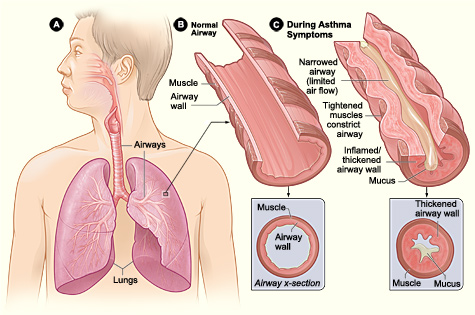
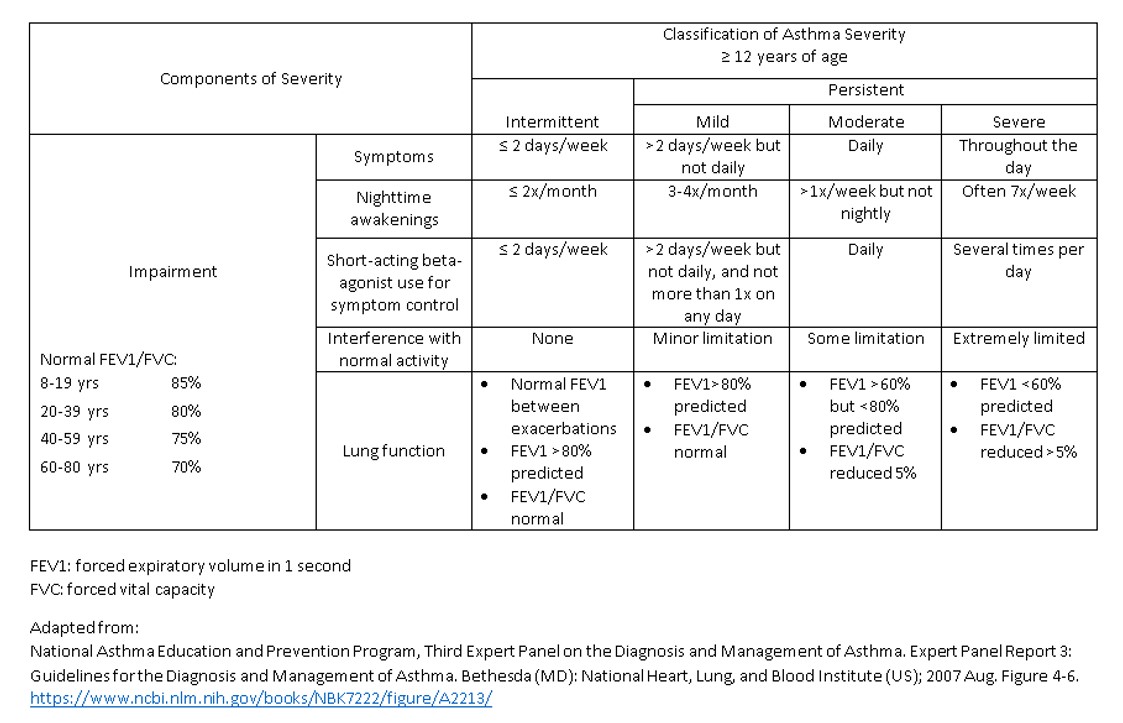
[1]
Mannino DM, Homa DM, Akinbami LJ, Moorman JE, Gwynn C, Redd SC. Surveillance for asthma--United States, 1980-1999. Morbidity and mortality weekly report. Surveillance summaries (Washington, D.C. : 2002). 2002 Mar 29:51(1):1-13
[PubMed PMID: 12420904]
[2]
Centers for Disease Control and Prevention (CDC). Vital signs: asthma prevalence, disease characteristics, and self-management education: United States, 2001--2009. MMWR. Morbidity and mortality weekly report. 2011 May 6:60(17):547-52
[PubMed PMID: 21544044]
[3]
Maio S, Baldacci S, Carrozzi L, Pistelli F, Angino A, Simoni M, Sarno G, Cerrai S, Martini F, Fresta M, Silvi P, Di Pede F, Guerriero M, Viegi G. Respiratory symptoms/diseases prevalence is still increasing: a 25-yr population study. Respiratory medicine. 2016 Jan:110():58-65. doi: 10.1016/j.rmed.2015.11.006. Epub 2015 Nov 14
[PubMed PMID: 26614595]
[5]
Burrows B, Martinez FD, Halonen M, Barbee RA, Cline MG. Association of asthma with serum IgE levels and skin-test reactivity to allergens. The New England journal of medicine. 1989 Feb 2:320(5):271-7
[PubMed PMID: 2911321]
[6]
Gilliland FD, Islam T, Berhane K, Gauderman WJ, McConnell R, Avol E, Peters JM. Regular smoking and asthma incidence in adolescents. American journal of respiratory and critical care medicine. 2006 Nov 15:174(10):1094-100
[PubMed PMID: 16973983]
[7]
Camargo CA Jr, Weiss ST, Zhang S, Willett WC, Speizer FE. Prospective study of body mass index, weight change, and risk of adult-onset asthma in women. Archives of internal medicine. 1999 Nov 22:159(21):2582-8
[PubMed PMID: 10573048]
[9]
Patwa A, Shah A. Anatomy and physiology of respiratory system relevant to anaesthesia. Indian journal of anaesthesia. 2015 Sep:59(9):533-41. doi: 10.4103/0019-5049.165849. Epub
[PubMed PMID: 26556911]
[11]
Grinnan DC, Truwit JD. Clinical review: respiratory mechanics in spontaneous and assisted ventilation. Critical care (London, England). 2005 Oct 5:9(5):472-84
[PubMed PMID: 16277736]
[12]
Liu MC, Hubbard WC, Proud D, Stealey BA, Galli SJ, Kagey-Sobotka A, Bleecker ER, Lichtenstein LM. Immediate and late inflammatory responses to ragweed antigen challenge of the peripheral airways in allergic asthmatics. Cellular, mediator, and permeability changes. The American review of respiratory disease. 1991 Jul:144(1):51-8
[PubMed PMID: 2064141]
[13]
Zhu Z, Homer RJ, Wang Z, Chen Q, Geba GP, Wang J, Zhang Y, Elias JA. Pulmonary expression of interleukin-13 causes inflammation, mucus hypersecretion, subepithelial fibrosis, physiologic abnormalities, and eotaxin production. The Journal of clinical investigation. 1999 Mar:103(6):779-88
[PubMed PMID: 10079098]
[14]
Stewart AG, Tomlinson PR, Fernandes DJ, Wilson JW, Harris T. Tumor necrosis factor alpha modulates mitogenic responses of human cultured airway smooth muscle. American journal of respiratory cell and molecular biology. 1995 Jan:12(1):110-9
[PubMed PMID: 7529028]
[15]
Doeing DC, Solway J. Airway smooth muscle in the pathophysiology and treatment of asthma. Journal of applied physiology (Bethesda, Md. : 1985). 2013 Apr:114(7):834-43. doi: 10.1152/japplphysiol.00950.2012. Epub 2013 Jan 10
[PubMed PMID: 23305987]
[16]
Brannan JD, Lougheed MD. Airway hyperresponsiveness in asthma: mechanisms, clinical significance, and treatment. Frontiers in physiology. 2012:3():460. doi: 10.3389/fphys.2012.00460. Epub 2012 Dec 10
[PubMed PMID: 23233839]
[17]
Chapman DG, Irvin CG. Mechanisms of airway hyper-responsiveness in asthma: the past, present and yet to come. Clinical and experimental allergy : journal of the British Society for Allergy and Clinical Immunology. 2015 Apr:45(4):706-19. doi: 10.1111/cea.12506. Epub
[PubMed PMID: 25651937]
[18]
Kudo M, Ishigatsubo Y, Aoki I. Pathology of asthma. Frontiers in microbiology. 2013 Sep 10:4():263. doi: 10.3389/fmicb.2013.00263. Epub 2013 Sep 10
[PubMed PMID: 24032029]
[19]
Limb SL, Brown KC, Wood RA, Wise RA, Eggleston PA, Tonascia J, Adkinson NF Jr. Irreversible lung function deficits in young adults with a history of childhood asthma. The Journal of allergy and clinical immunology. 2005 Dec:116(6):1213-9
[PubMed PMID: 16337448]
[20]
National Asthma Education and Prevention Program. Expert Panel Report 3 (EPR-3): Guidelines for the Diagnosis and Management of Asthma-Summary Report 2007. The Journal of allergy and clinical immunology. 2007 Nov:120(5 Suppl):S94-138
[PubMed PMID: 17983880]
[21]
Buels KS, Fryer AD. Muscarinic receptor antagonists: effects on pulmonary function. Handbook of experimental pharmacology. 2012:(208):317-41. doi: 10.1007/978-3-642-23274-9_14. Epub
[PubMed PMID: 22222705]
[22]
Moore KW, de Waal Malefyt R, Coffman RL, O'Garra A. Interleukin-10 and the interleukin-10 receptor. Annual review of immunology. 2001:19():683-765
[PubMed PMID: 11244051]
[23]
D'Amato M, Vitale C, Molino A, Lanza M, D'Amato G. Anticholinergic drugs in asthma therapy. Current opinion in pulmonary medicine. 2017 Jan:23(1):103-108
[PubMed PMID: 27820743]
Level 3 (low-level) evidence