Continuing Education Activity
Meningococcal disease is an infection caused by the bacterium Neisseria meningitidis. Meningococcal infections affect individuals of all ages, including healthy young adults. N meningitidis is the leading cause of bacterial meningitis in children and young adults, and the second-leading cause of community-acquired bacterial meningitis in adults. Although meningitis and meningococcemia are the classical presentations of meningococcal disease, a broad spectrum of clinical manifestations exists, spanning from asymptomatic nasopharyngeal colonization to fulminant disease. Prompt identification of patients with meningococcal disease, initiation of treatment, and prophylaxis for their close contacts are critical because meningococcal disease can rapidly progress and cause severe complications or death.
This educational activity reviews the etiology and epidemiology of N meningitidis infection. The need for a thorough history and clinical evaluation to develop tailored treatment strategies for patients with N meningitidis infection. This course highlights the need for interprofessional team collaboration to improve the management of meningococcal disease and patient outcomes.
Objectives:
Identify the clinical manifestations of Neisseria meningitidis infection.
Apply evidence-based diagnostic strategies for patients suspected of having Neisseria meningitidis infection.
Implement timely and appropriate treatment strategies for patients with Neisseria meningitidis infection.
Collaborate with interprofessional team members to improve the care coordination of those with Neisseria meningitidis infection, improving patient outcomes.
Introduction
Neisseria meningitidis was first discovered by Anton Weichselbaum in 1887 during his analysis of the cerebrospinal fluid (CSF) from a patient infected with meningitis.[1] N meningitidis is a human-specific bacterium that causes a multitude of illnesses, collectively termed as meningococcal disease. Up to 10% of the general population carries the bacteria in their nose and throat without experiencing any adverse effects. Risk factors for meningococcal carriage include being in the adolescent and young adult age group, being male, living in congregate conditions, being exposed to cigarette smoke, and frequently attending bars and parties.[2]
N meningitidis is associated with many infections. However, its association with fulminant meningococcemia and meningococcal meningitis has led to its significant impact. More importantly, despite recent vaccine campaigns to help reduce complications of this disease, its effects continue to impact at-risk populations.[3]
Etiology
N meningitidis is an aerobic or facultative anaerobic, gram-negative diplococcus that exclusively infects humans (see Image. Diplococci).[4] There are at least 12 serotypes based on unique capsular polysaccharides of N meningitidis, with serotypes A, B, C, W, X, and Y responsible for the majority of meningococcal infections. Serotypes A and C are the main serotypes responsible for meningococcal disease in Africa. Serotypes B, C, and Y are the main serotypes causing disease in Europe, the United States, and Canada.[5] Serotype W is responsible for epidemic outbreaks worldwide and is associated with the Hajj pilgrimages to Saudi Arabia.[6]
N meningitidis is a common and significant cause of community-acquired bacterial meningitis in the United States, affecting children and adults. It is a devastating infection with a high mortality rate and is the second most common cause of community-acquired adult bacterial meningitis after Streptococcus pneumoniae.[7] Other infections that N meningitidis can cause are meningococcemia (a bloodstream infection caused by the bacterium), pneumonia, septic arthritis, pericarditis, and urethritis. In addition, N meningitidis can also cause endemic and epidemic infections, affecting young, healthy adults.[5]
Epidemiology
The incidence of meningococcal disease has decreased with the routine administration of meningococcal vaccination. In the United States, the incidence of meningococcal disease decreased from 1.2 cases per 100,000 individuals in 1996 to 0.1 cases per 100,000 individuals in 2018.[5] The highest incidence of meningococcal disease appears in infants younger than 1 (2.45 cases per 100,000 individuals).[8] N meningitidis can cause endemic and epidemic outbreaks and is a significant cause of bacterial meningitis in sub-Saharan Africa, leading to the region being called the meningitis belt.[5] The mortality rate varies from approximately 10% to 14% among patients who receive treatment and can reach up to 50% among patients who do not receive treatment.[9]
Pathophysiology
Humans are the exclusive hosts of Neisseria meningitidis, carrying the pathogen in their nasopharynx before the onset of systemic infection. Colonized humans may be asymptomatic carriers.[2] Colonization of the nasopharynx puts individuals who are in close contact with infected patients, such as family members, college roommates, and military recruits, at an increased risk of contracting N meningitidis and possible infection. Individuals involved in laboratory work or medical care exposure, such as intubating an infected patient with no face shield or mask, are also at an increased risk of N meningitidis colonization of the nasopharynx through inhaling aerosolized particles containing the bacteria. The incubation period varies and ranges from 1 to 14 days.[10]
Once N meningitidis is colonized, several virulence factors help the bacterium in invading human cells and the immune system. After it invades the bloodstream, a robust immune response is triggered, leading to endothelial damage, capillary leakage, tissue necrosis, organ failure, and the development of meningococcal sepsis. Bloodstream invasion is considered to be the primary route to the brain, but N meningitidis can also cross the cribriform plate of the ethmoid bone.[11]
Similar to many other diseases, the immune system is essential in protecting the host. Certain health conditions that weaken the human immune system increase the risk of contracting meningococcal diseases, including meningococcemia and meningitis. These conditions include HIV infection, AIDS, asplenia, and complement deficiency.[4]
History and Physical
The clinical presentation of N meningitidis can vary widely and may initially appear benign, making it difficult to diagnose, especially in areas with no reported epidemics. Typically, the initial presentation of N meningitidis caused by meningitis can include sudden onset of fever, headache, nausea, vomiting, severe myalgias, nonspecific rash, sore throat, and other upper respiratory symptoms. These symptoms may be confused with a multitude of other illnesses. The potential for rapid disease progression, especially given the possibility of death within hours of symptom onset, makes early identification of meningococcal disease challenging due to its low incidence and nonspecific early symptoms. Later presentation of the meningococcal disease includes neck stiffness, photophobia, petechiae or hemorrhagic rash, altered mental status, shock, abnormal skin color, purpura fulminans, or even disseminated intravascular coagulation.[10]
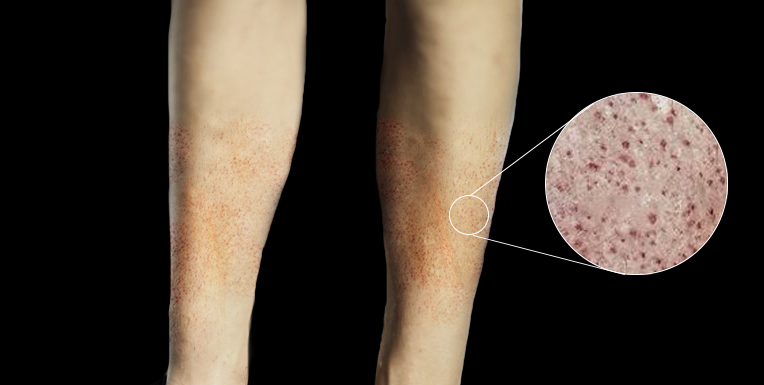
Vital signs may include hypotension and tachycardia, which indicate early signs of sepsis. A complete physical examination should be conducted, including a thorough skin inspection for any signs of a rash. The rash, a distinguishing feature, may initially appear as small lesions that may appear urticarial, macular, or papular. The rash can develop into petechiae, purpura, or ecchymosis later (see Image. Petechiae). These may be early signs of thrombocytopenia, purpura fulminans, and disseminated intravascular coagulation.[10] Although Kernig and Brudzinski signs are classic signs of meningitis, their respective sensitivities of 55.5% and 53.3% make them unreliable in excluding meningitis.[12] When considering signs and symptoms of the disease, it is important to note that the classic triad of fever, altered mental status, and nuchal rigidity is rare, occurring in only about 44% of cases. However, when 2 of the 4 symptoms, including headache, altered mental status, neck stiffness, and fever, were present, 95% of patients ultimately diagnosed with meningitis were identified.[13] Clinicians should consider N meningitidis infection in patients with sudden onset of fever who display early signs of sepsis, rapidly progressing disease, and rash.
Purpura fulminans, a complication of meningococcal disease resulting from vascular collapse, is characterized by cutaneous hemorrhage and skin necrosis due to vascular thrombosis and disseminated intravascular coagulation. Typically, petechiae and erythema are observed on the skin but can progress to ecchymosis and eventually painful areas of necrosis, with the development of bullae and vesicles. Gangrenous necrosis can occur and be severe, often requiring limb amputation. Atypical skin color, cold hands and feet, and painful legs may be the signs of vascular compromise.[10]
Although these infections are rarely caused by N meningitidis, meningococcal disease can also include pneumonia, septic arthritis, urethritis, and pericarditis.[5]
Evaluation
A lumbar puncture is the preferred diagnostic tool for identifying meningitis. Prioritizing a lumbar puncture ensures prompt evaluation and diagnosis. Evidence of elevated intracranial pressure, such as altered mental status, new-onset seizure within 1 week of symptoms, papilledema, an immunocompromised state, or focal neurologic deficits; coagulopathy; and cardiorespiratory insufficiency are contraindications to lumbar puncture. Antibiotic administration should occur promptly, even before lumbar puncture completion, for those patients when meningitis is highly likely to be the diagnosis.[9] Even in pretreated patients, blood cultures and CSF studies can still be useful. CSF analysis should include gram staining, culture, glucose, cell count, and protein count. CSF findings that suggest bacterial meningitis include decreased glucose levels, polymorphonuclear pleocytosis, and increased protein levels. CSF culture is the gold standard for the diagnosis of bacterial meningitis.[14]
CSF gram staining, polymerase chain reaction (PCR), and latex agglutination may aid in confirmation, particularly in patients with negative CSF cultures after antibiotic pretreatment. PCR detects strands of bacterial DNA, not requiring an intact bacterium for detection, whereas latex agglutination detects capsular antigens.[14]
Computed tomography (CT) imaging may assist in the diagnosis of patients with altered mental status, mainly to rule out other diagnoses under consideration.[9] If imaging is necessary, it is crucial to coordinate with the nursing team and imaging suite to avoid delaying antibiotic administration.
Treatment / Management
Early recognition and treatment of meningococcal infections are imperative in improving outcomes. The primary focus in treating meningococcal infection includes administering antibiotics, implementing isolation, contact precaution, consulting with infectious disease specialists, management in the intensive care unit (ICU), management of coagulopathies, and identifying individuals at risk who may have been exposed to N meningitidis.[9]
Because meningococcal meningitis can present similarly to other bacterial meningitides, empiric treatment should be initiated while awaiting culture results. This preemptive approach includes a third-generation cephalosporin such as ceftriaxone or cefotaxime. If culture identifies the organism as penicillin-susceptible, treatment can be switched to penicillin G. However, continuing third-generation cephalosporin treatment is also an option. For patients who have significant allergies to penicillin and other beta-lactams, chloramphenicol may be an alternative. The duration of antibiotic therapy is typically 5 to 6 days.[9][15]
Dosages
- Ceftriaxone dosing is 2 g for adults and 50 mg/kg for pediatric patients older than 1 month given intravenously (IV) every 12 hours, whereas cefotaxime dosing is 2 g every 4 to 6 hours for adults and 50 mg/kg for pediatric patients older than 1 month every 6 hours.[9]
- Third-generation cephalosporins are generally preferable due to their high efficacy and simpler dosing regimens.
- Penicillin G dosing is 4 million units every 4 hours IV for adults and pediatric patients older than 1 month.[9]
- Chloramphenicol dosing is 50 mg/kg IV 4 times per day.[9]
- Serum concentrations require monitoring because of the risk of chloramphenicol toxicity.[16]
- High-dose dexamethasone should be administered at initial suspicion of bacterial meningitis due to its protective benefits in pneumococcal meningitis.[9][17] Dexamethasone should be administered IV within 4 hours before but no more than 12 hours after the first dose of IV antibiotics. The dosage should be 0.15 mg/kg every 6 hours up to a maximum dosage of 10 mg every 6 hours.[9][15] However, dexamethasone has no therapeutic benefit in meningococcal meningitis and should be discontinued once this diagnosis is established.[17][15]
Patients with meningococcal infection should also be treated aggressively with supportive care, especially for sepsis or septic shock. This may include intravenous fluid resuscitation and the use of vasopressors such as norepinephrine.[15] Patients who show evidence of disseminated intravascular coagulation may require aggressive hydration, blood transfusions, platelet replacement, and possibly even coagulation factor replacement. Researchers have proposed protein C as an adjuvant treatment, but its use is controversial and currently not commonly used.[9]
Differential Diagnosis
S pneumoniae is the most common cause of bacterial meningitis in the United States and should be included in the differential diagnosis for patients suspected of having meningococcal meningitis. Less common causes of bacterial meningitis include Haemophilus influenzae, group B Streptococcus, and Listeria monocytogenes.[14] Other causes of meningitis include viral, tuberculous, eosinophilic, and fungal meningitis and noninfectious causes such as malignancy.[18]
Clinicians examining adults with a maculopapular rash and fever should consider alternate etiologies, including infectious mononucleosis, West Nile virus, Zika virus, HIV, Ebola virus, Rocky Mountain spotted fever, ehrlichiosis, and hypersensitivity drug reactions.[19] In addition, children with a maculopapular rash and fever should be considered for a differential diagnosis that includes Kawasaki disease, measles, scarlet fever, rubella, parvovirus B19, roseola, Epstein-Barr virus, and hand, foot, and mouth disease.[20]
Prognosis
Although the mortality rate of meningococcal infection can be as high as 50% in untreated patients, early and aggressive treatment can reduce the mortality rate to approximately 10% to 14%. Early administration of antibiotics is crucial in determining a good outcome of meningococcal infection. Even with treatment, long-term complications can still occur in 11% to 19% of survivors.[9]
Similar to other causes of bacterial meningitis, patients who survive meningococcal infections should undergo routine follow-up. Hearing tests are recommended within 4 weeks of hospital discharge. Orthopedic follow-up and prosthetic fitting are necessary for patients who have suffered limb amputations. Patients may experience psychological and psychiatric complications, including posttraumatic stress disorder, depression, and behavioral abnormalities, requiring follow-up with psychologists and psychiatrists.[15]
Complications
Common complications of meningococcal disease include chronic pain, skin scarring, limb amputation, and neurological impairments, ranging from hearing and visual impairments to motor function impairments. Hearing impairment and amputations occur in approximately 3% of cases, arthritis occurs in 10% of cases, and postinfection inflammatory syndrome occurs in 6% to 15% of cases. Up to one-third of survivors of meningococcal disease experience psychological disorders such as posttraumatic stress disorder, anxiety, and depression.[9] Urgent complications of meningococcal disease include septic shock, purpura fulminans, seizures, hydrocephalus, cerebral venous sinus thrombosis, and subdural empyema.[15]
Consultations
The interprofessional team managing meningococcal infection consists of internists, pediatricians, infectious disease specialists, hematologists, neurologists, and nursing staff.[15]
Deterrence and Patient Education
Early recognition and treatment of those infected are crucial, as meningococcal infection is a devastating infectious disease that has led to epidemics in the past and can do so again. Any meningococcal infection is reportable, and local health departments should be notified.[5]
Vaccination is the best strategy for preventing meningococcal disease, especially in populations at higher risk. The infectious disease nurse should educate patients about vaccination. Vaccines against N meningitidis serogroups A, C, W, Y, and B are routinely recommended in the United States for adolescents, young adults, and individuals at increased risk for meningococcal disease. The overall burden of meningococcal infection worldwide has diminished due to these vaccinations.[5]
Individuals at risk, including college students, military recruits, immunocompromised patients such as those with HIV/AIDS, complement deficiency, and those with asplenia, should be vaccinated. Individuals who work with N meningitidis in laboratories, travelers to endemic areas such as the meningitis belt, and those exposed to outbreaks of meningococcal vaccine should also be vaccinated based on local epidemiology.[5]
Table. Routine meningococcal vaccine recommendations for healthy adolescents and young adults aged 11 through 23 in the United States [5]
| Meningococcal Vaccine |
Primary Dosage(s) |
Booster Dosage(s) |
| MenACWY(for ages 11-21)* |
- Catch-up vaccination at ages 13-18 for adolescents who did not receive the primary dose at the recommended age
|
- 1 dose at age ≥16; the minimum interval between doses is 8 weeks.
- If the first dose was given after the age of 16, a booster is not needed unless the individual is at higher risk for meningococcal disease.
- College students living in residence halls should receive ≥1 dose of MenACWY ≤5 years before college entry.
|
| MenB(for ages 16-23) |
Preferred age: 16-18
- Either:
- MenB-4C: 2 doses, ≥1 month apart, or
- MenB-FHbp: 2 doses, ≥6 months apart
|
Not routinely recommended unless the individual becomes at risk.
|
*Although the Advisory Committee on Immunization Practices (ACIP) recommends routine MenACWY vaccination only for individuals aged 11 through 18, MenACWY may be given to individuals aged 19 through 21 if they have not been vaccinated previously.
Moreover, the MenACWY vaccine is mandatory for all individuals visiting Mecca for Hajj/Umrah.[5]
Pearls and Other Issues
Individuals who come into close contact with meningococcal-infected patients can themselves become infected and display symptoms within 14 days after initial contact. Close contact refers to proximity with the patient for more than 4 hours within the past 7 days. Examples of close contacts include family members, roommates, military recruits, and individuals in daycare centers.[10] Individuals who are exposed to the patient's oral secretions, such as through kissing, or respiratory secretions, such as during intubation, suctioning, and proximity with passengers on a long flight, are also at risk. If recognized, antimicrobial chemoprophylaxis should be initiated. Due to its high efficacy, chemoprophylaxis should be strongly considered for patients who may have had questionable close contact with an infected patient.
Several antimicrobial chemoprophylaxis options are listed below.[9]
- Ceftriaxone is administered at 250 mg intramuscularly as a one-time dose for adults and 125 mg for pediatric patients younger than 15.
- Rifampin is prescribed at 600 mg orally or intravenously twice daily for 2 days for adults. In pediatric patients younger than 1 month, the recommended dosage is 5 mg/kg, whereas for those older than 1 month, it is 10 mg/kg. Rifampin is not recommended for pregnant patients.
- Ciprofloxacin is given orally at 500 mg as a one-time dose for adults, and for pediatric patients older than 1 month, the dosage is 20 mg/kg (maximum 500 mg).
- Azithromycin, typically reserved as a last choice, is administered orally at 500 mg as a one-time dose in adults and 10 mg/kg (maximum 500 mg) in pediatric patients older than 1 month.
Enhancing Healthcare Team Outcomes
Meningococcal infection is a devastating, contagious disease that can progress rapidly. Recognizing the illness is perhaps the most challenging part of assessing and treating patients. This requires an interprofessional team consisting of physicians, infectious disease specialists, ICU nurses, pharmacists, adherence to proper isolation protocols, and collaboration with the local health department to treat infected patients. Meningococcal disease is reportable, and the local health department can assist in identifying individuals at risk and managing outbreaks.[5]
Clinicians who initiate treatment would benefit by enlisting the assistance of an infectious disease board-certified pharmacist. They can verify agent selection with the latest antibiogram data, confirm dosage, and check for drug interactions that may compromise therapy. Nurses typically administer these agents and should have a comprehensive understanding of appropriate isolation protocols. They also monitor for therapeutic effectiveness and watch for any adverse reactions, promptly notifying the healthcare team of any issues. The interprofessional team approach is the best way to optimize therapeutic results while minimizing the spread of infection and adverse reactions.