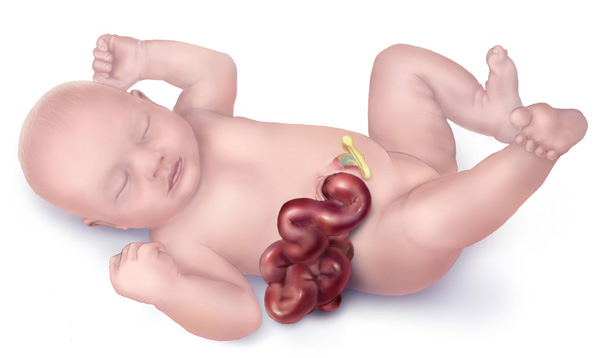
[1]
Robinson TD, Whyte C. Closed Gastroschisis. The New England journal of medicine. 2021 Apr 15:384(15):e56. doi: 10.1056/NEJMicm2029281. Epub 2021 Apr 10
[PubMed PMID: 33853208]
[2]
Feldkamp ML, Alder SC, Carey JC. A case control population-based study investigating smoking as a risk factor for gastroschisis in Utah, 1997-2005. Birth defects research. Part A, Clinical and molecular teratology. 2008 Nov:82(11):768-75. doi: 10.1002/bdra.20519. Epub
[PubMed PMID: 18985693]
Level 2 (mid-level) evidence
[3]
Goodman M, Mandel JS, DeSesso JM, Scialli AR. Atrazine and pregnancy outcomes: a systematic review of epidemiologic evidence. Birth defects research. Part B, Developmental and reproductive toxicology. 2014 Jun:101(3):215-36. doi: 10.1002/bdrb.21101. Epub 2014 May 2
[PubMed PMID: 24797711]
Level 1 (high-level) evidence
[4]
Agopian AJ, Langlois PH, Cai Y, Canfield MA, Lupo PJ. Maternal residential atrazine exposure and gastroschisis by maternal age. Maternal and child health journal. 2013 Dec:17(10):1768-75. doi: 10.1007/s10995-012-1196-3. Epub
[PubMed PMID: 23184502]
[5]
Baird PA, MacDonald EC. An epidemiologic study of congenital malformations of the anterior abdominal wall in more than half a million consecutive live births. American journal of human genetics. 1981 May:33(3):470-8
[PubMed PMID: 6454342]
[6]
Lap CC, Brizot ML, Pistorius LR, Kramer WL, Teeuwen IB, Eijkemans MJ, Brouwers HA, Pajkrt E, van Kaam AH, van Scheltema PN, Eggink AJ, van Heijst AF, Haak MC, van Weissenbruch MM, Sleeboom C, Willekes C, van der Hoeven MA, van Heurn EL, Bilardo CM, Dijk PH, van Baren R, Francisco RP, Tannuri AC, Visser GH, Manten GT. Outcome of isolated gastroschisis; an international study, systematic review and meta-analysis. Early human development. 2016 Dec:103():209-218. doi: 10.1016/j.earlhumdev.2016.10.002. Epub 2016 Oct 27
[PubMed PMID: 27825040]
Level 1 (high-level) evidence
[7]
Islam S. Clinical care outcomes in abdominal wall defects. Current opinion in pediatrics. 2008 Jun:20(3):305-10. doi: 10.1097/MOP.0b013e3282ffdc1e. Epub
[PubMed PMID: 18475100]
Level 3 (low-level) evidence
[8]
D'Antonio F, Virgone C, Rizzo G, Khalil A, Baud D, Cohen-Overbeek TE, Kuleva M, Salomon LJ, Flacco ME, Manzoli L, Giuliani S. Prenatal Risk Factors and Outcomes in Gastroschisis: A Meta-Analysis. Pediatrics. 2015 Jul:136(1):e159-69. doi: 10.1542/peds.2015-0017. Epub
[PubMed PMID: 26122809]
Level 1 (high-level) evidence
[9]
Page R, Ferraro ZM, Moretti F, Fung KF. Gastroschisis: antenatal sonographic predictors of adverse neonatal outcome. Journal of pregnancy. 2014:2014():239406. doi: 10.1155/2014/239406. Epub 2014 Dec 22
[PubMed PMID: 25587450]
[10]
Girsen AI, Do S, Davis AS, Hintz SR, Desai AK, Mansour T, Merritt TA, Oshiro BT, El-Sayed YY, Blumenfeld YJ. Peripartum and neonatal outcomes of small-for-gestational-age infants with gastroschisis. Prenatal diagnosis. 2015 May:35(5):477-82. doi: 10.1002/pd.4562. Epub
[PubMed PMID: 25613462]
[11]
Blumenfeld YJ, Do S, Girsen AI, Davis AS, Hintz SR, Desai AK, Mansour T, Merritt TA, Oshiro BT, El-Sayed YY, Shamshirsaz AA, Lee HC. Utility of third trimester sonographic measurements for predicting SGA in cases of fetal gastroschisis. Journal of perinatology : official journal of the California Perinatal Association. 2017 May:37(5):498-501. doi: 10.1038/jp.2016.275. Epub 2017 Jan 26
[PubMed PMID: 28125100]
Level 3 (low-level) evidence
[12]
Mastroiacovo P, Lisi A, Castilla EE, Martínez-Frías ML, Bermejo E, Marengo L, Kucik J, Siffel C, Halliday J, Gatt M, Annerèn G, Bianchi F, Canessa MA, Danderfer R, de Walle H, Harris J, Li Z, Lowry RB, McDonell R, Merlob P, Metneki J, Mutchinick O, Robert-Gnansia E, Scarano G, Sipek A, Pötzsch S, Szabova E, Yevtushok L. Gastroschisis and associated defects: an international study. American journal of medical genetics. Part A. 2007 Apr 1:143A(7):660-71
[PubMed PMID: 17357116]
[13]
Amin R, Domack A, Bartoletti J, Peterson E, Rink B, Bruggink J, Christensen M, Johnson A, Polzin W, Wagner AJ. National Practice Patterns for Prenatal Monitoring in Gastroschisis: Gastroschisis Outcomes of Delivery (GOOD) Provider Survey. Fetal diagnosis and therapy. 2019:45(2):125-130. doi: 10.1159/000487541. Epub 2018 May 23
[PubMed PMID: 29791899]
Level 3 (low-level) evidence
[14]
Japaraj RP, Hockey R, Chan FY. Gastroschisis: can prenatal sonography predict neonatal outcome? Ultrasound in obstetrics & gynecology : the official journal of the International Society of Ultrasound in Obstetrics and Gynecology. 2003 Apr:21(4):329-33
[PubMed PMID: 12704738]
[15]
Nicholas SS, Stamilio DM, Dicke JM, Gray DL, Macones GA, Odibo AO. Predicting adverse neonatal outcomes in fetuses with abdominal wall defects using prenatal risk factors. American journal of obstetrics and gynecology. 2009 Oct:201(4):383.e1-6. doi: 10.1016/j.ajog.2009.06.032. Epub 2009 Aug 29
[PubMed PMID: 19716531]
[16]
Santiago-Munoz PC, McIntire DD, Barber RG, Megison SM, Twickler DM, Dashe JS. Outcomes of pregnancies with fetal gastroschisis. Obstetrics and gynecology. 2007 Sep:110(3):663-8
[PubMed PMID: 17766615]
[17]
Overcash RT, DeUgarte DA, Stephenson ML, Gutkin RM, Norton ME, Parmar S, Porto M, Poulain FR, Schrimmer DB, University of California Fetal Consortium*. Factors associated with gastroschisis outcomes. Obstetrics and gynecology. 2014 Sep:124(3):551-557. doi: 10.1097/AOG.0000000000000425. Epub
[PubMed PMID: 25162255]
[18]
Logghe HL, Mason GC, Thornton JG, Stringer MD. A randomized controlled trial of elective preterm delivery of fetuses with gastroschisis. Journal of pediatric surgery. 2005 Nov:40(11):1726-31
[PubMed PMID: 16291160]
Level 1 (high-level) evidence
[19]
Zalles-Vidal C, Peñarrieta-Daher A, Bracho-Blanchet E, Ibarra-Rios D, Dávila-Perez R, Villegas-Silva R, Nieto-Zermeño J. A Gastroschisis bundle: effects of a quality improvement protocol on morbidity and mortality. Journal of pediatric surgery. 2018 Nov:53(11):2117-2122. doi: 10.1016/j.jpedsurg.2018.06.014. Epub 2018 Jun 19
[PubMed PMID: 30318281]
Level 2 (mid-level) evidence
[20]
Rosen O, Angert RM. Gastroschisis Simulation Model: Pre-surgical Management Technical Report. Cureus. 2017 Mar 22:9(3):e1109. doi: 10.7759/cureus.1109. Epub 2017 Mar 22
[PubMed PMID: 28439484]
[21]
Jansen LA, Safavi A, Lin Y, MacNab YC, Skarsgard ED, Canadian Pediatric Surgery Network (CAPSNet). Preclosure fluid resuscitation influences outcome in gastroschisis. American journal of perinatology. 2012 Apr:29(4):307-12. doi: 10.1055/s-0031-1295639. Epub 2011 Nov 17
[PubMed PMID: 22094919]
[22]
Poola AS, Aguayo P, Fraser JD, Hendrickson RJ, Weaver KL, Gonzalez KW, St Peter SD. Primary Closure versus Bedside Silo and Delayed Closure for Gastroschisis: A Truncated Prospective Randomized Trial. European journal of pediatric surgery : official journal of Austrian Association of Pediatric Surgery ... [et al] = Zeitschrift fur Kinderchirurgie. 2019 Apr:29(2):203-208. doi: 10.1055/s-0038-1627459. Epub 2018 Feb 19
[PubMed PMID: 29458229]
Level 1 (high-level) evidence
[23]
Bruzoni M, Jaramillo JD, Dunlap JL, Abrajano C, Stack SW, Hintz SR, Hernandez-Boussard T, Dutta S. Sutureless vs Sutured Gastroschisis Closure: A Prospective Randomized Controlled Trial. Journal of the American College of Surgeons. 2017 Jun:224(6):1091-1096.e1. doi: 10.1016/j.jamcollsurg.2017.02.014. Epub 2017 Mar 6
[PubMed PMID: 28279777]
Level 1 (high-level) evidence
[24]
Fraser JD, Deans KJ, Fallat ME, Helmrath MA, Kabre R, Leys CM, Burns RC, Corkum K, Dillon PA, Downard CD, Gadepalli SK, Grabowski JE, Hernandez E, Hirschl RB, Johnson KN, Kohler JE, Landman MP, Landisch RM, Lawrence AE, Mak GZ, Minneci PC, Rymeski B, Sato TT, Scannell M, Slater BJ, Wilkinson KH, Wright TN, St Peter SD, Midwest Pediatric Surgery Consortium. Sutureless vs sutured abdominal wall closure for gastroschisis: Operative characteristics and early outcomes from the Midwest Pediatric Surgery Consortium. Journal of pediatric surgery. 2020 Nov:55(11):2284-2288. doi: 10.1016/j.jpedsurg.2020.02.017. Epub 2020 Feb 20
[PubMed PMID: 32151403]
[25]
Olesevich M, Alexander F, Khan M, Cotman K. Gastroschisis revisited: role of intraoperative measurement of abdominal pressure. Journal of pediatric surgery. 2005 May:40(5):789-92
[PubMed PMID: 15937815]
[26]
Santos Schmidt AF, Goncalves A, Bustorff-Silva JM, Oliveira-Filho AG, Miranda ML, Oliveira ER, Marba S, Sbragia L. Monitoring intravesical pressure during gastroschisis closure. Does it help to decide between delayed primary or staged closure? The journal of maternal-fetal & neonatal medicine : the official journal of the European Association of Perinatal Medicine, the Federation of Asia and Oceania Perinatal Societies, the International Society of Perinatal Obstetricians. 2012 Aug:25(8):1438-41. doi: 10.3109/14767058.2011.640366. Epub 2012 Apr 25
[PubMed PMID: 22098652]
[27]
Ionescu S, Mocanu M, Andrei B, Bunea B, Carstoveanu C, Gurita A, Tabacaru R, Licsandru E, Stanescu D, Selleh M. Differential diagnosis of abdominal wall defects - omphalocele versus gastroschisis. Chirurgia (Bucharest, Romania : 1990). 2014 Jan-Feb:109(1):7-14
[PubMed PMID: 24524464]
[28]
Williams AP, Marayati R, Beierle EA. Pentalogy of Cantrell. Seminars in pediatric surgery. 2019 Apr:28(2):106-110. doi: 10.1053/j.sempedsurg.2019.04.006. Epub 2019 Apr 9
[PubMed PMID: 31072457]
[29]
Revels JW, Wang SS, Nasrullah A, Revzin M, Iyer RS, Deutsch G, Katz DS, Moshiri M. An Algorithmic Approach to Complex Fetal Abdominal Wall Defects. AJR. American journal of roentgenology. 2020 Jan:214(1):218-231. doi: 10.2214/AJR.19.21627. Epub 2019 Nov 12
[PubMed PMID: 31714849]
[30]
Lepigeon K, Van Mieghem T, Vasseur Maurer S, Giannoni E, Baud D. Gastroschisis--what should be told to parents? Prenatal diagnosis. 2014 Apr:34(4):316-26. doi: 10.1002/pd.4305. Epub 2014 Jan 20
[PubMed PMID: 24375446]
[31]
Fullerton BS, Velazco CS, Sparks EA, Morrow KA, Edwards EM, Soll RF, Modi BP, Horbar JD, Jaksic T. Contemporary Outcomes of Infants with Gastroschisis in North America: A Multicenter Cohort Study. The Journal of pediatrics. 2017 Sep:188():192-197.e6. doi: 10.1016/j.jpeds.2017.06.013. Epub 2017 Jul 13
[PubMed PMID: 28712519]
[32]
Arnold MA, Chang DC, Nabaweesi R, Colombani PM, Bathurst MA, Mon KS, Hosmane S, Abdullah F. Risk stratification of 4344 patients with gastroschisis into simple and complex categories. Journal of pediatric surgery. 2007 Sep:42(9):1520-5
[PubMed PMID: 17848242]
[33]
Bergholz R, Boettcher M, Reinshagen K, Wenke K. Complex gastroschisis is a different entity to simple gastroschisis affecting morbidity and mortality-a systematic review and meta-analysis. Journal of pediatric surgery. 2014 Oct:49(10):1527-32. doi: 10.1016/j.jpedsurg.2014.08.001. Epub 2014 Sep 4
[PubMed PMID: 25280661]
Level 1 (high-level) evidence
[34]
Bradnock TJ, Marven S, Owen A, Johnson P, Kurinczuk JJ, Spark P, Draper ES, Knight M, BAPS-CASS. Gastroschisis: one year outcomes from national cohort study. BMJ (Clinical research ed.). 2011 Nov 15:343():d6749. doi: 10.1136/bmj.d6749. Epub 2011 Nov 15
[PubMed PMID: 22089731]
[35]
Fawley JA, Abdelhafeez AH, Schultz JA, Ertl A, Cassidy LD, Peter SS, Wagner AJ. The risk of midgut volvulus in patients with abdominal wall defects: A multi-institutional study. Journal of pediatric surgery. 2017 Jan:52(1):26-29. doi: 10.1016/j.jpedsurg.2016.10.014. Epub 2016 Oct 25
[PubMed PMID: 27847120]
[36]
Koivusalo A, Lindahl H, Rintala RJ. Morbidity and quality of life in adult patients with a congenital abdominal wall defect: a questionnaire survey. Journal of pediatric surgery. 2002 Nov:37(11):1594-601
[PubMed PMID: 12407546]
Level 2 (mid-level) evidence
[37]
Yardley IE, Bostock E, Jones MO, Turnock RR, Corbett HJ, Losty PD. Congenital abdominal wall defects and testicular maldescent--a 10-year single-center experience. Journal of pediatric surgery. 2012 Jun:47(6):1118-22. doi: 10.1016/j.jpedsurg.2012.03.011. Epub
[PubMed PMID: 22703780]
[38]
Harris EL, Hart SJ, Minutillo C, Ravikumara M, Warner TM, Williams Y, Nathan EA, Dickinson JE. The long-term neurodevelopmental and psychological outcomes of gastroschisis: A cohort study. Journal of pediatric surgery. 2016 Apr:51(4):549-53. doi: 10.1016/j.jpedsurg.2015.08.062. Epub 2015 Sep 16
[PubMed PMID: 26490011]
[39]
Youssef F, Laberge JM, Puligandla P, Emil S, Canadian Pediatric Surgery Network (CAPSNet). Determinants of outcomes in patients with simple gastroschisis. Journal of pediatric surgery. 2017 May:52(5):710-714. doi: 10.1016/j.jpedsurg.2017.01.019. Epub 2017 Jan 28
[PubMed PMID: 28188037]
[40]
Youssef F, Cheong LH, Emil S, Canadian Pediatric Surgery Network (CAPSNet). Gastroschisis outcomes in North America: a comparison of Canada and the United States. Journal of pediatric surgery. 2016 Jun:51(6):891-5. doi: 10.1016/j.jpedsurg.2016.02.046. Epub 2016 Mar 3
[PubMed PMID: 27004440]
[41]
Emil S, Canvasser N, Chen T, Friedrich E, Su W. Contemporary 2-year outcomes of complex gastroschisis. Journal of pediatric surgery. 2012 Aug:47(8):1521-8. doi: 10.1016/j.jpedsurg.2011.12.023. Epub
[PubMed PMID: 22901911]
[42]
Abdullah F, Arnold MA, Nabaweesi R, Fischer AC, Colombani PM, Anderson KD, Lau H, Chang DC. Gastroschisis in the United States 1988-2003: analysis and risk categorization of 4344 patients. Journal of perinatology : official journal of the California Perinatal Association. 2007 Jan:27(1):50-5
[PubMed PMID: 17036030]
[43]
Alshehri A, Emil S, Laberge JM, Skarsgard E, Canadian Pediatric Surgery Network. Outcomes of early versus late intestinal operations in patients with gastroschisis and intestinal atresia: results from a prospective national database. Journal of pediatric surgery. 2013 Oct:48(10):2022-6. doi: 10.1016/j.jpedsurg.2013.04.003. Epub
[PubMed PMID: 24094951]
[44]
Houben C, Davenport M, Ade-Ajayi N, Flack N, Patel S. Closing gastroschisis: diagnosis, management, and outcomes. Journal of pediatric surgery. 2009 Feb:44(2):343-7. doi: 10.1016/j.jpedsurg.2008.10.084. Epub
[PubMed PMID: 19231531]