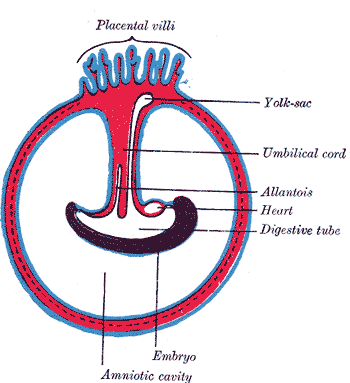
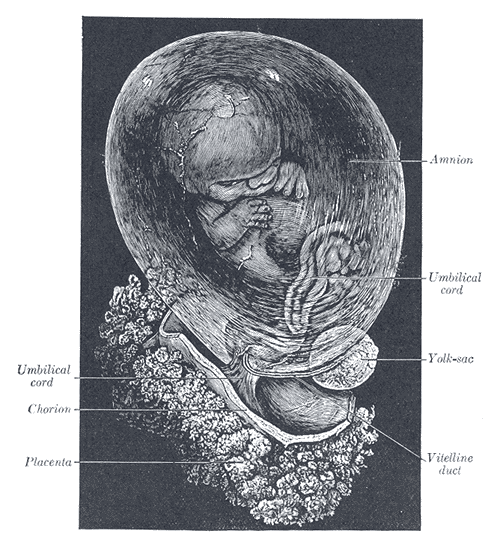
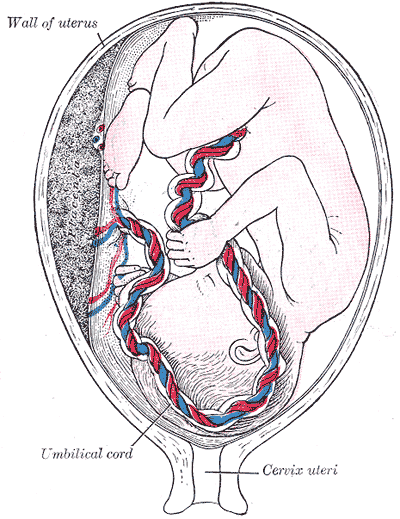
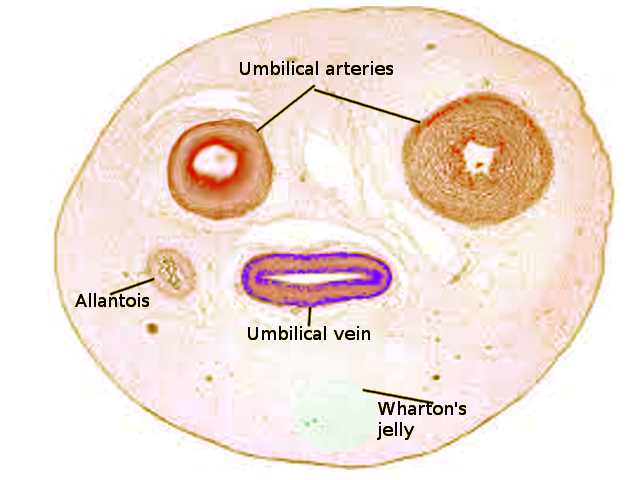
[1]
Persutte WH, Hobbins J. Single umbilical artery: a clinical enigma in modern prenatal diagnosis. Ultrasound in obstetrics & gynecology : the official journal of the International Society of Ultrasound in Obstetrics and Gynecology. 1995 Sep:6(3):216-29
[PubMed PMID: 8521073]
[2]
Hegazy AA. Anatomy and embryology of umbilicus in newborns: a review and clinical correlations. Frontiers of medicine. 2016 Sep:10(3):271-7. doi: 10.1007/s11684-016-0457-8. Epub 2016 Sep 7
[PubMed PMID: 27473223]
[4]
Moshiri M, Zaidi SF, Robinson TJ, Bhargava P, Siebert JR, Dubinsky TJ, Katz DS. Comprehensive imaging review of abnormalities of the umbilical cord. Radiographics : a review publication of the Radiological Society of North America, Inc. 2014 Jan-Feb:34(1):179-96. doi: 10.1148/rg.341125127. Epub
[PubMed PMID: 24428290]
[7]
Cheng T, Yang B, Li D, Ma S, Tian Y, Qu R, Zhang W, Zhang Y, Hu K, Guan F, Wang J. Wharton's Jelly Transplantation Improves Neurologic Function in a Rat Model of Traumatic Brain Injury. Cellular and molecular neurobiology. 2015 Jul:35(5):641-9. doi: 10.1007/s10571-015-0159-9. Epub 2015 Feb 1
[PubMed PMID: 25638565]
[9]
Mitchell KE, Weiss ML, Mitchell BM, Martin P, Davis D, Morales L, Helwig B, Beerenstrauch M, Abou-Easa K, Hildreth T, Troyer D, Medicetty S. Matrix cells from Wharton's jelly form neurons and glia. Stem cells (Dayton, Ohio). 2003:21(1):50-60
[PubMed PMID: 12529551]
[10]
Zhou C, Yang B, Tian Y, Jiao H, Zheng W, Wang J, Guan F. Immunomodulatory effect of human umbilical cord Wharton's jelly-derived mesenchymal stem cells on lymphocytes. Cellular immunology. 2011:272(1):33-8. doi: 10.1016/j.cellimm.2011.09.010. Epub 2011 Sep 29
[PubMed PMID: 22004796]
[11]
Safari F, Fani N, Eglin D, Alini M, Stoddart MJ, Baghaban Eslaminejad M. Human umbilical cord-derived scaffolds for cartilage tissue engineering. Journal of biomedical materials research. Part A. 2019 Aug:107(8):1793-1802. doi: 10.1002/jbm.a.36698. Epub 2019 Apr 24
[PubMed PMID: 30983084]
[12]
Corsello T, Amico G, Corrao S, Anzalone R, Timoneri F, Lo Iacono M, Russo E, Spatola GF, Uzzo ML, Giuffrè M, Caprnda M, Kubatka P, Kruzliak P, Conaldi PG, La Rocca G. Wharton's Jelly Mesenchymal Stromal Cells from Human Umbilical Cord: a Close-up on Immunomodulatory Molecules Featured In Situ and In Vitro. Stem cell reviews and reports. 2019 Dec:15(6):900-918. doi: 10.1007/s12015-019-09907-1. Epub
[PubMed PMID: 31741193]
[13]
Nishida F, Zappa Villar MF, Zanuzzi CN, Sisti MS, Camiña AE, Reggiani PC, Portiansky EL. Intracerebroventricular Delivery of Human Umbilical Cord Mesenchymal Stem Cells as a Promising Therapy for Repairing the Spinal Cord Injury Induced by Kainic Acid. Stem cell reviews and reports. 2020 Feb:16(1):167-180. doi: 10.1007/s12015-019-09934-y. Epub
[PubMed PMID: 31760626]
[16]
Saari H, Konttinen YT, Friman C, Sorsa T. Differential effects of reactive oxygen species on native synovial fluid and purified human umbilical cord hyaluronate. Inflammation. 1993 Aug:17(4):403-15
[PubMed PMID: 8406685]
[17]
Peesay M. Nuchal cord and its implications. Maternal health, neonatology and perinatology. 2017:3():28. doi: 10.1186/s40748-017-0068-7. Epub 2017 Dec 6
[PubMed PMID: 29234502]
[18]
Gupta A, El-Amin SF 3rd, Levy HJ, Sze-Tu R, Ibim SE, Maffulli N. Umbilical cord-derived Wharton's jelly for regenerative medicine applications. Journal of orthopaedic surgery and research. 2020 Feb 13:15(1):49. doi: 10.1186/s13018-020-1553-7. Epub 2020 Feb 13
[PubMed PMID: 32054483]
[19]
Meyer WW, Rumpelt HJ, Yao AC, Lind J. Structure and closure mechanism of the human umbilical artery. European journal of pediatrics. 1978 Jul 19:128(4):247-59
[PubMed PMID: 668732]
[22]
Damasceno EB, de Lima PP. Wharton's jelly absence: a possible cause of stillbirth. Autopsy & case reports. 2013 Oct-Dec:3(4):43-47. doi: 10.4322/acr.2013.038. Epub 2013 Dec 31
[PubMed PMID: 28584806]
[23]
Thorp JA, Rushing RS. Umbilical cord blood gas analysis. Obstetrics and gynecology clinics of North America. 1999 Dec:26(4):695-709
[PubMed PMID: 10587963]
[24]
Fleischer A, Schulman H, Farmakides G, Bracero L, Blattner P, Randolph G. Umbilical artery velocity waveforms and intrauterine growth retardation. American journal of obstetrics and gynecology. 1985 Feb 15:151(4):502-5
[PubMed PMID: 3976751]
[25]
Divon MY. Umbilical artery Doppler velocimetry: clinical utility in high-risk pregnancies. American journal of obstetrics and gynecology. 1996 Jan:174(1 Pt 1):10-4
[PubMed PMID: 8571990]
[26]
Murphy-Kaulbeck L, Dodds L, Joseph KS, Van den Hof M. Single umbilical artery risk factors and pregnancy outcomes. Obstetrics and gynecology. 2010 Oct:116(4):843-850. doi: 10.1097/AOG.0b013e3181f0bc08. Epub
[PubMed PMID: 20859147]
[27]
Ramesh S, Hariprasath S, Anandan G, Solomon PJ, Vijayakumar V. Single umbilical artery. Journal of pharmacy & bioallied sciences. 2015 Apr:7(Suppl 1):S83-4. doi: 10.4103/0975-7406.155815. Epub
[PubMed PMID: 26015760]
[28]
Hubinont C, Lewi L, Bernard P, Marbaix E, Debiève F, Jauniaux E. Anomalies of the placenta and umbilical cord in twin gestations. American journal of obstetrics and gynecology. 2015 Oct:213(4 Suppl):S91-S102. doi: 10.1016/j.ajog.2015.06.054. Epub
[PubMed PMID: 26428508]
[29]
Rocha J, Carvalho J, Costa F, Meireles I, do Carmo O. Velamentous cord insertion in a singleton pregnancy: an obscure cause of emergency cesarean-a case report. Case reports in obstetrics and gynecology. 2012:2012():308206. doi: 10.1155/2012/308206. Epub 2012 Nov 29
[PubMed PMID: 23243528]
Level 3 (low-level) evidence
[31]
Kim CJ, Romero R, Chaemsaithong P, Chaiyasit N, Yoon BH, Kim YM. Acute chorioamnionitis and funisitis: definition, pathologic features, and clinical significance. American journal of obstetrics and gynecology. 2015 Oct:213(4 Suppl):S29-52. doi: 10.1016/j.ajog.2015.08.040. Epub
[PubMed PMID: 26428501]
[32]
Sayed Ahmed WA, Hamdy MA. Optimal management of umbilical cord prolapse. International journal of women's health. 2018:10():459-465. doi: 10.2147/IJWH.S130879. Epub 2018 Aug 21
[PubMed PMID: 30174462]
[33]
Ballen KK, Gluckman E, Broxmeyer HE. Umbilical cord blood transplantation: the first 25 years and beyond. Blood. 2013 Jul 25:122(4):491-8. doi: 10.1182/blood-2013-02-453175. Epub 2013 May 14
[PubMed PMID: 23673863]