Introduction
The uterus is a hollow organ that provides protective and nutritive support to the ovum from the moment it is fertilized until it grows into a well-developed fetus ready for parturition. Its middle muscular layer is called the myometrium, which is known for its rhythmic contractions which result in 'endometrial waves' in the nonpregnant uterus, Braxton Hicks contractions during pregnancy, and true labor towards the end of the third trimester. Labor contractions are painful, regular, and present with a change in cervical dilation and/or effacement.
Issues of Concern
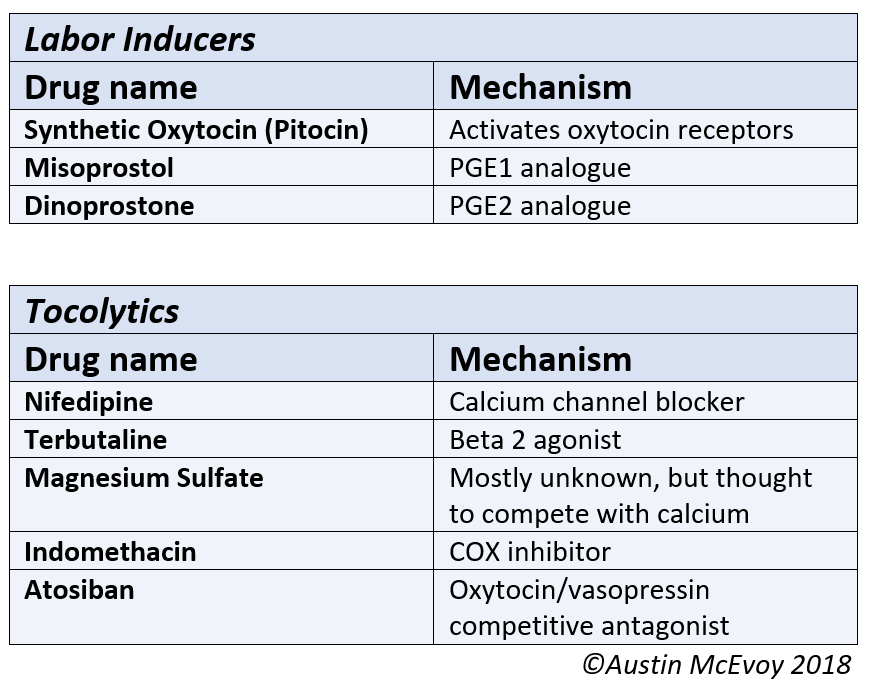
Since uterine contractions are always present in a thriving uterus, they are differentiated based on frequency, amplitude, duration, and direction of propagation. Although there have been recent advances in knowledge regarding uterine contractions, there remains a huge gap in understanding the related physiology at the cellular and molecular levels. Knowledge of the process of uterine contractions that eventually leads to the expulsion of the baby helps clinicians identify abnormalities that may lead to obstetrical complications, such as preterm labor and arrest of labor. This also aids the pharmacists in developing and improving the drugs used for labor augmentation, induction, and tocolysis (see Image. Medications Used to Manage Abnormal Labor). Uterine contractions also play an essential role in minimizing postpartum bleeding, which explains why many drugs used to treat this complication target the pathway involved in myometrial contractility.
Cellular Level
Uterine myocytes are smooth muscle cells packed heavily with myofilaments, dense bodies, and dense bands. These cells have more actin than myosin by a factor of 6. The dense bodies attach actin filaments, ensuring the contraction results in a force along the longitudinal direction of the myocyte. The actin filaments of the contractile machinery are attached to the cytoskeleton with the help of dense bands. This transmits the forces from the contractile units toward the plasma membrane, leading to a shortening of the myocyte.
The myosin present in these cells is classified as MII. It is a hexamer molecule of 2 myosin heavy chains (MHC) and 2 pairs of myosin light chains (MLC). It forms 3 basic domains. The ‘head’ region comprises the globular N-terminal end of the MHCs that protrudes laterally from the filament. It has the actin-binding region and the ATP hydrolysis site that gives the energy needed for contraction. This is converted into a larger movement due to the stiff 'neck' domain at the C-terminal end of the motor region.[1] The 'neck' domain is also where myosin light chains bind non-covalently. The ‘tail’ domain comprises the C-terminal ends of the myosin-heavy chains, which are intertwined in an α-helical rod and form the major constituents of the thick filaments of the myocytes.
Intermediate filaments present in myocytes give shape to the cell and help signal transduction and spatiotemporal organization of the cell, augmenting the tension created by the contractile action. The cells communicate with one another through connections called gap junctions which increase towards the end of pregnancy to help in synchronous contractions. As the uterus hypertrophies, the amount of actin and myosin in myocytes increases in pregnancy. Phosphocreatine, glycogen, and fatty acid deposits are increased to provide energy for contractions. The blood flow to the uterus during the pregnancy rises in sync with the growing need for perfusion but later lags towards the end of pregnancy.
Development
The myometrium has 2 layers. The circular layer is thinner and present at the innermost aspect of muscle fibers and is derived from the paramesonephric/Mullerian ducts. It is called sub-endometrial or junctional endometrium. The outer longitudinal layer comprises intertwined muscle bundles embedded in an extracellular matrix of highly vascular collagen fibers. This helps build the intense pressure needed for contractions. The longitudinal layer originates from non-Mullerian tissue. Both layers work together to expel the fetus from the uterine cavity. Some studies have suggested that myometrial cells exhibit "pacemaker" capabilities, such as those in the gut and urethra, that facilitate synchronous uterine contractions, although the evidence for this is inconsistent.[2]
Function
The primary function of uterine contractions is to expel the fetus from the uterine cavity. However, contractions also play an essential role in minimizing postpartum hemorrhage. Knowing the normal physiology of uterine contractions also allows clinicians to better differentiate between the true onset of labor and prodromal labor, also known as Braxton Hicks contractions. Braxton Hicks contractions occur sporadically and do not increase in strength. They are irregular in duration, frequency, and intensity, are unpredictable and non-rhythmic, and are more uncomfortable than painful. True labor consists of contractions at regular intervals. As labor progresses, these contractions become stronger, and the time between each contraction decreases. The first stage of labor is divided into 2 phases, defined by the degree of cervical dilation. The latent phase is during the dilation from 0 to 6 cm, while the active phase starts from 6 cm to full cervical dilation of 10 cm. The second stage of labor starts with cervical dilation of 10cm and ends with the delivery of the baby. The third stage of labor starts when the fetus is delivered and ends with the delivery of the placenta.
Mechanism
Electrophysiology of Contractions
Resting membrane potential is due to the electrochemical gradient, mainly created by the potassium ions concentrated inside the cell and, to a lesser extent, by the calcium, sodium, and chloride ions concentrated extracellularly. Two types of Action Potentials have been observed in uterine myocytes: depolarization followed by rapid repolarization and the other with initial depolarization with a sustained plateau. It is the release of calcium ions from intracellular stores, as well as the influx of calcium ions from the extracellular space, that initiates the action potentials. When the membrane potential is depolarized to about −40 mV, the L-type voltage-operated Calcium channels open, causing the influx of calcium ions. T-type Ca channels are also found in uterine myocytes and have a role in the propagation of action potentials.[3]
Interaction of the uterine agonists with the GPCR located on the plasma membrane of myocyte leads to a chain of events ultimately causing the IP–IPR mediated Ca release from the Sarcoplasmic Reticulum. This further elevated the concentration of calcium in the cytosol as well as the voltage of the plasma membrane. Another process causing the propagation of action potential is the positive feedback mechanism of calcium, known as the Ca-induced Ca release (CICR), whereby increasing the intracellular concentration of calcium stimulates other Ca channels to open. Perhaps the Store-operated Ca entry (SOCE) pathway is the most poorly understood. When the intracellular stores of Ca in the SR are emptied, a ‘calcium influx factor’ (CIF) is released, which causes the plasma membrane to allow the influx of extracellular Ca. Lipid rafts, termed as ‘caveolae’ which are stabilized by a scaffolding protein, caveolin, present on the plasma membrane of myocytes, are also involved in signal transduction and myocyte excitability.
After the influx of calcium ions and their subsequent binding to calmodulin, a conformational change in Myosin Light Chain Kinase leads to an increase in phosphorylation of myosin light chain at serine residue 19, which causes myosin cross-bridge to attach to the actin filament and bring about the contraction. The relaxation of myocytes is brought about by the efflux of calcium ions out of the cytosol into the SR and extracellular space. Plasma membrane Ca-ATPase (PMCA) and the SR/ER Ca-ATPase (SERCA) belong to the P-type Ca-ATPases family, are responsible for moving 1 Ca ion out of the intracellular compartment in exchange for an H+ restore the resting membrane potential. Another mechanism involves the Na/Ca exchanger (NCX), with the virtue of the electrochemical gradient of Na created by the Na/K ATPase. Interestingly, calcium itself causes calcium ion clearance: Calcium ions inhibit L-type calcium channels, and the calcium-calmodulin complex also stimulates CaM-Kinase II (CaMKII), which inhibits L-type calcium channels.
Calcium Sensitization and Desensitization
The increased sensitivity of regulatory and contractile proteins to calcium, eventually resulting in stronger contraction, is known as Calcium Sensitization after stimulation with an agonist. The vice versa is true for calcium desensitization. This is established through the RhoA/Rho-kinase signaling pathway. Activation of G-protein coupled receptors (GPCRs) causes the recruitment of RhoA to the plasma membrane after exchanging GDP for GTP. This activates the Rho-associated kinase (ROK), which phosphorylates the myosin targeting subunit (MYPT1) of Myosin Light Chain Phosphatase, preventing the dephosphorylation of Myosin Light Chain. The mRNAs for RhoA, ROK-1, and ROK-2 are present in the non-gravid uterus and increase during pregnancy. ROK inhibition hampers force development and promotes relaxation without altering the level of [Ca] in spontaneous and agonist-stimulated contractions. Similarly, cAMP causes phosphorylation of MLCK itself by a cAMP-dependent protein kinase, which decreases enzymatic activity by decreasing the affinity of the MLCK for the calmodulin-calcium complex. Several receptors are found on the surface of myometrial cells that affect contractility. These include:
- Oxytocin receptors: Contractility agonist
- Estrogen receptors: Contractility agonist
- Progesterone receptors: Contractility antagonist
- Beta2 adrenergic receptors cause an increase in cAMP levels, hence contractility antagonist
- Alpha-adrenergic receptors cause a decrease in cAMP levels, hence contractility agonist
These receptors are the targets of multiple drugs used to manage abnormal labor. The exact sequence of events that leads to uterine contractions is still largely unknown. Some studies suggest mechanical stretch and hormones work together to initiate contractions in normal labor.[2] However, due to the role of inflammation in preterm labor, other studies suggest that inflammatory mediators, such as cytokines and prostaglandins, initiate uterine contractions.[4]
Mechanical Stretch
Mechanical stretch refers to tension on myometrial cells as the uterus distends. The physical stretching of the uterus results in an influx of ions, namely sodium and calcium, that change the action potential across myometrial cells.[5][6] This change facilitates the onset of uterine contractions. The number of connexins (gap junctions) between myometrial cells increases before labor, allowing coordinated muscle contraction. These cell-to-cell contacts are less abundant early in pregnancy, favoring uterine quiescence.[7] There has also been a suggestion that uterine overdistention causes an "inflammatory pulse" that further activates myometrial contractility.[8]
Inflammatory Mediators
Many studies suggest that inflammation is a mediator of labor.[8][9] Inflammatory markers most notably include prostaglandins, which increase in concentration before the onset of labor due to a functional progesterone withdrawal, leading to an increased estrogen-to-progesterone ratio.[10] The amnion and chorion produce PGE2, and the decidua produces PGE2 and PGF2.
The 2 most studied prostaglandins involved in uterine contractions include prostaglandin E1 (PGE1) and prostaglandin E2 (PGE2.) They stimulate myometrial contractility, most likely by acting as calcium ionophores, leading to an increase in intracellular calcium. Misoprostol is the synthetic version of PGE1, which, although initially designed to prevent peptic ulcers, proved to have a dose-dependent effect on myometrial contractility.[10]
PGE2 also plays a role in uterine contractions by activating EP1 and EP3 receptors on myometrial cells.[11] However, the major physiological effect of PGE2 during labor is the activation of inflammatory mediators IL-8 and TNF-alpha that activate collagenases and MMPs, leading to the ripening of the cervix.[10]
Prostaglandin F2 alpha (PGF2a) is less studied, but it is thought to decrease progesterone levels and independently increase uterine contractility by stimulating smooth muscle cells.[12] Prostaglandins also play a role in uterine contractions after the delivery of the fetus. During this time, also known as stage 3 of labor, the placenta secretes prostaglandins that lead to its detachment from the endometrial cavity.[13] Contractions during this period also minimize postpartum bleeding. Lack of contractions during this period can occur due to uterine atony.
Hormones
The primary hormones involved include estrogen, progesterone, and oxytocin. Oxytocin is one of the most widely studied hormones involved in uterine contractions. It decreases Ca2+ efflux by inhibiting the Ca2+/ATPase of the myometrial cell membrane, which pumps calcium from the inside to the extracellular space, and increases Ca2+ influx, as well as causes the release of Ca2+ from the SR via IICR. Studies suggest that the increased estrogen-to-progesterone ratio that occurs before the onset of labor leads to an increased number of oxytocin receptors in the uterus.[14] Many animal studies also show an increase in oxytocin concentration before labor; however, there is limited evidence regarding this phenomenon in humans due to technical difficulties in obtaining oxytocin levels in laboring women.[15]
Oxytocin: The process starts with the hypothalamus, paraventricular and supraoptic nuclei. These nuclei generate and secrete oxytocin, the major contributor being the paraventricular nucleus. Oxytocin then travels to the posterior pituitary via the hypothalamic-hypophysial tract. After oxytocin arrives in the posterior pituitary, it accesses the systemic blood supply, which delivers the oxytocin molecule to oxytocin receptors on myometrial cells. The uterine epithelial lining also makes oxytocin during pregnancy, acting in an autocrine and paracrine fashion.[16][17] The positive feedback mechanism of oxytocin further catalyzes the onset of labor.[15][18][19]. These receptors are rhodopsin class 1 G proteins that couple with phospholipase C (PLC), which then activates inositol triphosphate (IP3) and diacylglycerol (DAG). Activated IP3 mobilizes calcium from the sarcoplasmic reticulum, which binds to myosin light chain kinases, resulting in smooth muscle contraction.[15]
Progesterone: It decreases permeability for calcium, sodium, and potassium, as well as modulates the intracellular calcium-binding that makes less calcium available for the calmodulin-MLCK system by increasing the rate of cAMP synthesis. It is vital for the maintenance of pregnancy as it causes uterine relaxation during early pregnancy, and its functional withdrawal leads to an increased estrogen-to-progesterone ratio that causes an increase in prostaglandin concentration, triggering labor.[20]
Related Testing
External tocometers and intrauterine pressure catheters are the primary clinical methods of monitoring myometrial activity. Although both devices allow visualization of contractions relative to the fetal heart rate, only intrauterine pressure catheters enable precise measurement of the strength of uterine contractions. There should be 3 to 5 contractions in the 10-minute window, each lasting 30 to 40 seconds. The monitoring of uterine contractions should be continuous during labor.
External Tocometer
Two probes are placed on the woman’s abdomen, 1 over the uterine fundus and the other in proximity to the fetal heart. This approach is the standard method of monitoring uterine contractions in laboring women.
Intrauterine Pressure Catheter
A thin catheter is inserted into the uterus and monitors changes in pressure. The standard unit of measurement is the Montevideo unit (MVU). Adequate contractions are defined as a total of 200 MVU within 10 minutes. Internal monitoring has limited use because it requires the rupture of fetal membranes for placement. It is commonly used with a fetal scalp electrode that monitors fetal heart rate.
Pathophysiology
Chorioamnionitis is an acute inflammation of the membranes and chorion of the placenta, typically due to ascending polymicrobial bacterial infection in the setting of membrane rupture.[21] It is associated with an elevation in prostaglandin levels. As PG stimulates myometrial contractility and softening of the cervix, it leads to the induction of preterm labor.[22]
Protracted stages of labor indicate that cervical change is occurring at a slower pace than expected in the presence or absence of adequate contractions. Arrest means complete cessation of labor's progress. It can be elaborated as the absence of cervical change for more than 4 hours in the presence of adequate contractions or the absence of cervical change for more than 6 hours in the presence of inadequate contractions. Abnormal third-stage labor is placenta retention for more than 30 minutes.
Clinical Significance
Understanding the physiology of uterine contractions allows clinicians to use targeted therapy for both the induction and cessation of labor. Commonly used medications for induction of labor include oxytocin, misoprostol, and dinoprostone. Because uterine contractions dually function to minimize postpartum bleeding, these are the same medications used to manage postpartum hemorrhage. Carboprost, an analog of PGF2a, is an additional medication used to manage postpartum hemorrhage, not used for the induction of labor.
Oxytocin is commonly used in modern obstetric practice to increase contractions when the labor process has failed to promote vaginal delivery.[23] Prostaglandins are active contractile agents, and their analogs are combined with mifepristone (an antiprogestin)for uterine evacuation during early and second-trimester pregnancy.[24][25][24] Misoprostol is cheaper than gemeprost and does not need refrigeration, so it is the prostaglandin of choice.
In 2013, the FDA approved the use of progesterone supplementation (hydroxyprogesterone caproate) during pregnancy to reduce the risk of recurrent preterm birth in women with a history of at least 1 prior spontaneous preterm delivery.[24] It is protective against recurrent preterm birth as the rate of recurrent preterm birth with its use decreases to 25% to 31% compared with 33% to 47% in placebo controls.[25]
Medications used to stop preterm labor antagonize uterine contractions. Commonly used tocolytics include nifedipine, terbutaline, magnesium sulfate, indomethacin, and atosiban. Figure 1 summarizes the medications used for induction of labor and tocolysis. Of these tocolytic agents, beta sympathomimetics, oxytocin receptor antagonists, and calcium channel blockers have the most promising data regarding efficacy.[26] Magnesium sulfate is a weak tocolytic but not used for tocolysis. It is administered to lower the risk of neurologic comorbidities in babies born at <32 weeks gestation. There is evidence that NSAIDs and CCBs are better than magnesium and beta sympathomimetics in terms of neonatal and maternal outcomes and maternal complications.[27] However, NSAIDs may lead to side effects such as oligohydramnios, renal failure, necrotizing enterocolitis, intraventricular hemorrhage, and closure of the patent ductus arteriosus.[28] Hence, calcium channel blockers are the drug of choice for tocolysis beyond 32 weeks gestation. CCBs can lead to maternal hypotension and tachycardia. Tocolytics are not recommended beyond 34 weeks gestation as their risks outweigh the risks of premature delivery.
Understanding the normal pattern of contractions is also useful in defining the arrest of labor, which has different management based on the stage. For example, an arrested latent phase is an indication of augmentation with oxytocin, while an arrested active phase is an indication of the cesarean section. Defining the true arrest of labor requires analysis of the adequacy of uterine contractions, which requires the use of an intrauterine pressure catheter. Active phase arrest has specific requirements, which include 4 or more hours of adequate contractions or 6 or more hours of inadequate contractions. These definitions are essential for clinicians because they dictate management algorithms.