Continuing Education Activity
Drug-induced gingival overgrowth (DIGO), also referred to as drug-induced gingival enlargement and previously known as drug-induced gingival hyperplasia, is a noted side effect of certain drugs given for non-dental uses where the gingival tissue is not the intended target organ. The key offending drug classes are anticonvulsants, immunosuppressants, and calcium channel blockers. This activity presents the causes, pathophysiology, and diagnosis of gingival overgrowth and highlights the role of the interprofessional team in its management.
Objectives:
Identify the types of drugs associated with gingival overgrowth.
Apply the pathophysiology of drug-induced gingival overgrowth.
Compare the treatment and management options available for gingival overgrowth.
Communicate some interprofessional team strategies for improving care coordination and outcomes in patients with gingival overgrowth.
Introduction
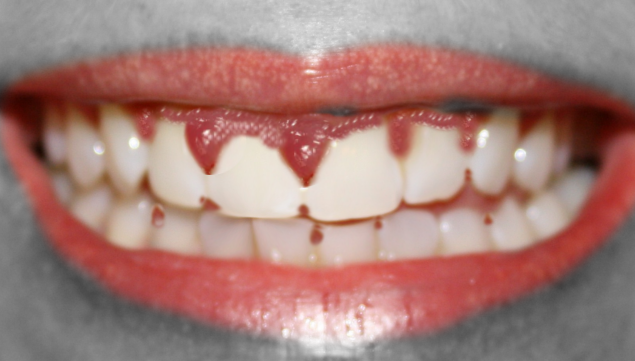
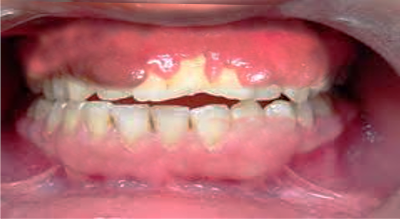
Drug-induced gingival overgrowth (DIGO), also referred to as drug-induced gingival enlargement and previously known as drug-induced gingival hyperplasia, is a side effect of certain drugs in which the gingival tissue is not the intended target organ. The key offending drug classes are anticonvulsants, immunosuppressants, and calcium channel blockers.[1] Gingival overgrowth impedes proper dental hygiene and, apart from the cosmetic damage, causes painful chewing and eating (See Images. Gingival Overgrowth From a Medication and Gingival Overgrowth).
A non-surgical approach is the first line of treatment, including appropriate plaque control and discontinuing or changing the inducing drug. In recent years, azithromycin has gained much attention due to its ability to improve gingival overgrowths in patients taking cyclosporin A, avoiding the need to change the drug to another agent. Surgical management comes into play only when medical treatment is unsuccessful. However, recurrences are common, and the results usually last 12 months.[2]
Etiology
Drugs are the most common reason behind gingival enlargements. DIGO is a side effect in patients taking anticonvulsants, immunosuppressants, or calcium channel blockers. It is associated with the patient's genetic predisposition and plaque or gingival inflammation.[3]
Anticonvulsants
Phenytoin (PHT, or 5,5-diphenylhydantoin), sodium valproate, phenobarbitone, vigabatrin, primidone, mephenytoin, and ethosuximide are some of the drugs that cause gingival hypertrophy. However, phenytoin is the anticonvulsant most associated with gingival enlargements.[4] It is used to treat temporal lobe, tonic-clonic, and psychomotor seizures.[4] Drugs like PHT, phenobarbitone, and primidone are metabolized to 5-(4-hydroxyphenyl) 5-phenyl hydantoin (4-HPPH), which is responsible for gingival tissue overgrowth. Multiple anticonvulsants are sometimes given together, acting synergistically and aggravating the condition.
Immunosuppressants
Immunosuppressants are prescribed after organ transplantation to prevent organ transplant rejection, such as renal transplants and treat some autoimmune diseases, like rheumatoid arthritis.[4] Cyclosporin, sirolimus, and tacrolimus are immunosuppressants linked to gingival enlargement. Cyclosporin is the most frequently prescribed immunosuppressive drug after an organ transplant, and the incidence of gingival overgrowth has been found in nearly 53% of patients taking cyclosporin after renal transplants.[5] Tacrolimus, on the other hand, is less toxic than cyclosporin, causing less severe gingival overgrowth and hepatic and renal toxicity.[6][7][6] Sirolimus is another immunosuppressant that has shown a predisposition to gingival enlargement.[8][9][10][8]
Calcium Channel Blocker
Calcium channel blockers are indicated for treating hypertension, angina pectoris, and peripheral vascular disease.[4] These include nifedipine, nitrendipine, felodipine, amlodipine, nisoldipine, verapamil, and diltiazem. Seymour et al reported the first case of gingival overgrowth attributed to amlodipine in 1994. Lafzi et al reported gingival hypertrophy in patients receiving 10 mg of amlodipine daily within 2 months of the onset of treatment.[11]
Renal transplant patients taking immunosuppressants like cyclosporin show a greater propensity to develop gingival hypertrophy when put on nifedipine or diltiazem. However, the extent of hypertrophy is more significant with cyclosporin.[4] Combinations of these drugs could act synergistically, causing exaggerated hypertrophy of gingival tissue.
Epidemiology
Drug-induced gingival overgrowth is most commonly seen in male children and adolescents, with a most prevalent location in the anterior gingival tissue. The individual's genotype affects the development of DIGO [12], and the extent and degree of overgrowth depend on the drug. Phenytoin, cyclosporin, and nifedipine are the most common causes of gingival overgrowth, and phenytoin has the highest prevalence. It is estimated that 50% of adults treated with phenytoin experience gingival enlargement, 30% with cyclosporin, and 20% with nifedipine.[12]
Pathophysiology
In 1996, Seymour et al postulated the theory of genetic predisposition for the etiopathology of DIGO.[3][13] This is substantiated because some individuals develop gingival overgrowth, and some do not while on the same drug. The usual inflammatory response of gingival fibroblasts and subsequent proliferation of connective tissue matrix emphasize the heterogeneous character of the individual’s gingival fibroblasts in response to the inducing drugs.
The common mechanism of action at the cellular level of all these 3 dissimilar drugs appears to inhibit cation influx, particularly sodium and calcium ions. Gingival overgrowth is multifactorial.[13][14][4] Bacterial plaque appears to be a contributory factor, and the severity of gingival overgrowth is directly proportional to the degree of plaque buildup and inflammation. Decreased cation-dependant folic acid (FA) active transport within gingival fibroblasts causes reduced FA uptake by the cells, causing changes in the metabolism of matrix metalloproteinases and the inability to activate collagenase. This results in an accumulation of connective tissue and collagen due to a lack of collagenase.
Gingival Fibroblasts and Cellular Folate Uptake
Inducing drugs trigger the activation and proliferation of gingival fibroblasts, causing an increased production of GAGs (glycosaminoglycans) in the connective tissue. These drugs decrease the cellular uptake of folate by genetically predisposing fibroblasts. Reduced intracellular folate decreases the synthesis or activation of MMPs (matrix metalloproteinases) required to convert inactive collagenase to active collagenase, allowing an excess of connective tissue build-up. Brown et al (1991) postulated that bacterial plaque contributes to gingival inflammation, which completes the vicious cycle.
Matrix Metalloproteinases
More than 20 enzymes degrade connective tissue and cause tissue remodeling. These include collagenases, gelatinases, and stromelysins. Inhibition of their activation can result in the accumulation of extracellular matrix and collagen and cause DIGO.
Inflammatory Cytokines
Inflamed gingival tissue exhibits higher levels of interleukin-1 beta (IL-1beta), a proinflammatory cytokine. Likewise, IL-6 causes fibroblastic proliferation and increased production of collagen and GAG synthesis.[4]
Na+/ Ca2+ Ion Flux Drug Mechanisms
Fugi and Kobayashi (1990) reported that PHT and several calcium channel blockers (CCBAs) inhibit Ca2+ uptake within gingival fibroblasts. Thomas and Petrou (2013) reported reduced Na+ channel availability and decreased action potential amplitude. This causes reduced Ca2+ entry and a decrease in Ca2+-activated K+ channels. All 3 types of DIGO-inducing drugs act on Ca2+ flux similarly.
Plaque Buildup
The concentrated drug in crevicular gingival fluid or bacterial plaque exerts a direct toxic effect on the gingival tissue. Dental plaque induces inflammation, which causes gingival overgrowth. Inflammation causes the upregulation of transforming growth factor-beta 1 (TGF-beta 1). Hence, dental plaque control is needed in treating and preventing DIGO over time.
Histopathology
In DIGO, the pathological changes lie in the connective tissue and not the epithelial cells of the gingiva. There is an excessive accumulation of extracellular matrix-like collagen with varying amounts of inflammatory infiltrates, predominantly plasma cells. Fibroblastic proliferation may not be evident. Erratic columns of collagen fibers are seen interspersed with penetrating epithelial ridges.[4]
History and Physical
Patients with DIGO report a past medical history of hypertension, angina, epilepsy, or a recent organ transplant. They typically present with gingival enlargement, pain during mastication, and cosmetic disfigurement 1 to 3 months after starting treatment with 1 of the drugs associated with the disease.[4] The gingival enlargement caused by the drug makes oral hygiene challenging, leading to increased plaque accumulation and secondary inflammation.[15]
Not all drug-induced gingival overgrowth lesions are fibrotic, it was previously believed. According to histological and histomorphometric analyses, phenytoin-induced lesions are the most fibrotic. Cyclosporin-induced lesions are more inflamed and show a low fibrotic component, and nifedipine lesions are mixed.[16]
Clinical Findings
- Firm, painless, nodular enlargement of the interdental papilla, limited to the keratinized portions of the gingiva and extending to the facial and lingual gingival margins.
- A huge fold of hypertrophied gingival tissue is observed covering the crowns in severe cases.
- If no secondary inflammation is present, it appears firm and pale pink with minute lobulations, pouting from underneath the gingival margin. It is delineated by a groove of tissue that does not bleed on touch.
- If secondary inflammation exists, the gingiva appears smooth and red or bluish-red.
- The enlargement is generalized but usually greater in the anterior regions.
- Typically, it is not seen in edentulous areas.[15]
- The gingival overgrowth disappears when teeth are extracted.[15]
Evaluation
The diagnosis of drug-induced gingival overgrowth is made by clinical examination and the patient's past medical history.
- A periodontal examination is necessary to evaluate for the presence of periodontal disease.
- Before beginning any treatment, full mouth periapical radiographs and orthopantomography are required to rule out periodontal disease.
- Complete blood count (CBC) is indicated in patients with gingival enlargement if there is a presence of profuse gingival bleeding, even if it is drug-induced, to rule out anemia and leukemia.
- A tissue biopsy should be carried out in case the presentation of the disease is unusual.
- Histopathological examination of persisting overgrowths is mandatory to evaluate malignant changes [17]
- Candidiasis and other infections may be ruled out by taking a culture.
Treatment / Management
Treatment in DIGO aims to alleviate the patient's discomfort, enabling them to do simple acts like eating and chewing pain-free, treat the inflammation, reduce the swelling, and improve the gingiva's cosmetic appearance. The modalities of treatment are medical and surgical. Medical management is the first line of therapy, whereas surgery is reserved for recurrences or cases that persist despite good medical treatment.
Discontinuing or changing the medication must be placed under consideration.[18][12] An alternative to phenytoin includes carbamazepine and valproic acid, which have shown a lower rate of gingival enlargement. Diltiazem and verapamil exhibit a lower prevalence of gingival enlargement compared to nifedipine. Cyclosporin substitution is more complicated because there are limited options available. Cyclosporin can be substituted by tacrolimus, and azithromycin in combination with cyclosporin has shown a decrease in the severity of DIGO.[19]
Plaque control should be the first step in treating DIGO, correct oral hygiene, and professional plaque removal, including tooth surface cleaning and periodic scaling.[20][21] Control of inflammation, including non-steroidal anti-inflammatory agents, antibiotics to control infection, and topical antifungal medication like nystatin, may also be utilized. Folate supplementation has also been used.[22]
A sufficient period of 6 to 12 months should be allowed to elapse after discontinuing the inducing drug before surgery is considered.[19]The surgical methods include gingivectomy and periodontal flap surgery. Electrocautery may be used in difficult cases, children, or where the gingiva is fragile and likely to bleed. The CO2 laser has a wavelength of 10600 nm; hence, it is readily absorbed by water and is effective in the surgery of soft tissues with high water content, like the gingiva. Blood vessels up to a diameter of 0.5 mm can be sealed effectively and provide a dry field for better surgical field visibility. A laser is preferred over the scalpel as it has strong bactericidal and hemostatic effects.[20][2]
The Role of Azithromycin in Cyclosporin-Induced Gingival Overgrowth
In the last decade, researchers have found that azithromycin can help reverse gingival overgrowth caused by cyclosporine A in renal transplant patients.[23] Interestingly, azithromycin does not alter the cyclosporin A blood levels or renal function, avoiding changing this drug to another anticonvulsant due to DIGO.[23] The effect of azithromycin on cyclosporine A-induced collagen turnover and cell proliferation was studied by Kim et al. They concluded that azithromycin inhibits the proliferation of human gingival fibroblasts and collagen accumulation - both induced by cyclosporin A – activates MMP-2 in renal transplant and normal fibroblast, inhibits mRNA levels of type 1 collagen and raises levels of MMP-2.[23]
Differential Diagnosis
The differential diagnoses for drug-induced gingival overgrowth include the following:
- False enlargement of gingival tissue: pseudo-enlargement of the gingiva caused by an enlarged underlying bony tissue. The gingiva here is normal.
- Inflammation: chronically inflamed gingival tissue is red or violaceous, smooth, and tends to bleed to the touch.
- Familial or hereditary conditions: examples include familial fibromatosis, idiopathic fibromatosis, gingivomatosis, and hereditary gingival hyperplasia. The gingiva is pink and non-tender, involves the attached gingiva, the gingival margin, and the interdental papillae, and has a firm and leathery consistency.
- Physiological states: puberty and pregnancy are associated with gingival enlargement.
- Scurvy: vitamin C deficiency can produce very tender bleeding gingiva.
- Systemic diseases: leukemia, tuberculosis, sarcoidosis - their diagnosis can be corroborated hematologically.
A thorough history, physical examination, and investigations, including biopsy, may be required to confirm the etiology of all these conditions.
Conditions Similar to Gingival Enlargement
- Fibrous epulis/peripheral fibroma
- Angiogranuloma/Pyogenic granuloma
- Gingival cysts
- Neoplasms: They can be benign or malignant. Benign lesions include fibroma, peripheral and central giant cell granuloma, papilloma, leukoplakia, nevus, hemangioma, leukoplakia, nevus, myoblastoma, hemangioma, neurilemoma, neurofibroma, ameloblastoma. Malignant tumors include squamous cell carcinoma and Kaposi sarcoma, among others.[24]
Prognosis
Suppose the medication inducing the gingival overgrowth can be changed to a drug that is less associated with this complication. In that case, the gingival tissue may return to normal with adequate plaque control. The gingival enlargement can persist despite drug substitution and good plaque control, and in this case, surgical management to restore normal gingival contours is required. These modalities of treatment, although effective, do not always prevent the recurrence of the enlargement. DIGO recurrence in surgically treated cases may appear as soon as 3 to 6 months after the procedure. In general, the results last for at least 12 months.[2]
Complications
The complications that can manifest with drug-induced gingival overgrowth are as follows:
- Functional difficulties
- Esthetic concerns
- Increased incidence of caries
- Delayed eruption of teeth
- Pathological drifting of teeth
- Prolonged retention of the primary dentition
- Diastemas and spacing problems
- Poor plaque control leads to periodontal complications [2]
Deterrence and Patient Education
Some studies indicate that bacterial plaque is a prerequisite for the disease to occur. On the contrary, others believe that the presence of dental plaque is a consequence of the difficulty that the gingival enlargement represents in dental hygiene, which then exacerbates the severity of the overgrowth.[2] In any case, 1 thing is certain: bacterial plaque plays an essential role in developing DIGO, and patients must be educated about adequate dental brushing and flossing techniques that chlorhexidine gluconate rinses may accompany to control the levels of bacterial plaque. Regular appointments with their dentist must be accomplished to perform professional teeth cleaning. These measures can prevent or decrease the rate and the degree to which recurrence occurs.
Enhancing Healthcare Team Outcomes
Since the condition is entirely drug-induced, patient education about the side effects of the implicated drugs before starting treatment is warranted, and the importance of good dental hygiene should be emphasized [2]. Drug-induced gingival enlargement has a good prognosis and is generally reversible on stopping or substituting the offending drug. An interdisciplinary approach is needed as the primary clinician, dentist, and healthcare team should adequately address the patient's signs and symptoms.