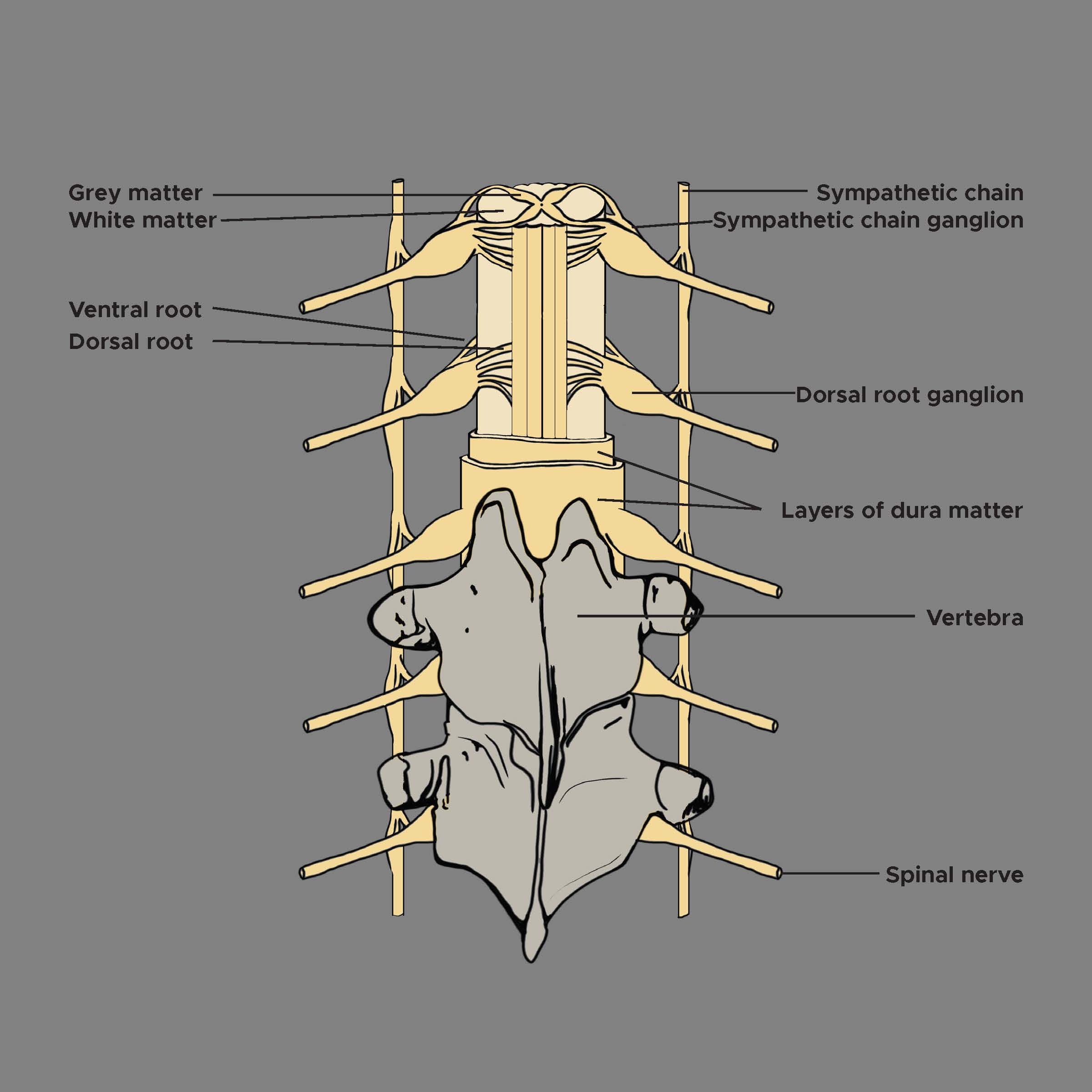
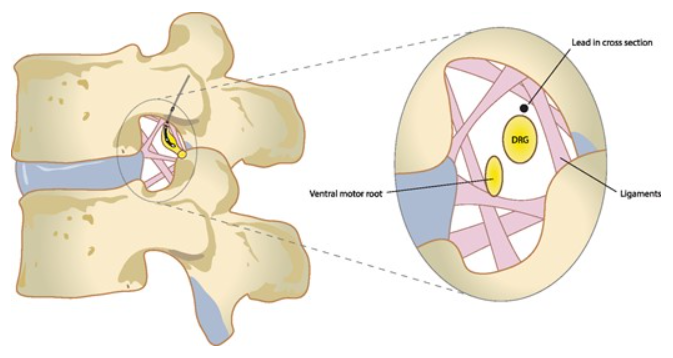
[1]
SORESI AL. Control of intractable pain by spinal ganglia block. American journal of surgery. 1949 Jan:77(1):72-8
[PubMed PMID: 18122204]
[2]
Deer TR, Levy RM, Kramer J, Poree L, Amirdelfan K, Grigsby E, Staats P, Burton AW, Burgher AH, Obray J, Scowcroft J, Golovac S, Kapural L, Paicius R, Kim C, Pope J, Yearwood T, Samuel S, McRoberts WP, Cassim H, Netherton M, Miller N, Schaufele M, Tavel E, Davis T, Davis K, Johnson L, Mekhail N. Dorsal root ganglion stimulation yielded higher treatment success rate for complex regional pain syndrome and causalgia at 3 and 12 months: a randomized comparative trial. Pain. 2017 Apr:158(4):669-681. doi: 10.1097/j.pain.0000000000000814. Epub
[PubMed PMID: 28030470]
Level 2 (mid-level) evidence
[3]
Liem L, Russo M, Huygen FJ, Van Buyten JP, Smet I, Verrills P, Cousins M, Brooker C, Levy R, Deer T, Kramer J. One-year outcomes of spinal cord stimulation of the dorsal root ganglion in the treatment of chronic neuropathic pain. Neuromodulation : journal of the International Neuromodulation Society. 2015 Jan:18(1):41-8; discussion 48-9. doi: 10.1111/ner.12228. Epub 2014 Aug 21
[PubMed PMID: 25145467]
[4]
Liem L, Russo M, Huygen FJ, Van Buyten JP, Smet I, Verrills P, Cousins M, Brooker C, Levy R, Deer T, Kramer J. A multicenter, prospective trial to assess the safety and performance of the spinal modulation dorsal root ganglion neurostimulator system in the treatment of chronic pain. Neuromodulation : journal of the International Neuromodulation Society. 2013 Sep-Oct:16(5):471-82; discussion 482. doi: 10.1111/ner.12072. Epub 2013 May 13
[PubMed PMID: 23668228]
[5]
Noh MC, Mikler B, Joy T, Smith PA. Time Course of Inflammation in Dorsal Root Ganglia Correlates with Differential Reversibility of Mechanical Allodynia. Neuroscience. 2020 Jan 21:428():199-216. doi: 10.1016/j.neuroscience.2019.12.040. Epub 2020 Jan 7
[PubMed PMID: 31918012]
[6]
Hogan QH. Labat lecture: the primary sensory neuron: where it is, what it does, and why it matters. Regional anesthesia and pain medicine. 2010 May-Jun:35(3):306-11. doi: 10.1097/AAP.0b013e3181d2375e. Epub
[PubMed PMID: 20460965]
[7]
BRIERLEY JB. The penetration of particulate matter from the cerebrospinal fluid into the spinal ganglia, peripheral nerves, and perivascular spaces of the central nervous system. Journal of neurology, neurosurgery, and psychiatry. 1950 Aug:13(3):203-15
[PubMed PMID: 14774743]
[8]
Devor M. Unexplained peculiarities of the dorsal root ganglion. Pain. 1999 Aug:Suppl 6():S27-S35. doi: 10.1016/S0304-3959(99)00135-9. Epub
[PubMed PMID: 10491970]
[9]
Znaor L, Lovrić S, Hogan Q, Sapunar D. Association of neural inflammation with hyperalgesia following spinal nerve ligation. Croatian medical journal. 2007 Feb:48(1):35-42
[PubMed PMID: 17309137]
[10]
Hanani M. Satellite glial cells in sensory ganglia: from form to function. Brain research. Brain research reviews. 2005 Jun:48(3):457-76
[PubMed PMID: 15914252]
[11]
Leijnse JN, D'Herde K. Revisiting the segmental organization of the human spinal cord. Journal of anatomy. 2016 Sep:229(3):384-93. doi: 10.1111/joa.12493. Epub 2016 May 12
[PubMed PMID: 27173936]
[13]
Hunter CW, Sayed D, Lubenow T, Davis T, Carlson J, Rowe J, Justiz R, McJunkin T, Deer T, Mehta P, Falowski S, Kapural L, Pope J, Mekhail N. DRG FOCUS: A Multicenter Study Evaluating Dorsal Root Ganglion Stimulation and Predictors for Trial Success. Neuromodulation : journal of the International Neuromodulation Society. 2019 Jan:22(1):61-79. doi: 10.1111/ner.12796. Epub 2018 Aug 7
[PubMed PMID: 30085382]
Level 2 (mid-level) evidence
[14]
Huygen F, Liem L, Cusack W, Kramer J. Stimulation of the L2-L3 Dorsal Root Ganglia Induces Effective Pain Relief in the Low Back. Pain practice : the official journal of World Institute of Pain. 2018 Feb:18(2):205-213. doi: 10.1111/papr.12591. Epub 2017 Dec 6
[PubMed PMID: 28486758]
[15]
Hasegawa T, Mikawa Y, Watanabe R, An HS. Morphometric analysis of the lumbosacral nerve roots and dorsal root ganglia by magnetic resonance imaging. Spine. 1996 May 1:21(9):1005-9
[PubMed PMID: 8724082]
[16]
Ebraheim NA,Lu J, Morphometric evaluation of the sacral dorsal root ganglia. A cadaveric study. Surgical and radiologic anatomy : SRA. 1998;
[PubMed PMID: 9658528]
[17]
Lirk P, Poroli M, Rigaud M, Fuchs A, Fillip P, Huang CY, Ljubkovic M, Sapunar D, Hogan Q. Modulators of calcium influx regulate membrane excitability in rat dorsal root ganglion neurons. Anesthesia and analgesia. 2008 Aug:107(2):673-85. doi: 10.1213/ane.0b013e31817b7a73. Epub
[PubMed PMID: 18633052]
[18]
Rush AM, Dib-Hajj SD, Liu S, Cummins TR, Black JA, Waxman SG. A single sodium channel mutation produces hyper- or hypoexcitability in different types of neurons. Proceedings of the National Academy of Sciences of the United States of America. 2006 May 23:103(21):8245-50
[PubMed PMID: 16702558]
[19]
Kovalsky Y, Amir R, Devor M. Simulation in sensory neurons reveals a key role for delayed Na+ current in subthreshold oscillations and ectopic discharge: implications for neuropathic pain. Journal of neurophysiology. 2009 Sep:102(3):1430-42. doi: 10.1152/jn.00005.2009. Epub 2009 Jul 1
[PubMed PMID: 19571204]
[20]
Teillet MA,Kalcheim C,Le Douarin NM, Formation of the dorsal root ganglia in the avian embryo: segmental origin and migratory behavior of neural crest progenitor cells. Developmental biology. 1987 Apr
[PubMed PMID: 3549390]
[21]
Utzschneider D, Kocsis J, Devor M. Mutual excitation among dorsal root ganglion neurons in the rat. Neuroscience letters. 1992 Oct 26:146(1):53-6
[PubMed PMID: 1475049]
Level 3 (low-level) evidence
[22]
Kent AR, Min X, Hogan QH, Kramer JM. Mechanisms of Dorsal Root Ganglion Stimulation in Pain Suppression: A Computational Modeling Analysis. Neuromodulation : journal of the International Neuromodulation Society. 2018 Apr:21(3):234-246. doi: 10.1111/ner.12754. Epub 2018 Jan 29
[PubMed PMID: 29377442]
[23]
Pinto V, Szûcs P, Derkach VA, Safronov BV. Monosynaptic convergence of C- and Adelta-afferent fibres from different segmental dorsal roots on to single substantia gelatinosa neurones in the rat spinal cord. The Journal of physiology. 2008 Sep 1:586(17):4165-77. doi: 10.1113/jphysiol.2008.154898. Epub 2008 Jul 17
[PubMed PMID: 18635648]
[24]
Kalcheim C,Le Douarin NM, Requirement of a neural tube signal for the differentiation of neural crest cells into dorsal root ganglia. Developmental biology. 1986 Aug
[PubMed PMID: 3525281]
[25]
Parke WW, Whalen JL. The vascular pattern of the human dorsal root ganglion and its probable bearing on a compartment syndrome. Spine. 2002 Feb 15:27(4):347-52
[PubMed PMID: 11840098]
[26]
Haberberger RV, Barry C, Dominguez N, Matusica D. Human Dorsal Root Ganglia. Frontiers in cellular neuroscience. 2019:13():271. doi: 10.3389/fncel.2019.00271. Epub 2019 Jun 19
[PubMed PMID: 31293388]
[27]
Krames ES. The role of the dorsal root ganglion in the development of neuropathic pain. Pain medicine (Malden, Mass.). 2014 Oct:15(10):1669-85. doi: 10.1111/pme.12413. Epub 2014 Mar 18
[PubMed PMID: 24641192]
[28]
Han C, Hoeijmakers JG, Liu S, Gerrits MM, te Morsche RH, Lauria G, Dib-Hajj SD, Drenth JP, Faber CG, Merkies IS, Waxman SG. Functional profiles of SCN9A variants in dorsal root ganglion neurons and superior cervical ganglion neurons correlate with autonomic symptoms in small fibre neuropathy. Brain : a journal of neurology. 2012 Sep:135(Pt 9):2613-28. doi: 10.1093/brain/aws187. Epub 2012 Jul 22
[PubMed PMID: 22826602]
[29]
Pope JE, Deer TR, Kramer J. A systematic review: current and future directions of dorsal root ganglion therapeutics to treat chronic pain. Pain medicine (Malden, Mass.). 2013 Oct:14(10):1477-96. doi: 10.1111/pme.12171. Epub 2013 Jun 26
[PubMed PMID: 23802747]
Level 3 (low-level) evidence
[30]
Liem L, van Dongen E, Huygen FJ, Staats P, Kramer J. The Dorsal Root Ganglion as a Therapeutic Target for Chronic Pain. Regional anesthesia and pain medicine. 2016 Jul-Aug:41(4):511-9. doi: 10.1097/AAP.0000000000000408. Epub
[PubMed PMID: 27224659]
[31]
Liu CN, Wall PD, Ben-Dor E, Michaelis M, Amir R, Devor M. Tactile allodynia in the absence of C-fiber activation: altered firing properties of DRG neurons following spinal nerve injury. Pain. 2000 Apr:85(3):503-521. doi: 10.1016/S0304-3959(00)00251-7. Epub
[PubMed PMID: 10781925]
[32]
Hu P, McLachlan EM. Distinct functional types of macrophage in dorsal root ganglia and spinal nerves proximal to sciatic and spinal nerve transections in the rat. Experimental neurology. 2003 Dec:184(2):590-605
[PubMed PMID: 14769352]
[33]
Sapunar D, Kostic S, Banozic A, Puljak L. Dorsal root ganglion - a potential new therapeutic target for neuropathic pain. Journal of pain research. 2012:5():31-8. doi: 10.2147/JPR.S26603. Epub 2012 Feb 16
[PubMed PMID: 22375099]
[34]
Harrison C, Epton S, Bojanic S, Green AL, FitzGerald JJ. The Efficacy and Safety of Dorsal Root Ganglion Stimulation as a Treatment for Neuropathic Pain: A Literature Review. Neuromodulation : journal of the International Neuromodulation Society. 2018 Apr:21(3):225-233. doi: 10.1111/ner.12685. Epub 2017 Sep 28
[PubMed PMID: 28960653]
[35]
Kanno T, Yaguchi T, Nishizaki T. Noradrenaline stimulates ATP release from DRG neurons by targeting beta(3) adrenoceptors as a factor of neuropathic pain. Journal of cellular physiology. 2010 Aug:224(2):345-51. doi: 10.1002/jcp.22114. Epub
[PubMed PMID: 20432431]
[36]
Amir R, Michaelis M, Devor M. Membrane potential oscillations in dorsal root ganglion neurons: role in normal electrogenesis and neuropathic pain. The Journal of neuroscience : the official journal of the Society for Neuroscience. 1999 Oct 1:19(19):8589-96
[PubMed PMID: 10493758]
[37]
Nakamura SI, Myers RR. Injury to dorsal root ganglia alters innervation of spinal cord dorsal horn lamina involved in nociception. Spine. 2000 Mar 1:25(5):537-42
[PubMed PMID: 10749628]
[38]
Kajander KC, Wakisaka S, Bennett GJ. Spontaneous discharge originates in the dorsal root ganglion at the onset of a painful peripheral neuropathy in the rat. Neuroscience letters. 1992 Apr 27:138(2):225-8
[PubMed PMID: 1319012]
Level 3 (low-level) evidence
[39]
Ma C, LaMotte RH. Multiple sites for generation of ectopic spontaneous activity in neurons of the chronically compressed dorsal root ganglion. The Journal of neuroscience : the official journal of the Society for Neuroscience. 2007 Dec 19:27(51):14059-68
[PubMed PMID: 18094245]
[40]
Devor M. Ectopic discharge in Abeta afferents as a source of neuropathic pain. Experimental brain research. 2009 Jun:196(1):115-28. doi: 10.1007/s00221-009-1724-6. Epub 2009 Feb 26
[PubMed PMID: 19242687]
[41]
Deer TR, Grigsby E, Weiner RL, Wilcosky B, Kramer JM. A prospective study of dorsal root ganglion stimulation for the relief of chronic pain. Neuromodulation : journal of the International Neuromodulation Society. 2013 Jan-Feb:16(1):67-71; discussion 71-2. doi: 10.1111/ner.12013. Epub 2012 Dec 14
[PubMed PMID: 23240657]
[42]
Van Buyten JP, Smet I, Liem L, Russo M, Huygen F. Stimulation of dorsal root ganglia for the management of complex regional pain syndrome: a prospective case series. Pain practice : the official journal of World Institute of Pain. 2015 Mar:15(3):208-16. doi: 10.1111/papr.12170. Epub 2014 Jan 23
[PubMed PMID: 24451048]
Level 2 (mid-level) evidence
[43]
Maino P, Koetsier E, Kaelin-Lang A, Gobbi C, Perez R. Efficacious Dorsal Root Ganglion Stimulation for Painful Small Fiber Neuropathy: A Case Report. Pain physician. 2017 Mar:20(3):E459-E463
[PubMed PMID: 28339448]
Level 3 (low-level) evidence
[44]
Liem L, Mekhail N. Management of Postherniorrhaphy Chronic Neuropathic Groin Pain: A Role for Dorsal Root Ganglion Stimulation. Pain practice : the official journal of World Institute of Pain. 2016 Sep:16(7):915-23. doi: 10.1111/papr.12424. Epub 2016 Feb 23
[PubMed PMID: 26914499]
[45]
Zuidema X, Breel J, Wille F. Paresthesia mapping: a practical workup for successful implantation of the dorsal root ganglion stimulator in refractory groin pain. Neuromodulation : journal of the International Neuromodulation Society. 2014 Oct:17(7):665-9; discussion 669. doi: 10.1111/ner.12113. Epub 2014 Feb 26
[PubMed PMID: 24571400]
[46]
Schu S, Gulve A, ElDabe S, Baranidharan G, Wolf K, Demmel W, Rasche D, Sharma M, Klase D, Jahnichen G, Wahlstedt A, Nijhuis H, Liem L. Spinal cord stimulation of the dorsal root ganglion for groin pain-a retrospective review. Pain practice : the official journal of World Institute of Pain. 2015 Apr:15(4):293-9. doi: 10.1111/papr.12194. Epub 2014 Apr 1
[PubMed PMID: 24690212]
Level 2 (mid-level) evidence
[47]
Feirabend HK, Choufoer H, Ploeger S, Holsheimer J, van Gool JD. Morphometry of human superficial dorsal and dorsolateral column fibres: significance to spinal cord stimulation. Brain : a journal of neurology. 2002 May:125(Pt 5):1137-49
[PubMed PMID: 11960902]
[48]
Eldabe S, Burger K, Moser H, Klase D, Schu S, Wahlstedt A, Vanderick B, Francois E, Kramer J, Subbaroyan J. Dorsal Root Ganglion (DRG) Stimulation in the Treatment of Phantom Limb Pain (PLP). Neuromodulation : journal of the International Neuromodulation Society. 2015 Oct:18(7):610-6; discussion 616-7. doi: 10.1111/ner.12338. Epub 2015 Aug 13
[PubMed PMID: 26268453]
[49]
Hunter CW, Yang A, Davis T. Selective Radiofrequency Stimulation of the Dorsal Root Ganglion (DRG) as a Method for Predicting Targets for Neuromodulation in Patients With Post Amputation Pain: A Case Series. Neuromodulation : journal of the International Neuromodulation Society. 2017 Oct:20(7):708-718. doi: 10.1111/ner.12595. Epub 2017 Mar 23
[PubMed PMID: 28337820]
Level 2 (mid-level) evidence
[50]
Lynch PJ, McJunkin T, Eross E, Gooch S, Maloney J. Case report: successful epiradicular peripheral nerve stimulation of the C2 dorsal root ganglion for postherpetic neuralgia. Neuromodulation : journal of the International Neuromodulation Society. 2011 Jan:14(1):58-61; discussion 61. doi: 10.1111/j.1525-1403.2010.00307.x. Epub 2010 Nov 4
[PubMed PMID: 21992163]
Level 3 (low-level) evidence
[51]
Eldabe S, Espinet A, Wahlstedt A, Kang P, Liem L, Patel NK, Vesper J, Kimber A, Cusack W, Kramer J. Retrospective Case Series on the Treatment of Painful Diabetic Peripheral Neuropathy With Dorsal Root Ganglion Stimulation. Neuromodulation : journal of the International Neuromodulation Society. 2018 Dec:21(8):787-792. doi: 10.1111/ner.12767. Epub 2018 Mar 25
[PubMed PMID: 29575331]
Level 2 (mid-level) evidence
[52]
Zuidema X, Breel J, Wille F. S3 Dorsal Root Ganglion/Nerve Root Stimulation for Refractory Postsurgical Perineal Pain: Technical Aspects of Anchorless Sacral Transforaminal Lead Placement. Case reports in neurological medicine. 2016:2016():8926578. doi: 10.1155/2016/8926578. Epub 2016 Mar 30
[PubMed PMID: 27123351]
Level 3 (low-level) evidence
[53]
Morgalla MH, Fortunato M, Lepski G, Chander BS. Dorsal Root Ganglion Stimulation (DRGS) for the Treatment of Chronic Neuropathic Pain: A Single-Center Study with Long-Term Prospective Results in 62 Cases. Pain physician. 2018 Jul:21(4):E377-E387
[PubMed PMID: 30045604]
Level 3 (low-level) evidence
[54]
Deer TR, Mekhail N, Provenzano D, Pope J, Krames E, Leong M, Levy RM, Abejon D, Buchser E, Burton A, Buvanendran A, Candido K, Caraway D, Cousins M, DeJongste M, Diwan S, Eldabe S, Gatzinsky K, Foreman RD, Hayek S, Kim P, Kinfe T, Kloth D, Kumar K, Rizvi S, Lad SP, Liem L, Linderoth B, Mackey S, McDowell G, McRoberts P, Poree L, Prager J, Raso L, Rauck R, Russo M, Simpson B, Slavin K, Staats P, Stanton-Hicks M, Verrills P, Wellington J, Williams K, North R, Neuromodulation Appropriateness Consensus Committee. The appropriate use of neurostimulation of the spinal cord and peripheral nervous system for the treatment of chronic pain and ischemic diseases: the Neuromodulation Appropriateness Consensus Committee. Neuromodulation : journal of the International Neuromodulation Society. 2014 Aug:17(6):515-50; discussion 550. doi: 10.1111/ner.12208. Epub
[PubMed PMID: 25112889]
Level 3 (low-level) evidence