[1]
Sellers D,Chess-Williams R,Michel MC, Modulation of lower urinary tract smooth muscle contraction and relaxation by the urothelium. Naunyn-Schmiedeberg's archives of pharmacology. 2018 Jul;
[PubMed PMID: 29808232]
[3]
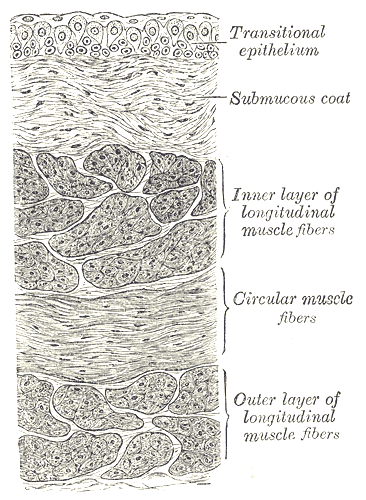
Andersson KE, Arner A. Urinary bladder contraction and relaxation: physiology and pathophysiology. Physiological reviews. 2004 Jul:84(3):935-86
[PubMed PMID: 15269341]
[4]
Caione P,Capozza N,Zavaglia D,Palombaro G,Boldrini R, In vivo bladder regeneration using small intestinal submucosa: experimental study. Pediatric surgery international. 2006 Jul
[PubMed PMID: 16773371]
[5]
Abelson B,Sun D,Que L,Nebel RA,Baker D,Popiel P,Amundsen CL,Chai T,Close C,DiSanto M,Fraser MO,Kielb SJ,Kuchel G,Mueller ER,Palmer MH,Parker-Autry C,Wolfe AJ,Damaser MS, Sex differences in lower urinary tract biology and physiology. Biology of sex differences. 2018 Oct 22;
[PubMed PMID: 30343668]
[6]
Walz T,Häner M,Wu XR,Henn C,Engel A,Sun TT,Aebi U, Towards the molecular architecture of the asymmetric unit membrane of the mammalian urinary bladder epithelium: a closed
[PubMed PMID: 7760330]
[7]
Andersson KE,McCloskey KD, Lamina propria: the functional center of the bladder? Neurourology and urodynamics. 2014 Jan;
[PubMed PMID: 23847015]
[8]
Drumm BT,Koh SD,Andersson KE,Ward SM, Calcium signalling in Cajal-like interstitial cells of the lower urinary tract. Nature reviews. Urology. 2014 Oct;
[PubMed PMID: 25224445]
[9]
Sánchez Freire V,Burkhard FC,Schmitz A,Kessler TM,Monastyrskaya K, Structural differences between the bladder dome and trigone revealed by mRNA expression analysis of cold-cut biopsies. BJU international. 2011 Jul;
[PubMed PMID: 21244608]
[10]
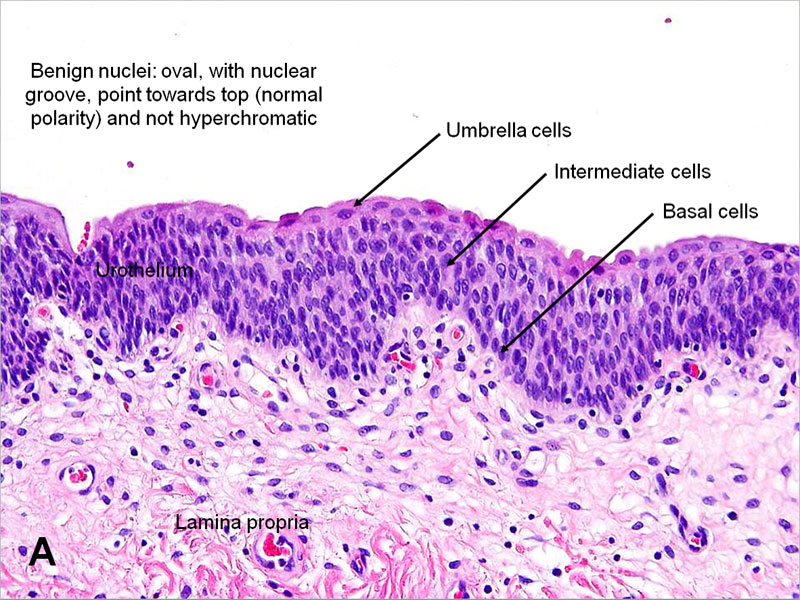
Khandelwal P, Abraham SN, Apodaca G. Cell biology and physiology of the uroepithelium. American journal of physiology. Renal physiology. 2009 Dec:297(6):F1477-501. doi: 10.1152/ajprenal.00327.2009. Epub 2009 Jul 8
[PubMed PMID: 19587142]
[11]
Vannucchi MG,Traini C,Guasti D,Del Popolo G,Faussone-Pellegrini MS, Telocytes subtypes in human urinary bladder. Journal of cellular and molecular medicine. 2014 Oct;
[PubMed PMID: 25139461]
[12]
Butros SR,McCarthy CJ,Karaosmanoğlu AD,Shenoy-Bhangle AS,Arellano RS, Feasibility and effectiveness of image-guided percutaneous biopsy of the urinary bladder. Abdominal imaging. 2015 Aug;
[PubMed PMID: 25875861]
Level 2 (mid-level) evidence
[13]
Wang G,McKenney JK, Urinary Bladder Pathology: World Health Organization (WHO) Classification and American Joint Committee on Cancer (AJCC) Staging Update. Archives of pathology
[PubMed PMID: 30044124]
[14]
Jhang JF,Ho HC,Jiang YH,Lee CL,Hsu YH,Kuo HC, Electron microscopic characteristics of interstitial cystitis/bladder pain syndrome and their association with clinical condition. PloS one. 2018
[PubMed PMID: 29879217]
[15]
Hodges KB, Lopez-Beltran A, Maclennan GT, Montironi R, Cheng L. Urothelial lesions with inverted growth patterns: histogenesis, molecular genetic findings, differential diagnosis and clinical management. BJU international. 2011 Feb:107(4):532-7. doi: 10.1111/j.1464-410X.2010.09853.x. Epub 2010 Nov 23
[PubMed PMID: 21091975]
[16]
Castillo-Martin M,Domingo-Domenech J,Karni-Schmidt O,Matos T,Cordon-Cardo C, Molecular pathways of urothelial development and bladder tumorigenesis. Urologic oncology. 2010 Jul-Aug
[PubMed PMID: 20610278]