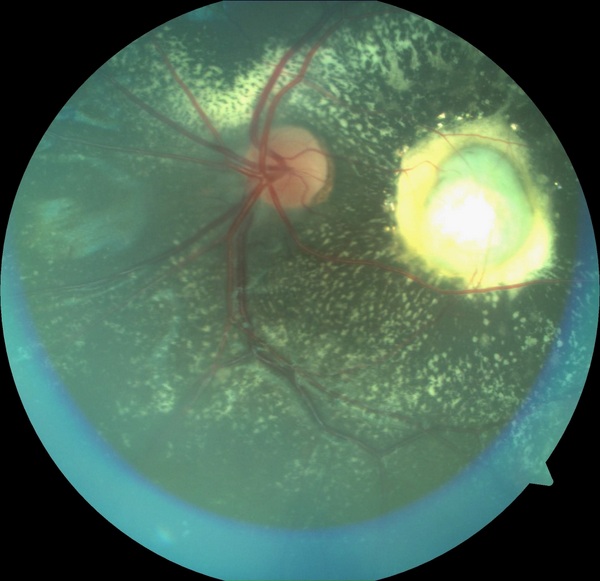
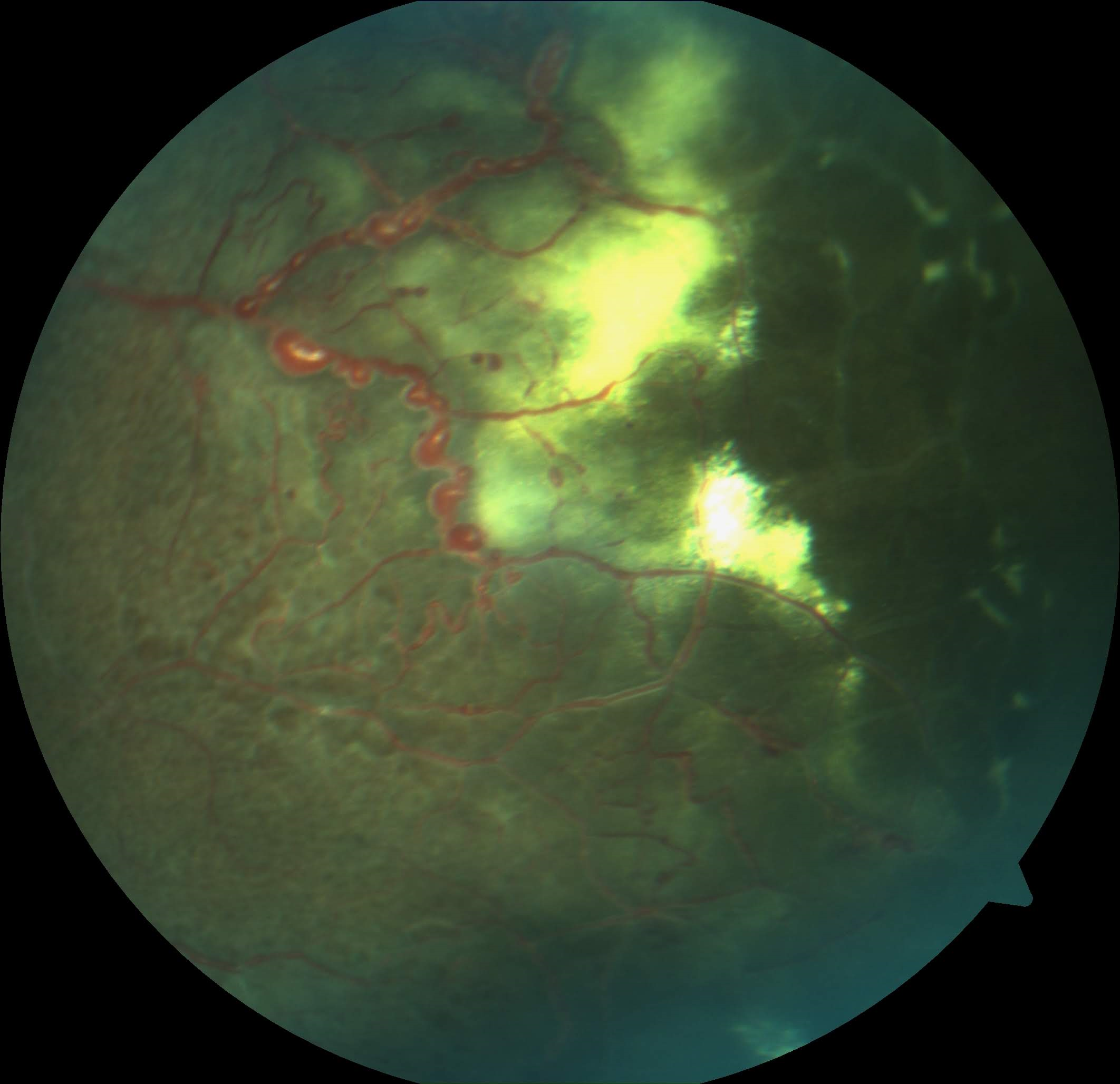
[2]
Shields JA,Shields CL,Honavar SG,Demirci H, Clinical variations and complications of Coats disease in 150 cases: the 2000 Sanford Gifford Memorial Lecture. American journal of ophthalmology. 2001 May;
[PubMed PMID: 11336930]
Level 3 (low-level) evidence
[4]
Sen M, Shields CL, Honavar SG, Shields JA. Coats disease: An overview of classification, management and outcomes. Indian journal of ophthalmology. 2019 Jun:67(6):763-771. doi: 10.4103/ijo.IJO_841_19. Epub
[PubMed PMID: 31124484]
Level 3 (low-level) evidence
[5]
Shields JA,Shields CL,Honavar SG,Demirci H,Cater J, Classification and management of Coats disease: the 2000 Proctor Lecture. American journal of ophthalmology. 2001 May;
[PubMed PMID: 11336931]
[6]
Sigler EJ,Randolph JC,Calzada JI,Wilson MW,Haik BG, Current management of Coats disease. Survey of ophthalmology. 2014 Jan-Feb
[PubMed PMID: 24138893]
Level 3 (low-level) evidence
[7]
Ye X,Wang Y,Cahill H,Yu M,Badea TC,Smallwood PM,Peachey NS,Nathans J, Norrin, frizzled-4, and Lrp5 signaling in endothelial cells controls a genetic program for retinal vascularization. Cell. 2009 Oct 16;
[PubMed PMID: 19837032]
[9]
Dickinson JL,Sale MM,Passmore A,FitzGerald LM,Wheatley CM,Burdon KP,Craig JE,Tengtrisorn S,Carden SM,Maclean H,Mackey DA, Mutations in the NDP gene: contribution to Norrie disease, familial exudative vitreoretinopathy and retinopathy of prematurity. Clinical
[PubMed PMID: 16970763]
[10]
Warden SM,Andreoli CM,Mukai S, The Wnt signaling pathway in familial exudative vitreoretinopathy and Norrie disease. Seminars in ophthalmology. 2007 Oct-Dec;
[PubMed PMID: 18097984]
[11]
Qin M,Hayashi H,Oshima K,Tahira T,Hayashi K,Kondo H, Complexity of the genotype-phenotype correlation in familial exudative vitreoretinopathy with mutations in the LRP5 and/or FZD4 genes. Human mutation. 2005 Aug;
[PubMed PMID: 15981244]
Level 2 (mid-level) evidence
[12]
Jiao X,Ventruto V,Trese MT,Shastry BS,Hejtmancik JF, Autosomal recessive familial exudative vitreoretinopathy is associated with mutations in LRP5. American journal of human genetics. 2004 Nov;
[PubMed PMID: 15346351]
[13]
Chen ZY,Battinelli EM,Fielder A,Bundey S,Sims K,Breakefield XO,Craig IW, A mutation in the Norrie disease gene (NDP) associated with X-linked familial exudative vitreoretinopathy. Nature genetics. 1993 Oct;
[PubMed PMID: 8252044]
[14]
Cremers FP,Maugeri A,den Hollander AI,Hoyng CB, The expanding roles of ABCA4 and CRB1 in inherited blindness. Novartis Foundation symposium. 2004;
[PubMed PMID: 14750597]
[15]
Black GC,Perveen R,Bonshek R,Cahill M,Clayton-Smith J,Lloyd IC,McLeod D, Coats' disease of the retina (unilateral retinal telangiectasis) caused by somatic mutation in the NDP gene: a role for norrin in retinal angiogenesis. Human molecular genetics. 1999 Oct;
[PubMed PMID: 10484772]
[16]
Shastry BS,Trese MT, Overproduction and partial purification of the Norrie disease gene product, norrin, from a recombinant baculovirus. Biochemical and biophysical research communications. 2003 Dec 5;
[PubMed PMID: 14630047]
[17]
Berger W,van de Pol D,Bächner D,Oerlemans F,Winkens H,Hameister H,Wieringa B,Hendriks W,Ropers HH, An animal model for Norrie disease (ND): gene targeting of the mouse ND gene. Human molecular genetics. 1996 Jan;
[PubMed PMID: 8789439]
Level 3 (low-level) evidence
[18]
Luhmann UF,Lin J,Acar N,Lammel S,Feil S,Grimm C,Seeliger MW,Hammes HP,Berger W, Role of the Norrie disease pseudoglioma gene in sprouting angiogenesis during development of the retinal vasculature. Investigative ophthalmology
[PubMed PMID: 16123442]
[19]
Ohlmann A,Scholz M,Goldwich A,Chauhan BK,Hudl K,Ohlmann AV,Zrenner E,Berger W,Cvekl A,Seeliger MW,Tamm ER, Ectopic norrin induces growth of ocular capillaries and restores normal retinal angiogenesis in Norrie disease mutant mice. The Journal of neuroscience : the official journal of the Society for Neuroscience. 2005 Feb 16;
[PubMed PMID: 15716406]
[20]
Sohn EH,Michaelides M,Bird AC,Roberts CJ,Moore AT,Smyth D,Brady AF,Hungerford JL, Novel mutation in PANK2 associated with retinal telangiectasis. The British journal of ophthalmology. 2011 Jan
[PubMed PMID: 20974629]
[21]
Morris B,Foot B,Mulvihill A, A population-based study of Coats disease in the United Kingdom I: epidemiology and clinical features at diagnosis. Eye (London, England). 2010 Dec;
[PubMed PMID: 20865031]
[22]
Campbell FP, Coats' disease and congenital vascular retinopathy. Transactions of the American Ophthalmological Society. 1976;
[PubMed PMID: 325857]
[23]
Rishi E,Rishi P,Appukuttan B,Uparkar M,Sharma T,Gopal L, Coats' disease of adult-onset in 48 eyes. Indian journal of ophthalmology. 2016 Jul
[PubMed PMID: 27609165]
[24]
Egbert PR,Chan CC,Winter FC, Flat preparations of the retinal vessels in Coats' disease. Journal of pediatric ophthalmology. 1976 Nov-Dec;
[PubMed PMID: 798026]
[25]
Fernandes BF,Odashiro AN,Maloney S,Zajdenweber ME,Lopes AG,Burnier MN Jr, Clinical-histopathological correlation in a case of Coats' disease. Diagnostic pathology. 2006 Aug 30;
[PubMed PMID: 16942617]
Level 3 (low-level) evidence
[26]
Tripathi R,Ashton N, Electron microscopical study of Coat's disease. The British journal of ophthalmology. 1971 May;
[PubMed PMID: 4102954]
[27]
DUKE JR,WOODS AC, COATS'S DISEASE. II. STUDIES ON THE IDENTITY OF THE LIPIDS CONCERNED, AND THE PROBABLE ROLE OF MUCOPOLYSACCHARIDES IN ITS PATHOGENESIS. The British journal of ophthalmology. 1963 Jul;
[PubMed PMID: 14189711]
[28]
Chang MM,McLean IW,Merritt JC, Coats' disease: a study of 62 histologically confirmed cases. Journal of pediatric ophthalmology and strabismus. 1984 Sep-Oct;
[PubMed PMID: 6502405]
Level 3 (low-level) evidence
[29]
Farkas TG,Potts AM,Boone C, Some pathologic and biochemical aspects of Coats' disease. American journal of ophthalmology. 1973 Feb;
[PubMed PMID: 4348894]
[31]
Kase S,Rao NA,Yoshikawa H,Fukuhara J,Noda K,Kanda A,Ishida S, Expression of vascular endothelial growth factor in eyes with Coats' disease. Investigative ophthalmology
[PubMed PMID: 23221067]
[32]
Gupta N,Beri S,D'souza P, Cholesterolosis Bulbi of the Anterior Chamber in Coats Disease. Journal of pediatric ophthalmology and strabismus. 2009 Jun 25;
[PubMed PMID: 19645389]
[33]
Shields JA,Eagle RC Jr,Fammartino J,Shields CL,De Potter P, Coats' disease as a cause of anterior chamber cholesterolosis. Archives of ophthalmology (Chicago, Ill. : 1960). 1995 Aug;
[PubMed PMID: 7639669]
[34]
Reichstein DA,Recchia FM, Coats disease and exudative retinopathy. International ophthalmology clinics. 2011 Winter;
[PubMed PMID: 21139479]
[35]
Friedenwald H,Friedenwald JS, Terminal Stage in a Case of Retinitis with Massive Exudation. Transactions of the American Ophthalmological Society. 1929;
[PubMed PMID: 16692828]
Level 3 (low-level) evidence
[36]
Naumann GD,Portwich E, [Etiology and final clinical cause for 1000 enucleations. (A clinico-pathologic study) (author's transl)]. Klinische Monatsblatter fur Augenheilkunde. 1976 May;
[PubMed PMID: 957559]
[37]
Tripathy K,Chawla R,Temkar S,Sagar P,Kashyap S,Pushker N,Sharma YR, Phthisis Bulbi-a Clinicopathological Perspective. Seminars in ophthalmology. 2018
[PubMed PMID: 29902388]
Level 3 (low-level) evidence
[39]
Cunha-Vaz JG, The blood-retinal barriers. Documenta ophthalmologica. Advances in ophthalmology. 1976 Oct 15;
[PubMed PMID: 1009819]
Level 3 (low-level) evidence
[40]
Ridley ME,Shields JA,Brown GC,Tasman W, Coats' disease. Evaluation of management. Ophthalmology. 1982 Dec;
[PubMed PMID: 6891766]
[41]
Tarkkanen A,Laatikainen L, Coat's disease: clinical, angiographic, histopathological findings and clinical management. The British journal of ophthalmology. 1983 Nov;
[PubMed PMID: 6685529]
[42]
Kang KB,Wessel MM,Tong J,D'Amico DJ,Chan RV, Ultra-widefield imaging for the management of pediatric retinal diseases. Journal of pediatric ophthalmology and strabismus. 2013 Sep-Oct;
[PubMed PMID: 23739460]
[43]
Shields CL,Udyaver S,Dalvin LA,Lim LS,Atalay HT,L Khoo CT,Mazloumi M,Shields JA, Coats disease in 351 eyes: Analysis of features and outcomes over 45 years (by decade) at a single center. Indian journal of ophthalmology. 2019 Jun;
[PubMed PMID: 31124485]
[44]
Ong SS,Mruthyunjaya P,Stinnett S,Vajzovic L,Toth CA, Macular Features on Spectral-Domain Optical Coherence Tomography Imaging Associated With Visual Acuity in Coats' Disease. Investigative ophthalmology & visual science. 2018 Jun 1
[PubMed PMID: 30025132]
[45]
Ashkenazy N,Acon D,Kalavar M,Berrocal AM, Optical coherence tomography angiography and multimodal imaging in the management of coats' disease. American journal of ophthalmology case reports. 2021 Sep
[PubMed PMID: 34381924]
Level 3 (low-level) evidence
[46]
Hautz W,Gołębiewska J,Kocyła-Karczmarewicz B, Optical Coherence Tomography and Optical Coherence Tomography Angiography in Monitoring Coats' Disease. Journal of ophthalmology. 2017
[PubMed PMID: 28377823]
[47]
Muakkassa NW,de Carlo TE,Choudhry N,Duker JS,Baumal CR, Optical Coherence Tomography Angiography Findings in Coats' Disease. Ophthalmic surgery, lasers & imaging retina. 2016 Jul 1
[PubMed PMID: 27434894]
[48]
Eisenberg L,Castillo M,Kwock L,Mukherji SK,Wallace DK, Proton MR spectroscopy in Coats disease. AJNR. American journal of neuroradiology. 1997 Apr;
[PubMed PMID: 9127038]
[49]
Potter PD,Shields CL,Shields JA,Flanders AE, The role of magnetic resonance imaging in children with intraocular tumors and simulating lesions. Ophthalmology. 1996 Nov;
[PubMed PMID: 8942869]
[50]
Senft SH,Hidayat AA,Cavender JC, Atypical presentation of Coats disease. Retina (Philadelphia, Pa.). 1994;
[PubMed PMID: 8016459]
[51]
Schefler AC,Berrocal AM,Murray TG, Advanced Coats' disease. Management with repetitive aggressive laser ablation therapy. Retina (Philadelphia, Pa.). 2008 Mar;
[PubMed PMID: 18317343]
[52]
Spitznas M,Joussen F,Wessing A, Treatment of Coats' disease with photocoagulation. Albrecht von Graefes Archiv fur klinische und experimentelle Ophthalmologie. Albrecht von Graefe's archive for clinical and experimental ophthalmology. 1976 Apr 1;
[PubMed PMID: 1083682]
[53]
Shapiro MJ,Chow CC,Karth PA,Kiernan DF,Blair MP, Effects of green diode laser in the treatment of pediatric Coats disease. American journal of ophthalmology. 2011 Apr;
[PubMed PMID: 21257148]
[54]
Shields JA,Shields CL, Review: coats disease: the 2001 LuEsther T. Mertz lecture. Retina (Philadelphia, Pa.). 2002 Feb;
[PubMed PMID: 11884883]
[55]
Harris GS, Coats' disease, diagnosis and treatment. Canadian journal of ophthalmology. Journal canadien d'ophtalmologie. 1970 Oct;
[PubMed PMID: 5481542]
[56]
Othman IS,Moussa M,Bouhaimed M, Management of lipid exudates in Coats disease by adjuvant intravitreal triamcinolone: effects and complications. The British journal of ophthalmology. 2010 May;
[PubMed PMID: 19955197]
[57]
Jarin RR,Teoh SC,Lim TH, Resolution of severe macular oedema in adult Coat's syndrome with high-dose intravitreal triamcinolone acetonide. Eye (London, England). 2006 Feb;
[PubMed PMID: 15723038]
[58]
Lei S,Lam WC, Efficacy and safety of dexamethasone intravitreal implant for refractory macular edema in children. Canadian journal of ophthalmology. Journal canadien d'ophtalmologie. 2015 Jun;
[PubMed PMID: 26040225]
[59]
Ghazi NG,Al Shamsi H,Larsson J,Abboud E, Intravitreal triamcinolone in Coats' disease. Ophthalmology. 2012 Mar;
[PubMed PMID: 22385486]
[60]
Entezari M,Ramezani A,Safavizadeh L,Bassirnia N, Resolution of macular edema in Coats' disease with intravitreal bevacizumab. Indian journal of ophthalmology. 2010 Jan-Feb;
[PubMed PMID: 20029156]
[61]
Alvarez-Rivera LG,Abraham-Marín ML,Flores-Orta HJ,Mayorquín-Ruiz M,Cortés-Luna CF, [Coat's disease treated with bevacizumab (Avastin)]. Archivos de la Sociedad Espanola de Oftalmologia. 2008 May;
[PubMed PMID: 18464184]
[62]
Lin CJ,Hwang JF,Chen YT,Chen SN, The effect of intravitreal bevacizumab in the treatment of Coats disease in children. Retina (Philadelphia, Pa.). 2010 Apr;
[PubMed PMID: 19996822]
[63]
Gaillard MC,Mataftsi A,Balmer A,Houghton S,Munier FL, ranibizumab in the management of advanced Coats disease Stages 3B and 4: long-term outcomes. Retina (Philadelphia, Pa.). 2014 Nov;
[PubMed PMID: 25075562]
[64]
Zheng XX,Jiang YR, The effect of intravitreal bevacizumab injection as the initial treatment for Coats' disease. Graefe's archive for clinical and experimental ophthalmology = Albrecht von Graefes Archiv fur klinische und experimentelle Ophthalmologie. 2014 Jan;
[PubMed PMID: 23873253]
[65]
Li S,Deng G,Liu J,Ma Y,Lu H, The effects of a treatment combination of anti-VEGF injections, laser coagulation and cryotherapy on patients with type 3 Coat's disease. BMC ophthalmology. 2017 May 22
[PubMed PMID: 28532448]
[69]
Jiang Y,Lim J,Janowicz M, Cholesterol Crystals Secondary to Coats-Like Response With Retinitis Pigmentosa. JAMA ophthalmology. 2017 Oct 12
[PubMed PMID: 29049676]
[70]
Verma S,Bhatia I,Banerjee M,Kumar V, Coats like response in healed choroiditis. Ocular immunology and inflammation. 2021 Apr 2
[PubMed PMID: 33797302]
[71]
Nair N,Kummamuri S,Sudharshan S,Khetan V, Coats-like response in tubercular subretinal abscess masquerading as melanoma as the initial manifestation of miliary tuberculosis. Indian journal of ophthalmology. 2020 Sep
[PubMed PMID: 32823473]
[72]
Chaudhary P,Varshney A,Videkar C, Secondary Coats' response to retinal vasculitis managed with bevacizumab. Indian journal of ophthalmology. 2018 Sep
[PubMed PMID: 30127171]
[73]
Suh DW,Pulido JS,Jampol LM,Chong LP,Thomas M, Coats'-like response in pars planitis. Retina (Philadelphia, Pa.). 1999;
[PubMed PMID: 10048380]
[75]
Chen PP,Chong LP, Coats'-like response in a patient with pars planitis. The British journal of ophthalmology. 1996 Jul
[PubMed PMID: 8795387]
[76]
Kumar V,Tewari R,Chandra P,Kumar A, Ultra wide field imaging of coats like response in Leber's congenital amaurosis. Saudi journal of ophthalmology : official journal of the Saudi Ophthalmological Society. 2017 Apr-Jun
[PubMed PMID: 28559726]
[77]
Agarwal A,Pilania RK,Anjani G,Choudhary H,Gupta A,Gupta V, Retinal Vasculitis With Coats-Like Response in a Young Girl With Parry-Romberg Syndrome. Journal of clinical rheumatology : practical reports on rheumatic & musculoskeletal diseases. 2021 Dec 1
[PubMed PMID: 31124919]
[78]
Fan KC,McAllister MA,Yannuzzi NA,Patel NA,Prakhunhungsit S,Negron CI,Capó H,Berrocal AM, X-Linked Retinoschisis and a Coats-Like Response in the Setting of Retinopathy of Prematurity. Journal of vitreoretinal diseases. 2020 Nov 1;
[PubMed PMID: 34622118]
[79]
Gelisken F,Sherif Adel S,Inhoffen W,Bartz-Schmidt KU, Coats'-like response in blood-filled senile retinoschisis. Ophthalmologica. Journal international d'ophtalmologie. International journal of ophthalmology. Zeitschrift fur Augenheilkunde. 2002 Sep-Oct;
[PubMed PMID: 12424408]
[80]
Behboudi H,Zayeni H,Haji-Abbasi A,Moravvej Z,Azaripour E,Alizadeh Y,Soltani-Moghadam R, Coats'-like Response Associated with Linear Scleroderma. Journal of ophthalmic & vision research. 2022 Jan-Mar
[PubMed PMID: 35194504]
[81]
George MK,Bernardino CR,Huang JJ, Coats-like response in linear en coup de sabre scleroderma. Retinal cases & brief reports. 2011 Fall
[PubMed PMID: 25390412]
Level 3 (low-level) evidence
[82]
Gass JD,Blodi BA, Idiopathic juxtafoveolar retinal telangiectasis. Update of classification and follow-up study. Ophthalmology. 1993 Oct
[PubMed PMID: 8414413]
[83]
Yannuzzi LA,Bardal AM,Freund KB,Chen KJ,Eandi CM,Blodi B, Idiopathic macular telangiectasia. Archives of ophthalmology (Chicago, Ill. : 1960). 2006 Apr
[PubMed PMID: 16606869]
[84]
Tran L,Papasavvas I,Fleischhauer J,Herbort CP Jr, Peripheral Inflammatory Yellow Exudative Retinal Coats-Like Vitreoretinopathy Misdiagnosed as Acute Retinal Necrosis in a Retinitis Pigmentosa Patient after Cataract Surgery. Case reports in ophthalmology. 2021 May-Aug
[PubMed PMID: 34248592]
Level 3 (low-level) evidence
[86]
Fitzsimons RB,Gurwin EB,Bird AC, Retinal vascular abnormalities in facioscapulohumeral muscular dystrophy. A general association with genetic and therapeutic implications. Brain : a journal of neurology. 1987 Jun
[PubMed PMID: 3580827]
[87]
Vance SK,Wald KJ,Sherman J,Freund KB, Subclinical facioscapulohumeral muscular dystrophy masquerading as bilateral Coats disease in a woman. Archives of ophthalmology (Chicago, Ill. : 1960). 2011 Jun
[PubMed PMID: 21670353]
[88]
Cahill M,O'Keefe M,Acheson R,Mulvihill A,Wallace D,Mooney D, Classification of the spectrum of Coats' disease as subtypes of idiopathic retinal telangiectasis with exudation. Acta ophthalmologica Scandinavica. 2001 Dec;
[PubMed PMID: 11782226]
[89]
Shields CL,Udyaver S,Dalvin LA,Lim LS,Atalay HT,Khoo C,Mazloumi M,Shields JA, Visual acuity outcomes in Coats disease by classification stage in 160 patients. The British journal of ophthalmology. 2020 Mar;
[PubMed PMID: 31177186]
[90]
Egerer I,Tasman W,Tomer TT, Coats disease. Archives of ophthalmology (Chicago, Ill. : 1960). 1974 Aug;
[PubMed PMID: 4847500]
[92]
Shanmugam MP,Shah PN,Sagar P,Ramanjulu R,Mishra DK, Coats' disease - Prognostic factors for globe and vision salvage in children, a long-term experience. Indian journal of ophthalmology. 2022 Feb
[PubMed PMID: 35086222]
[93]
Judisch GF,Apple DJ, Orbital cellulitis in an infant secondary to Coats' disease. Archives of ophthalmology (Chicago, Ill. : 1960). 1980 Nov
[PubMed PMID: 7436836]