRD5 is associated with blepharospasm. Neurology. 2002 Jan 8:58(1):124-6 [PubMed PMID: 11781417]
Corvol JC, Studler JM, Schonn JS, Girault JA, Hervé D. Galpha(olf) is necessary for coupling D1 and A2a receptors to adenylyl cyclase in the striatum. Journal of neurochemistry. 2001 Mar:76(5):1585-8 [PubMed PMID: 11238742]
Level 3 (low-level) evidenceFuchs T, Saunders-Pullman R, Masuho I, Luciano MS, Raymond D, Factor S, Lang AE, Liang TW, Trosch RM, White S, Ainehsazan E, Hervé D, Sharma N, Ehrlich ME, Martemyanov KA, Bressman SB, Ozelius LJ. Mutations in GNAL cause primary torsion dystonia. Nature genetics. 2013 Jan:45(1):88-92. doi: 10.1038/ng.2496. Epub 2012 Dec 9 [PubMed PMID: 23222958]
Mitsui K, Matsumoto A, Ohtsuka S, Ohtsubo M, Yoshimura A. Cloning and characterization of a novel p21(Cip1/Waf1)-interacting zinc finger protein, ciz1. Biochemical and biophysical research communications. 1999 Oct 22:264(2):457-64 [PubMed PMID: 10529385]
Konakova M, Huynh DP, Yong W, Pulst SM. Cellular distribution of torsin A and torsin B in normal human brain. Archives of neurology. 2001 Jun:58(6):921-7 [PubMed PMID: 11405807]
Level 3 (low-level) evidenceAugood SJ, Penney JB Jr, Friberg IK, Breakefield XO, Young AB, Ozelius LJ, Standaert DG. Expression of the early-onset torsion dystonia gene (DYT1) in human brain. Annals of neurology. 1998 May:43(5):669-73 [PubMed PMID: 9585364]
Hewett J, Gonzalez-Agosti C, Slater D, Ziefer P, Li S, Bergeron D, Jacoby DJ, Ozelius LJ, Ramesh V, Breakefield XO. Mutant torsinA, responsible for early-onset torsion dystonia, forms membrane inclusions in cultured neural cells. Human molecular genetics. 2000 May 22:9(9):1403-13 [PubMed PMID: 10814722]
Level 3 (low-level) evidenceOzelius LJ, Hewett JW, Page CE, Bressman SB, Kramer PL, Shalish C, de Leon D, Brin MF, Raymond D, Corey DP, Fahn S, Risch NJ, Buckler AJ, Gusella JF, Breakefield XO. The early-onset torsion dystonia gene (DYT1) encodes an ATP-binding protein. Nature genetics. 1997 Sep:17(1):40-8 [PubMed PMID: 9288096]
Level 3 (low-level) evidenceGrandy DK, Zhang YA, Bouvier C, Zhou QY, Johnson RA, Allen L, Buck K, Bunzow JR, Salon J, Civelli O. Multiple human D5 dopamine receptor genes: a functional receptor and two pseudogenes. Proceedings of the National Academy of Sciences of the United States of America. 1991 Oct 15:88(20):9175-9 [PubMed PMID: 1833775]
Level 3 (low-level) evidencePolymeropoulos MH, Xiao H, Merril CR. The human D5 dopamine receptor (DRD5) maps on chromosome 4. Genomics. 1991 Nov:11(3):777-8 [PubMed PMID: 1774076]
Level 3 (low-level) evidenceSteeves TD, Day L, Dykeman J, Jette N, Pringsheim T. The prevalence of primary dystonia: a systematic review and meta-analysis. Movement disorders : official journal of the Movement Disorder Society. 2012 Dec:27(14):1789-96. doi: 10.1002/mds.25244. Epub 2012 Oct 31 [PubMed PMID: 23114997]
Level 1 (high-level) evidenceMartino D, Livrea P, Giorelli M, Masi G, Aniello MS, Defazio G. Menopause and menarche in patients with primary blepharospasm: an exploratory case-control study. European neurology. 2002:47(3):161-4 [PubMed PMID: 11914554]
Level 2 (mid-level) evidenceDefazio G, Livrea P. Epidemiology of primary blepharospasm. Movement disorders : official journal of the Movement Disorder Society. 2002 Jan:17(1):7-12 [PubMed PMID: 11835433]
Snir M, Weinberger D, Bourla D, Kristal-Shalit O, Dotan G, Axer-Siegel R. Quantitative changes in botulinum toxin a treatment over time in patients with essential blepharospasm and idiopathic hemifacial spasm. American journal of ophthalmology. 2003 Jul:136(1):99-105 [PubMed PMID: 12834676]
Level 2 (mid-level) evidenceValls-Sole J, Defazio G. Blepharospasm: Update on Epidemiology, Clinical Aspects, and Pathophysiology. Frontiers in neurology. 2016:7():45. doi: 10.3389/fneur.2016.00045. Epub 2016 Mar 31 [PubMed PMID: 27064462]
Evinger C. Benign Essential Blepharospasm is a Disorder of Neuroplasticity: Lessons From Animal Models. Journal of neuro-ophthalmology : the official journal of the North American Neuro-Ophthalmology Society. 2015 Dec:35(4):374-9. doi: 10.1097/WNO.0000000000000317. Epub [PubMed PMID: 26576017]
Level 3 (low-level) evidenceLee JM, Baek JS, Choi HS, Kim SJ, Jang JW. Clinical Features of Benign Essential Blepharospasm in Korean Patients. Korean journal of ophthalmology : KJO. 2018 Oct:32(5):339-343. doi: 10.3341/kjo.2018.0038. Epub [PubMed PMID: 30311455]
Hallett M. Blepharospasm: recent advances. Neurology. 2002 Nov 12:59(9):1306-12 [PubMed PMID: 12434791]
Level 3 (low-level) evidenceJankovic J, Havins WE, Wilkins RB. Blinking and blepharospasm. Mechanism, diagnosis, and management. JAMA. 1982 Dec 17:248(23):3160-4 [PubMed PMID: 7143695]
Hwang CJ, Eftekhari K. Benign Essential Blepharospasm: What We Know and What We Don't. International ophthalmology clinics. 2018 Winter:58(1):11-24. doi: 10.1097/IIO.0000000000000207. Epub [PubMed PMID: 29239874]
Suzuki Y, Mizoguchi S, Kiyosawa M, Mochizuki M, Ishiwata K, Wakakura M, Ishii K. Glucose hypermetabolism in the thalamus of patients with essential blepharospasm. Journal of neurology. 2007 Jul:254(7):890-6 [PubMed PMID: 17325818]
Etgen T, Mühlau M, Gaser C, Sander D. Bilateral grey-matter increase in the putamen in primary blepharospasm. Journal of neurology, neurosurgery, and psychiatry. 2006 Sep:77(9):1017-20 [PubMed PMID: 16690695]
Level 2 (mid-level) evidenceBaker RS, Andersen AH, Morecraft RJ, Smith CD. A functional magnetic resonance imaging study in patients with benign essential blepharospasm. Journal of neuro-ophthalmology : the official journal of the North American Neuro-Ophthalmology Society. 2003 Mar:23(1):11-5 [PubMed PMID: 12616082]
Level 2 (mid-level) evidenceHorie C, Suzuki Y, Kiyosawa M, Mochizuki M, Wakakura M, Oda K, Ishiwata K, Ishii K. Decreased dopamine D receptor binding in essential blepharospasm. Acta neurologica Scandinavica. 2009 Jan:119(1):49-54. doi: 10.1111/j.1600-0404.2008.01053.x. Epub 2008 Jun 5 [PubMed PMID: 18540899]
Jinnah HA, Berardelli A, Comella C, Defazio G, Delong MR, Factor S, Galpern WR, Hallett M, Ludlow CL, Perlmutter JS, Rosen AR, Dystonia Coalition Investigators. The focal dystonias: current views and challenges for future research. Movement disorders : official journal of the Movement Disorder Society. 2013 Jun 15:28(7):926-43. doi: 10.1002/mds.25567. Epub [PubMed PMID: 23893450]
Wakakura M, Yamagami A, Iwasa M. Blepharospasm in Japan: A Clinical Observational Study From a Large Referral Hospital in Tokyo. Neuro-ophthalmology (Aeolus Press). 2018 Oct:42(5):275-283. doi: 10.1080/01658107.2017.1409770. Epub 2018 Jan 9 [PubMed PMID: 30258472]
Level 2 (mid-level) evidenceKrack P, Marion MH. "Apraxia of lid opening," a focal eyelid dystonia: clinical study of 32 patients. Movement disorders : official journal of the Movement Disorder Society. 1994 Nov:9(6):610-5 [PubMed PMID: 7845400]
Elston JS, Marsden CD, Grandas F, Quinn NP. The significance of ophthalmological symptoms in idiopathic blepharospasm. Eye (London, England). 1988:2 ( Pt 4)():435-9 [PubMed PMID: 3253136]
Defazio G, Hallett M, Jinnah HA, Stebbins GT, Gigante AF, Ferrazzano G, Conte A, Fabbrini G, Berardelli A. Development and validation of a clinical scale for rating the severity of blepharospasm. Movement disorders : official journal of the Movement Disorder Society. 2015 Apr:30(4):525-30. doi: 10.1002/mds.26156. Epub [PubMed PMID: 25847472]
Level 1 (high-level) evidenceDefazio G, Hallett M, Jinnah HA, Berardelli A. Development and validation of a clinical guideline for diagnosing blepharospasm. Neurology. 2013 Jul 16:81(3):236-40. doi: 10.1212/WNL.0b013e31829bfdf6. Epub 2013 Jun 14 [PubMed PMID: 23771487]
Level 1 (high-level) evidenceDefazio G, Conte A, Gigante AF, Fabbrini G, Berardelli A. Is tremor in dystonia a phenotypic feature of dystonia? Neurology. 2015 Mar 10:84(10):1053-9. doi: 10.1212/WNL.0000000000001341. Epub 2015 Feb 6 [PubMed PMID: 25663232]
Martino D, Defazio G, Alessio G, Abbruzzese G, Girlanda P, Tinazzi M, Fabbrini G, Marinelli L, Majorana G, Buccafusca M, Vacca L, Livrea P, Berardelli A. Relationship between eye symptoms and blepharospasm: a multicenter case-control study. Movement disorders : official journal of the Movement Disorder Society. 2005 Dec:20(12):1564-70 [PubMed PMID: 16092106]
Level 2 (mid-level) evidenceJankovic J, Orman J. Botulinum A toxin for cranial-cervical dystonia: a double-blind, placebo-controlled study. Neurology. 1987 Apr:37(4):616-23 [PubMed PMID: 3561773]
Level 1 (high-level) evidenceKimura J. Disorder of interneurons in Parkinsonism. The orbicularis oculi reflex to paired stimuli. Brain : a journal of neurology. 1973:96(1):87-96 [PubMed PMID: 4695726]
Yen MT. Developments in the treatment of benign essential blepharospasm. Current opinion in ophthalmology. 2018 Sep:29(5):440-444. doi: 10.1097/ICU.0000000000000500. Epub [PubMed PMID: 29916840]
Level 3 (low-level) evidenceHerz NL, Yen MT. Modulation of sensory photophobia in essential blepharospasm with chromatic lenses. Ophthalmology. 2005 Dec:112(12):2208-11 [PubMed PMID: 16242188]
Level 2 (mid-level) evidenceBlackburn MK, Lamb RD, Digre KB, Smith AG, Warner JE, McClane RW, Nandedkar SD, Langeberg WJ, Holubkov R, Katz BJ. FL-41 tint improves blink frequency, light sensitivity, and functional limitations in patients with benign essential blepharospasm. Ophthalmology. 2009 May:116(5):997-1001. doi: 10.1016/j.ophtha.2008.12.031. Epub [PubMed PMID: 19410958]
Level 2 (mid-level) evidenceAbabneh OH, Cetinkaya A, Kulwin DR. Long-term efficacy and safety of botulinum toxin A injections to treat blepharospasm and hemifacial spasm. Clinical & experimental ophthalmology. 2014 Apr:42(3):254-61. doi: 10.1111/ceo.12165. Epub 2013 Aug 4 [PubMed PMID: 23844601]
Level 2 (mid-level) evidenceHellman A, Torres-Russotto D. Botulinum toxin in the management of blepharospasm: current evidence and recent developments. Therapeutic advances in neurological disorders. 2015 Mar:8(2):82-91. doi: 10.1177/1756285614557475. Epub [PubMed PMID: 25922620]
Level 3 (low-level) evidenceAnwar MS, Zafar H. Efficacy of botulinum toxin in benign essential Blepharospasm: Desirable & undesirable effects. Pakistan journal of medical sciences. 2013 Nov:29(6):1389-93 [PubMed PMID: 24550960]
Ramirez-Castaneda J, Jankovic J. Long-term efficacy and safety of botulinum toxin injections in dystonia. Toxins. 2013 Feb 4:5(2):249-66. doi: 10.3390/toxins5020249. Epub 2013 Feb 4 [PubMed PMID: 23381141]
Yang H, Lu J, Zhao X, Ding X, Wang Z, Cai X, Luo Y, Lu L. Comparison of Two Botulinum Neurotoxin A Injection Patterns with or without the Medial Lower Eyelid in the Treatment of Blepharospasm. Journal of ophthalmology. 2016:2016():5957812. doi: 10.1155/2016/5957812. Epub 2016 Jan 14 [PubMed PMID: 26885381]
Scott AB, Kennedy RA, Stubbs HA. Botulinum A toxin injection as a treatment for blepharospasm. Archives of ophthalmology (Chicago, Ill. : 1960). 1985 Mar:103(3):347-50 [PubMed PMID: 3977705]
Naumann M, Albanese A, Heinen F, Molenaers G, Relja M. Safety and efficacy of botulinum toxin type A following long-term use. European journal of neurology. 2006 Dec:13 Suppl 4():35-40 [PubMed PMID: 17112348]
Level 3 (low-level) evidenceHassell TJW, Charles D. Treatment of Blepharospasm and Oromandibular Dystonia with Botulinum Toxins. Toxins. 2020 Apr 22:12(4):. doi: 10.3390/toxins12040269. Epub 2020 Apr 22 [PubMed PMID: 32331272]
Anderson RL, Patel BC, Holds JB, Jordan DR. Blepharospasm: past, present, and future. Ophthalmic plastic and reconstructive surgery. 1998 Sep:14(5):305-17 [PubMed PMID: 9783280]
Level 2 (mid-level) evidenceGillum WN, Anderson RL. Blepharospasm surgery. An anatomical approach. Archives of ophthalmology (Chicago, Ill. : 1960). 1981 Jun:99(6):1056-62 [PubMed PMID: 7236103]
Kerty E, Eidal K. Apraxia of eyelid opening: clinical features and therapy. European journal of ophthalmology. 2006 Mar-Apr:16(2):204-8 [PubMed PMID: 16703535]
Rosenstengel C, Matthes M, Baldauf J, Fleck S, Schroeder H. Hemifacial spasm: conservative and surgical treatment options. Deutsches Arzteblatt international. 2012 Oct:109(41):667-73. doi: 10.3238/arztebl.2012.0667. Epub 2012 Oct 12 [PubMed PMID: 23264807]
Tan NC, Chan LL, Tan EK. Hemifacial spasm and involuntary facial movements. QJM : monthly journal of the Association of Physicians. 2002 Aug:95(8):493-500 [PubMed PMID: 12145388]
Wang A, Jankovic J. Hemifacial spasm: clinical findings and treatment. Muscle & nerve. 1998 Dec:21(12):1740-7 [PubMed PMID: 9843077]
Chang WS, Kim BS, Lee JE, Jung HH, Kim K, Kwon HC, Lee YH, Chang JW. 'Is tinnitus accompanied by hemifacial spasm in normal-hearing patients also a type of hyperactive neurovascular compression syndrome? : A magnetoencephalography study. BMC neurology. 2013 May 8:13():42. doi: 10.1186/1471-2377-13-42. Epub 2013 May 8 [PubMed PMID: 23651913]
Lefaucheur JP, Ben Daamer N, Sangla S, Le Guerinel C. Diagnosis of primary hemifacial spasm. Neuro-Chirurgie. 2018 May:64(2):82-86. doi: 10.1016/j.neuchi.2017.12.003. Epub 2018 Apr 16 [PubMed PMID: 29673578]
Oge AE, Yayla V, Demir GA, Eraksoy M. Excitability of facial nucleus and related brain-stem reflexes in hemifacial spasm, post-facial palsy synkinesis and facial myokymia. Clinical neurophysiology : official journal of the International Federation of Clinical Neurophysiology. 2005 Jul:116(7):1542-54 [PubMed PMID: 15953558]
Salles AG, da Costa EF, Ferreira MC, do Nascimento Remigio AF, Moraes LB, Gemperli R. Epidemiologic Overview of Synkinesis in 353 Patients with Longstanding Facial Paralysis under Treatment with Botulinum Toxin for 11 Years. Plastic and reconstructive surgery. 2015 Dec:136(6):1289-1298. doi: 10.1097/PRS.0000000000001802. Epub [PubMed PMID: 26595022]
Level 3 (low-level) evidencePourmomeny AA, Asadi S. Management of synkinesis and asymmetry in facial nerve palsy: a review article. Iranian journal of otorhinolaryngology. 2014 Oct:26(77):251-6 [PubMed PMID: 25320703]
Tan EK, Jankovic J. Psychogenic hemifacial spasm. The Journal of neuropsychiatry and clinical neurosciences. 2001 Summer:13(3):380-4 [PubMed PMID: 11514645]
Level 3 (low-level) evidenceTucha O, Naumann M, Berg D, Alders GL, Lange KW. Quality of life in patients with blepharospasm. Acta neurologica Scandinavica. 2001 Jan:103(1):49-52 [PubMed PMID: 11153888]
Level 2 (mid-level) evidenceHall TA, McGwin G Jr, Searcey K, Xie A, Hupp SL, Owsley C, Kline LB. Health-related quality of life and psychosocial characteristics of patients with benign essential blepharospasm. Archives of ophthalmology (Chicago, Ill. : 1960). 2006 Jan:124(1):116-9 [PubMed PMID: 16401794]
Level 2 (mid-level) evidenceCastelbuono A, Miller NR. Spontaneous remission in patients with essential blepharospasm and Meige syndrome. American journal of ophthalmology. 1998 Sep:126(3):432-5 [PubMed PMID: 9744377]
Level 2 (mid-level) evidence
