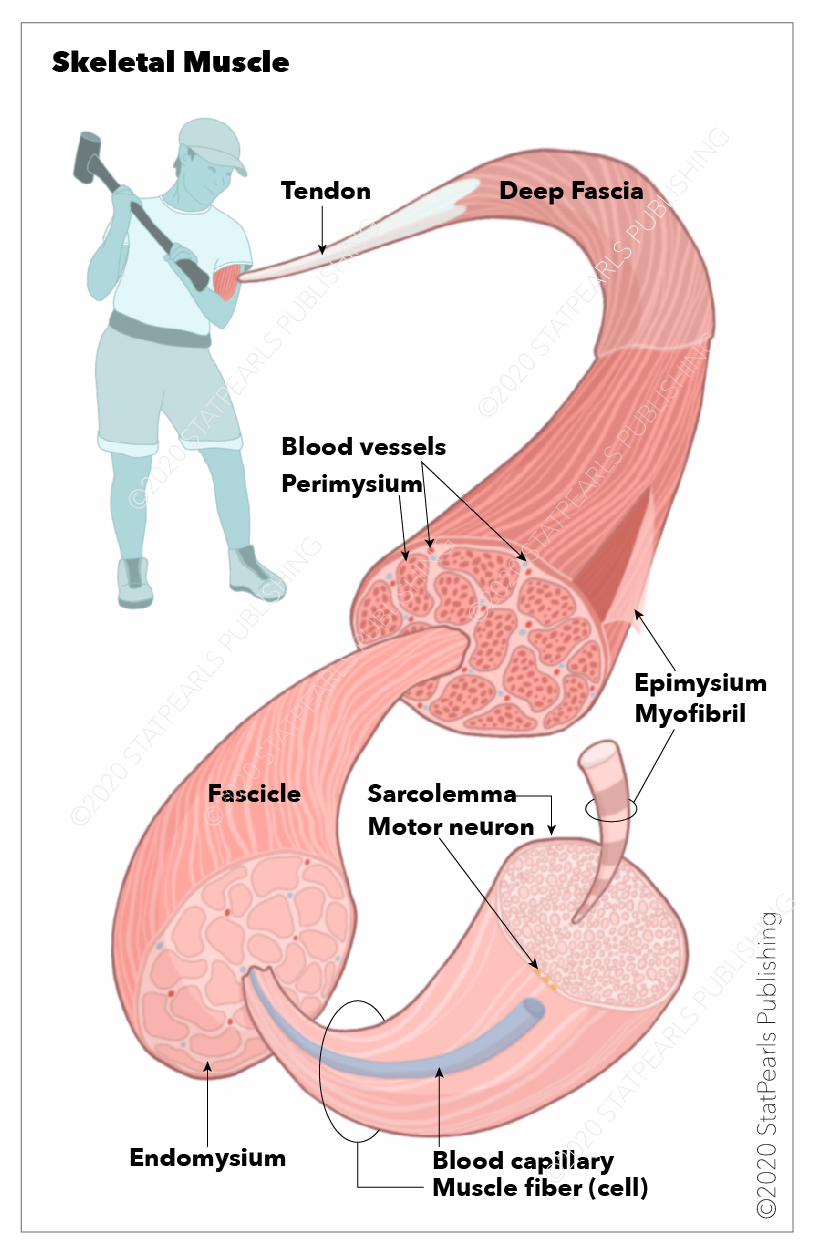
[1]
Goodman CA, Hornberger TA, Robling AG. Bone and skeletal muscle: Key players in mechanotransduction and potential overlapping mechanisms. Bone. 2015 Nov:80():24-36. doi: 10.1016/j.bone.2015.04.014. Epub
[PubMed PMID: 26453495]
[2]
Wilke J, Engeroff T, Nürnberger F, Vogt L, Banzer W. Anatomical study of the morphological continuity between iliotibial tract and the fibularis longus fascia. Surgical and radiologic anatomy : SRA. 2016 Apr:38(3):349-52. doi: 10.1007/s00276-015-1585-6. Epub 2015 Nov 2
[PubMed PMID: 26522465]
[7]
Bourne M, Talkad A, Varacallo M. Anatomy, Bony Pelvis and Lower Limb, Foot Fascia. StatPearls. 2023 Jan:():
[PubMed PMID: 30252299]
[9]
Frontera WR, Ochala J. Skeletal muscle: a brief review of structure and function. Calcified tissue international. 2015 Mar:96(3):183-95. doi: 10.1007/s00223-014-9915-y. Epub 2014 Oct 8
[PubMed PMID: 25294644]
[10]
Hikida RS. Aging changes in satellite cells and their functions. Current aging science. 2011 Dec:4(3):279-97
[PubMed PMID: 21529324]
[12]
Wolfe RR. The underappreciated role of muscle in health and disease. The American journal of clinical nutrition. 2006 Sep:84(3):475-82
[PubMed PMID: 16960159]
[13]
Buckingham M, Rigby PW. Gene regulatory networks and transcriptional mechanisms that control myogenesis. Developmental cell. 2014 Feb 10:28(3):225-38. doi: 10.1016/j.devcel.2013.12.020. Epub
[PubMed PMID: 24525185]
[14]
Hernández-Hernández JM, García-González EG, Brun CE, Rudnicki MA. The myogenic regulatory factors, determinants of muscle development, cell identity and regeneration. Seminars in cell & developmental biology. 2017 Dec:72():10-18. doi: 10.1016/j.semcdb.2017.11.010. Epub 2017 Nov 15
[PubMed PMID: 29127045]
[15]
Borycki AG, Brunk B, Tajbakhsh S, Buckingham M, Chiang C, Emerson CP Jr. Sonic hedgehog controls epaxial muscle determination through Myf5 activation. Development (Cambridge, England). 1999 Sep:126(18):4053-63
[PubMed PMID: 10457014]
[16]
Kablar B, Krastel K, Ying C, Asakura A, Tapscott SJ, Rudnicki MA. MyoD and Myf-5 differentially regulate the development of limb versus trunk skeletal muscle. Development (Cambridge, England). 1997 Dec:124(23):4729-38
[PubMed PMID: 9428409]
[17]
Bagher P, Segal SS. Regulation of blood flow in the microcirculation: role of conducted vasodilation. Acta physiologica (Oxford, England). 2011 Jul:202(3):271-84. doi: 10.1111/j.1748-1716.2010.02244.x. Epub 2011 Mar 1
[PubMed PMID: 21199397]
[18]
Segal SS. Integration of blood flow control to skeletal muscle: key role of feed arteries. Acta physiologica Scandinavica. 2000 Apr:168(4):511-8
[PubMed PMID: 10759588]
[19]
Dodd LR, Johnson PC. Diameter changes in arteriolar networks of contracting skeletal muscle. The American journal of physiology. 1991 Mar:260(3 Pt 2):H662-70
[PubMed PMID: 2000963]
[20]
Heuser JE, Salpeter SR. Organization of acetylcholine receptors in quick-frozen, deep-etched, and rotary-replicated Torpedo postsynaptic membrane. The Journal of cell biology. 1979 Jul:82(1):150-73
[PubMed PMID: 479296]
[21]
Slater CR. The Structure of Human Neuromuscular Junctions: Some Unanswered Molecular Questions. International journal of molecular sciences. 2017 Oct 19:18(10):. doi: 10.3390/ijms18102183. Epub 2017 Oct 19
[PubMed PMID: 29048368]
[23]
Wu H, Xiong WC, Mei L. To build a synapse: signaling pathways in neuromuscular junction assembly. Development (Cambridge, England). 2010 Apr:137(7):1017-33. doi: 10.1242/dev.038711. Epub
[PubMed PMID: 20215342]
[24]
Ottenheijm CA, Granzier H. Lifting the nebula: novel insights into skeletal muscle contractility. Physiology (Bethesda, Md.). 2010 Oct:25(5):304-10. doi: 10.1152/physiol.00016.2010. Epub
[PubMed PMID: 20940435]
[25]
Jayasinghe I, Launikonis BS. Three-dimensional reconstruction and analysis of the tubular system of vertebrate skeletal muscle. Journal of cell science. 2013 Sep 1:126(Pt 17):4048-58. doi: 10.1242/jcs.131565. Epub 2013 Jun 26
[PubMed PMID: 23813954]
[27]
Giuriato G, Pedrinolla A, Schena F, Venturelli M. Muscle cramps: A comparison of the two-leading hypothesis. Journal of electromyography and kinesiology : official journal of the International Society of Electrophysiological Kinesiology. 2018 Aug:41():89-95. doi: 10.1016/j.jelekin.2018.05.006. Epub 2018 May 26
[PubMed PMID: 29857264]
[28]
Bordoni B, Jozsa F, Varacallo M. Anatomy, Head and Neck, Sternocleidomastoid Muscle. StatPearls. 2023 Jan:():
[PubMed PMID: 30422476]