[1]
COON WW, WILLIS PW. Deep venous thrombosis and pulmonary embolism: prediction, prevention and treatment. The American journal of cardiology. 1959 Nov:4():611-21
[PubMed PMID: 13811755]
[2]
Goldhaber SZ, Grodstein F, Stampfer MJ, Manson JE, Colditz GA, Speizer FE, Willett WC, Hennekens CH. A prospective study of risk factors for pulmonary embolism in women. JAMA. 1997 Feb 26:277(8):642-5
[PubMed PMID: 9039882]
[3]
Gohil R, Peck G, Sharma P. The genetics of venous thromboembolism. A meta-analysis involving approximately 120,000 cases and 180,000 controls. Thrombosis and haemostasis. 2009 Aug:102(2):360-70. doi: 10.1160/TH09-01-0013. Epub
[PubMed PMID: 19652888]
Level 3 (low-level) evidence
[4]
Rogers MA, Levine DA, Blumberg N, Flanders SA, Chopra V, Langa KM. Triggers of hospitalization for venous thromboembolism. Circulation. 2012 May 1:125(17):2092-9. doi: 10.1161/CIRCULATIONAHA.111.084467. Epub 2012 Apr 3
[PubMed PMID: 22474264]
[5]
Anderson FA Jr, Spencer FA. Risk factors for venous thromboembolism. Circulation. 2003 Jun 17:107(23 Suppl 1):I9-16
[PubMed PMID: 12814980]
[6]
Blom JW, Doggen CJ, Osanto S, Rosendaal FR. Malignancies, prothrombotic mutations, and the risk of venous thrombosis. JAMA. 2005 Feb 9:293(6):715-22
[PubMed PMID: 15701913]
[7]
Clayton TC, Gaskin M, Meade TW. Recent respiratory infection and risk of venous thromboembolism: case-control study through a general practice database. International journal of epidemiology. 2011 Jun:40(3):819-27. doi: 10.1093/ije/dyr012. Epub 2011 Feb 15
[PubMed PMID: 21324940]
Level 2 (mid-level) evidence
[8]
Prandoni P, Pesavento R, Sørensen HT, Gennaro N, Dalla Valle F, Minotto I, Perina F, Pengo V, Pagnan A. Prevalence of heart diseases in patients with pulmonary embolism with and without peripheral venous thrombosis: findings from a cross-sectional survey. European journal of internal medicine. 2009 Sep:20(5):470-3. doi: 10.1016/j.ejim.2009.06.001. Epub 2009 Jul 4
[PubMed PMID: 19712846]
Level 2 (mid-level) evidence
[9]
Sørensen HT, Horvath-Puho E, Pedersen L, Baron JA, Prandoni P. Venous thromboembolism and subsequent hospitalisation due to acute arterial cardiovascular events: a 20-year cohort study. Lancet (London, England). 2007 Nov 24:370(9601):1773-9
[PubMed PMID: 18037081]
[10]
Wendelboe AM, Raskob GE. Global Burden of Thrombosis: Epidemiologic Aspects. Circulation research. 2016 Apr 29:118(9):1340-7. doi: 10.1161/CIRCRESAHA.115.306841. Epub
[PubMed PMID: 27126645]
[11]
Raskob GE, Angchaisuksiri P, Blanco AN, Buller H, Gallus A, Hunt BJ, Hylek EM, Kakkar A, Konstantinides SV, McCumber M, Ozaki Y, Wendelboe A, Weitz JI, ISTH Steering Committee for World Thrombosis Day. Thrombosis: a major contributor to global disease burden. Arteriosclerosis, thrombosis, and vascular biology. 2014 Nov:34(11):2363-71. doi: 10.1161/ATVBAHA.114.304488. Epub
[PubMed PMID: 25304324]
[12]
Horlander KT, Mannino DM, Leeper KV. Pulmonary embolism mortality in the United States, 1979-1998: an analysis using multiple-cause mortality data. Archives of internal medicine. 2003 Jul 28:163(14):1711-7
[PubMed PMID: 12885687]
[14]
Nakos G, Kitsiouli EI, Lekka ME. Bronchoalveolar lavage alterations in pulmonary embolism. American journal of respiratory and critical care medicine. 1998 Nov:158(5 Pt 1):1504-10
[PubMed PMID: 9817700]
[15]
Morrone D, Morrone V. Acute Pulmonary Embolism: Focus on the Clinical Picture. Korean circulation journal. 2018 May:48(5):365-381. doi: 10.4070/kcj.2017.0314. Epub
[PubMed PMID: 29737640]
[16]
Carson JL, Kelley MA, Duff A, Weg JG, Fulkerson WJ, Palevsky HI, Schwartz JS, Thompson BT, Popovich J Jr, Hobbins TE. The clinical course of pulmonary embolism. The New England journal of medicine. 1992 May 7:326(19):1240-5
[PubMed PMID: 1560799]
[17]
Nijkeuter M, Söhne M, Tick LW, Kamphuisen PW, Kramer MH, Laterveer L, van Houten AA, Kruip MJ, Leebeek FW, Büller HR, Huisman MV, Christopher Study Investigators. The natural course of hemodynamically stable pulmonary embolism: Clinical outcome and risk factors in a large prospective cohort study. Chest. 2007 Feb:131(2):517-23
[PubMed PMID: 17296656]
Level 2 (mid-level) evidence
[18]
Stein PD, Henry JW. Clinical characteristics of patients with acute pulmonary embolism stratified according to their presenting syndromes. Chest. 1997 Oct:112(4):974-9
[PubMed PMID: 9377961]
[19]
Stein PD, Beemath A, Matta F, Weg JG, Yusen RD, Hales CA, Hull RD, Leeper KV Jr, Sostman HD, Tapson VF, Buckley JD, Gottschalk A, Goodman LR, Wakefied TW, Woodard PK. Clinical characteristics of patients with acute pulmonary embolism: data from PIOPED II. The American journal of medicine. 2007 Oct:120(10):871-9
[PubMed PMID: 17904458]
[20]
Barco S, Ende-Verhaar YM, Becattini C, Jimenez D, Lankeit M, Huisman MV, Konstantinides SV, Klok FA. Differential impact of syncope on the prognosis of patients with acute pulmonary embolism: a systematic review and meta-analysis. European heart journal. 2018 Dec 14:39(47):4186-4195. doi: 10.1093/eurheartj/ehy631. Epub
[PubMed PMID: 30339253]
Level 1 (high-level) evidence
[21]
Courtney DM, Sasser HC, Pincus CL, Kline JA. Pulseless electrical activity with witnessed arrest as a predictor of sudden death from massive pulmonary embolism in outpatients. Resuscitation. 2001 Jun:49(3):265-72
[PubMed PMID: 11719120]
[23]
Kiely DG, Kennedy NS, Pirzada O, Batchelor SA, Struthers AD, Lipworth BJ. Elevated levels of natriuretic peptides in patients with pulmonary thromboembolism. Respiratory medicine. 2005 Oct:99(10):1286-91
[PubMed PMID: 16099151]
[24]
Henzler T, Roeger S, Meyer M, Schoepf UJ, Nance JW Jr, Haghi D, Kaminski WE, Neumaier M, Schoenberg SO, Fink C. Pulmonary embolism: CT signs and cardiac biomarkers for predicting right ventricular dysfunction. The European respiratory journal. 2012 Apr:39(4):919-26. doi: 10.1183/09031936.00088711. Epub 2011 Sep 29
[PubMed PMID: 21965223]
[25]
Meyer T, Binder L, Hruska N, Luthe H, Buchwald AB. Cardiac troponin I elevation in acute pulmonary embolism is associated with right ventricular dysfunction. Journal of the American College of Cardiology. 2000 Nov 1:36(5):1632-6
[PubMed PMID: 11079669]
[26]
Konstantinides S, Geibel A, Olschewski M, Kasper W, Hruska N, Jäckle S, Binder L. Importance of cardiac troponins I and T in risk stratification of patients with acute pulmonary embolism. Circulation. 2002 Sep 3:106(10):1263-8
[PubMed PMID: 12208803]
[27]
Horlander KT, Leeper KV. Troponin levels as a guide to treatment of pulmonary embolism. Current opinion in pulmonary medicine. 2003 Sep:9(5):374-7
[PubMed PMID: 12904706]
Level 3 (low-level) evidence
[28]
Righini M, Van Es J, Den Exter PL, Roy PM, Verschuren F, Ghuysen A, Rutschmann OT, Sanchez O, Jaffrelot M, Trinh-Duc A, Le Gall C, Moustafa F, Principe A, Van Houten AA, Ten Wolde M, Douma RA, Hazelaar G, Erkens PM, Van Kralingen KW, Grootenboers MJ, Durian MF, Cheung YW, Meyer G, Bounameaux H, Huisman MV, Kamphuisen PW, Le Gal G. Age-adjusted D-dimer cutoff levels to rule out pulmonary embolism: the ADJUST-PE study. JAMA. 2014 Mar 19:311(11):1117-24. doi: 10.1001/jama.2014.2135. Epub
[PubMed PMID: 24643601]
[29]
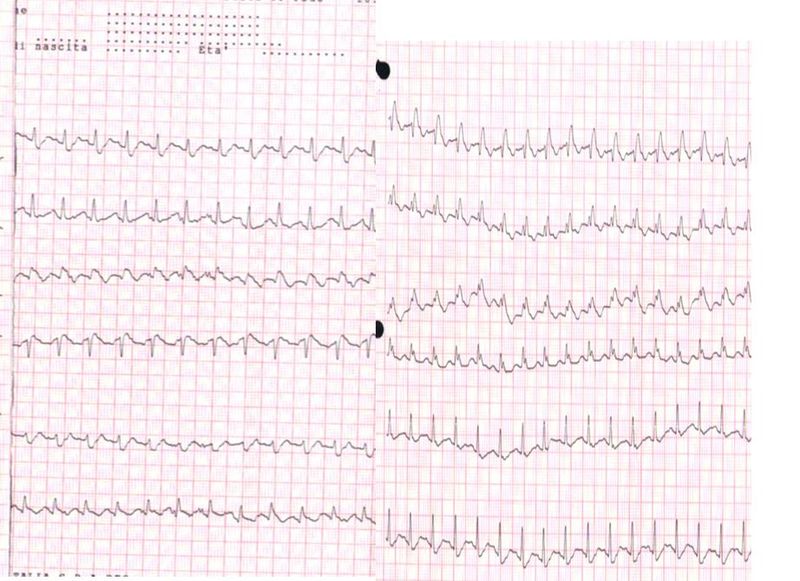
Rodger M, Makropoulos D, Turek M, Quevillon J, Raymond F, Rasuli P, Wells PS. Diagnostic value of the electrocardiogram in suspected pulmonary embolism. The American journal of cardiology. 2000 Oct 1:86(7):807-9, A10
[PubMed PMID: 11018210]
[30]
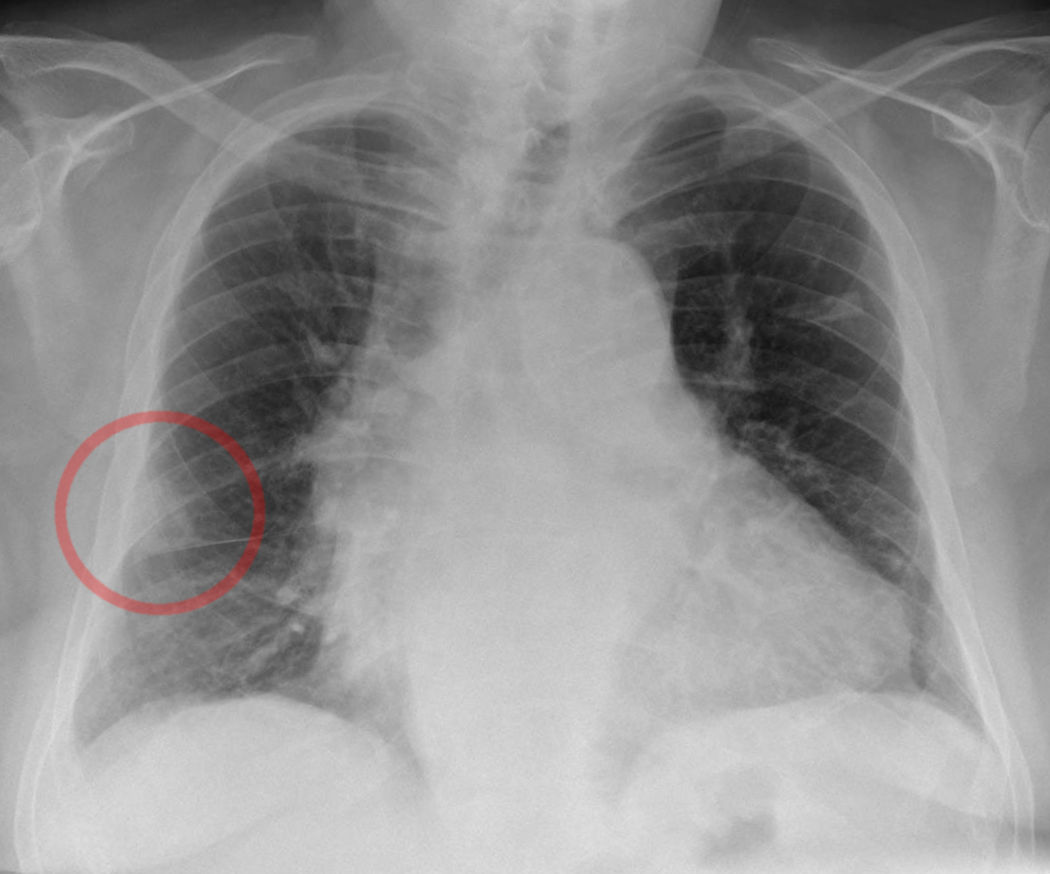
Worsley DF, Alavi A, Aronchick JM, Chen JT, Greenspan RH, Ravin CE. Chest radiographic findings in patients with acute pulmonary embolism: observations from the PIOPED Study. Radiology. 1993 Oct:189(1):133-6
[PubMed PMID: 8372182]
[31]
Ghaye B, Szapiro D, Mastora I, Delannoy V, Duhamel A, Remy J, Remy-Jardin M. Peripheral pulmonary arteries: how far in the lung does multi-detector row spiral CT allow analysis? Radiology. 2001 Jun:219(3):629-36
[PubMed PMID: 11376246]
[32]
Stein PD, Fowler SE, Goodman LR, Gottschalk A, Hales CA, Hull RD, Leeper KV Jr, Popovich J Jr, Quinn DA, Sos TA, Sostman HD, Tapson VF, Wakefield TW, Weg JG, Woodard PK, PIOPED II Investigators. Multidetector computed tomography for acute pulmonary embolism. The New England journal of medicine. 2006 Jun 1:354(22):2317-27
[PubMed PMID: 16738268]
[33]
Becattini C, Agnelli G, Vedovati MC, Pruszczyk P, Casazza F, Grifoni S, Salvi A, Bianchi M, Douma R, Konstantinides S, Lankeit M, Duranti M. Multidetector computed tomography for acute pulmonary embolism: diagnosis and risk stratification in a single test. European heart journal. 2011 Jul:32(13):1657-63. doi: 10.1093/eurheartj/ehr108. Epub 2011 Apr 18
[PubMed PMID: 21504936]
[34]
Reid JH, Coche EE, Inoue T, Kim EE, Dondi M, Watanabe N, Mariani G, International Atomic Energy Agency Consultants' Group. Is the lung scan alive and well? Facts and controversies in defining the role of lung scintigraphy for the diagnosis of pulmonary embolism in the era of MDCT. European journal of nuclear medicine and molecular imaging. 2009 Mar:36(3):505-21. doi: 10.1007/s00259-008-1014-8. Epub 2009 Jan 27
[PubMed PMID: 19172269]
[35]
Glaser JE, Chamarthy M, Haramati LB, Esses D, Freeman LM. Successful and safe implementation of a trinary interpretation and reporting strategy for V/Q lung scintigraphy. Journal of nuclear medicine : official publication, Society of Nuclear Medicine. 2011 Oct:52(10):1508-12. doi: 10.2967/jnumed.111.090753. Epub 2011 Jul 29
[PubMed PMID: 21803837]
[36]
Anderson DR, Kahn SR, Rodger MA, Kovacs MJ, Morris T, Hirsch A, Lang E, Stiell I, Kovacs G, Dreyer J, Dennie C, Cartier Y, Barnes D, Burton E, Pleasance S, Skedgel C, O'Rouke K, Wells PS. Computed tomographic pulmonary angiography vs ventilation-perfusion lung scanning in patients with suspected pulmonary embolism: a randomized controlled trial. JAMA. 2007 Dec 19:298(23):2743-53. doi: 10.1001/jama.298.23.2743. Epub
[PubMed PMID: 18165667]
Level 1 (high-level) evidence
[37]
Sostman HD, Stein PD, Gottschalk A, Matta F, Hull R, Goodman L. Acute pulmonary embolism: sensitivity and specificity of ventilation-perfusion scintigraphy in PIOPED II study. Radiology. 2008 Mar:246(3):941-6. doi: 10.1148/radiol.2463070270. Epub 2008 Jan 14
[PubMed PMID: 18195380]
[38]
PIOPED Investigators. Value of the ventilation/perfusion scan in acute pulmonary embolism. Results of the prospective investigation of pulmonary embolism diagnosis (PIOPED). JAMA. 1990 May 23-30:263(20):2753-9
[PubMed PMID: 2332918]
[39]
Wittram C, Waltman AC, Shepard JA, Halpern E, Goodman LR. Discordance between CT and angiography in the PIOPED II study. Radiology. 2007 Sep:244(3):883-9
[PubMed PMID: 17664436]
[40]
Stein PD, Chenevert TL, Fowler SE, Goodman LR, Gottschalk A, Hales CA, Hull RD, Jablonski KA, Leeper KV Jr, Naidich DP, Sak DJ, Sostman HD, Tapson VF, Weg JG, Woodard PK, PIOPED III (Prospective Investigation of Pulmonary Embolism Diagnosis III) Investigators. Gadolinium-enhanced magnetic resonance angiography for pulmonary embolism: a multicenter prospective study (PIOPED III). Annals of internal medicine. 2010 Apr 6:152(7):434-43, W142-3. doi: 10.7326/0003-4819-152-7-201004060-00008. Epub
[PubMed PMID: 20368649]
[41]
Grifoni S, Olivotto I, Cecchini P, Pieralli F, Camaiti A, Santoro G, Conti A, Agnelli G, Berni G. Short-term clinical outcome of patients with acute pulmonary embolism, normal blood pressure, and echocardiographic right ventricular dysfunction. Circulation. 2000 Jun 20:101(24):2817-22
[PubMed PMID: 10859287]
Level 2 (mid-level) evidence
[42]
Torbicki A, Kurzyna M, Ciurzynski M, Pruszczyk P, Pacho R, Kuch-Wocial A, Szulc M. Proximal pulmonary emboli modify right ventricular ejection pattern. The European respiratory journal. 1999 Mar:13(3):616-21
[PubMed PMID: 10232436]
[43]
Bova C, Greco F, Misuraca G, Serafini O, Crocco F, Greco A, Noto A. Diagnostic utility of echocardiography in patients with suspected pulmonary embolism. The American journal of emergency medicine. 2003 May:21(3):180-3
[PubMed PMID: 12811708]
[44]
Wolfe MW, Lee RT, Feldstein ML, Parker JA, Come PC, Goldhaber SZ. Prognostic significance of right ventricular hypokinesis and perfusion lung scan defects in pulmonary embolism. American heart journal. 1994 May:127(5):1371-5
[PubMed PMID: 8172067]
[45]
Kurzyna M, Torbicki A, Pruszczyk P, Burakowska B, Fijałkowska A, Kober J, Oniszh K, Kuca P, Tomkowski W, Burakowski J, Wawrzyńska L. Disturbed right ventricular ejection pattern as a new Doppler echocardiographic sign of acute pulmonary embolism. The American journal of cardiology. 2002 Sep 1:90(5):507-11
[PubMed PMID: 12208411]
[46]
Pruszczyk P, Goliszek S, Lichodziejewska B, Kostrubiec M, Ciurzyński M, Kurnicka K, Dzikowska-Diduch O, Palczewski P, Wyzgal A. Prognostic value of echocardiography in normotensive patients with acute pulmonary embolism. JACC. Cardiovascular imaging. 2014 Jun:7(6):553-60. doi: 10.1016/j.jcmg.2013.11.004. Epub 2014 Jan 8
[PubMed PMID: 24412192]
[47]
Hull RD, Hirsh J, Carter CJ, Jay RM, Dodd PE, Ockelford PA, Coates G, Gill GJ, Turpie AG, Doyle DJ, Buller HR, Raskob GE. Pulmonary angiography, ventilation lung scanning, and venography for clinically suspected pulmonary embolism with abnormal perfusion lung scan. Annals of internal medicine. 1983 Jun:98(6):891-9
[PubMed PMID: 6859705]
[48]
Kearon C, Ginsberg JS, Hirsh J. The role of venous ultrasonography in the diagnosis of suspected deep venous thrombosis and pulmonary embolism. Annals of internal medicine. 1998 Dec 15:129(12):1044-9
[PubMed PMID: 9867760]
[49]
Le Gal G, Righini M, Sanchez O, Roy PM, Baba-Ahmed M, Perrier A, Bounameaux H. A positive compression ultrasonography of the lower limb veins is highly predictive of pulmonary embolism on computed tomography in suspected patients. Thrombosis and haemostasis. 2006 Jun:95(6):963-6
[PubMed PMID: 16732375]
[50]
van Rossum AB, van Houwelingen HC, Kieft GJ, Pattynama PM. Prevalence of deep vein thrombosis in suspected and proven pulmonary embolism: a meta-analysis. The British journal of radiology. 1998 Dec:71(852):1260-5
[PubMed PMID: 10318998]
Level 1 (high-level) evidence
[51]
Kline JA, Mitchell AM, Kabrhel C, Richman PB, Courtney DM. Clinical criteria to prevent unnecessary diagnostic testing in emergency department patients with suspected pulmonary embolism. Journal of thrombosis and haemostasis : JTH. 2004 Aug:2(8):1247-55
[PubMed PMID: 15304025]
[52]
Coutance G, Cauderlier E, Ehtisham J, Hamon M, Hamon M. The prognostic value of markers of right ventricular dysfunction in pulmonary embolism: a meta-analysis. Critical care (London, England). 2011:15(2):R103. doi: 10.1186/cc10119. Epub 2011 Mar 28
[PubMed PMID: 21443777]
Level 1 (high-level) evidence
[53]
Hugli O, Righini M, Le Gal G, Roy PM, Sanchez O, Verschuren F, Meyer G, Bounameaux H, Aujesky D. The pulmonary embolism rule-out criteria (PERC) rule does not safely exclude pulmonary embolism. Journal of thrombosis and haemostasis : JTH. 2011 Feb:9(2):300-4. doi: 10.1111/j.1538-7836.2010.04147.x. Epub
[PubMed PMID: 21091866]
[54]
Stein PD, Hull RD, Matta F, Yaekoub AY, Liang J. Incidence of thrombocytopenia in hospitalized patients with venous thromboembolism. The American journal of medicine. 2009 Oct:122(10):919-30. doi: 10.1016/j.amjmed.2009.03.026. Epub 2009 Aug 13
[PubMed PMID: 19682670]
[55]
Cossette B, Pelletier ME, Carrier N, Turgeon M, Leclair C, Charron P, Echenberg D, Fayad T, Farand P. Evaluation of bleeding risk in patients exposed to therapeutic unfractionated or low-molecular-weight heparin: a cohort study in the context of a quality improvement initiative. The Annals of pharmacotherapy. 2010 Jun:44(6):994-1002. doi: 10.1345/aph.1M615. Epub 2010 May 4
[PubMed PMID: 20442353]
Level 2 (mid-level) evidence
[56]
Goldhaber SZ, Haire WD, Feldstein ML, Miller M, Toltzis R, Smith JL, Taveira da Silva AM, Come PC, Lee RT, Parker JA. Alteplase versus heparin in acute pulmonary embolism: randomised trial assessing right-ventricular function and pulmonary perfusion. Lancet (London, England). 1993 Feb 27:341(8844):507-11
[PubMed PMID: 8094768]
Level 1 (high-level) evidence
[57]
Dalla-Volta S, Palla A, Santolicandro A, Giuntini C, Pengo V, Visioli O, Zonzin P, Zanuttini D, Barbaresi F, Agnelli G. PAIMS 2: alteplase combined with heparin versus heparin in the treatment of acute pulmonary embolism. Plasminogen activator Italian multicenter study 2. Journal of the American College of Cardiology. 1992 Sep:20(3):520-6
[PubMed PMID: 1512328]
Level 2 (mid-level) evidence
[58]
Daniels LB, Parker JA, Patel SR, Grodstein F, Goldhaber SZ. Relation of duration of symptoms with response to thrombolytic therapy in pulmonary embolism. The American journal of cardiology. 1997 Jul 15:80(2):184-8
[PubMed PMID: 9230156]
[59]
Marti C, John G, Konstantinides S, Combescure C, Sanchez O, Lankeit M, Meyer G, Perrier A. Systemic thrombolytic therapy for acute pulmonary embolism: a systematic review and meta-analysis. European heart journal. 2015 Mar 7:36(10):605-14. doi: 10.1093/eurheartj/ehu218. Epub 2014 Jun 10
[PubMed PMID: 24917641]
Level 1 (high-level) evidence
[60]
Meyer G, Vicaut E, Danays T, Agnelli G, Becattini C, Beyer-Westendorf J, Bluhmki E, Bouvaist H, Brenner B, Couturaud F, Dellas C, Empen K, Franca A, Galiè N, Geibel A, Goldhaber SZ, Jimenez D, Kozak M, Kupatt C, Kucher N, Lang IM, Lankeit M, Meneveau N, Pacouret G, Palazzini M, Petris A, Pruszczyk P, Rugolotto M, Salvi A, Schellong S, Sebbane M, Sobkowicz B, Stefanovic BS, Thiele H, Torbicki A, Verschuren F, Konstantinides SV, PEITHO Investigators. Fibrinolysis for patients with intermediate-risk pulmonary embolism. The New England journal of medicine. 2014 Apr 10:370(15):1402-11. doi: 10.1056/NEJMoa1302097. Epub
[PubMed PMID: 24716681]
[61]
Konstantinides SV, Meyer G, Becattini C, Bueno H, Geersing GJ, Harjola VP, Huisman MV, Humbert M, Jennings CS, Jiménez D, Kucher N, Lang IM, Lankeit M, Lorusso R, Mazzolai L, Meneveau N, Áinle FN, Prandoni P, Pruszczyk P, Righini M, Torbicki A, Van Belle E, Zamorano JL, The Task Force for the diagnosis and management of acute pulmonary embolism of the European Society of Cardiology (ESC). 2019 ESC Guidelines for the diagnosis and management of acute pulmonary embolism developed in collaboration with the European Respiratory Society (ERS): The Task Force for the diagnosis and management of acute pulmonary embolism of the European Society of Cardiology (ESC). The European respiratory journal. 2019 Sep:54(3):. pii: 1901647. doi: 10.1183/13993003.01647-2019. Epub 2019 Oct 9
[PubMed PMID: 31473594]
[62]
Tafur AJ, Shamoun FE, Patel SI, Tafur D, Donna F, Murad MH. Catheter-Directed Treatment of Pulmonary Embolism: A Systematic Review and Meta-Analysis of Modern Literature. Clinical and applied thrombosis/hemostasis : official journal of the International Academy of Clinical and Applied Thrombosis/Hemostasis. 2017 Oct:23(7):821-829. doi: 10.1177/1076029616661414. Epub 2016 Aug 1
[PubMed PMID: 27481877]
Level 1 (high-level) evidence
[63]
Bajaj NS, Kalra R, Arora P, Ather S, Guichard JL, Lancaster WJ, Patel N, Raman F, Arora G, Al Solaiman F, Clark DT 3rd, Dell'Italia LJ, Leesar MA, Davies JE, McGiffin DC, Ahmed MI. Catheter-directed treatment for acute pulmonary embolism: Systematic review and single-arm meta-analyses. International journal of cardiology. 2016 Dec 15:225():128-139. doi: 10.1016/j.ijcard.2016.09.036. Epub 2016 Sep 20
[PubMed PMID: 27718446]
Level 1 (high-level) evidence
[64]
Lee T, Itagaki S, Chiang YP, Egorova NN, Adams DH, Chikwe J. Survival and recurrence after acute pulmonary embolism treated with pulmonary embolectomy or thrombolysis in New York State, 1999 to 2013. The Journal of thoracic and cardiovascular surgery. 2018 Mar:155(3):1084-1090.e12. doi: 10.1016/j.jtcvs.2017.07.074. Epub 2017 Aug 31
[PubMed PMID: 28942971]
[65]
Keeling WB, Sundt T, Leacche M, Okita Y, Binongo J, Lasajanak Y, Aklog L, Lattouf OM, SPEAR Working Group. Outcomes After Surgical Pulmonary Embolectomy for Acute Pulmonary Embolus: A Multi-Institutional Study. The Annals of thoracic surgery. 2016 Nov:102(5):1498-1502. doi: 10.1016/j.athoracsur.2016.05.004. Epub 2016 Jun 30
[PubMed PMID: 27373187]
[66]
Pasrija C, Kronfli A, Rouse M, Raithel M, Bittle GJ, Pousatis S, Ghoreishi M, Gammie JS, Griffith BP, Sanchez PG, Kon ZN. Outcomes after surgical pulmonary embolectomy for acute submassive and massive pulmonary embolism: A single-center experience. The Journal of thoracic and cardiovascular surgery. 2018 Mar:155(3):1095-1106.e2. doi: 10.1016/j.jtcvs.2017.10.139. Epub 2017 Dec 6
[PubMed PMID: 29452460]
[67]
PREPIC Study Group. Eight-year follow-up of patients with permanent vena cava filters in the prevention of pulmonary embolism: the PREPIC (Prevention du Risque d'Embolie Pulmonaire par Interruption Cave) randomized study. Circulation. 2005 Jul 19:112(3):416-22
[PubMed PMID: 16009794]
Level 1 (high-level) evidence
[68]
Agnelli G, Prandoni P, Becattini C, Silingardi M, Taliani MR, Miccio M, Imberti D, Poggio R, Ageno W, Pogliani E, Porro F, Zonzin P, Warfarin Optimal Duration Italian Trial Investigators. Extended oral anticoagulant therapy after a first episode of pulmonary embolism. Annals of internal medicine. 2003 Jul 1:139(1):19-25
[PubMed PMID: 12834314]
[69]
Campbell IA, Bentley DP, Prescott RJ, Routledge PA, Shetty HG, Williamson IJ. Anticoagulation for three versus six months in patients with deep vein thrombosis or pulmonary embolism, or both: randomised trial. BMJ (Clinical research ed.). 2007 Mar 31:334(7595):674
[PubMed PMID: 17289685]
Level 1 (high-level) evidence
[70]
Schulman S, Rhedin AS, Lindmarker P, Carlsson A, Lärfars G, Nicol P, Loogna E, Svensson E, Ljungberg B, Walter H. A comparison of six weeks with six months of oral anticoagulant therapy after a first episode of venous thromboembolism. Duration of Anticoagulation Trial Study Group. The New England journal of medicine. 1995 Jun 22:332(25):1661-5
[PubMed PMID: 7760866]
[71]
Boutitie F, Pinede L, Schulman S, Agnelli G, Raskob G, Julian J, Hirsh J, Kearon C. Influence of preceding length of anticoagulant treatment and initial presentation of venous thromboembolism on risk of recurrence after stopping treatment: analysis of individual participants' data from seven trials. BMJ (Clinical research ed.). 2011 May 24:342():d3036. doi: 10.1136/bmj.d3036. Epub 2011 May 24
[PubMed PMID: 21610040]
[72]
Poli D, Miniati M. The incidence of recurrent venous thromboembolism and chronic thromboembolic pulmonary hypertension following a first episode of pulmonary embolism. Current opinion in pulmonary medicine. 2011 Sep:17(5):392-7. doi: 10.1097/MCP.0b013e328349289a. Epub
[PubMed PMID: 21743331]
Level 3 (low-level) evidence
[73]
Young AM, Marshall A, Thirlwall J, Chapman O, Lokare A, Hill C, Hale D, Dunn JA, Lyman GH, Hutchinson C, MacCallum P, Kakkar A, Hobbs FDR, Petrou S, Dale J, Poole CJ, Maraveyas A, Levine M. Comparison of an Oral Factor Xa Inhibitor With Low Molecular Weight Heparin in Patients With Cancer With Venous Thromboembolism: Results of a Randomized Trial (SELECT-D). Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2018 Jul 10:36(20):2017-2023. doi: 10.1200/JCO.2018.78.8034. Epub 2018 May 10
[PubMed PMID: 29746227]
Level 1 (high-level) evidence
[74]
Rezende SM. Barriers in the diagnosis of pulmonary embolism. The Lancet. Haematology. 2023 Jan:10(1):e11. doi: 10.1016/S2352-3026(22)00359-3. Epub
[PubMed PMID: 36566044]
[75]
Aujesky D, Obrosky DS, Stone RA, Auble TE, Perrier A, Cornuz J, Roy PM, Fine MJ. Derivation and validation of a prognostic model for pulmonary embolism. American journal of respiratory and critical care medicine. 2005 Oct 15:172(8):1041-6
[PubMed PMID: 16020800]
Level 1 (high-level) evidence
[76]
Jiménez D, Aujesky D, Moores L, Gómez V, Lobo JL, Uresandi F, Otero R, Monreal M, Muriel A, Yusen RD, RIETE Investigators. Simplification of the pulmonary embolism severity index for prognostication in patients with acute symptomatic pulmonary embolism. Archives of internal medicine. 2010 Aug 9:170(15):1383-9. doi: 10.1001/archinternmed.2010.199. Epub
[PubMed PMID: 20696966]
[77]
Goliszek S, Wiśniewska M, Kurnicka K, Lichodziejewska B, Ciurzyński M, Kostrubiec M, Gołębiowski M, Babiuch M, Paczynska M, Koć M, Palczewski P, Wyzgał A, Pruszczyk P. Patent foramen ovale increases the risk of acute ischemic stroke in patients with acute pulmonary embolism leading to right ventricular dysfunction. Thrombosis research. 2014 Nov:134(5):1052-6. doi: 10.1016/j.thromres.2014.09.013. Epub 2014 Sep 21
[PubMed PMID: 25282541]
[78]
Sista AK, Miller LE, Kahn SR, Kline JA. Persistent right ventricular dysfunction, functional capacity limitation, exercise intolerance, and quality of life impairment following pulmonary embolism: Systematic review with meta-analysis. Vascular medicine (London, England). 2017 Feb:22(1):37-43. doi: 10.1177/1358863X16670250. Epub 2016 Oct 5
[PubMed PMID: 27707980]
Level 2 (mid-level) evidence
[79]
Højen AA, Nielsen PB, Overvad TF, Albertsen IE, Klok FA, Rolving N, Søgaard M, Ording AG. Long-Term Management of Pulmonary Embolism: A Review of Consequences, Treatment, and Rehabilitation. Journal of clinical medicine. 2022 Oct 10:11(19):. doi: 10.3390/jcm11195970. Epub 2022 Oct 10
[PubMed PMID: 36233833]
[80]
Rosovsky R, Zhao K, Sista A, Rivera-Lebron B, Kabrhel C. Pulmonary embolism response teams: Purpose, evidence for efficacy, and future research directions. Research and practice in thrombosis and haemostasis. 2019 Jul:3(3):315-330. doi: 10.1002/rth2.12216. Epub 2019 Jun 9
[PubMed PMID: 31294318]