Definition/Introduction
The heart is a specialized vascular structure with an intrinsic pumping mechanism to direct oxygenated blood and nutrients to the tissues of the body. Basic physics informs us that pressure gradients provide the physical impetus for fluid flow and the force generated by myocardial contraction drives blood flow into systemic and pulmonary circulation. Myocardial relaxation generates a drop in pressure that promotes retrograde blood flow. Prevention of this retrograde blood flow is by the presence of heart valves which open and close due to changes in pressure. The heart has four valves: the mitral valve, aortic valve, tricuspid valve, and pulmonic valve. The mitral and tricuspid valves are collectively known as the atrioventricular valves due to their anatomical location at the junction of the atrium and ventricle. Their primary function is to prevent retrograde blood flow into the atria during ventricular systole. The aortic and pulmonic valves are collectively known as the semilunar valves, and their primary function is to prevent retrograde blood flow into the ventricles during diastole. They are at the junction of the ventricle and their respective great artery.
The atrioventricular valves are composed of flat and non-homogenous tissue leaflets (or cusps) with the mitral valve consisting of two leaflets while the tricuspid valve is composed three leaflets. The leaflets of the atrioventricular valves extend into the ventricle via fibrous chordae tendinae (literally “heart-strings”). The chordae tendinae attach the apical aspects of the atrioventricular valves to muscular projections along the ventricular wall. These muscular projections are called papillary muscles, and they serve as a point of anchoring stability for the atrioventricular valves.
The presence of heart valves is to facilitate unidirectional blood flow. The driver behind the functionality of the heart valves is pressure gradients rather than electrical conduction. Heart valve disease can broadly classify as either valvular stenosis and valvular regurgitation (also known as incompetence or insufficiency). Valvular stenosis is the narrowing of the heart valve orifice. As such, valvular stenosis can accurately be described as an incomplete opening of the valvular orifice. In contrast, valvular regurgitation is an incomplete closure of the valvular orifice. Patients often describe this condition as having a “leaky valve.”
The cardiac cycle is the best context by which to understand heart valve disease. During ventricular diastole, the heart is in a relaxed state, and the mitral valve is open, allowing passive blood flow from the left atrium into the left ventricle. The increased blood flow into the left ventricle increases the interventricular pressure. With atrial systole, additional blood is actively directed into the ventricle, further increasing the ventricular pressure. The start of ventricular contraction leads to an abrupt increase in interventricular pressure. As the ventricular volume of blood increases, the volume pushes against the inferior aspects of the mitral valve, effectively closing them as the valve cusps coapt. The closure of the mitral valve leads to an abrupt and physiologic increase in interventricular pressure that, when coupled with the ventricular systole, exceeds the pressure in the aorta. As the interventricular pressure exceeds the aortic pressure, the aortic valve opens, leading to a blood jet into the ascending aorta. As ventricular systole ends, the driving pressure that directs blood flow into the ascending aorta decreases. When the interventricular pressure drops below the aortic pressure, the aortic valve closes. Considering only the left side of the heart, the aortic valve should be fully open during systole and completely closed during diastole. The mitral valve, on the other hand, should be fully closed during systole and open during diastole. Any deviation from this mechanism is likely representative of a heart valve pathology.
As the heart continuously beat throughout a lifetime, there is cyclic stress and strain on the native heart valves. The left side of the heart, in particular, is associated with higher pressures which range between approximately 100 mmHg to 150 mmHg. As a result, the left-sided valves (i.e., aortic and mitral) have a higher predisposition to disease compared to their right-sided counterparts. A diseased valve that can cause hemodynamic compromise or affect the quality of life will often warrant evaluation regarding possible prosthesis.
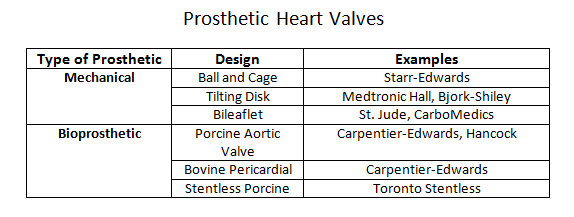
Prosthetic heart valves can broadly classify into a few general categories: mechanical heart valves, bioprosthetic heart valves, and homograft valves.Mechanical valves are composed of three major components: an occluder, an occluder restraint, and a sewing ring. Throughout the evolution of mechanical valve design, the artificial mechanical valves fall into three basic classes: ball and cage, disc valves (with tilting and non-tilting variants), and bileaflet valves. The prototypical ball and cage valve is the Starr-Edwards valve which, despite its longevity of use from the early 1960s, was retired in 2007. The occluder in this valve is a ball retained in a silicone-coated stainless steel cage. The sewing cuff is composed a Teflon ring with the intent minimizing thrombosis. During diastole (or systole for the mitral valve), the occluder ball would situate itself into the sewing ring, forming a seal to prevent retrograde flow. The major drawback of this design is the subpar hemodynamics. The ball occluder of the ball and cage design disrupted the central flow of blood found in a native heart valve. Further designs were developed to help improve the hemodynamics. The non-tilting disc valve, composed of a flat, circular disc within a cage, did not necessarily offer improved fluid dynamics but later bileaflet design performed better in emulating the natural hemodynamics of the native heart valve.Bioprosthetic valves are composed of three xenograft tissue leaflets of either porcine valvular leaflets or bovine pericardial tissue. Both xenografts are treated with glutaraldehyde solution to reduce thromboembolic risk due as glutaraldehyde inhibits collagen denaturation. In a stented bioprosthetic valve, the leaflets get sewn to a cloth-covered (typically dacron) metallic support structure, similar to their mechanical counterparts. The non-stented bioprosthetics require the prosthetic leaflets to attach directly into the aortic annulus. The bioprosthetic valves are hemodynamically improved compared to the mechanical valves but generate greater pressure gradients due to their smaller annulus.Homograft valves are valves obtained from human cadavers. As such valves no longer have regenerative capabilities, they are more susceptible to cyclic stress. Homograft valves are usable in conjunction with an autograft valve. An autograft is a heart valve transplant from one anatomic point to a separate location within the same individual. The Ross procedure is an example of a surgical intervention utilizing a homograft/autograft procedure.[1] The Ross procedure uses a cadaver valve (homograft) to replace a functional pulmonary valve. The healthy pulmonary valve is then transferred to replace the diseased aortic valve (autograft). The rationale behind the autograft component of the Ross procedure is due to the higher pressure system of the left side of the heart. A healthy pulmonary valve has enough regenerative capability to withstand the high-pressure environment compared to a homograft.The choice of the prosthetic type is patient-centered and depends on two risk factors: the concern for anticoagulation-associated bleeding risk, and valve deterioration.[2] The durability of the tissue is inversely correlated with age, reflecting the increased level of deterioration in younger patients compared to the older population. Thus, bioprosthetics tend to be a more suitable choice in older patients who may be less tolerant of anticoagulation while mechanical valves may be a better option in younger patients where bioprosthetic valves may have accelerated deterioration. Although older patients may be opted for a bioprosthetic valve to avoid complications with anticoagulation, the development of atrial fibrillation in the same demographic may effectively cancel any benefit of this management approach.[3]The delivery method of the prosthetic valve (surgical vs. transcatheter) should also play a role in the discussion of management. SAVR (surgical aortic valve replacement) is a highly invasive procedure that is accompanied by the usual risk associated with an open thoracotomy. TAVR (transcatheter aortic valve replacement) is considerably less invasive but offers its particular share of disadvantages, including valve oversizing, the difficulty of intravascular manipulation, and positioning. Furthermore, any injury occurring during prosthesis deployment may have adverse effects on the lifespan of the prosthesis.[4] Perioperative imaging can help minimize these adverse effects although they do not comletely eliminate the risks. CT imaging has utility as an imaging tool for all stages of TAVR.[5]