[1]
Ostrom QT,Cioffi G,Gittleman H,Patil N,Waite K,Kruchko C,Barnholtz-Sloan JS, CBTRUS Statistical Report: Primary Brain and Other Central Nervous System Tumors Diagnosed in the United States in 2012-2016. Neuro-oncology. 2019 Nov 1
[PubMed PMID: 31675094]
[2]
Lim YS,Kim MK,Park BJ,Kim TS,Lim YJ, Long term clinical outcomes of malignant meningiomas. Brain tumor research and treatment. 2013 Oct;
[PubMed PMID: 24904897]
Level 2 (mid-level) evidence
[3]
Wiemels J,Wrensch M,Claus EB, Epidemiology and etiology of meningioma. Journal of neuro-oncology. 2010 Sep;
[PubMed PMID: 20821343]
[4]
Buerki RA,Horbinski CM,Kruser T,Horowitz PM,James CD,Lukas RV, An overview of meningiomas. Future oncology (London, England). 2018 Sep;
[PubMed PMID: 30084265]
Level 3 (low-level) evidence
[5]
Shao C,Bai LP,Qi ZY,Hui GZ,Wang Z, Overweight, obesity and meningioma risk: a meta-analysis. PloS one. 2014;
[PubMed PMID: 24587258]
Level 2 (mid-level) evidence
[6]
Gurcay AG,Bozkurt I,Senturk S,Kazanci A,Gurcan O,Turkoglu OF,Beskonakli E, Diagnosis, Treatment, and Management Strategy of Meningioma during Pregnancy. Asian journal of neurosurgery. 2018 Jan-Mar;
[PubMed PMID: 29492130]
[7]
Connolly ID,Cole T,Veeravagu A,Popat R,Ratliff J,Li G, Craniotomy for Resection of Meningioma: An Age-Stratified Analysis of the MarketScan Longitudinal Database. World neurosurgery. 2015 Dec
[PubMed PMID: 26318633]
[8]
de Robles P,Fiest KM,Frolkis AD,Pringsheim T,Atta C,St Germaine-Smith C,Day L,Lam D,Jette N, The worldwide incidence and prevalence of primary brain tumors: a systematic review and meta-analysis. Neuro-oncology. 2015 Jun;
[PubMed PMID: 25313193]
Level 1 (high-level) evidence
[9]
Hortobágyi T,Bencze J,Varkoly G,Kouhsari MC,Klekner Á, Meningioma recurrence. Open medicine (Warsaw, Poland). 2016;
[PubMed PMID: 28352788]
[10]
Shibuya M, Pathology and molecular genetics of meningioma: recent advances. Neurologia medico-chirurgica. 2015;
[PubMed PMID: 25744347]
Level 3 (low-level) evidence
[11]
Varlotto J,Flickinger J,Pavelic MT,Specht CS,Sheehan JM,Timek DT,Glantz MJ,Sogge S,Dimaio C,Moser R,Yunus S,Fitzgerald TJ,Upadhyay U,Rava P,Tangel M,Yao A,Kanekar S, Distinguishing grade I meningioma from higher grade meningiomas without biopsy. Oncotarget. 2015 Nov 10;
[PubMed PMID: 26472106]
[12]
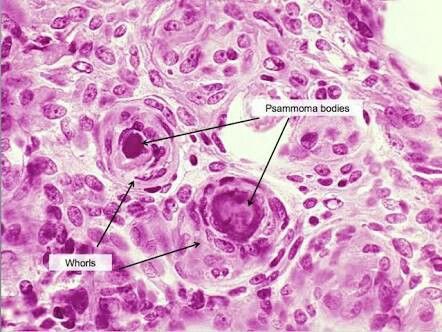
Backer-Grøndahl T,Moen BH,Torp SH, The histopathological spectrum of human meningiomas. International journal of clinical and experimental pathology. 2012;
[PubMed PMID: 22558478]
[13]
Westwick HJ,Shamji MF, Effects of sex on the incidence and prognosis of spinal meningiomas: a Surveillance, Epidemiology, and End Results study. Journal of neurosurgery. Spine. 2015 Sep;
[PubMed PMID: 26023898]
[14]
Louis DN,Perry A,Reifenberger G,von Deimling A,Figarella-Branger D,Cavenee WK,Ohgaki H,Wiestler OD,Kleihues P,Ellison DW, The 2016 World Health Organization Classification of Tumors of the Central Nervous System: a summary. Acta neuropathologica. 2016 Jun
[PubMed PMID: 27157931]
[15]
Sotiriadis C,Vo QD,Ciarpaglini R,Hoogewoud HM, Cystic meningioma: diagnostic difficulties and utility of MRI in diagnosis and management. BMJ case reports. 2015 Mar 26;
[PubMed PMID: 25814028]
Level 3 (low-level) evidence
[16]
Mittal A,Layton KF,Finn SS,Snipes GJ,Opatowsky MJ, Cystic meningioma: unusual imaging appearance of a common intracranial tumor. Proceedings (Baylor University. Medical Center). 2010 Oct;
[PubMed PMID: 21240328]
[19]
Simas NM,Farias JP, Sphenoid Wing en plaque meningiomas: Surgical results and recurrence rates. Surgical neurology international. 2013;
[PubMed PMID: 23956929]
[20]
Basu K,Majumdar K,Chatterjee U,Ganguli M,Chatterjee S, En plaque meningioma with angioinvasion. Indian journal of pathology
[PubMed PMID: 20551544]
[21]
Baek JU,Cho YD,Yoo JC, An osteolytic meningioma en plaque of the sphenoid ridge. Journal of Korean Neurosurgical Society. 2008 Jan;
[PubMed PMID: 19096543]
[22]
Matschke J,Addo J,Bernreuther C,Zustin J, Osseous changes in meningioma en plaque. Anticancer research. 2011 Feb;
[PubMed PMID: 21378343]
[24]
Mansouri A,Badhiwala J,Mansouri S,Zadeh G, The evolving role of radiosurgery in the management of radiation-induced meningiomas: a review of current advances and future directions. BioMed research international. 2014;
[PubMed PMID: 25136551]
Level 3 (low-level) evidence
[25]
Goto Y,Yamada S,Yamada SM,Nakaguchi H,Hoya K,Murakami M,Yamazaki K,Ishida Y,Matsuno A, Radiation-induced meningiomas in multiple regions, showing rapid recurrence and a high MIB 1 labeling index: a case report and review of the literature. World journal of surgical oncology. 2014 Apr 26;
[PubMed PMID: 24767145]
Level 3 (low-level) evidence
[26]
Oikonomou A,Birbilis T,Daskalogiannakis G,Prassopoulos P, Meningioma of the conus medullaris mimicking neurofibroma--possibly radiation induced. The spine journal : official journal of the North American Spine Society. 2011 Feb;
[PubMed PMID: 21193353]
[27]
Murakami T,Tanishima S,Takeda C,Kato S,Nagashima H, Ossified Metaplastic Spinal Meningioma Without Psammomatous Calcification: A Case Report. Yonago acta medica. 2019 Jun;
[PubMed PMID: 31320828]
Level 3 (low-level) evidence
[28]
Kitagawa M,Nakamura T,Aida T,Iwasaki Y,Abe H,Nagashima K, [Clinicopathologic analysis of ossification in spinal meningioma]. Noshuyo byori = Brain tumor pathology. 1994;
[PubMed PMID: 8162148]
[29]
Bhat AR,Wani MA,Kirmani AR,Ramzan AU, Histological-subtypes and anatomical location correlated in meningeal brain tumors (meningiomas). Journal of neurosciences in rural practice. 2014 Jul;
[PubMed PMID: 25002762]
[30]
Marchand AA,O'Shaughnessy J, Subtle clinical signs of a meningioma in an adult: a case report. Chiropractic
[PubMed PMID: 24490991]
Level 3 (low-level) evidence
[31]
Rodríguez-Porcel F,Hughes I,Anderson D,Lee J,Biller J, Foster Kennedy Syndrome Due to Meningioma Growth during Pregnancy. Frontiers in neurology. 2013
[PubMed PMID: 24273529]
[32]
Hekmatpanah J, Evidence-based treatment of cavernous sinus meningioma. Surgical neurology international. 2019
[PubMed PMID: 31819821]
[33]
Gadgil N,Hansen D,Barry J,Chang R,Lam S, Posterior fossa syndrome in children following tumor resection: Knowledge update. Surgical neurology international. 2016
[PubMed PMID: 27057398]
[35]
Elwatidy SM,Albakr AA,Al Towim AA,Malik SH, Tumors of the lateral and third ventricle: surgical management and outcome analysis in 42 cases. Neurosciences (Riyadh, Saudi Arabia). 2017 Oct;
[PubMed PMID: 29057852]
Level 3 (low-level) evidence
[36]
Slentz DH,Bellur S,Taheri MR,Almira-Suarez MI,Sherman JH,Mansour TN, Orbital malignant meningioma: a unique presentation of a rare entity. Orbit (Amsterdam, Netherlands). 2018 Dec
[PubMed PMID: 29485367]
[38]
Samartzis D,Gillis CC,Shih P,O'Toole JE,Fessler RG, Intramedullary Spinal Cord Tumors: Part I-Epidemiology, Pathophysiology, and Diagnosis. Global spine journal. 2015 Oct;
[PubMed PMID: 26430598]
[39]
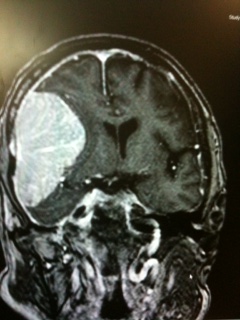
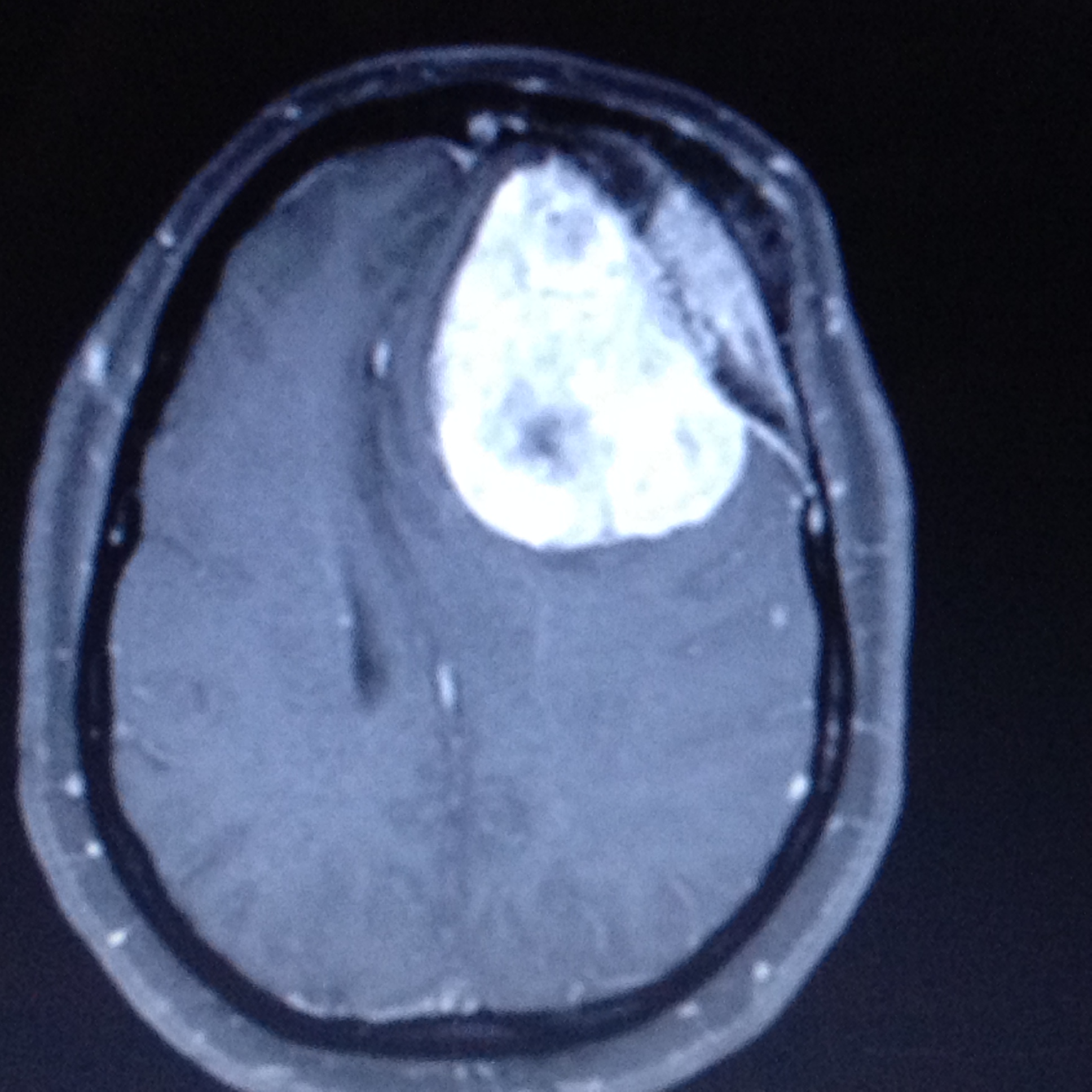
Watts J,Box G,Galvin A,Brotchie P,Trost N,Sutherland T, Magnetic resonance imaging of meningiomas: a pictorial review. Insights into imaging. 2014 Feb;
[PubMed PMID: 24399610]
[40]
Hou J,Kshettry VR,Selman WR,Bambakidis NC, Peritumoral brain edema in intracranial meningiomas: the emergence of vascular endothelial growth factor-directed therapy. Neurosurgical focus. 2013 Dec;
[PubMed PMID: 24289127]
[41]
Kim BW,Kim MS,Kim SW,Chang CH,Kim OL, Peritumoral brain edema in meningiomas : correlation of radiologic and pathologic features. Journal of Korean Neurosurgical Society. 2011 Jan;
[PubMed PMID: 21494359]
[42]
George AE,Russell EJ,Kricheff II, White matter buckling: CT sign of extraaxial intracranial mass. AJR. American journal of roentgenology. 1980 Nov;
[PubMed PMID: 6778144]
[43]
Papacci F,Pedicelli A,Montano N, The role of preoperative angiography in the management of giant meningiomas associated to vascular malformation. Surgical neurology international. 2015;
[PubMed PMID: 26167366]
[44]
Tsuchiya K,Katase S,Yoshino A,Hachiya J, MR digital subtraction angiography in the diagnosis of meningiomas. European journal of radiology. 2003 May;
[PubMed PMID: 12714229]
[46]
Galldiks N,Albert NL,Sommerauer M,Grosu AL,Ganswindt U,Law I,Preusser M,Le Rhun E,Vogelbaum MA,Zadeh G,Dhermain F,Weller M,Langen KJ,Tonn JC, PET imaging in patients with meningioma-report of the RANO/PET Group. Neuro-oncology. 2017 Nov 29;
[PubMed PMID: 28605532]
[47]
Nowosielski M,Galldiks N,Iglseder S,Kickingereder P,von Deimling A,Bendszus M,Wick W,Sahm F, Diagnostic challenges in meningioma. Neuro-oncology. 2017 Nov 29;
[PubMed PMID: 28531331]
[48]
Yamaguchi S,Takeda M,Takahashi T,Yamahata H,Mitsuhara T,Niiro T,Hanakita J,Hida K,Arita K,Kurisu K, Ginkgo leaf sign: a highly predictive imaging feature of spinal meningioma. Journal of neurosurgery. Spine. 2015 Nov;
[PubMed PMID: 26230423]
[49]
Quddusi A,Shamim MS, Simpson grading as predictor of meningioma recurrence. JPMA. The Journal of the Pakistan Medical Association. 2018 May;
[PubMed PMID: 29885194]
[50]
Nanda A,Bir SC,Maiti TK,Konar SK,Missios S,Guthikonda B, Relevance of Simpson grading system and recurrence-free survival after surgery for World Health Organization Grade I meningioma. Journal of neurosurgery. 2017 Jan;
[PubMed PMID: 27058201]
[51]
Gousias K,Schramm J,Simon M, The Simpson grading revisited: aggressive surgery and its place in modern meningioma management. Journal of neurosurgery. 2016 Sep
[PubMed PMID: 26824369]
[53]
Bartek J Jr,Sjåvik K,Förander P,Solheim O,Gulati S,Weber C,Ingebrigtsen T,Jakola AS, Predictors of severe complications in intracranial meningioma surgery: a population-based multicenter study. World neurosurgery. 2015 May
[PubMed PMID: 25655686]
Level 2 (mid-level) evidence
[54]
Walcott BP,Nahed BV,Brastianos PK,Loeffler JS, Radiation Treatment for WHO Grade II and III Meningiomas. Frontiers in oncology. 2013 Sep 2;
[PubMed PMID: 24032107]
[56]
Pisćević I,Villa A,Milićević M,Ilić R,Nikitović M,Cavallo LM,Grujičić D, The Influence of Adjuvant Radiotherapy in Atypical and Anaplastic Meningiomas: A Series of 88 Patients in a Single Institution. World neurosurgery. 2015 Jun
[PubMed PMID: 25769488]
[57]
Altering patients' responses to surgery: an extension and replication., Johnson JE,Fuller SS,Endress MP,Rice VH,, Research in nursing & health, 1978 Oct
[PubMed PMID: 25535067]
[58]
Aizer AA,Arvold ND,Catalano P,Claus EB,Golby AJ,Johnson MD,Al-Mefty O,Wen PY,Reardon DA,Lee EQ,Nayak L,Rinne ML,Beroukhim R,Weiss SE,Ramkissoon SH,Abedalthagafi M,Santagata S,Dunn IF,Alexander BM, Adjuvant radiation therapy, local recurrence, and the need for salvage therapy in atypical meningioma. Neuro-oncology. 2014 Nov
[PubMed PMID: 24891451]
[59]
Samblas J,Luis Lopez Guerra J,Bustos J,Angel Gutierrez-Diaz J,Wolski M,Peraza C,Marsiglia H,Sallabanda K, Stereotactic radiosurgery in patients with multiple intracranial meningiomas. Journal of B.U.ON. : official journal of the Balkan Union of Oncology. 2014 Jan-Mar;
[PubMed PMID: 24659672]
[60]
Wang WH,Lee CC,Yang HC,Liu KD,Wu HM,Shiau CY,Guo WY,Pan DH,Chung WY,Chen MT, Gamma Knife Radiosurgery for Atypical and Anaplastic Meningiomas. World neurosurgery. 2016 Mar
[PubMed PMID: 26485417]
[61]
Puchner MJ,Hans VH,Harati A,Lohmann F,Glas M,Herrlinger U, Bevacizumab-induced regression of anaplastic meningioma. Annals of oncology : official journal of the European Society for Medical Oncology. 2010 Dec;
[PubMed PMID: 21041375]
[62]
Chourmouzi D,Potsi S,Moumtzouoglou A,Papadopoulou E,Drevelegas K,Zaraboukas T,Drevelegas A, Dural lesions mimicking meningiomas: A pictorial essay. World journal of radiology. 2012 Mar 28;
[PubMed PMID: 22468187]
[64]
Ostrom QT,Gittleman H,Truitt G,Boscia A,Kruchko C,Barnholtz-Sloan JS, CBTRUS Statistical Report: Primary Brain and Other Central Nervous System Tumors Diagnosed in the United States in 2011-2015. Neuro-oncology. 2018 Oct 1
[PubMed PMID: 30445539]
[65]
Development of an instrument to measure exercise of self-care agency., Kearney BY,Fleischer BJ,, Research in nursing & health, 1979 Mar
[PubMed PMID: 27274498]
[66]
Aizer AA,Bi WL,Kandola MS,Lee EQ,Nayak L,Rinne ML,Norden AD,Beroukhim R,Reardon DA,Wen PY,Al-Mefty O,Arvold ND,Dunn IF,Alexander BM, Extent of resection and overall survival for patients with atypical and malignant meningioma. Cancer. 2015 Dec 15
[PubMed PMID: 26308667]
[68]
Sughrue ME,Rutkowski MJ,Shangari G,Chang HQ,Parsa AT,Berger MS,McDermott MW, Risk factors for the development of serious medical complications after resection of meningiomas. Clinical article. Journal of neurosurgery. 2011 Mar;
[PubMed PMID: 20653395]
[69]
Shaikh N,Dixit K,Raizer J, Recent advances in managing/understanding meningioma. F1000Research. 2018;
[PubMed PMID: 29770198]
Level 3 (low-level) evidence
[70]
Rajaraman P, Hunting for the causes of meningioma--obesity is a suspect. Cancer prevention research (Philadelphia, Pa.). 2011 Sep;
[PubMed PMID: 21893497]
[71]
Michaud DS,Bové G,Gallo V,Schlehofer B,Tjønneland A,Olsen A,Overvad K,Dahm CC,Teucher B,Boeing H,Steffen A,Trichopoulou A,Bamia C,Kyrozis A,Sacerdote C,Agnoli C,Palli D,Tumino R,Mattiello A,Bueno-de-Mesquita HB,Peeters PH,May AM,Barricarte A,Chirlaque MD,Dorronsoro M,José Sánchez M,Rodríguez L,Duell EJ,Hallmans G,Melin BS,Manjer J,Borgquist S,Khaw KT,Wareham N,Allen NE,Travis RC,Romieu I,Vineis P,Riboli E, Anthropometric measures, physical activity, and risk of glioma and meningioma in a large prospective cohort study. Cancer prevention research (Philadelphia, Pa.). 2011 Sep;
[PubMed PMID: 21685234]
[72]
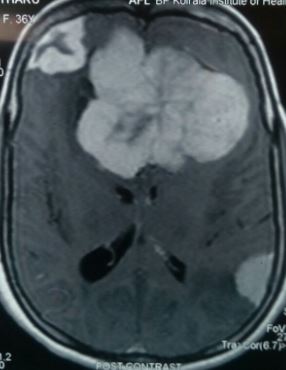
Silva DP,Carvalho SD,Marçal N,Dias L, Giant meningioma in paranasal sinuses: an atypical nasal occupation. BMJ case reports. 2018 Jan 4;
[PubMed PMID: 29301819]
Level 3 (low-level) evidence