Continuing Education Activity
Pulsus paradoxus, characterized by a systolic pressure drop exceeding 10 mm Hg during inspiration, stems from altered mechanical forces affecting cardiac chambers and pulmonary vasculature. Pulsus paradoxus is a manifestation of an underlying disease process rather than a disease state. While commonly associated with pericardial diseases, notably cardiac tamponade, and constrictive pericarditis, it is crucial to recognize its occurrence in non-pericardial cardiac conditions and noncardiac contexts. In the emergency department, early identification of pulsus paradoxus proves pivotal for swift diagnosis of cardiac tamponade. In addition, its measurement aids in assessing the severity of acute asthma and gauging therapeutic responses.
This activity reviews the diverse etiologies, pathophysiology, evaluation, and treatment of pulsus paradoxus, empowering healthcare professionals with the necessary knowledge and tools to recognize pulsus paradoxus early in emergency settings, aiding in the rapid diagnosis of conditions such as cardiac tamponade. The importance of improving coordination among the interprofessional team to facilitate timely intervention and optimize care is highlighted.
Objectives:
Determine the underlying etiology of pulsus paradoxus.
Evaluate the presentation of a patient with pulsus paradoxus.
Select appropriate interventions for pulsus paradoxus.
Identify the importance of improving coordination among the interprofessional team to facilitate timely intervention and management for patients with pulsus paradoxus.
Introduction
Pulsus paradoxus is an exaggerated drop in a patient’s blood pressure by more than 10 mmHg during inspiration. Pulsus paradoxus arises from changes in the mechanical forces imposed on the chambers of the heart and pulmonary vasculature. These changes are often due to pericardial diseases, particularly cardiac tamponade and, to a lesser degree, constrictive pericarditis. However, it is important to understand that pulsus paradoxus may be observed in non-pericardial cardiac diseases such as right ventricular myocardial infarction and restrictive cardiomyopathy. In addition, noncardiac disease states, including pulmonary diseases such as severe chronic obstructive pulmonary disease, asthma, tension pneumothorax, large bilateral pleural effusions, and pulmonary embolism, and any condition that causes cardiac compression such as iatrogenic injuries during surgery, marked obesity, and pectus excavatum, can occasionally lead to pulsus paradoxus. Finally, pulsus paradoxus may also manifest secondary to severe hypovolemic shock.[1][2][3][4]
Recognizing pulsus paradoxus early in the emergency department can help diagnose cardiac tamponade rapidly. The measurement of pulsus paradoxus is also helpful in assessing the severity of acute asthma and its response to therapy. In addition, noninvasive devices are now available to measure variations in arterial pressure, improving the assessment of pulsus paradoxus at the bedside.[5]
Etiology
The causes of pulsus paradoxus can be categorized into cardiac, pulmonary, and other conditions. As discussed previously in the Introduction section, pulsus paradoxus typically manifests in patients with pericardial diseases, notably cardiac tamponade and constrictive pericarditis. Other cardiac causes include right ventricular infarction and restrictive cardiomyopathy. In addition, noncardiac disease states, including pulmonary diseases such as severe chronic obstructive pulmonary disease, asthma, tension pneumothorax, large bilateral pleural effusions, and pulmonary embolism, and any condition that causes cardiac compression such as iatrogenic injuries during surgery, marked obesity, and pectus excavatum, can also lead to pulsus paradoxus.[6][7][8]
Cardiac: Individuals with certain cardiac conditions, including cardiac tamponade or pericardial effusion, acute myocardial infarction,[9] constrictive pericarditis (rare),[10] cardiomyopathy, pulmonary embolism, right ventricular infarct,[11] tricuspid atresia, interrupted aortic arch,[12] mitral stenosis,[13] and endocardial fibroelastosis, can cause pulsus paradoxus.
Pulmonary: Individuals with certain pulmonary conditions, including acute asthma, acute exacerbation of chronic obstructive pulmonary disease, tension pneumothorax, large compressive pleural effusion, and obstructive sleep apnea, can cause pulsus paradoxus.
Other conditions: Other conditions contributing to pulsus paradoxus may include obesity, superior vena caval obstruction, diaphragmatic hernia, volvulus of the stomach[14], large compressive pleural effusion, thoracic outlet syndrome, and tense ascites.
Epidemiology
Defining the epidemiology of pulsus paradoxus is challenging due to the heterogeneity of the diseases that lead to its manifestation. The incidence of cardiac tamponade leading to pulsus paradoxus has been poorly documented. In a single-center retrospective study involving 136 patients admitted with cardiac tamponade, the primary causes identified were malignancy (32%), infection (24%), idiopathic diseases (16%), iatrogenic injuries (15%), postmyocardial infarction (7%), uremia (4%), and other miscellaneous causes (2%). However, the study did not specify the number of patients who developed pulsus paradoxus. Although the statistics regarding pulsus paradoxus in cardiac tamponade are limited, it is believed to occur in most cases. In a prospective study involving 15 patients with cardiac tamponade, pulsus paradoxus was present in 10 out of 15 patients (66.6%).
Pathophysiology
Examining the normal respirophasic effects of chest mechanics on blood pressure is crucial for understanding the pathophysiology of pulsus paradoxus. In healthy individuals, normal phasic changes in cardiac output occur with respiration. During inspiration, there is a small decrease in systemic arterial pressure of less than 10 mmHg. While inhaling, intrapleural pressure drops; there is a decrease in intrathoracic pressure that promotes venous inflow into the chest, thereby increasing right heart filling. However, this does not equate to an increased filling of the left heart during inspiration. This is because while inhaling, the lungs expand, exerting radial traction on the pulmonary vasculature, which increases its capacitance. This momentarily sequesters blood in the chest, reducing blood flow to the left heart and decreasing preload, consequently leading to decreased cardiac output. The opposite occurs during expiration; thus, systolic pressure normally decreases during inspiration and increases during expiration. So, why the term pulsus paradoxus implies the drop in blood pressure during inspiration is paradoxical. The term "pulsus paradoxus" was coined by the historic German physician Adolph Kussmaul. He used it to describe the palpated pulse of affected patients, which exhibited variable strength despite regular precordial activity.[15][16][17]
The pathophysiology of pulsus paradoxus is complex and varies depending on the etiology, involving several mechanisms. In cases of cardiac tamponade resulting in pulsus paradoxus, the physiologic drop in cardiac output is exaggerated for several reasons; however, the most significant is enhanced ventricular chamber interaction, often referred to as ventricular interdependence. The increased pericardial pressure limits the ability of the right ventricular free wall to expand and accommodate the inflow of blood during inspiration. The outcome is an increased bowing of the ventricular septum into the left ventricle as blood fills the right heart, leading to a decrease in lower left ventricular end-diastolic volume, stroke volume, and systolic pressure. In simpler terms, in a non-compliant pericardial space, the left heart must fill less to allow the right heart to fill more in inspiration. As pericardial pressure increases, the compliance of the ventricles decreases until, under extreme pressure, the effective compliance of all chambers meets that of the pericardial space. In advanced tamponade, intrapericardial pressure is the critical factor determining diastolic cardiac pressures. This is why clinicians observe equalization of chamber pressures during diastole in cardiac tamponade.[18]
In constrictive pericarditis, a separate mechanism may be involved. During normal inspiration, both intrapleural pressure and intrapericardial pressure decrease in tandem to roughly the same degree, and this decrease in pressure is transmitted to the cardiac chambers. In constrictive pericarditis, the thickened pericardium will prevent the normal decrease in pressure from reaching the ventricles; thus, the normal decrease in filling pressures is blunted by the thick pericardium. This is important because the pressure drop in the pulmonary veins (extrapericardial) is more significant compared to the pressure drop in the left ventricular diastole (intrapericardial). The gradient for left ventricular filling is decreased, resulting in decreased left ventricular filling. The opposite occurs during expiration.
Another mechanism to consider is the effect of inspiration on left ventricular transmural pressure. The left ventricular transmural pressure is the sum of the intracavitary pressure minus the negative intrathoracic pressure generated during inspiration. During inspiration, the left ventricle contracts against negative intrathoracic pressure. This increases left ventricular wall stress and afterload, effectively decreasing systolic aortic pressure. This mechanism is exaggerated in severe pulmonary diseases, such as chronic obstructive pulmonary disease, asthma, and obstructive sleep apnea, that require significant negative intrathoracic pressures during inspiration.
History and Physical
Pulsus paradoxus is a clinical exam finding that may be observed in various disease processes and is a crucial exam finding that may herald cardiac tamponade. In pericardial effusion, the presence of pulsus paradoxus has a sensitivity of greater than 80% for tamponade and strongly increases the likelihood of tamponade presence. However, the absence of pulsus paradoxus does not exclude a diagnosis of cardiac tamponade. If pulsus paradoxus is present, it may indicate imminent hemodynamic collapse; thus, all patients with suspected tamponade and pulsus paradoxus should be evaluated for urgent or emergent pericardial drainage.
Disease states in which cardiac tamponade may occur without pulsus paradoxus include the following factors:
- Intracardiac shunts or moderate-to-severe valvular insufficiency
- A coexisting disease, such as severe systemic hypertension, aortic stenosis, or cor pulmonale, significantly increases left or right ventricular diastolic pressure
- Aortic dissection resulting in pericardial effusion/tamponade[19]
- Cardiac tamponade in hypovolemia[20]
Pulsus paradoxus may be observed in constrictive pericarditis, though less frequently compared to that in tamponade. A notable exception is constrictive pericarditis with pericardial effusion (effusive-constrictive pericarditis), where a severe pulsus paradoxus is likely to be observed.
Many disease states apart from pericardial pathology may lead to pulsus paradoxus; the most common of these is severe asthma or chronic obstructive pulmonary disease.[21] In these disease states, the negative intrathoracic pressure generated is significantly increased compared to that in healthy individuals, sometimes close to 40 mmHg below atmospheric pressure. These wide pressure variations will increase left ventricular transmural pressure and effectively increase left ventricular afterload. In addition, the force is transmitted to the intrathoracic aorta and downstream vessels. This may manifest as pulsus paradoxus.
When differentiating between pericardial and non-pericardial diseases while assessing pulsus paradoxus, it is crucial to note that in non-pericardial diseases, pulsus paradoxus typically presents with a decrease in both systolic and diastolic pressures. In contrast, pulsus paradoxus in pericardial diseases presents with a decrease primarily in systolic pressure with a minimum impact on diastolic pressure, resulting in narrow pulse pressure.
Evaluation
Measuring Pulsus Paradoxus
For patients without indwelling arterial access, the most accurate method to measure pulsus paradoxus is by using a manual sphygmomanometer and stethoscope. Automatic blood pressure cuffs cannot accurately measure pulsus paradoxus. Assessment is conducted by inflating the cuff until all Korotkoff sounds disappear, and then slowly releasing pressure from the cuff. The initial sounds auscultated are heard only during expiration, and it is important to take note of this pressure. Next, as the cuff pressure is dropped further, the pressure should be noted when Korotkoff sounds are heard during inspiration and expiration. The variation between these two systolic pressures is what quantifies pulsus paradoxus. Severe pulsus paradoxus may be appreciated as a weakening or disappearance of the palpated pulse during inspiration. Under certain circumstances, pulsus paradoxus may also be observed with respirophasic variations in the patient’s pulse oximetry waveform.[21] The measurement of pulsus paradoxus using the pulse oximeter–pulse plethysmographic waveform is a simple and noninvasive method to assess patients with airway obstruction.[22]
When assessing for pulsus paradoxus, it is crucial to ensure that patients maintain normal tidal volume breathing. Patients should not be instructed to change their breathing patterns because the depth of respiration affects the magnitude of the pulsus paradoxus and is amplified in patients with pulmonary diseases.
For patients with indwelling arterial access, measuring pulsus paradoxus is as simple as observing the waveform and noting systolic pressure differences during the respiratory cycle. Because the drop in blood pressure is secondary to a drop in left ventricular stroke volume, the change in pressure noted during pulsus paradoxus will primarily indicate a decrease in both systolic and pulse pressures, with a minimum impact on diastolic pressure.
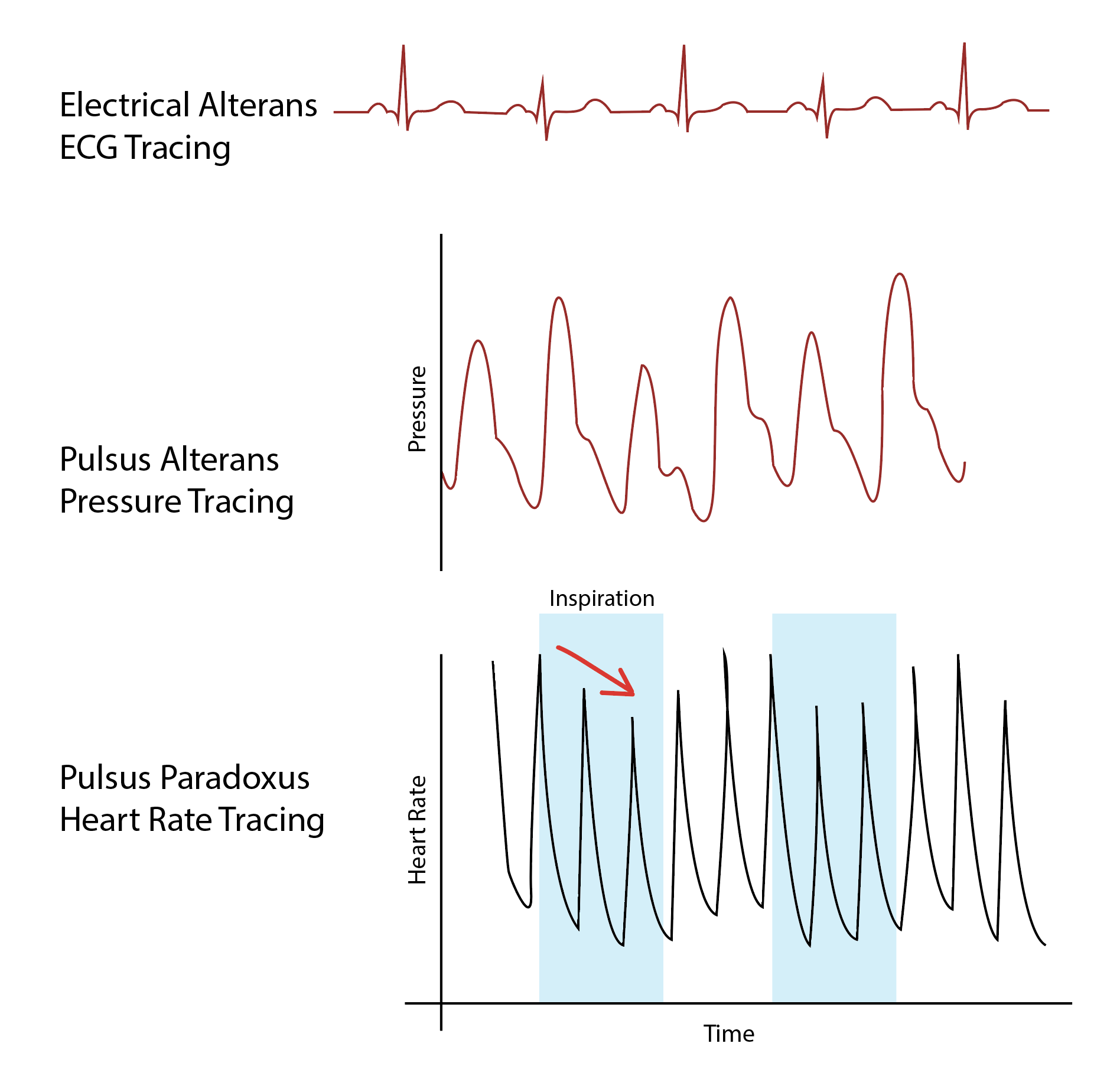
Further diagnosis of patients with pulsus paradoxus will depend on the underlying cause. The diagnosis of cardiac tamponade should be considered if there is known or suspected pericardial effusion. In such cases, obtaining an electrocardiogram (ECG), chest radiography, and trans-thoracic echocardiography is necessary in the absence of hemodynamic compromise. If hemodynamic instability is present, emergent drainage of pericardial fluid should be considered. See Image. Diagram of Electrical Alterans, Pulsus Alterans, and Pulsus Paradoxus.
Treatment / Management
Pulsus paradoxus is not a disease state but rather a physiologic manifestation of an underlying disease process, and treatment should address the underlying pathology. If cardiac tamponade is suspected and pulsus paradoxus is present, urgent or emergent pericardiocentesis should be considered. An exception is pericardial effusion secondary to aortic dissection or myocardial rupture, as these patients require emergency surgical intervention.[23][5]
If a patient has pulsus paradoxus due to obstructive lung disease, immediate bronchodilator therapy and treatment of the underlying cause, such as an infective exacerbation, are required. If there is an underlying tension pneumothorax, immediate treatment is necessary, which involves allowing trapped air to escape and creating a valve opening in the chest until a surgical repair can be performed.
If pulsus paradoxus is caused by a valvular abnormality, consultation with cardiologists and cardiothoracic surgeons is necessary to develop a treatment plan for addressing underlying valvular heart disease. In such cases, the treatment plan is tailored based on the patient's ongoing clinical state.
Differential Diagnosis
The etiological factors outlined above could be considered as differential diagnoses of pulsus paradoxus, as pulsus paradoxus represents an underlying pathology. Healthcare providers should consider differential diagnoses such as cardiogenic shock, constrictive pericarditis, effusive-constrictive pericarditis, pneumothorax, and pulmonary embolism when examining a patient with pulsus paradoxus.
Prognosis
Pulsus paradoxus is not a disease entity; therefore, the prognosis of patients presenting with pulsus paradoxus is determined by the underlying pathological process. For example, patients with pulsus paradoxus due to cardiac tamponade face typically critical conditions, and without prompt treatment, they are likely to have grave outcomes. The key is the timing of intervention; the longer the delay, the worse the outcomes. Similarly, tension pneumothorax arises from many causes and rapidly progresses to respiratory insufficiency, cardiovascular collapse, and ultimately leading to death if not recognized and treated. Uncomplicated pneumothoraces have the potential to recur within a timeframe ranging from 6 months to 3 years. Recurrences are more common in smokers, individuals with chronic obstructive pulmonary disease, and patients with AIDS.[24]
Pulsus paradoxus associated with underlying aortic dissection represents a life-threatening situation with an unfavorable prognosis. In Stanford type A aortic dissection, the prognosis is poor without prompt surgical intervention because the delay can lead to a mortality rate of 50% within the first 48 hours.[25]
Complications
Detection of pulsus paradoxus earlier in the course of illness is critical because it could indicate a serious underlying cause or progression of the disease process. Prompt treatment of the underlying cause could prevent the patient from developing complications. For example, a patient with constrictive pericarditis could develop pulmonary hypertension, shock, hypoxia, renal failure, and ultimately leading to death.[26]
Deterrence and Patient Education
Awareness regarding general health and signs of various illnesses could aid in preventing the severe complications of certain diseases. In addition, compliance with treatment in cases such as asthma and chronic obstructive pulmonary disease can mitigate the risk of patients reaching a stage where pulsus paradoxus may develop.
Enhancing Healthcare Team Outcomes
Pulsus paradoxus refers to an abnormal drop in systolic blood pressure during inspiration, commonly seen in cardiac and respiratory conditions. Clinicians should measure blood pressure during both inspiration and expiration, noting a difference exceeding 10 mm Hg as indicative of pulsus paradoxus. This phenomenon suggests impaired cardiac function or pericardial involvement. Common causes include cardiac tamponade, severe asthma, and chronic obstructive pulmonary disease. Timely recognition of pulsus paradoxus aids in the prompt diagnosis of underlying conditions, guiding appropriate interventions. Monitoring this subtle but clinically significant sign enhances the clinician's ability to assess cardiovascular and respiratory health, facilitating targeted management strategies for improved patient outcomes.
Pulsus paradoxus is best managed by an interprofessional team. Pulsus paradoxus is not a disease state but rather a physiologic manifestation of an underlying disease process, and treatment should address the underlying pathology. If cardiac tamponade is suspected and pulsus paradoxus is present, urgent or emergent pericardiocentesis should be considered.