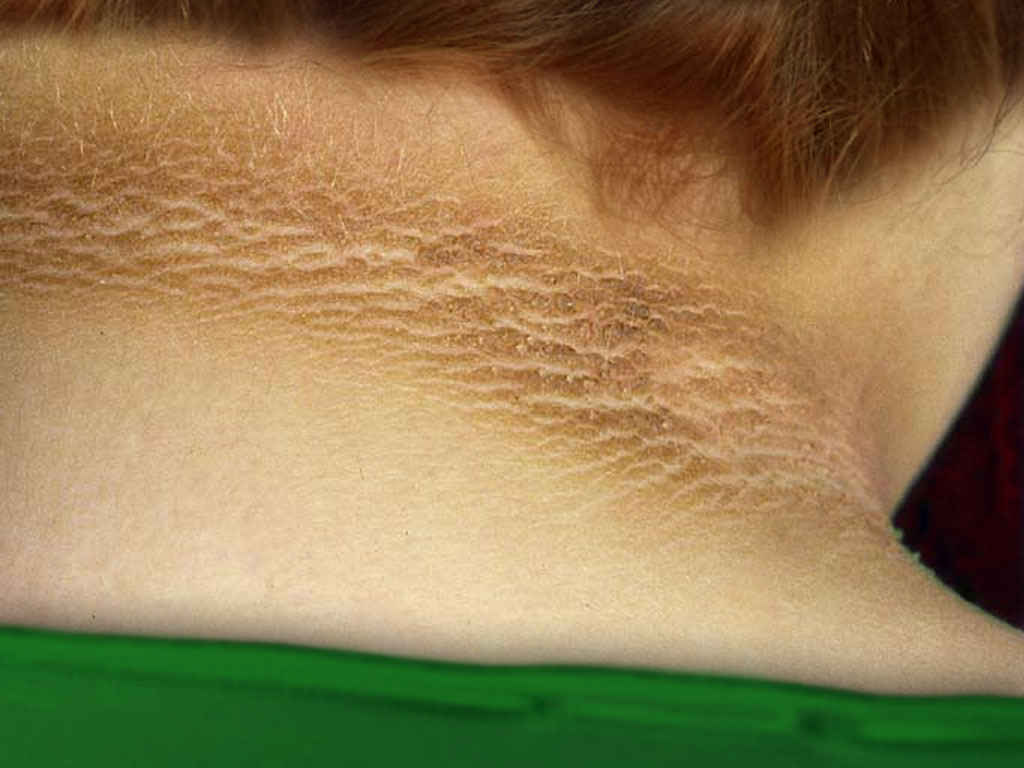
[1]
Seong J, Kang JY, Sun JS, Kim KW. Hypothalamic inflammation and obesity: a mechanistic review. Archives of pharmacal research. 2019 May:42(5):383-392. doi: 10.1007/s12272-019-01138-9. Epub 2019 Mar 5
[PubMed PMID: 30835074]
[2]
Brown JC, Harhay MO, Harhay MN. The Value of Anthropometric Measures in Nutrition and Metabolism: Comment on Anthropometrically Predicted Visceral Adipose Tissue and Blood-Based Biomarkers: A Cross-Sectional Analysis. Nutrition and metabolic insights. 2019:12():1178638819831712. doi: 10.1177/1178638819831712. Epub 2019 Feb 27
[PubMed PMID: 30833814]
Level 2 (mid-level) evidence
[3]
Nolan CJ, Prentki M. Insulin resistance and insulin hypersecretion in the metabolic syndrome and type 2 diabetes: Time for a conceptual framework shift. Diabetes & vascular disease research. 2019 Mar:16(2):118-127. doi: 10.1177/1479164119827611. Epub 2019 Feb 15
[PubMed PMID: 30770030]
[4]
Deacon CF. Physiology and Pharmacology of DPP-4 in Glucose Homeostasis and the Treatment of Type 2 Diabetes. Frontiers in endocrinology. 2019:10():80. doi: 10.3389/fendo.2019.00080. Epub 2019 Feb 15
[PubMed PMID: 30828317]
[5]
Thomas DD, Corkey BE, Istfan NW, Apovian CM. Hyperinsulinemia: An Early Indicator of Metabolic Dysfunction. Journal of the Endocrine Society. 2019 Sep 1:3(9):1727-1747. doi: 10.1210/js.2019-00065. Epub 2019 Jul 24
[PubMed PMID: 31528832]
[6]
Hossan T, Kundu S, Alam SS, Nagarajan S. Epigenetic Modifications Associated with the Pathogenesis of Type 2 Diabetes Mellitus. Endocrine, metabolic & immune disorders drug targets. 2019:19(6):775-786. doi: 10.2174/1871530319666190301145545. Epub
[PubMed PMID: 30827271]
[7]
Bothou C, Beuschlein F, Spyroglou A. Links between aldosterone excess and metabolic complications: A comprehensive review. Diabetes & metabolism. 2020 Feb:46(1):1-7. doi: 10.1016/j.diabet.2019.02.003. Epub 2019 Feb 27
[PubMed PMID: 30825519]
[8]
Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985 Jul:28(7):412-9
[PubMed PMID: 3899825]
[9]
Levy JC, Matthews DR, Hermans MP. Correct homeostasis model assessment (HOMA) evaluation uses the computer program. Diabetes care. 1998 Dec:21(12):2191-2
[PubMed PMID: 9839117]
[10]
Katz A, Nambi SS, Mather K, Baron AD, Follmann DA, Sullivan G, Quon MJ. Quantitative insulin sensitivity check index: a simple, accurate method for assessing insulin sensitivity in humans. The Journal of clinical endocrinology and metabolism. 2000 Jul:85(7):2402-10
[PubMed PMID: 10902785]
[11]
Kim-Dorner SJ, Deuster PA, Zeno SA, Remaley AT, Poth M. Should triglycerides and the triglycerides to high-density lipoprotein cholesterol ratio be used as surrogates for insulin resistance? Metabolism: clinical and experimental. 2010 Feb:59(2):299-304. doi: 10.1016/j.metabol.2009.07.027. Epub 2009 Sep 30
[PubMed PMID: 19796777]
[12]
Tobin GS, Cavaghan MK, Hoogwerf BJ, McGill JB. Addition of exenatide twice daily to basal insulin for the treatment of type 2 diabetes: clinical studies and practical approaches to therapy. International journal of clinical practice. 2012 Dec:66(12):1147-57. doi: 10.1111/ijcp.12032. Epub 2012 Oct 14
[PubMed PMID: 23061886]
[13]
Abdul-Ghani M, DeFronzo RA. Insulin Resistance and Hyperinsulinemia: the Egg and the Chicken. The Journal of clinical endocrinology and metabolism. 2021 Mar 25:106(4):e1897-e1899. doi: 10.1210/clinem/dgaa364. Epub
[PubMed PMID: 33522574]
[14]
Laursen TL, Hagemann CA, Wei C, Kazankov K, Thomsen KL, Knop FK, Grønbæk H. Bariatric surgery in patients with non-alcoholic fatty liver disease - from pathophysiology to clinical effects. World journal of hepatology. 2019 Feb 27:11(2):138-149. doi: 10.4254/wjh.v11.i2.138. Epub
[PubMed PMID: 30820265]
[15]
Pennings N, Jaber J, Ahiawodzi P. Ten-year weight gain is associated with elevated fasting insulin levels and precedes glucose elevation. Diabetes/metabolism research and reviews. 2018 May:34(4):e2986. doi: 10.1002/dmrr.2986. Epub 2018 Mar 15
[PubMed PMID: 29392827]
[16]
Church TJ, Haines ST. Treatment Approach to Patients With Severe Insulin Resistance. Clinical diabetes : a publication of the American Diabetes Association. 2016 Apr:34(2):97-104. doi: 10.2337/diaclin.34.2.97. Epub
[PubMed PMID: 27092020]
[17]
Diamanti-Kandarakis E, Dunaif A. Insulin resistance and the polycystic ovary syndrome revisited: an update on mechanisms and implications. Endocrine reviews. 2012 Dec:33(6):981-1030. doi: 10.1210/er.2011-1034. Epub 2012 Oct 12
[PubMed PMID: 23065822]
[18]
Engin A. The Definition and Prevalence of Obesity and Metabolic Syndrome. Advances in experimental medicine and biology. 2017:960():1-17. doi: 10.1007/978-3-319-48382-5_1. Epub
[PubMed PMID: 28585193]
Level 3 (low-level) evidence
[19]
Tanase DM, Gosav EM, Costea CF, Ciocoiu M, Lacatusu CM, Maranduca MA, Ouatu A, Floria M. The Intricate Relationship between Type 2 Diabetes Mellitus (T2DM), Insulin Resistance (IR), and Nonalcoholic Fatty Liver Disease (NAFLD). Journal of diabetes research. 2020:2020():3920196. doi: 10.1155/2020/3920196. Epub 2020 Jul 31
[PubMed PMID: 32832560]
[20]
Nellaiappan K, Preeti K, Khatri DK, Singh SB. Diabetic Complications: An Update on Pathobiology and Therapeutic Strategies. Current diabetes reviews. 2022:18(1):e030821192146. doi: 10.2174/1573399817666210309104203. Epub
[PubMed PMID: 33745424]
[21]
Reaven GM. The metabolic syndrome: is this diagnosis necessary? The American journal of clinical nutrition. 2006 Jun:83(6):1237-47
[PubMed PMID: 16762930]
[22]
McCormick N, O'Connor MJ, Yokose C, Merriman TR, Mount DB, Leong A, Choi HK. Assessing the Causal Relationships Between Insulin Resistance and Hyperuricemia and Gout Using Bidirectional Mendelian Randomization. Arthritis & rheumatology (Hoboken, N.J.). 2021 Nov:73(11):2096-2104. doi: 10.1002/art.41779. Epub 2021 Sep 26
[PubMed PMID: 33982892]
[23]
Deshpande AD, Harris-Hayes M, Schootman M. Epidemiology of diabetes and diabetes-related complications. Physical therapy. 2008 Nov:88(11):1254-64. doi: 10.2522/ptj.20080020. Epub 2008 Sep 18
[PubMed PMID: 18801858]
[24]
Knowler WC, Barrett-Connor E, Fowler SE, Hamman RF, Lachin JM, Walker EA, Nathan DM, Diabetes Prevention Program Research Group. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. The New England journal of medicine. 2002 Feb 7:346(6):393-403
[PubMed PMID: 11832527]
[25]
Samuel VT, Shulman GI. The pathogenesis of insulin resistance: integrating signaling pathways and substrate flux. The Journal of clinical investigation. 2016 Jan:126(1):12-22. doi: 10.1172/JCI77812. Epub 2016 Jan 4
[PubMed PMID: 26727229]
[26]
Perreault L, Pan Q, Schroeder EB, Kalyani RR, Bray GA, Dagogo-Jack S, White NH, Goldberg RB, Kahn SE, Knowler WC, Mathioudakis N, Dabelea D, Diabetes Prevention Program Research Group. Regression From Prediabetes to Normal Glucose Regulation and Prevalence of Microvascular Disease in the Diabetes Prevention Program Outcomes Study (DPPOS). Diabetes care. 2019 Sep:42(9):1809-1815. doi: 10.2337/dc19-0244. Epub 2019 Jul 18
[PubMed PMID: 31320445]
[27]
Ogawa W, Araki E, Ishigaki Y, Hirota Y, Maegawa H, Yamauchi T, Yorifuji T, Katagiri H. New classification and diagnostic criteria for insulin resistance syndrome. Endocrine journal. 2022 Feb 28:69(2):107-113. doi: 10.1507/endocrj.EJ21-0725. Epub 2022 Feb 1
[PubMed PMID: 35110500]
[28]
Park YW, Zhu S, Palaniappan L, Heshka S, Carnethon MR, Heymsfield SB. The metabolic syndrome: prevalence and associated risk factor findings in the US population from the Third National Health and Nutrition Examination Survey, 1988-1994. Archives of internal medicine. 2003 Feb 24:163(4):427-36
[PubMed PMID: 12588201]
Level 3 (low-level) evidence
[29]
Parcha V, Heindl B, Kalra R, Li P, Gower B, Arora G, Arora P. Insulin Resistance and Cardiometabolic Risk Profile Among Nondiabetic American Young Adults: Insights From NHANES. The Journal of clinical endocrinology and metabolism. 2022 Jan 1:107(1):e25-e37. doi: 10.1210/clinem/dgab645. Epub
[PubMed PMID: 34473288]
[30]
Petersen MC, Shulman GI. Mechanisms of Insulin Action and Insulin Resistance. Physiological reviews. 2018 Oct 1:98(4):2133-2223. doi: 10.1152/physrev.00063.2017. Epub
[PubMed PMID: 30067154]
[31]
Kim JK. Hyperinsulinemic-euglycemic clamp to assess insulin sensitivity in vivo. Methods in molecular biology (Clifton, N.J.). 2009:560():221-38. doi: 10.1007/978-1-59745-448-3_15. Epub
[PubMed PMID: 19504253]
[32]
Legro RS, Finegood D, Dunaif A. A fasting glucose to insulin ratio is a useful measure of insulin sensitivity in women with polycystic ovary syndrome. The Journal of clinical endocrinology and metabolism. 1998 Aug:83(8):2694-8
[PubMed PMID: 9709933]
[33]
McAuley KA, Williams SM, Mann JI, Walker RJ, Lewis-Barned NJ, Temple LA, Duncan AW. Diagnosing insulin resistance in the general population. Diabetes care. 2001 Mar:24(3):460-4
[PubMed PMID: 11289468]
[34]
Stumvoll M, Van Haeften T, Fritsche A, Gerich J. Oral glucose tolerance test indexes for insulin sensitivity and secretion based on various availabilities of sampling times. Diabetes care. 2001 Apr:24(4):796-7
[PubMed PMID: 11315860]
[35]
Matsuda M, DeFronzo RA. Insulin sensitivity indices obtained from oral glucose tolerance testing: comparison with the euglycemic insulin clamp. Diabetes care. 1999 Sep:22(9):1462-70
[PubMed PMID: 10480510]
[36]
Gutt M, Davis CL, Spitzer SB, Llabre MM, Kumar M, Czarnecki EM, Schneiderman N, Skyler JS, Marks JB. Validation of the insulin sensitivity index (ISI(0,120)): comparison with other measures. Diabetes research and clinical practice. 2000 Mar:47(3):177-84
[PubMed PMID: 10741566]
Level 1 (high-level) evidence
[37]
Sumner AE, Finley KB, Genovese DJ, Criqui MH, Boston RC. Fasting triglyceride and the triglyceride-HDL cholesterol ratio are not markers of insulin resistance in African Americans. Archives of internal medicine. 2005 Jun 27:165(12):1395-400
[PubMed PMID: 15983289]
[38]
Alberti KG, Eckel RH, Grundy SM, Zimmet PZ, Cleeman JI, Donato KA, Fruchart JC, James WP, Loria CM, Smith SC Jr, International Diabetes Federation Task Force on Epidemiology and Prevention, Hational Heart, Lung, and Blood Institute, American Heart Association, World Heart Federation, International Atherosclerosis Society, International Association for the Study of Obesity. Harmonizing the metabolic syndrome: a joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation. 2009 Oct 20:120(16):1640-5. doi: 10.1161/CIRCULATIONAHA.109.192644. Epub 2009 Oct 5
[PubMed PMID: 19805654]
[39]
Einhorn D, Reaven GM, Cobin RH, Ford E, Ganda OP, Handelsman Y, Hellman R, Jellinger PS, Kendall D, Krauss RM, Neufeld ND, Petak SM, Rodbard HW, Seibel JA, Smith DA, Wilson PW. American College of Endocrinology position statement on the insulin resistance syndrome. Endocrine practice : official journal of the American College of Endocrinology and the American Association of Clinical Endocrinologists. 2003 May-Jun:9(3):237-52
[PubMed PMID: 12924350]
[40]
Rácz O, Linková M, Jakubowski K, Link R, Kuzmová D. [Barriers of the initiation of insulin treatment in type 2 diabetic patients - conquering the "psychological insulin resistance"]. Orvosi hetilap. 2019 Jan:160(3):93-97. doi: 10.1556/650.2019.31269. Epub
[PubMed PMID: 30640530]
[41]
Yaribeygi H, Atkin SL, Simental-Mendía LE, Sahebkar A. Molecular mechanisms by which aerobic exercise induces insulin sensitivity. Journal of cellular physiology. 2019 Aug:234(8):12385-12392. doi: 10.1002/jcp.28066. Epub 2019 Jan 3
[PubMed PMID: 30605232]
[42]
He X, Wu D, Hu C, Xu T, Liu Y, Liu C, Xu B, Tang W. Role of Metformin in the Treatment of Patients with Thyroid Nodules and Insulin Resistance: A Systematic Review and Meta-Analysis. Thyroid : official journal of the American Thyroid Association. 2019 Mar:29(3):359-367. doi: 10.1089/thy.2017.0707. Epub 2019 Feb 1
[PubMed PMID: 30595105]
Level 1 (high-level) evidence
[43]
Zhou J, Massey S, Story D, Li L. Metformin: An Old Drug with New Applications. International journal of molecular sciences. 2018 Sep 21:19(10):. doi: 10.3390/ijms19102863. Epub 2018 Sep 21
[PubMed PMID: 30241400]
[44]
Mottl AK, Alicic R, Argyropoulos C, Brosius FC, Mauer M, Molitch M, Nelson RG, Perreault L, Nicholas SB. KDOQI US Commentary on the KDIGO 2020 Clinical Practice Guideline for Diabetes Management in CKD. American journal of kidney diseases : the official journal of the National Kidney Foundation. 2022 Apr:79(4):457-479. doi: 10.1053/j.ajkd.2021.09.010. Epub 2022 Feb 7
[PubMed PMID: 35144840]
Level 1 (high-level) evidence
[45]
American Geriatrics Society Expert Panel on Care of Older Adults with Diabetes Mellitus, Moreno G, Mangione CM, Kimbro L, Vaisberg E. Guidelines abstracted from the American Geriatrics Society Guidelines for Improving the Care of Older Adults with Diabetes Mellitus: 2013 update. Journal of the American Geriatrics Society. 2013 Nov:61(11):2020-6. doi: 10.1111/jgs.12514. Epub
[PubMed PMID: 24219204]
[46]
Mehta A, Marso SP, Neeland IJ. Liraglutide for weight management: a critical review of the evidence. Obesity science & practice. 2017 Mar:3(1):3-14. doi: 10.1002/osp4.84. Epub 2016 Dec 19
[PubMed PMID: 28392927]
[47]
Slomski A. Semaglutide's Weight-Loss Benefits Were Sustained in a 2-Year Study. JAMA. 2021 Dec 28:326(24):2464. doi: 10.1001/jama.2021.21717. Epub
[PubMed PMID: 34962530]
[48]
Jastreboff AM, Aronne LJ, Ahmad NN, Wharton S, Connery L, Alves B, Kiyosue A, Zhang S, Liu B, Bunck MC, Stefanski A, SURMOUNT-1 Investigators. Tirzepatide Once Weekly for the Treatment of Obesity. The New England journal of medicine. 2022 Jul 21:387(3):205-216. doi: 10.1056/NEJMoa2206038. Epub 2022 Jun 4
[PubMed PMID: 35658024]
[49]
Zheng H, Liu M, Li S, Shi Q, Zhang S, Zhou Y, Su N. Sodium-Glucose Co-Transporter-2 Inhibitors in Non-Diabetic Adults With Overweight or Obesity: A Systematic Review and Meta-Analysis. Frontiers in endocrinology. 2021:12():706914. doi: 10.3389/fendo.2021.706914. Epub 2021 Aug 16
[PubMed PMID: 34484120]
Level 1 (high-level) evidence
[50]
Neeland IJ, McGuire DK, Chilton R, Crowe S, Lund SS, Woerle HJ, Broedl UC, Johansen OE. Empagliflozin reduces body weight and indices of adipose distribution in patients with type 2 diabetes mellitus. Diabetes & vascular disease research. 2016 Mar:13(2):119-26. doi: 10.1177/1479164115616901. Epub
[PubMed PMID: 26873905]
[51]
Nesto RW, Bell D, Bonow RO, Fonseca V, Grundy SM, Horton ES, Le Winter M, Porte D, Semenkovich CF, Smith S, Young LH, Kahn R. Thiazolidinedione use, fluid retention, and congestive heart failure: a consensus statement from the American Heart Association and American Diabetes Association. Diabetes care. 2004 Jan:27(1):256-63
[PubMed PMID: 14693998]
Level 3 (low-level) evidence
[52]
Lebovitz HE. Thiazolidinediones: the Forgotten Diabetes Medications. Current diabetes reports. 2019 Nov 27:19(12):151. doi: 10.1007/s11892-019-1270-y. Epub 2019 Nov 27
[PubMed PMID: 31776781]
[53]
Schauer PR, Bhatt DL, Kirwan JP, Wolski K, Aminian A, Brethauer SA, Navaneethan SD, Singh RP, Pothier CE, Nissen SE, Kashyap SR, STAMPEDE Investigators. Bariatric Surgery versus Intensive Medical Therapy for Diabetes - 5-Year Outcomes. The New England journal of medicine. 2017 Feb 16:376(7):641-651. doi: 10.1056/NEJMoa1600869. Epub
[PubMed PMID: 28199805]
[54]
McGlone ER, Carey I, Veličković V, Chana P, Mahawar K, Batterham RL, Hopkins J, Walton P, Kinsman R, Byrne J, Somers S, Kerrigan D, Menon V, Borg C, Ahmed A, Sgromo B, Cheruvu C, Bano G, Leonard C, Thom H, le Roux CW, Reddy M, Welbourn R, Small P, Khan OA. Bariatric surgery for patients with type 2 diabetes mellitus requiring insulin: Clinical outcome and cost-effectiveness analyses. PLoS medicine. 2020 Dec:17(12):e1003228. doi: 10.1371/journal.pmed.1003228. Epub 2020 Dec 7
[PubMed PMID: 33285553]
Level 2 (mid-level) evidence
[55]
Purnell JQ, Dewey EN, Laferrère B, Selzer F, Flum DR, Mitchell JE, Pomp A, Pories WJ, Inge T, Courcoulas A, Wolfe BM. Diabetes Remission Status During Seven-year Follow-up of the Longitudinal Assessment of Bariatric Surgery Study. The Journal of clinical endocrinology and metabolism. 2021 Mar 8:106(3):774-788. doi: 10.1210/clinem/dgaa849. Epub
[PubMed PMID: 33270130]
[56]
Brown RJ, Araujo-Vilar D, Cheung PT, Dunger D, Garg A, Jack M, Mungai L, Oral EA, Patni N, Rother KI, von Schnurbein J, Sorkina E, Stanley T, Vigouroux C, Wabitsch M, Williams R, Yorifuji T. The Diagnosis and Management of Lipodystrophy Syndromes: A Multi-Society Practice Guideline. The Journal of clinical endocrinology and metabolism. 2016 Dec:101(12):4500-4511
[PubMed PMID: 27710244]
Level 1 (high-level) evidence
[57]
Aronne LJ. Classification of obesity and assessment of obesity-related health risks. Obesity research. 2002 Dec:10 Suppl 2():105S-115S
[PubMed PMID: 12490659]
[58]
Whelton PK, Carey RM, Aronow WS, Casey DE Jr, Collins KJ, Dennison Himmelfarb C, DePalma SM, Gidding S, Jamerson KA, Jones DW, MacLaughlin EJ, Muntner P, Ovbiagele B, Smith SC Jr, Spencer CC, Stafford RS, Taler SJ, Thomas RJ, Williams KA Sr, Williamson JD, Wright JT Jr. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults: Executive Summary: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Hypertension (Dallas, Tex. : 1979). 2018 Jun:71(6):1269-1324. doi: 10.1161/HYP.0000000000000066. Epub 2017 Nov 13
[PubMed PMID: 29133354]
Level 1 (high-level) evidence
[59]
Wang B, Li F, Guo J, Wang C, Xu D, Li C. Effects of liver function, insulin resistance and inflammatory factors on vascular endothelial dilation function and prognosis of coronary heart disease patients complicated with NAFLD. Experimental and therapeutic medicine. 2019 Feb:17(2):1306-1311. doi: 10.3892/etm.2018.7043. Epub 2018 Dec 4
[PubMed PMID: 30680007]
[60]
Piercy KL, Troiano RP, Ballard RM, Carlson SA, Fulton JE, Galuska DA, George SM, Olson RD. The Physical Activity Guidelines for Americans. JAMA. 2018 Nov 20:320(19):2020-2028. doi: 10.1001/jama.2018.14854. Epub
[PubMed PMID: 30418471]
[61]
Mosher AL, Piercy KL, Webber BJ, Goodwin SK, Casavale KO, Olson RD. Dietary Guidelines for Americans: Implications for Primary Care Providers. American journal of lifestyle medicine. 2016 Jan-Feb:10(1):23-35. doi: 10.1177/1559827614521755. Epub 2014 Feb 6
[PubMed PMID: 30202257]
[62]
Hamdy O, Ganda OP, Maryniuk M, Gabbay RA, Members of the Joslin Clinical Oversight Committee. CHAPTER 2. Clinical nutrition guideline for overweight and obese adults with type 2 diabetes (T2D) or prediabetes, or those at high risk for developing T2D. The American journal of managed care. 2018 Jun:24(7 Spec No.):SP226-SP231
[PubMed PMID: 29938995]
[63]
Carlson AL, Mullen DM, Bergenstal RM. Clinical Use of Continuous Glucose Monitoring in Adults with Type 2 Diabetes. Diabetes technology & therapeutics. 2017 May:19(S2):S4-S11. doi: 10.1089/dia.2017.0024. Epub
[PubMed PMID: 28541137]
[64]
Jackson MA, Ahmann A, Shah VN. Type 2 Diabetes and the Use of Real-Time Continuous Glucose Monitoring. Diabetes technology & therapeutics. 2021 Mar:23(S1):S27-S34. doi: 10.1089/dia.2021.0007. Epub
[PubMed PMID: 33534631]
[65]
Dearborn JL, Viscoli CM, Inzucchi SE, Young LH, Kernan WN. Metabolic syndrome identifies normal weight insulin-resistant stroke patients at risk for recurrent vascular disease. International journal of stroke : official journal of the International Stroke Society. 2019 Aug:14(6):639-645. doi: 10.1177/1747493018816425. Epub 2018 Dec 3
[PubMed PMID: 30507360]