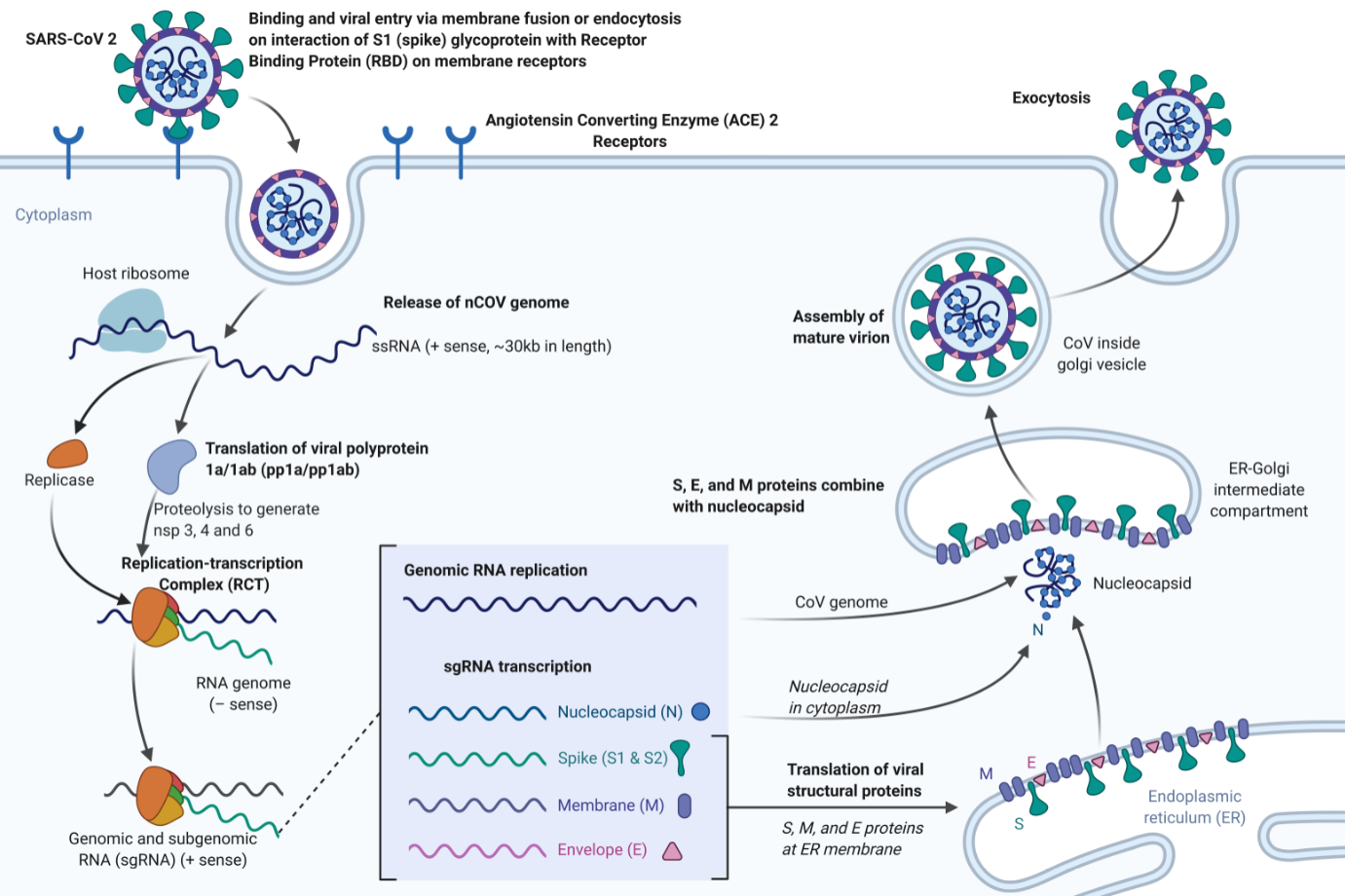
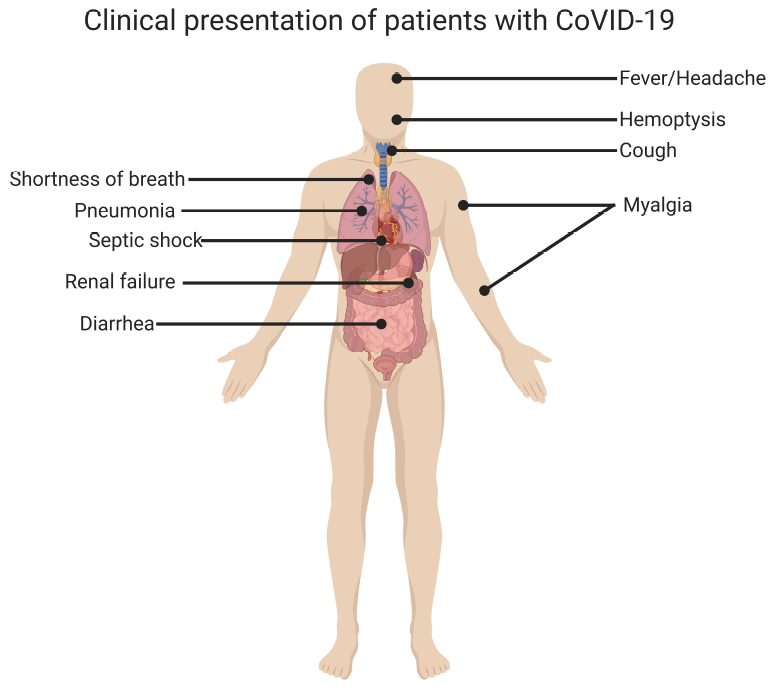
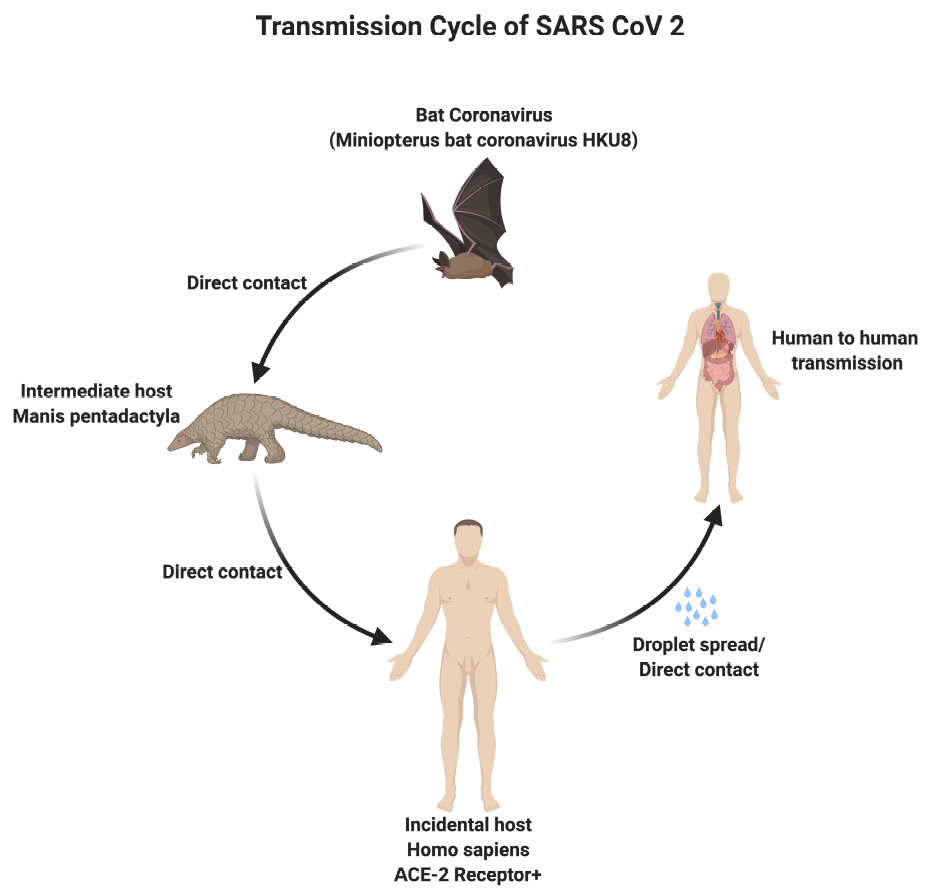
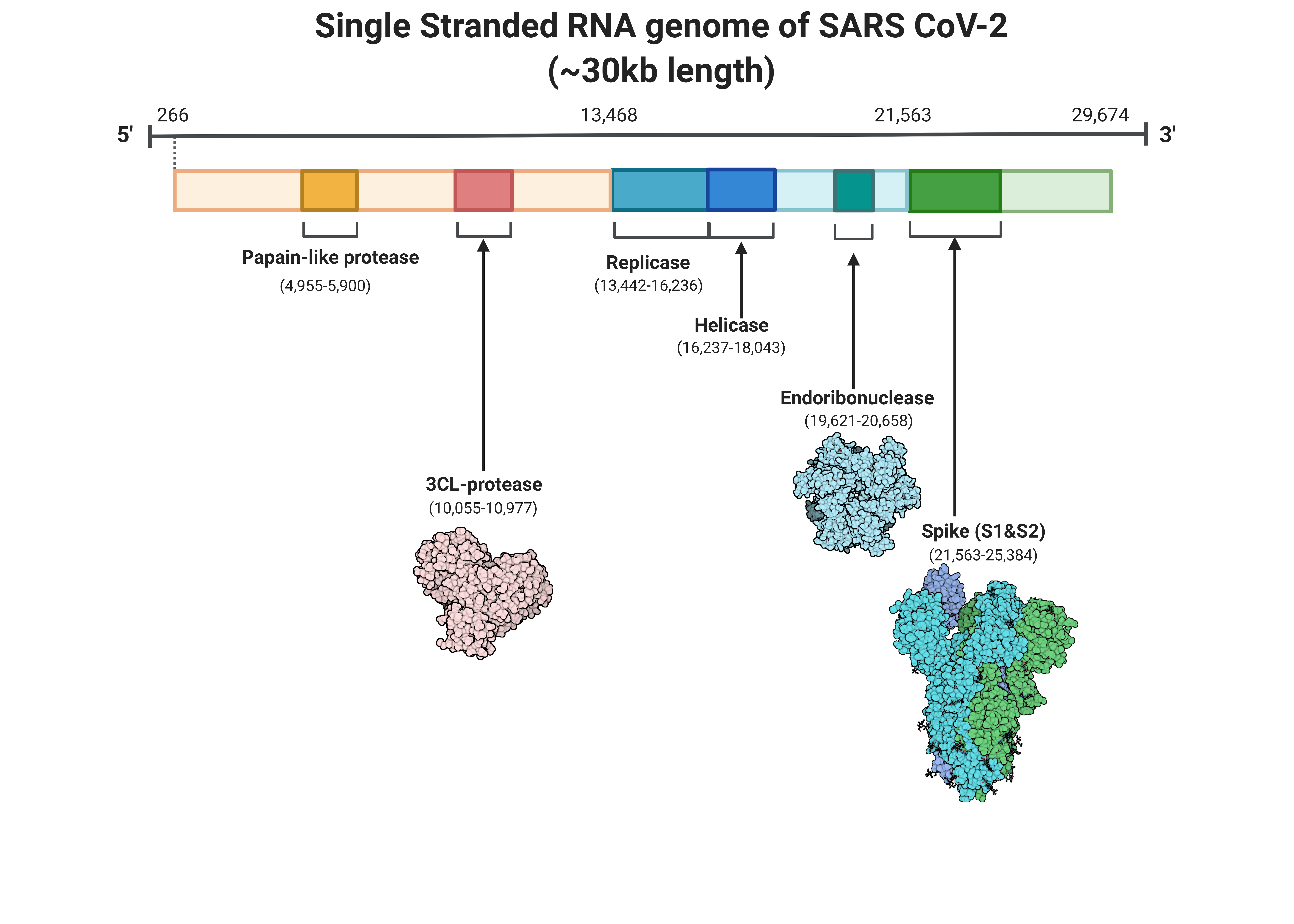
[1]
Sharma A, Ahmad Farouk I, Lal SK. COVID-19: A Review on the Novel Coronavirus Disease Evolution, Transmission, Detection, Control and Prevention. Viruses. 2021 Jan 29:13(2):. doi: 10.3390/v13020202. Epub 2021 Jan 29
[PubMed PMID: 33572857]
[2]
Raman R, Patel KJ, Ranjan K. COVID-19: Unmasking Emerging SARS-CoV-2 Variants, Vaccines and Therapeutic Strategies. Biomolecules. 2021 Jul 6:11(7):. doi: 10.3390/biom11070993. Epub 2021 Jul 6
[PubMed PMID: 34356617]
[3]
Chenchula S, Karunakaran P, Sharma S, Chavan M. Current evidence on efficacy of COVID-19 booster dose vaccination against the Omicron variant: A systematic review. Journal of medical virology. 2022 Jul:94(7):2969-2976. doi: 10.1002/jmv.27697. Epub 2022 Mar 14
[PubMed PMID: 35246846]
Level 1 (high-level) evidence
[4]
Chan JF, To KK, Tse H, Jin DY, Yuen KY. Interspecies transmission and emergence of novel viruses: lessons from bats and birds. Trends in microbiology. 2013 Oct:21(10):544-55. doi: 10.1016/j.tim.2013.05.005. Epub 2013 Jun 14
[PubMed PMID: 23770275]
[5]
Lei J, Kusov Y, Hilgenfeld R. Nsp3 of coronaviruses: Structures and functions of a large multi-domain protein. Antiviral research. 2018 Jan:149():58-74. doi: 10.1016/j.antiviral.2017.11.001. Epub 2017 Nov 8
[PubMed PMID: 29128390]
[6]
Chan JF, Kok KH, Zhu Z, Chu H, To KK, Yuan S, Yuen KY. Genomic characterization of the 2019 novel human-pathogenic coronavirus isolated from a patient with atypical pneumonia after visiting Wuhan. Emerging microbes & infections. 2020:9(1):221-236. doi: 10.1080/22221751.2020.1719902. Epub 2020 Jan 28
[PubMed PMID: 31987001]
[7]
Biryukov J, Boydston JA, Dunning RA, Yeager JJ, Wood S, Ferris A, Miller D, Weaver W, Zeitouni NE, Freeburger D, Dabisch P, Wahl V, Hevey MC, Altamura LA. SARS-CoV-2 is rapidly inactivated at high temperature. Environmental chemistry letters. 2021:19(2):1773-1777. doi: 10.1007/s10311-021-01187-x. Epub 2021 Feb 3
[PubMed PMID: 33551702]
Level 3 (low-level) evidence
[8]
Andersen KG, Rambaut A, Lipkin WI, Holmes EC, Garry RF. The proximal origin of SARS-CoV-2. Nature medicine. 2020 Apr:26(4):450-452. doi: 10.1038/s41591-020-0820-9. Epub
[PubMed PMID: 32284615]
[9]
Zhang T, Wu Q, Zhang Z. Probable Pangolin Origin of SARS-CoV-2 Associated with the COVID-19 Outbreak. Current biology : CB. 2020 Apr 6:30(7):1346-1351.e2. doi: 10.1016/j.cub.2020.03.022. Epub 2020 Mar 19
[PubMed PMID: 32197085]
[10]
Oreshkova N, Molenaar RJ, Vreman S, Harders F, Oude Munnink BB, Hakze-van der Honing RW, Gerhards N, Tolsma P, Bouwstra R, Sikkema RS, Tacken MG, de Rooij MM, Weesendorp E, Engelsma MY, Bruschke CJ, Smit LA, Koopmans M, van der Poel WH, Stegeman A. SARS-CoV-2 infection in farmed minks, the Netherlands, April and May 2020. Euro surveillance : bulletin Europeen sur les maladies transmissibles = European communicable disease bulletin. 2020 Jun:25(23):. doi: 10.2807/1560-7917.ES.2020.25.23.2001005. Epub
[PubMed PMID: 32553059]
[11]
Korber B, Fischer WM, Gnanakaran S, Yoon H, Theiler J, Abfalterer W, Hengartner N, Giorgi EE, Bhattacharya T, Foley B, Hastie KM, Parker MD, Partridge DG, Evans CM, Freeman TM, de Silva TI, Sheffield COVID-19 Genomics Group, McDanal C, Perez LG, Tang H, Moon-Walker A, Whelan SP, LaBranche CC, Saphire EO, Montefiori DC. Tracking Changes in SARS-CoV-2 Spike: Evidence that D614G Increases Infectivity of the COVID-19 Virus. Cell. 2020 Aug 20:182(4):812-827.e19. doi: 10.1016/j.cell.2020.06.043. Epub 2020 Jul 3
[PubMed PMID: 32697968]
[12]
Galloway SE, Paul P, MacCannell DR, Johansson MA, Brooks JT, MacNeil A, Slayton RB, Tong S, Silk BJ, Armstrong GL, Biggerstaff M, Dugan VG. Emergence of SARS-CoV-2 B.1.1.7 Lineage - United States, December 29, 2020-January 12, 2021. MMWR. Morbidity and mortality weekly report. 2021 Jan 22:70(3):95-99. doi: 10.15585/mmwr.mm7003e2. Epub 2021 Jan 22
[PubMed PMID: 33476315]
[13]
Volz E, Mishra S, Chand M, Barrett JC, Johnson R, Geidelberg L, Hinsley WR, Laydon DJ, Dabrera G, O'Toole Á, Amato R, Ragonnet-Cronin M, Harrison I, Jackson B, Ariani CV, Boyd O, Loman NJ, McCrone JT, Gonçalves S, Jorgensen D, Myers R, Hill V, Jackson DK, Gaythorpe K, Groves N, Sillitoe J, Kwiatkowski DP, COVID-19 Genomics UK (COG-UK) consortium, Flaxman S, Ratmann O, Bhatt S, Hopkins S, Gandy A, Rambaut A, Ferguson NM. Assessing transmissibility of SARS-CoV-2 lineage B.1.1.7 in England. Nature. 2021 May:593(7858):266-269. doi: 10.1038/s41586-021-03470-x. Epub 2021 Mar 25
[PubMed PMID: 33767447]
[14]
Wu K, Werner AP, Moliva JI, Koch M, Choi A, Stewart-Jones GBE, Bennett H, Boyoglu-Barnum S, Shi W, Graham BS, Carfi A, Corbett KS, Seder RA, Edwards DK. mRNA-1273 vaccine induces neutralizing antibodies against spike mutants from global SARS-CoV-2 variants. bioRxiv : the preprint server for biology. 2021 Jan 25:():. pii: 2021.01.25.427948. doi: 10.1101/2021.01.25.427948. Epub 2021 Jan 25
[PubMed PMID: 33501442]
[15]
Davies NG, Abbott S, Barnard RC, Jarvis CI, Kucharski AJ, Munday JD, Pearson CAB, Russell TW, Tully DC, Washburne AD, Wenseleers T, Gimma A, Waites W, Wong KLM, van Zandvoort K, Silverman JD, CMMID COVID-19 Working Group, COVID-19 Genomics UK (COG-UK) Consortium, Diaz-Ordaz K, Keogh R, Eggo RM, Funk S, Jit M, Atkins KE, Edmunds WJ. Estimated transmissibility and impact of SARS-CoV-2 lineage B.1.1.7 in England. Science (New York, N.Y.). 2021 Apr 9:372(6538):. doi: 10.1126/science.abg3055. Epub 2021 Mar 3
[PubMed PMID: 33658326]
[16]
Walensky RP, Walke HT, Fauci AS. SARS-CoV-2 Variants of Concern in the United States-Challenges and Opportunities. JAMA. 2021 Mar 16:325(11):1037-1038. doi: 10.1001/jama.2021.2294. Epub
[PubMed PMID: 33595644]
[17]
Davies NG, Barnard RC, Jarvis CI, Russell TW, Semple MG, Jit M, Edmunds WJ, Centre for Mathematical Modelling of Infectious Diseases COVID-19 Working Group, ISARIC4C investigators. Association of tiered restrictions and a second lockdown with COVID-19 deaths and hospital admissions in England: a modelling study. The Lancet. Infectious diseases. 2021 Apr:21(4):482-492. doi: 10.1016/S1473-3099(20)30984-1. Epub 2020 Dec 24
[PubMed PMID: 33357518]
[18]
Challen R, Brooks-Pollock E, Read JM, Dyson L, Tsaneva-Atanasova K, Danon L. Risk of mortality in patients infected with SARS-CoV-2 variant of concern 202012/1: matched cohort study. BMJ (Clinical research ed.). 2021 Mar 9:372():n579. doi: 10.1136/bmj.n579. Epub 2021 Mar 9
[PubMed PMID: 33687922]
[19]
Davies NG, Jarvis CI, CMMID COVID-19 Working Group, Edmunds WJ, Jewell NP, Diaz-Ordaz K, Keogh RH. Increased mortality in community-tested cases of SARS-CoV-2 lineage B.1.1.7. Nature. 2021 May:593(7858):270-274. doi: 10.1038/s41586-021-03426-1. Epub 2021 Mar 15
[PubMed PMID: 33723411]
Level 3 (low-level) evidence
[20]
Grint DJ, Wing K, Williamson E, McDonald HI, Bhaskaran K, Evans D, Evans SJ, Walker AJ, Hickman G, Nightingale E, Schultze A, Rentsch CT, Bates C, Cockburn J, Curtis HJ, Morton CE, Bacon S, Davy S, Wong AY, Mehrkar A, Tomlinson L, Douglas IJ, Mathur R, Blomquist P, MacKenna B, Ingelsby P, Croker R, Parry J, Hester F, Harper S, DeVito NJ, Hulme W, Tazare J, Goldacre B, Smeeth L, Eggo RM. Case fatality risk of the SARS-CoV-2 variant of concern B.1.1.7 in England, 16 November to 5 February. Euro surveillance : bulletin Europeen sur les maladies transmissibles = European communicable disease bulletin. 2021 Mar:26(11):. doi: 10.2807/1560-7917.ES.2021.26.11.2100256. Epub
[PubMed PMID: 33739254]
Level 3 (low-level) evidence
[21]
Tegally H, Wilkinson E, Giovanetti M, Iranzadeh A, Fonseca V, Giandhari J, Doolabh D, Pillay S, San EJ, Msomi N, Mlisana K, von Gottberg A, Walaza S, Allam M, Ismail A, Mohale T, Glass AJ, Engelbrecht S, Van Zyl G, Preiser W, Petruccione F, Sigal A, Hardie D, Marais G, Hsiao NY, Korsman S, Davies MA, Tyers L, Mudau I, York D, Maslo C, Goedhals D, Abrahams S, Laguda-Akingba O, Alisoltani-Dehkordi A, Godzik A, Wibmer CK, Sewell BT, Lourenço J, Alcantara LCJ, Kosakovsky Pond SL, Weaver S, Martin D, Lessells RJ, Bhiman JN, Williamson C, de Oliveira T. Detection of a SARS-CoV-2 variant of concern in South Africa. Nature. 2021 Apr:592(7854):438-443. doi: 10.1038/s41586-021-03402-9. Epub 2021 Mar 9
[PubMed PMID: 33690265]
[22]
Wibmer CK, Ayres F, Hermanus T, Madzivhandila M, Kgagudi P, Oosthuysen B, Lambson BE, de Oliveira T, Vermeulen M, van der Berg K, Rossouw T, Boswell M, Ueckermann V, Meiring S, von Gottberg A, Cohen C, Morris L, Bhiman JN, Moore PL. SARS-CoV-2 501Y.V2 escapes neutralization by South African COVID-19 donor plasma. bioRxiv : the preprint server for biology. 2021 Mar 1:():. pii: 2021.01.18.427166. doi: 10.1101/2021.01.18.427166. Epub 2021 Mar 1
[PubMed PMID: 33501446]
[23]
Mwenda M, Saasa N, Sinyange N, Busby G, Chipimo PJ, Hendry J, Kapona O, Yingst S, Hines JZ, Minchella P, Simulundu E, Changula K, Nalubamba KS, Sawa H, Kajihara M, Yamagishi J, Kapin'a M, Kapata N, Fwoloshi S, Zulu P, Mulenga LB, Agolory S, Mukonka V, Bridges DJ. Detection of B.1.351 SARS-CoV-2 Variant Strain - Zambia, December 2020. MMWR. Morbidity and mortality weekly report. 2021 Feb 26:70(8):280-282. doi: 10.15585/mmwr.mm7008e2. Epub 2021 Feb 26
[PubMed PMID: 33630820]
[24]
Wang P, Casner RG, Nair MS, Wang M, Yu J, Cerutti G, Liu L, Kwong PD, Huang Y, Shapiro L, Ho DD. Increased Resistance of SARS-CoV-2 Variant P.1 to Antibody Neutralization. bioRxiv : the preprint server for biology. 2021 Apr 9:():. pii: 2021.03.01.433466. doi: 10.1101/2021.03.01.433466. Epub 2021 Apr 9
[PubMed PMID: 33688656]
[25]
Faria NR, Mellan TA, Whittaker C, Claro IM, Candido DDS, Mishra S, Crispim MAE, Sales FC, Hawryluk I, McCrone JT, Hulswit RJG, Franco LAM, Ramundo MS, de Jesus JG, Andrade PS, Coletti TM, Ferreira GM, Silva CAM, Manuli ER, Pereira RHM, Peixoto PS, Kraemer MU, Gaburo N Jr, Camilo CDC, Hoeltgebaum H, Souza WM, Rocha EC, de Souza LM, de Pinho MC, Araujo LJT, Malta FSV, de Lima AB, Silva JDP, Zauli DAG, de S Ferreira AC, Schnekenberg RP, Laydon DJ, Walker PGT, Schlüter HM, Dos Santos ALP, Vidal MS, Del Caro VS, Filho RMF, Dos Santos HM, Aguiar RS, Modena JLP, Nelson B, Hay JA, Monod M, Miscouridou X, Coupland H, Sonabend R, Vollmer M, Gandy A, Suchard MA, Bowden TA, Pond SLK, Wu CH, Ratmann O, Ferguson NM, Dye C, Loman NJ, Lemey P, Rambaut A, Fraiji NA, Carvalho MDPSS, Pybus OG, Flaxman S, Bhatt S, Sabino EC. Genomics and epidemiology of a novel SARS-CoV-2 lineage in Manaus, Brazil. medRxiv : the preprint server for health sciences. 2021 Mar 3:():. pii: 2021.02.26.21252554. doi: 10.1101/2021.02.26.21252554. Epub 2021 Mar 3
[PubMed PMID: 33688664]
[26]
Vaughan A. Omicron emerges. New scientist (1971). 2021 Dec 4:252(3363):7. doi: 10.1016/S0262-4079(21)02140-0. Epub 2021 Dec 3
[PubMed PMID: 34876769]
[27]
Callaway E. Heavily mutated Omicron variant puts scientists on alert. Nature. 2021 Dec:600(7887):21. doi: 10.1038/d41586-021-03552-w. Epub
[PubMed PMID: 34824381]
[28]
Gu H, Krishnan P, Ng DYM, Chang LDJ, Liu GYZ, Cheng SSM, Hui MMY, Fan MCY, Wan JHL, Lau LHK, Cowling BJ, Peiris M, Poon LLM. Probable Transmission of SARS-CoV-2 Omicron Variant in Quarantine Hotel, Hong Kong, China, November 2021. Emerging infectious diseases. 2022 Feb:28(2):460-462. doi: 10.3201/eid2802.212422. Epub 2021 Dec 3
[PubMed PMID: 34860154]
[29]
van Doremalen N, Bushmaker T, Morris DH, Holbrook MG, Gamble A, Williamson BN, Tamin A, Harcourt JL, Thornburg NJ, Gerber SI, Lloyd-Smith JO, de Wit E, Munster VJ. Aerosol and Surface Stability of SARS-CoV-2 as Compared with SARS-CoV-1. The New England journal of medicine. 2020 Apr 16:382(16):1564-1567. doi: 10.1056/NEJMc2004973. Epub 2020 Mar 17
[PubMed PMID: 32182409]
[30]
Riddell S, Goldie S, Hill A, Eagles D, Drew TW. The effect of temperature on persistence of SARS-CoV-2 on common surfaces. Virology journal. 2020 Oct 7:17(1):145. doi: 10.1186/s12985-020-01418-7. Epub 2020 Oct 7
[PubMed PMID: 33028356]
[31]
Guo ZD, Wang ZY, Zhang SF, Li X, Li L, Li C, Cui Y, Fu RB, Dong YZ, Chi XY, Zhang MY, Liu K, Cao C, Liu B, Zhang K, Gao YW, Lu B, Chen W. Aerosol and Surface Distribution of Severe Acute Respiratory Syndrome Coronavirus 2 in Hospital Wards, Wuhan, China, 2020. Emerging infectious diseases. 2020 Jul:26(7):1583-1591. doi: 10.3201/eid2607.200885. Epub 2020 Jun 21
[PubMed PMID: 32275497]
[32]
Yeo C, Kaushal S, Yeo D. Enteric involvement of coronaviruses: is faecal-oral transmission of SARS-CoV-2 possible? The lancet. Gastroenterology & hepatology. 2020 Apr:5(4):335-337. doi: 10.1016/S2468-1253(20)30048-0. Epub 2020 Feb 20
[PubMed PMID: 32087098]
[33]
Kotlyar AM, Grechukhina O, Chen A, Popkhadze S, Grimshaw A, Tal O, Taylor HS, Tal R. Vertical transmission of coronavirus disease 2019: a systematic review and meta-analysis. American journal of obstetrics and gynecology. 2021 Jan:224(1):35-53.e3. doi: 10.1016/j.ajog.2020.07.049. Epub 2020 Jul 31
[PubMed PMID: 32739398]
Level 1 (high-level) evidence
[34]
Ahmad FB, Cisewski JA, Miniño A, Anderson RN. Provisional Mortality Data - United States, 2020. MMWR. Morbidity and mortality weekly report. 2021 Apr 9:70(14):519-522. doi: 10.15585/mmwr.mm7014e1. Epub 2021 Apr 9
[PubMed PMID: 33830988]
[35]
Stokes EK, Zambrano LD, Anderson KN, Marder EP, Raz KM, El Burai Felix S, Tie Y, Fullerton KE. Coronavirus Disease 2019 Case Surveillance - United States, January 22-May 30, 2020. MMWR. Morbidity and mortality weekly report. 2020 Jun 19:69(24):759-765. doi: 10.15585/mmwr.mm6924e2. Epub 2020 Jun 19
[PubMed PMID: 32555134]
Level 3 (low-level) evidence
[36]
Gebhard C, Regitz-Zagrosek V, Neuhauser HK, Morgan R, Klein SL. Impact of sex and gender on COVID-19 outcomes in Europe. Biology of sex differences. 2020 May 25:11(1):29. doi: 10.1186/s13293-020-00304-9. Epub 2020 May 25
[PubMed PMID: 32450906]
[37]
Jin JM, Bai P, He W, Wu F, Liu XF, Han DM, Liu S, Yang JK. Gender Differences in Patients With COVID-19: Focus on Severity and Mortality. Frontiers in public health. 2020:8():152. doi: 10.3389/fpubh.2020.00152. Epub 2020 Apr 29
[PubMed PMID: 32411652]
[38]
Finelli L, Gupta V, Petigara T, Yu K, Bauer KA, Puzniak LA. Mortality Among US Patients Hospitalized With SARS-CoV-2 Infection in 2020. JAMA network open. 2021 Apr 1:4(4):e216556. doi: 10.1001/jamanetworkopen.2021.6556. Epub 2021 Apr 1
[PubMed PMID: 33830226]
[39]
Romano SD, Blackstock AJ, Taylor EV, El Burai Felix S, Adjei S, Singleton CM, Fuld J, Bruce BB, Boehmer TK. Trends in Racial and Ethnic Disparities in COVID-19 Hospitalizations, by Region - United States, March-December 2020. MMWR. Morbidity and mortality weekly report. 2021 Apr 16:70(15):560-565. doi: 10.15585/mmwr.mm7015e2. Epub 2021 Apr 16
[PubMed PMID: 33857068]
[40]
Sze S, Pan D, Nevill CR, Gray LJ, Martin CA, Nazareth J, Minhas JS, Divall P, Khunti K, Abrams KR, Nellums LB, Pareek M. Ethnicity and clinical outcomes in COVID-19: A systematic review and meta-analysis. EClinicalMedicine. 2020 Dec:29():100630. doi: 10.1016/j.eclinm.2020.100630. Epub 2020 Nov 12
[PubMed PMID: 33200120]
Level 2 (mid-level) evidence
[41]
Heslin KC, Hall JE. Sexual Orientation Disparities in Risk Factors for Adverse COVID-19-Related Outcomes, by Race/Ethnicity - Behavioral Risk Factor Surveillance System, United States, 2017-2019. MMWR. Morbidity and mortality weekly report. 2021 Feb 5:70(5):149-154. doi: 10.15585/mmwr.mm7005a1. Epub 2021 Feb 5
[PubMed PMID: 33539330]
[42]
Jiang S, Hillyer C, Du L. Neutralizing Antibodies against SARS-CoV-2 and Other Human Coronaviruses: (Trends in Immunology 41, 355-359; 2020). Trends in immunology. 2020 Jun:41(6):545. doi: 10.1016/j.it.2020.04.008. Epub 2020 Apr 24
[PubMed PMID: 32362491]
[43]
Du L, He Y, Zhou Y, Liu S, Zheng BJ, Jiang S. The spike protein of SARS-CoV--a target for vaccine and therapeutic development. Nature reviews. Microbiology. 2009 Mar:7(3):226-36. doi: 10.1038/nrmicro2090. Epub 2009 Feb 9
[PubMed PMID: 19198616]
[44]
Jiang S, Hillyer C, Du L. Neutralizing Antibodies against SARS-CoV-2 and Other Human Coronaviruses. Trends in immunology. 2020 May:41(5):355-359. doi: 10.1016/j.it.2020.03.007. Epub 2020 Apr 2
[PubMed PMID: 32249063]
[45]
Song W, Gui M, Wang X, Xiang Y. Cryo-EM structure of the SARS coronavirus spike glycoprotein in complex with its host cell receptor ACE2. PLoS pathogens. 2018 Aug:14(8):e1007236. doi: 10.1371/journal.ppat.1007236. Epub 2018 Aug 13
[PubMed PMID: 30102747]
[46]
de Abajo FJ, Rodríguez-Martín S, Lerma V, Mejía-Abril G, Aguilar M, García-Luque A, Laredo L, Laosa O, Centeno-Soto GA, Ángeles Gálvez M, Puerro M, González-Rojano E, Pedraza L, de Pablo I, Abad-Santos F, Rodríguez-Mañas L, Gil M, Tobías A, Rodríguez-Miguel A, Rodríguez-Puyol D, MED-ACE2-COVID19 study group. Use of renin-angiotensin-aldosterone system inhibitors and risk of COVID-19 requiring admission to hospital: a case-population study. Lancet (London, England). 2020 May 30:395(10238):1705-1714. doi: 10.1016/S0140-6736(20)31030-8. Epub 2020 May 14
[PubMed PMID: 32416785]
Level 3 (low-level) evidence
[47]
Xu H, Zhong L, Deng J, Peng J, Dan H, Zeng X, Li T, Chen Q. High expression of ACE2 receptor of 2019-nCoV on the epithelial cells of oral mucosa. International journal of oral science. 2020 Feb 24:12(1):8. doi: 10.1038/s41368-020-0074-x. Epub 2020 Feb 24
[PubMed PMID: 32094336]
[48]
Hoffmann M, Kleine-Weber H, Schroeder S, Krüger N, Herrler T, Erichsen S, Schiergens TS, Herrler G, Wu NH, Nitsche A, Müller MA, Drosten C, Pöhlmann S. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell. 2020 Apr 16:181(2):271-280.e8. doi: 10.1016/j.cell.2020.02.052. Epub 2020 Mar 5
[PubMed PMID: 32142651]
[49]
Wang J, Jiang M, Chen X, Montaner LJ. Cytokine storm and leukocyte changes in mild versus severe SARS-CoV-2 infection: Review of 3939 COVID-19 patients in China and emerging pathogenesis and therapy concepts. Journal of leukocyte biology. 2020 Jul:108(1):17-41. doi: 10.1002/JLB.3COVR0520-272R. Epub 2020 Jun 13
[PubMed PMID: 32534467]
[50]
Azkur AK, Akdis M, Azkur D, Sokolowska M, van de Veen W, Brüggen MC, O'Mahony L, Gao Y, Nadeau K, Akdis CA. Immune response to SARS-CoV-2 and mechanisms of immunopathological changes in COVID-19. Allergy. 2020 Jul:75(7):1564-1581. doi: 10.1111/all.14364. Epub
[PubMed PMID: 32396996]
[51]
Teuwen LA, Geldhof V, Pasut A, Carmeliet P. COVID-19: the vasculature unleashed. Nature reviews. Immunology. 2020 Jul:20(7):389-391. doi: 10.1038/s41577-020-0343-0. Epub
[PubMed PMID: 32439870]
[52]
Ackermann M, Verleden SE, Kuehnel M, Haverich A, Welte T, Laenger F, Vanstapel A, Werlein C, Stark H, Tzankov A, Li WW, Li VW, Mentzer SJ, Jonigk D. Pulmonary Vascular Endothelialitis, Thrombosis, and Angiogenesis in Covid-19. The New England journal of medicine. 2020 Jul 9:383(2):120-128. doi: 10.1056/NEJMoa2015432. Epub 2020 May 21
[PubMed PMID: 32437596]
[53]
van de Veerdonk FL, Netea MG, van Deuren M, van der Meer JW, de Mast Q, Brüggemann RJ, van der Hoeven H. Kallikrein-kinin blockade in patients with COVID-19 to prevent acute respiratory distress syndrome. eLife. 2020 Apr 27:9():. doi: 10.7554/eLife.57555. Epub 2020 Apr 27
[PubMed PMID: 32338605]
[54]
Conti P, Ronconi G, Caraffa A, Gallenga CE, Ross R, Frydas I, Kritas SK. Induction of pro-inflammatory cytokines (IL-1 and IL-6) and lung inflammation by Coronavirus-19 (COVI-19 or SARS-CoV-2): anti-inflammatory strategies. Journal of biological regulators and homeostatic agents. 2020 March-April,:34(2):327-331. doi: 10.23812/CONTI-E. Epub
[PubMed PMID: 32171193]
[55]
Coopersmith CM, Antonelli M, Bauer SR, Deutschman CS, Evans LE, Ferrer R, Hellman J, Jog S, Kesecioglu J, Kissoon N, Martin-Loeches I, Nunnally ME, Prescott HC, Rhodes A, Talmor D, Tissieres P, De Backer D. The Surviving Sepsis Campaign: Research Priorities for Coronavirus Disease 2019 in Critical Illness. Critical care medicine. 2021 Apr 1:49(4):598-622. doi: 10.1097/CCM.0000000000004895. Epub
[PubMed PMID: 33591008]
[56]
Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, Zhang L, Fan G, Xu J, Gu X, Cheng Z, Yu T, Xia J, Wei Y, Wu W, Xie X, Yin W, Li H, Liu M, Xiao Y, Gao H, Guo L, Xie J, Wang G, Jiang R, Gao Z, Jin Q, Wang J, Cao B. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet (London, England). 2020 Feb 15:395(10223):497-506. doi: 10.1016/S0140-6736(20)30183-5. Epub 2020 Jan 24
[PubMed PMID: 31986264]
[57]
Guo T, Fan Y, Chen M, Wu X, Zhang L, He T, Wang H, Wan J, Wang X, Lu Z. Cardiovascular Implications of Fatal Outcomes of Patients With Coronavirus Disease 2019 (COVID-19). JAMA cardiology. 2020 Jul 1:5(7):811-818. doi: 10.1001/jamacardio.2020.1017. Epub
[PubMed PMID: 32219356]
[58]
Tan L, Wang Q, Zhang D, Ding J, Huang Q, Tang YQ, Wang Q, Miao H. Lymphopenia predicts disease severity of COVID-19: a descriptive and predictive study. Signal transduction and targeted therapy. 2020 Mar 27:5(1):33. doi: 10.1038/s41392-020-0148-4. Epub 2020 Mar 27
[PubMed PMID: 32296069]
[59]
Abou-Ismail MY, Diamond A, Kapoor S, Arafah Y, Nayak L. The hypercoagulable state in COVID-19: Incidence, pathophysiology, and management. Thrombosis research. 2020 Oct:194():101-115. doi: 10.1016/j.thromres.2020.06.029. Epub 2020 Jun 20
[PubMed PMID: 32788101]
[60]
Amgalan A, Othman M. Exploring possible mechanisms for COVID-19 induced thrombocytopenia: Unanswered questions. Journal of thrombosis and haemostasis : JTH. 2020 Jun:18(6):1514-1516. doi: 10.1111/jth.14832. Epub 2020 May 6
[PubMed PMID: 32278338]
[61]
Lauer SA, Grantz KH, Bi Q, Jones FK, Zheng Q, Meredith HR, Azman AS, Reich NG, Lessler J. The Incubation Period of Coronavirus Disease 2019 (COVID-19) From Publicly Reported Confirmed Cases: Estimation and Application. Annals of internal medicine. 2020 May 5:172(9):577-582. doi: 10.7326/M20-0504. Epub 2020 Mar 10
[PubMed PMID: 32150748]
Level 3 (low-level) evidence
[62]
Mizumoto K, Kagaya K, Zarebski A, Chowell G. Estimating the asymptomatic proportion of coronavirus disease 2019 (COVID-19) cases on board the Diamond Princess cruise ship, Yokohama, Japan, 2020. Euro surveillance : bulletin Europeen sur les maladies transmissibles = European communicable disease bulletin. 2020 Mar:25(10):. doi: 10.2807/1560-7917.ES.2020.25.10.2000180. Epub
[PubMed PMID: 32183930]
Level 3 (low-level) evidence
[63]
Nishiura H, Kobayashi T, Miyama T, Suzuki A, Jung SM, Hayashi K, Kinoshita R, Yang Y, Yuan B, Akhmetzhanov AR, Linton NM. Estimation of the asymptomatic ratio of novel coronavirus infections (COVID-19). International journal of infectious diseases : IJID : official publication of the International Society for Infectious Diseases. 2020 May:94():154-155. doi: 10.1016/j.ijid.2020.03.020. Epub 2020 Mar 14
[PubMed PMID: 32179137]
[64]
Zhu J, Zhong Z, Ji P, Li H, Li B, Pang J, Zhang J, Zhao C. Clinicopathological characteristics of 8697 patients with COVID-19 in China: a meta-analysis. Family medicine and community health. 2020 Apr:8(2):. doi: 10.1136/fmch-2020-000406. Epub
[PubMed PMID: 32371463]
Level 1 (high-level) evidence
[65]
Li J, Huang DQ, Zou B, Yang H, Hui WZ, Rui F, Yee NTS, Liu C, Nerurkar SN, Kai JCY, Teng MLP, Li X, Zeng H, Borghi JA, Henry L, Cheung R, Nguyen MH. Epidemiology of COVID-19: A systematic review and meta-analysis of clinical characteristics, risk factors, and outcomes. Journal of medical virology. 2021 Mar:93(3):1449-1458. doi: 10.1002/jmv.26424. Epub 2020 Aug 25
[PubMed PMID: 32790106]
Level 1 (high-level) evidence
[66]
Yang AP, Liu JP, Tao WQ, Li HM. The diagnostic and predictive role of NLR, d-NLR and PLR in COVID-19 patients. International immunopharmacology. 2020 Jul:84():106504. doi: 10.1016/j.intimp.2020.106504. Epub 2020 Apr 13
[PubMed PMID: 32304994]
[67]
ARDS Definition Task Force, Ranieri VM, Rubenfeld GD, Thompson BT, Ferguson ND, Caldwell E, Fan E, Camporota L, Slutsky AS. Acute respiratory distress syndrome: the Berlin Definition. JAMA. 2012 Jun 20:307(23):2526-33. doi: 10.1001/jama.2012.5669. Epub
[PubMed PMID: 22797452]
[68]
Ferrando C, Suarez-Sipmann F, Mellado-Artigas R, Hernández M, Gea A, Arruti E, Aldecoa C, Martínez-Pallí G, Martínez-González MA, Slutsky AS, Villar J, COVID-19 Spanish ICU Network. Clinical features, ventilatory management, and outcome of ARDS caused by COVID-19 are similar to other causes of ARDS. Intensive care medicine. 2020 Dec:46(12):2200-2211. doi: 10.1007/s00134-020-06192-2. Epub 2020 Jul 29
[PubMed PMID: 32728965]
[69]
Martinez-Rojas MA, Vega-Vega O, Bobadilla NA. Is the kidney a target of SARS-CoV-2? American journal of physiology. Renal physiology. 2020 Jun 1:318(6):F1454-F1462. doi: 10.1152/ajprenal.00160.2020. Epub 2020 May 15
[PubMed PMID: 32412303]
[70]
Hirsch JS, Ng JH, Ross DW, Sharma P, Shah HH, Barnett RL, Hazzan AD, Fishbane S, Jhaveri KD, Northwell COVID-19 Research Consortium, Northwell Nephrology COVID-19 Research Consortium. Acute kidney injury in patients hospitalized with COVID-19. Kidney international. 2020 Jul:98(1):209-218. doi: 10.1016/j.kint.2020.05.006. Epub 2020 May 16
[PubMed PMID: 32416116]
[71]
Gupta A, Madhavan MV, Sehgal K, Nair N, Mahajan S, Sehrawat TS, Bikdeli B, Ahluwalia N, Ausiello JC, Wan EY, Freedberg DE, Kirtane AJ, Parikh SA, Maurer MS, Nordvig AS, Accili D, Bathon JM, Mohan S, Bauer KA, Leon MB, Krumholz HM, Uriel N, Mehra MR, Elkind MSV, Stone GW, Schwartz A, Ho DD, Bilezikian JP, Landry DW. Extrapulmonary manifestations of COVID-19. Nature medicine. 2020 Jul:26(7):1017-1032. doi: 10.1038/s41591-020-0968-3. Epub 2020 Jul 10
[PubMed PMID: 32651579]
[72]
Hessami A, Shamshirian A, Heydari K, Pourali F, Alizadeh-Navaei R, Moosazadeh M, Abrotan S, Shojaie L, Sedighi S, Shamshirian D, Rezaei N. Cardiovascular diseases burden in COVID-19: Systematic review and meta-analysis. The American journal of emergency medicine. 2021 Aug:46():382-391. doi: 10.1016/j.ajem.2020.10.022. Epub 2020 Oct 16
[PubMed PMID: 33268238]
Level 1 (high-level) evidence
[73]
Tariq R, Saha S, Furqan F, Hassett L, Pardi D, Khanna S. Prevalence and Mortality of COVID-19 Patients With Gastrointestinal Symptoms: A Systematic Review and Meta-analysis. Mayo Clinic proceedings. 2020 Aug:95(8):1632-1648. doi: 10.1016/j.mayocp.2020.06.003. Epub 2020 Jun 10
[PubMed PMID: 32753138]
Level 1 (high-level) evidence
[74]
Azouz E, Yang S, Monnier-Cholley L, Arrivé L. Systemic arterial thrombosis and acute mesenteric ischemia in a patient with COVID-19. Intensive care medicine. 2020 Jul:46(7):1464-1465. doi: 10.1007/s00134-020-06079-2. Epub 2020 May 18
[PubMed PMID: 32424482]
[75]
Xu L, Liu J, Lu M, Yang D, Zheng X. Liver injury during highly pathogenic human coronavirus infections. Liver international : official journal of the International Association for the Study of the Liver. 2020 May:40(5):998-1004. doi: 10.1111/liv.14435. Epub 2020 Mar 30
[PubMed PMID: 32170806]
[76]
Toscano G, Palmerini F, Ravaglia S, Ruiz L, Invernizzi P, Cuzzoni MG, Franciotta D, Baldanti F, Daturi R, Postorino P, Cavallini A, Micieli G. Guillain-Barré Syndrome Associated with SARS-CoV-2. The New England journal of medicine. 2020 Jun 25:382(26):2574-2576. doi: 10.1056/NEJMc2009191. Epub 2020 Apr 17
[PubMed PMID: 32302082]
[77]
Zubair AS, McAlpine LS, Gardin T, Farhadian S, Kuruvilla DE, Spudich S. Neuropathogenesis and Neurologic Manifestations of the Coronaviruses in the Age of Coronavirus Disease 2019: A Review. JAMA neurology. 2020 Aug 1:77(8):1018-1027. doi: 10.1001/jamaneurol.2020.2065. Epub
[PubMed PMID: 32469387]
[78]
Daneshgaran G, Dubin DP, Gould DJ. Cutaneous Manifestations of COVID-19: An Evidence-Based Review. American journal of clinical dermatology. 2020 Oct:21(5):627-639. doi: 10.1007/s40257-020-00558-4. Epub
[PubMed PMID: 32865778]
[79]
Wiersinga WJ, Rhodes A, Cheng AC, Peacock SJ, Prescott HC. Pathophysiology, Transmission, Diagnosis, and Treatment of Coronavirus Disease 2019 (COVID-19): A Review. JAMA. 2020 Aug 25:324(8):782-793. doi: 10.1001/jama.2020.12839. Epub
[PubMed PMID: 32648899]
[80]
Gandhi RT, Lynch JB, Del Rio C. Mild or Moderate Covid-19. The New England journal of medicine. 2020 Oct 29:383(18):1757-1766. doi: 10.1056/NEJMcp2009249. Epub 2020 Apr 24
[PubMed PMID: 32329974]
[81]
Bao C, Liu X, Zhang H, Li Y, Liu J. Coronavirus Disease 2019 (COVID-19) CT Findings: A Systematic Review and Meta-analysis. Journal of the American College of Radiology : JACR. 2020 Jun:17(6):701-709. doi: 10.1016/j.jacr.2020.03.006. Epub 2020 Mar 25
[PubMed PMID: 32283052]
Level 1 (high-level) evidence
[82]
Wong HYF, Lam HYS, Fong AH, Leung ST, Chin TW, Lo CSY, Lui MM, Lee JCY, Chiu KW, Chung TW, Lee EYP, Wan EYF, Hung IFN, Lam TPW, Kuo MD, Ng MY. Frequency and Distribution of Chest Radiographic Findings in Patients Positive for COVID-19. Radiology. 2020 Aug:296(2):E72-E78. doi: 10.1148/radiol.2020201160. Epub 2020 Mar 27
[PubMed PMID: 32216717]
[83]
RECOVERY Collaborative Group, Horby P, Lim WS, Emberson JR, Mafham M, Bell JL, Linsell L, Staplin N, Brightling C, Ustianowski A, Elmahi E, Prudon B, Green C, Felton T, Chadwick D, Rege K, Fegan C, Chappell LC, Faust SN, Jaki T, Jeffery K, Montgomery A, Rowan K, Juszczak E, Baillie JK, Haynes R, Landray MJ. Dexamethasone in Hospitalized Patients with Covid-19. The New England journal of medicine. 2021 Feb 25:384(8):693-704. doi: 10.1056/NEJMoa2021436. Epub 2020 Jul 17
[PubMed PMID: 32678530]
[84]
Hammond J, Leister-Tebbe H, Gardner A, Abreu P, Bao W, Wisemandle W, Baniecki M, Hendrick VM, Damle B, Simón-Campos A, Pypstra R, Rusnak JM, EPIC-HR Investigators. Oral Nirmatrelvir for High-Risk, Nonhospitalized Adults with Covid-19. The New England journal of medicine. 2022 Apr 14:386(15):1397-1408. doi: 10.1056/NEJMoa2118542. Epub 2022 Feb 16
[PubMed PMID: 35172054]
[85]
REMAP-CAP Investigators, Gordon AC, Mouncey PR, Al-Beidh F, Rowan KM, Nichol AD, Arabi YM, Annane D, Beane A, van Bentum-Puijk W, Berry LR, Bhimani Z, Bonten MJM, Bradbury CA, Brunkhorst FM, Buzgau A, Cheng AC, Detry MA, Duffy EJ, Estcourt LJ, Fitzgerald M, Goossens H, Haniffa R, Higgins AM, Hills TE, Horvat CM, Lamontagne F, Lawler PR, Leavis HL, Linstrum KM, Litton E, Lorenzi E, Marshall JC, Mayr FB, McAuley DF, McGlothlin A, McGuinness SP, McVerry BJ, Montgomery SK, Morpeth SC, Murthy S, Orr K, Parke RL, Parker JC, Patanwala AE, Pettilä V, Rademaker E, Santos MS, Saunders CT, Seymour CW, Shankar-Hari M, Sligl WI, Turgeon AF, Turner AM, van de Veerdonk FL, Zarychanski R, Green C, Lewis RJ, Angus DC, McArthur CJ, Berry S, Webb SA, Derde LPG. Interleukin-6 Receptor Antagonists in Critically Ill Patients with Covid-19. The New England journal of medicine. 2021 Apr 22:384(16):1491-1502. doi: 10.1056/NEJMoa2100433. Epub 2021 Feb 25
[PubMed PMID: 33631065]
[86]
Ni YN, Luo J, Yu H, Liu D, Liang BM, Liang ZA. The effect of high-flow nasal cannula in reducing the mortality and the rate of endotracheal intubation when used before mechanical ventilation compared with conventional oxygen therapy and noninvasive positive pressure ventilation. A systematic review and meta-analysis. The American journal of emergency medicine. 2018 Feb:36(2):226-233. doi: 10.1016/j.ajem.2017.07.083. Epub 2017 Jul 28
[PubMed PMID: 28780231]
Level 1 (high-level) evidence
[87]
Hui DS, Chow BK, Lo T, Tsang OTY, Ko FW, Ng SS, Gin T, Chan MTV. Exhaled air dispersion during high-flow nasal cannula therapy versus CPAP via different masks. The European respiratory journal. 2019 Apr:53(4):. pii: 1802339. doi: 10.1183/13993003.02339-2018. Epub 2019 Apr 11
[PubMed PMID: 30705129]
[88]
Grieco DL, Menga LS, Cesarano M, Rosà T, Spadaro S, Bitondo MM, Montomoli J, Falò G, Tonetti T, Cutuli SL, Pintaudi G, Tanzarella ES, Piervincenzi E, Bongiovanni F, Dell'Anna AM, Delle Cese L, Berardi C, Carelli S, Bocci MG, Montini L, Bello G, Natalini D, De Pascale G, Velardo M, Volta CA, Ranieri VM, Conti G, Maggiore SM, Antonelli M, COVID-ICU Gemelli Study Group. Effect of Helmet Noninvasive Ventilation vs High-Flow Nasal Oxygen on Days Free of Respiratory Support in Patients With COVID-19 and Moderate to Severe Hypoxemic Respiratory Failure: The HENIVOT Randomized Clinical Trial. JAMA. 2021 May 4:325(17):1731-1743. doi: 10.1001/jama.2021.4682. Epub
[PubMed PMID: 33764378]
Level 1 (high-level) evidence
[89]
WHO Solidarity Trial Consortium. Remdesivir and three other drugs for hospitalised patients with COVID-19: final results of the WHO Solidarity randomised trial and updated meta-analyses. Lancet (London, England). 2022 May 21:399(10339):1941-1953. doi: 10.1016/S0140-6736(22)00519-0. Epub 2022 May 2
[PubMed PMID: 35512728]
Level 1 (high-level) evidence
[90]
Joyner MJ, Senefeld JW, Klassen SA, Mills JR, Johnson PW, Theel ES, Wiggins CC, Bruno KA, Klompas AM, Lesser ER, Kunze KL, Sexton MA, Diaz Soto JC, Baker SE, Shepherd JRA, van Helmond N, van Buskirk CM, Winters JL, Stubbs JR, Rea RF, Hodge DO, Herasevich V, Whelan ER, Clayburn AJ, Larson KF, Ripoll JG, Andersen KJ, Buras MR, Vogt MNP, Dennis JJ, Regimbal RJ, Bauer PR, Blair JE, Paneth NS, Fairweather D, Wright RS, Carter RE, Casadevall A. Effect of Convalescent Plasma on Mortality among Hospitalized Patients with COVID-19: Initial Three-Month Experience. medRxiv : the preprint server for health sciences. 2020 Aug 12:():. pii: 2020.08.12.20169359. doi: 10.1101/2020.08.12.20169359. Epub 2020 Aug 12
[PubMed PMID: 32817978]
[91]
Joyner MJ, Bruno KA, Klassen SA, Kunze KL, Johnson PW, Lesser ER, Wiggins CC, Senefeld JW, Klompas AM, Hodge DO, Shepherd JRA, Rea RF, Whelan ER, Clayburn AJ, Spiegel MR, Baker SE, Larson KF, Ripoll JG, Andersen KJ, Buras MR, Vogt MNP, Herasevich V, Dennis JJ, Regimbal RJ, Bauer PR, Blair JE, van Buskirk CM, Winters JL, Stubbs JR, van Helmond N, Butterfield BP, Sexton MA, Diaz Soto JC, Paneth NS, Verdun NC, Marks P, Casadevall A, Fairweather D, Carter RE, Wright RS. Safety Update: COVID-19 Convalescent Plasma in 20,000 Hospitalized Patients. Mayo Clinic proceedings. 2020 Sep:95(9):1888-1897. doi: 10.1016/j.mayocp.2020.06.028. Epub 2020 Jul 19
[PubMed PMID: 32861333]
[92]
Simonovich VA, Burgos Pratx LD, Scibona P, Beruto MV, Vallone MG, Vázquez C, Savoy N, Giunta DH, Pérez LG, Sánchez MDL, Gamarnik AV, Ojeda DS, Santoro DM, Camino PJ, Antelo S, Rainero K, Vidiella GP, Miyazaki EA, Cornistein W, Trabadelo OA, Ross FM, Spotti M, Funtowicz G, Scordo WE, Losso MH, Ferniot I, Pardo PE, Rodriguez E, Rucci P, Pasquali J, Fuentes NA, Esperatti M, Speroni GA, Nannini EC, Matteaccio A, Michelangelo HG, Follmann D, Lane HC, Belloso WH, PlasmAr Study Group. A Randomized Trial of Convalescent Plasma in Covid-19 Severe Pneumonia. The New England journal of medicine. 2021 Feb 18:384(7):619-629. doi: 10.1056/NEJMoa2031304. Epub 2020 Nov 24
[PubMed PMID: 33232588]
Level 1 (high-level) evidence
[93]
Li L, Zhang W, Hu Y, Tong X, Zheng S, Yang J, Kong Y, Ren L, Wei Q, Mei H, Hu C, Tao C, Yang R, Wang J, Yu Y, Guo Y, Wu X, Xu Z, Zeng L, Xiong N, Chen L, Wang J, Man N, Liu Y, Xu H, Deng E, Zhang X, Li C, Wang C, Su S, Zhang L, Wang J, Wu Y, Liu Z. Effect of Convalescent Plasma Therapy on Time to Clinical Improvement in Patients With Severe and Life-threatening COVID-19: A Randomized Clinical Trial. JAMA. 2020 Aug 4:324(5):460-470. doi: 10.1001/jama.2020.10044. Epub
[PubMed PMID: 32492084]
Level 1 (high-level) evidence
[94]
. Convalescent plasma in the management of moderate covid-19 in adults in India: open label phase II multicentre randomised controlled trial (PLACID Trial). BMJ (Clinical research ed.). 2020 Nov 3:371():m4232. doi: 10.1136/bmj.m4232. Epub 2020 Nov 3
[PubMed PMID: 33144278]
Level 1 (high-level) evidence
[95]
Levin MJ, Ustianowski A, De Wit S, Launay O, Avila M, Templeton A, Yuan Y, Seegobin S, Ellery A, Levinson DJ, Ambery P, Arends RH, Beavon R, Dey K, Garbes P, Kelly EJ, Koh GCKW, Near KA, Padilla KW, Psachoulia K, Sharbaugh A, Streicher K, Pangalos MN, Esser MT, PROVENT Study Group. Intramuscular AZD7442 (Tixagevimab-Cilgavimab) for Prevention of Covid-19. The New England journal of medicine. 2022 Jun 9:386(23):2188-2200. doi: 10.1056/NEJMoa2116620. Epub 2022 Apr 20
[PubMed PMID: 35443106]
[96]
Dong J, Zost SJ, Greaney AJ, Starr TN, Dingens AS, Chen EC, Chen RE, Case JB, Sutton RE, Gilchuk P, Rodriguez J, Armstrong E, Gainza C, Nargi RS, Binshtein E, Xie X, Zhang X, Shi PY, Logue J, Weston S, McGrath ME, Frieman MB, Brady T, Tuffy KM, Bright H, Loo YM, McTamney PM, Esser MT, Carnahan RH, Diamond MS, Bloom JD, Crowe JE Jr. Genetic and structural basis for SARS-CoV-2 variant neutralization by a two-antibody cocktail. Nature microbiology. 2021 Oct:6(10):1233-1244. doi: 10.1038/s41564-021-00972-2. Epub 2021 Sep 21
[PubMed PMID: 34548634]
Level 3 (low-level) evidence
[97]
Zost SJ, Gilchuk P, Case JB, Binshtein E, Chen RE, Nkolola JP, Schäfer A, Reidy JX, Trivette A, Nargi RS, Sutton RE, Suryadevara N, Martinez DR, Williamson LE, Chen EC, Jones T, Day S, Myers L, Hassan AO, Kafai NM, Winkler ES, Fox JM, Shrihari S, Mueller BK, Meiler J, Chandrashekar A, Mercado NB, Steinhardt JJ, Ren K, Loo YM, Kallewaard NL, McCune BT, Keeler SP, Holtzman MJ, Barouch DH, Gralinski LE, Baric RS, Thackray LB, Diamond MS, Carnahan RH, Crowe JE Jr. Potently neutralizing and protective human antibodies against SARS-CoV-2. Nature. 2020 Aug:584(7821):443-449. doi: 10.1038/s41586-020-2548-6. Epub 2020 Jul 15
[PubMed PMID: 32668443]
Level 3 (low-level) evidence
[98]
COVID-ICU Group on behalf of the REVA Network and the COVID-ICU Investigators. Clinical characteristics and day-90 outcomes of 4244 critically ill adults with COVID-19: a prospective cohort study. Intensive care medicine. 2021 Jan:47(1):60-73. doi: 10.1007/s00134-020-06294-x. Epub 2020 Oct 29
[PubMed PMID: 33211135]
[99]
Kaafarani HMA, El Moheb M, Hwabejire JO, Naar L, Christensen MA, Breen K, Gaitanidis A, Alser O, Mashbari H, Bankhead-Kendall B, Mokhtari A, Maurer L, Kapoen C, Langeveld K, El Hechi MW, Lee J, Mendoza AE, Saillant NN, Parks J, Fawley J, King DR, Fagenholz PJ, Velmahos GC. Gastrointestinal Complications in Critically Ill Patients With COVID-19. Annals of surgery. 2020 Aug:272(2):e61-e62. doi: 10.1097/SLA.0000000000004004. Epub
[PubMed PMID: 32675498]
[100]
Zhou X, Cheng Z, Luo L, Zhu Y, Lin W, Ming Z, Chen W, Hu Y. Incidence and impact of disseminated intravascular coagulation in COVID-19 a systematic review and meta-analysis. Thrombosis research. 2021 May:201():23-29. doi: 10.1016/j.thromres.2021.02.010. Epub 2021 Feb 17
[PubMed PMID: 33631519]
Level 1 (high-level) evidence
[101]
Huang C, Huang L, Wang Y, Li X, Ren L, Gu X, Kang L, Guo L, Liu M, Zhou X, Luo J, Huang Z, Tu S, Zhao Y, Chen L, Xu D, Li Y, Li C, Peng L, Li Y, Xie W, Cui D, Shang L, Fan G, Xu J, Wang G, Wang Y, Zhong J, Wang C, Wang J, Zhang D, Cao B. 6-month consequences of COVID-19 in patients discharged from hospital: a cohort study. Lancet (London, England). 2021 Jan 16:397(10270):220-232. doi: 10.1016/S0140-6736(20)32656-8. Epub 2021 Jan 8
[PubMed PMID: 33428867]
[102]
Taquet M, Geddes JR, Husain M, Luciano S, Harrison PJ. 6-month neurological and psychiatric outcomes in 236 379 survivors of COVID-19: a retrospective cohort study using electronic health records. The lancet. Psychiatry. 2021 May:8(5):416-427. doi: 10.1016/S2215-0366(21)00084-5. Epub 2021 Apr 6
[PubMed PMID: 33836148]
Level 2 (mid-level) evidence