[1]
Hoit BD. Anatomy and Physiology of the Pericardium. Cardiology clinics. 2017 Nov:35(4):481-490. doi: 10.1016/j.ccl.2017.07.002. Epub
[PubMed PMID: 29025540]
[3]
Imazio M, Gaita F, LeWinter M. Evaluation and Treatment of Pericarditis: A Systematic Review. JAMA. 2015 Oct 13:314(14):1498-506. doi: 10.1001/jama.2015.12763. Epub
[PubMed PMID: 26461998]
Level 1 (high-level) evidence
[5]
Imazio M, Lazaros G, Brucato A, Gaita F. Recurrent pericarditis: new and emerging therapeutic options. Nature reviews. Cardiology. 2016 Feb:13(2):99-105. doi: 10.1038/nrcardio.2015.115. Epub 2015 Aug 11
[PubMed PMID: 26259934]
[6]
Maisch B. [Management of pericarditis and pericardial effusion, constrictive and effusive-constrictive pericarditis]. Herz. 2018 Nov:43(7):663-678. doi: 10.1007/s00059-018-4744-9. Epub
[PubMed PMID: 30315402]
[7]
Khandaker MH, Espinosa RE, Nishimura RA, Sinak LJ, Hayes SN, Melduni RM, Oh JK. Pericardial disease: diagnosis and management. Mayo Clinic proceedings. 2010 Jun:85(6):572-93. doi: 10.4065/mcp.2010.0046. Epub
[PubMed PMID: 20511488]
[8]
Oh JK, Hatle LK, Seward JB, Danielson GK, Schaff HV, Reeder GS, Tajik AJ. Diagnostic role of Doppler echocardiography in constrictive pericarditis. Journal of the American College of Cardiology. 1994 Jan:23(1):154-62
[PubMed PMID: 8277074]
[9]
Adler Y, Charron P. The 2015 ESC Guidelines on the diagnosis and management of pericardial diseases. European heart journal. 2015 Nov 7:36(42):2873-4. doi: 10.1093/eurheartj/ehv479. Epub
[PubMed PMID: 26547486]
[10]
Imazio M, Brucato A, Derosa FG, Lestuzzi C, Bombana E, Scipione F, Leuzzi S, Cecchi E, Trinchero R, Adler Y. Aetiological diagnosis in acute and recurrent pericarditis: when and how. Journal of cardiovascular medicine (Hagerstown, Md.). 2009 Mar:10(3):217-30. doi: 10.2459/JCM.0b013e328322f9b1. Epub
[PubMed PMID: 19262208]
[12]
Ntsekhe M, Mayosi BM. Tuberculous pericarditis with and without HIV. Heart failure reviews. 2013 May:18(3):367-73. doi: 10.1007/s10741-012-9310-6. Epub
[PubMed PMID: 22427006]
[13]
Petcu CP, Dilof R, Bătăiosu C, Petcu PD. Purulent pericardial effusions with pericardial tamponade - diagnosis and treatment issues. Current health sciences journal. 2013 Jan:39(1):53-6
[PubMed PMID: 24778855]
[14]
Oladele RO, Ayanlowo OO, Richardson MD, Denning DW. Histoplasmosis in Africa: An emerging or a neglected disease? PLoS neglected tropical diseases. 2018 Jan:12(1):e0006046. doi: 10.1371/journal.pntd.0006046. Epub 2018 Jan 18
[PubMed PMID: 29346384]
[15]
Imazio M, Negro A, Belli R, Beqaraj F, Forno D, Giammaria M, Trinchero R, Adler Y, Spodick D. Frequency and prognostic significance of pericarditis following acute myocardial infarction treated by primary percutaneous coronary intervention. The American journal of cardiology. 2009 Jun 1:103(11):1525-9. doi: 10.1016/j.amjcard.2009.01.366. Epub 2009 Apr 8
[PubMed PMID: 19463510]
[16]
Jaworska-Wilczynska M, Abramczuk E, Hryniewiecki T. Postcardiac injury syndrome. Medical science monitor : international medical journal of experimental and clinical research. 2011 Nov:17(11):CQ13-14
[PubMed PMID: 22037738]
[17]
Wessman DE, Stafford CM. The postcardiac injury syndrome: case report and review of the literature. Southern medical journal. 2006 Mar:99(3):309-14
[PubMed PMID: 16553111]
Level 3 (low-level) evidence
[18]
Katz U, Zandman-Goddard G. Drug-induced lupus: an update. Autoimmunity reviews. 2010 Nov:10(1):46-50. doi: 10.1016/j.autrev.2010.07.005. Epub 2010 Jul 23
[PubMed PMID: 20656071]
[19]
Altan M, Toki MI, Gettinger SN, Carvajal-Hausdorf DE, Zugazagoitia J, Sinard JH, Herbst RS, Rimm DL. Immune Checkpoint Inhibitor-Associated Pericarditis. Journal of thoracic oncology : official publication of the International Association for the Study of Lung Cancer. 2019 Jun:14(6):1102-1108. doi: 10.1016/j.jtho.2019.02.026. Epub 2019 Mar 7
[PubMed PMID: 30851443]
[20]
Wyplosz B, Marijon E, Dougados J, Pouchot J. Sarcoidosis: an unusual cause of acute pericarditis. Acta cardiologica. 2010 Feb:65(1):83-4
[PubMed PMID: 20306895]
[21]
Imazio M. Noninfectious pericarditis: management challenges for cardiologists. Kardiologia polska. 2020 May 25:78(5):396-403. doi: 10.33963/KP.15353. Epub 2020 May 11
[PubMed PMID: 32394692]
[22]
Kim HJ, Cho YS, Cho GY, Choi SI. Congenital absence of the pericardium. Journal of cardiovascular ultrasound. 2014 Mar:22(1):36-9. doi: 10.4250/jcu.2014.22.1.36. Epub 2014 Mar 31
[PubMed PMID: 24753808]
[23]
Shah AB, Kronzon I. Congenital defects of the pericardium: a review. European heart journal. Cardiovascular Imaging. 2015 Aug:16(8):821-7. doi: 10.1093/ehjci/jev119. Epub 2015 May 23
[PubMed PMID: 26003149]
[24]
Ben-Horin S, Bank I, Guetta V, Livneh A. Large symptomatic pericardial effusion as the presentation of unrecognized cancer: a study in 173 consecutive patients undergoing pericardiocentesis. Medicine. 2006 Jan:85(1):49-53. doi: 10.1097/01.md.0000199556.69588.8e. Epub
[PubMed PMID: 16523053]
[25]
LeWinter MM. Clinical practice. Acute pericarditis. The New England journal of medicine. 2014 Dec 18:371(25):2410-6. doi: 10.1056/NEJMcp1404070. Epub
[PubMed PMID: 25517707]
[26]
Imazio M, Trinchero R. Myopericarditis: Etiology, management, and prognosis. International journal of cardiology. 2008 Jun 23:127(1):17-26. doi: 10.1016/j.ijcard.2007.10.053. Epub 2008 Jan 24
[PubMed PMID: 18221804]
[27]
Goodacre S, Locker T, Morris F, Campbell S. How useful are clinical features in the diagnosis of acute, undifferentiated chest pain? Academic emergency medicine : official journal of the Society for Academic Emergency Medicine. 2002 Mar:9(3):203-8
[PubMed PMID: 11874776]
[28]
Launbjerg J, Fruergaard P, Hesse B, Jørgensen F, Elsborg L, Petri A. Long-term risk of death, cardiac events and recurrent chest pain in patients with acute chest pain of different origin. Cardiology. 1996 Jan-Feb:87(1):60-6
[PubMed PMID: 8631047]
[29]
Zayas R, Anguita M, Torres F, Giménez D, Bergillos F, Ruiz M, Ciudad M, Gallardo A, Vallés F. Incidence of specific etiology and role of methods for specific etiologic diagnosis of primary acute pericarditis. The American journal of cardiology. 1995 Feb 15:75(5):378-82
[PubMed PMID: 7856532]
[30]
Sarkar M, Madabhavi I, Niranjan N, Dogra M. Auscultation of the respiratory system. Annals of thoracic medicine. 2015 Jul-Sep:10(3):158-68. doi: 10.4103/1817-1737.160831. Epub
[PubMed PMID: 26229557]
[31]
Imazio M, Gaita F. Diagnosis and treatment of pericarditis. Heart (British Cardiac Society). 2015 Jul:101(14):1159-68. doi: 10.1136/heartjnl-2014-306362. Epub 2015 Apr 8
[PubMed PMID: 25855795]
[32]
Rossello X, Wiegerinck RF, Alguersuari J, Bardají A, Worner F, Sutil M, Ferrero A, Cinca J. New electrocardiographic criteria to differentiate acute pericarditis and myocardial infarction. The American journal of medicine. 2014 Mar:127(3):233-9. doi: 10.1016/j.amjmed.2013.11.006. Epub 2013 Nov 25
[PubMed PMID: 24287008]
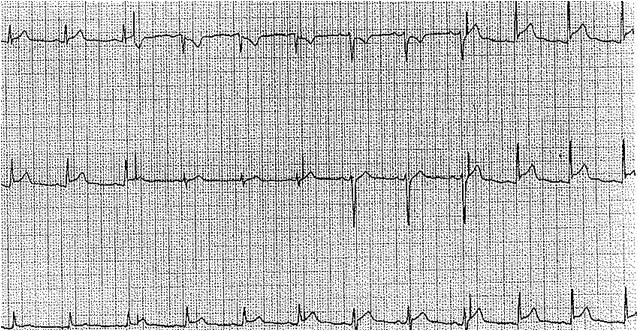
[33]
Spodick DH. Differential characteristics of the electrocardiogram in early repolarization and acute pericarditis. The New England journal of medicine. 1976 Sep 2:295(10):523-6
[PubMed PMID: 950958]
[34]
Buppajamrntham T, Palavutitotai N, Katchamart W. Clinical manifestation, diagnosis, management, and treatment outcome of pericarditis in patients with systemic lupus erythematosus. Journal of the Medical Association of Thailand = Chotmaihet thangphaet. 2014 Dec:97(12):1234-40
[PubMed PMID: 25764628]
[35]
Bentata Y, Hamdi F, Chemlal A, Haddiya I, Ismaili N, El Ouafi N. Uremic pericarditis in patients with End Stage Renal Disease: Prevalence, symptoms and outcome in 2017. The American journal of emergency medicine. 2018 Mar:36(3):464-466. doi: 10.1016/j.ajem.2017.11.048. Epub 2017 Nov 21
[PubMed PMID: 29248269]
[36]
Imazio M, Spodick DH, Brucato A, Trinchero R, Adler Y. Controversial issues in the management of pericardial diseases. Circulation. 2010 Feb 23:121(7):916-28. doi: 10.1161/CIRCULATIONAHA.108.844753. Epub
[PubMed PMID: 20177006]
[37]
Klein AL, Abbara S, Agler DA, Appleton CP, Asher CR, Hoit B, Hung J, Garcia MJ, Kronzon I, Oh JK, Rodriguez ER, Schaff HV, Schoenhagen P, Tan CD, White RD. American Society of Echocardiography clinical recommendations for multimodality cardiovascular imaging of patients with pericardial disease: endorsed by the Society for Cardiovascular Magnetic Resonance and Society of Cardiovascular Computed Tomography. Journal of the American Society of Echocardiography : official publication of the American Society of Echocardiography. 2013 Sep:26(9):965-1012.e15. doi: 10.1016/j.echo.2013.06.023. Epub
[PubMed PMID: 23998693]
[38]
Cosyns B, Plein S, Nihoyanopoulos P, Smiseth O, Achenbach S, Andrade MJ, Pepi M, Ristic A, Imazio M, Paelinck B, Lancellotti P, European Association of Cardiovascular Imaging (EACVI), European Society of Cardiology Working Group (ESC WG) on Myocardial and Pericardial diseases. European Association of Cardiovascular Imaging (EACVI) position paper: Multimodality imaging in pericardial disease. European heart journal. Cardiovascular Imaging. 2015 Jan:16(1):12-31. doi: 10.1093/ehjci/jeu128. Epub 2014 Sep 23
[PubMed PMID: 25248336]
[39]
Bogaert J, Francone M. Cardiovascular magnetic resonance in pericardial diseases. Journal of cardiovascular magnetic resonance : official journal of the Society for Cardiovascular Magnetic Resonance. 2009 May 4:11(1):14. doi: 10.1186/1532-429X-11-14. Epub 2009 May 4
[PubMed PMID: 19413898]
[40]
Maisch B, Seferović PM, Ristić AD, Erbel R, Rienmüller R, Adler Y, Tomkowski WZ, Thiene G, Yacoub MH, Grupo de Trabajo para el Diagnóstico y Tratamiento de las Enfermedades del Pericardio de la Sociedad Europea de Cardiología. [Guidelines on the diagnosis and management of pericardial diseases. Executive summary]. Revista espanola de cardiologia. 2004 Nov:57(11):1090-114
[PubMed PMID: 15544758]
[41]
Ben-Horin S, Bank I, Shinfeld A, Kachel E, Guetta V, Livneh A. Diagnostic value of the biochemical composition of pericardial effusions in patients undergoing pericardiocentesis. The American journal of cardiology. 2007 May 1:99(9):1294-7
[PubMed PMID: 17478160]
[42]
Mayosi BM, Ntsekhe M, Volmink JA, Commerford PJ. Interventions for treating tuberculous pericarditis. The Cochrane database of systematic reviews. 2002:(4):CD000526
[PubMed PMID: 12519546]
Level 1 (high-level) evidence
[43]
Mayosi BM, Ntsekhe M, Bosch J, Pandie S, Jung H, Gumedze F, Pogue J, Thabane L, Smieja M, Francis V, Joldersma L, Thomas KM, Thomas B, Awotedu AA, Magula NP, Naidoo DP, Damasceno A, Chitsa Banda A, Brown B, Manga P, Kirenga B, Mondo C, Mntla P, Tsitsi JM, Peters F, Essop MR, Russell JB, Hakim J, Matenga J, Barasa AF, Sani MU, Olunuga T, Ogah O, Ansa V, Aje A, Danbauchi S, Ojji D, Yusuf S, IMPI Trial Investigators. Prednisolone and Mycobacterium indicus pranii in tuberculous pericarditis. The New England journal of medicine. 2014 Sep 18:371(12):1121-30. doi: 10.1056/NEJMoa1407380. Epub 2014 Sep 1
[PubMed PMID: 25178809]
[44]
Karatolios K, Pankuweit S, Maisch B. Diagnostic value of biochemical biomarkers in malignant and non-malignant pericardial effusion. Heart failure reviews. 2013 May:18(3):337-44. doi: 10.1007/s10741-012-9327-x. Epub
[PubMed PMID: 22638889]
[45]
Alpert MA, Ravenscraft MD. Pericardial involvement in end-stage renal disease. The American journal of the medical sciences. 2003 Apr:325(4):228-36
[PubMed PMID: 12695728]
[46]
Wood JE, Mahnensmith RL. Pericarditis associated with renal failure: evolution and management. Seminars in dialysis. 2001 Jan-Feb:14(1):61-6
[PubMed PMID: 11208042]
[47]
Seidenberg PH, Haynes J. Pericarditis: diagnosis, management, and return to play. Current sports medicine reports. 2006 Apr:5(2):74-9
[PubMed PMID: 16529677]
[48]
Imazio M, Cecchi E, Demichelis B, Ierna S, Demarie D, Ghisio A, Pomari F, Coda L, Belli R, Trinchero R. Indicators of poor prognosis of acute pericarditis. Circulation. 2007 May 29:115(21):2739-44
[PubMed PMID: 17502574]
[49]
Imazio M, Brucato A, Trinchero R, Spodick D, Adler Y. Individualized therapy for pericarditis. Expert review of cardiovascular therapy. 2009 Aug:7(8):965-75. doi: 10.1586/erc.09.82. Epub
[PubMed PMID: 19673674]
[50]
Imazio M, Bobbio M, Cecchi E, Demarie D, Demichelis B, Pomari F, Moratti M, Gaschino G, Giammaria M, Ghisio A, Belli R, Trinchero R. Colchicine in addition to conventional therapy for acute pericarditis: results of the COlchicine for acute PEricarditis (COPE) trial. Circulation. 2005 Sep 27:112(13):2012-6
[PubMed PMID: 16186437]
[51]
Imazio M, Bobbio M, Cecchi E, Demarie D, Pomari F, Moratti M, Ghisio A, Belli R, Trinchero R. Colchicine as first-choice therapy for recurrent pericarditis: results of the CORE (COlchicine for REcurrent pericarditis) trial. Archives of internal medicine. 2005 Sep 26:165(17):1987-91
[PubMed PMID: 16186468]
[52]
Lotrionte M, Biondi-Zoccai G, Imazio M, Castagno D, Moretti C, Abbate A, Agostoni P, Brucato AL, Di Pasquale P, Raatikka M, Sangiorgi G, Laudito A, Sheiban I, Gaita F. International collaborative systematic review of controlled clinical trials on pharmacologic treatments for acute pericarditis and its recurrences. American heart journal. 2010 Oct:160(4):662-70. doi: 10.1016/j.ahj.2010.06.015. Epub
[PubMed PMID: 20934560]
Level 1 (high-level) evidence
[53]
Imazio M, Brucato A, Cumetti D, Brambilla G, Demichelis B, Ferro S, Maestroni S, Cecchi E, Belli R, Palmieri G, Trinchero R. Corticosteroids for recurrent pericarditis: high versus low doses: a nonrandomized observation. Circulation. 2008 Aug 5:118(6):667-71. doi: 10.1161/CIRCULATIONAHA.107.761064. Epub 2008 Jul 21
[PubMed PMID: 18645054]
Level 2 (mid-level) evidence
[54]
Finetti M, Insalaco A, Cantarini L, Meini A, Breda L, Alessio M, D'Alessandro M, Picco P, Martini A, Gattorno M. Long-term efficacy of interleukin-1 receptor antagonist (anakinra) in corticosteroid-dependent and colchicine-resistant recurrent pericarditis. The Journal of pediatrics. 2014 Jun:164(6):1425-31.e1. doi: 10.1016/j.jpeds.2014.01.065. Epub 2014 Mar 12
[PubMed PMID: 24630353]
[55]
Khandaker MH, Schaff HV, Greason KL, Anavekar NS, Espinosa RE, Hayes SN, Nishimura RA, Oh JK. Pericardiectomy vs medical management in patients with relapsing pericarditis. Mayo Clinic proceedings. 2012 Nov:87(11):1062-70. doi: 10.1016/j.mayocp.2012.05.024. Epub
[PubMed PMID: 23127733]
[56]
Imazio M, Brucato A, Maestroni S, Cumetti D, Belli R, Trinchero R, Adler Y. Risk of constrictive pericarditis after acute pericarditis. Circulation. 2011 Sep 13:124(11):1270-5. doi: 10.1161/CIRCULATIONAHA.111.018580. Epub 2011 Aug 15
[PubMed PMID: 21844077]
[57]
Soler-Soler J, Sagristà-Sauleda J, Permanyer-Miralda G. Relapsing pericarditis. Heart (British Cardiac Society). 2004 Nov:90(11):1364-8
[PubMed PMID: 15486149]
[58]
Kabukcu M, Demircioglu F, Yanik E, Basarici I, Ersel F. Pericardial tamponade and large pericardial effusions: causal factors and efficacy of percutaneous catheter drainage in 50 patients. Texas Heart Institute journal. 2004:31(4):398-403
[PubMed PMID: 15745292]