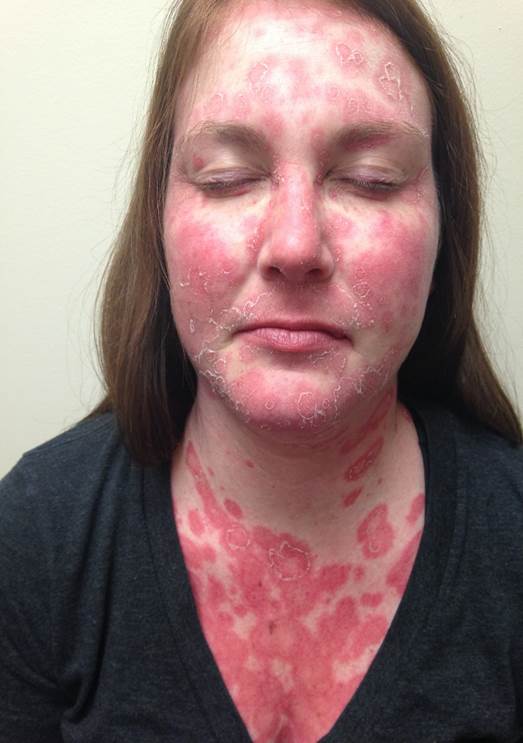
[1]
Parodi A, Caproni M, Cardinali C, Bernacchi E, Fuligni A, De Panfilis G, Zane C, Papini M, Veller FC, Vaccaro M, Fabbri P. Clinical, histological and immunopathological features of 58 patients with subacute cutaneous lupus erythematosus. A review by the Italian group of immunodermatology. Dermatology (Basel, Switzerland). 2000:200(1):6-10
[PubMed PMID: 10681606]
[2]
Biazar C, Sigges J, Patsinakidis N, Ruland V, Amler S, Bonsmann G, Kuhn A, EUSCLE co-authors. Cutaneous lupus erythematosus: first multicenter database analysis of 1002 patients from the European Society of Cutaneous Lupus Erythematosus (EUSCLE). Autoimmunity reviews. 2013 Jan:12(3):444-54. doi: 10.1016/j.autrev.2012.08.019. Epub 2012 Sep 18
[PubMed PMID: 23000206]
[3]
Millard TP, Kondeatis E, Cox A, Wilson AG, Grabczynska SA, Carey BS, Lewis CM, Khamashta MA, Duff GW, Hughes GR, Hawk JL, Vaughan RW, McGregor JM. A candidate gene analysis of three related photosensitivity disorders: cutaneous lupus erythematosus, polymorphic light eruption and actinic prurigo. The British journal of dermatology. 2001 Aug:145(2):229-36
[PubMed PMID: 11531784]
[4]
Sontheimer RD, Stastny P, Gilliam JN. Human histocompatibility antigen associations in subacute cutaneous lupus erythematosus. The Journal of clinical investigation. 1981 Jan:67(1):312-6
[PubMed PMID: 7451656]
[5]
Watson RM, Talwar P, Alexander E, Bias WB, Provost TT. Subacute cutaneous lupus erythematosus-immunogenetic associations. Journal of autoimmunity. 1991 Feb:4(1):73-85
[PubMed PMID: 2031665]
[6]
Callen JP, Hodge SJ, Kulick KB, Stelzer G, Buchino JJ. Subacute cutaneous lupus erythematosus in multiple members of a family with C2 deficiency. Archives of dermatology. 1987 Jan:123(1):66-70
[PubMed PMID: 3467658]
[7]
Bennion SD, Norris DA. Ultraviolet light modulation of autoantigens, epidermal cytokines and adhesion molecules as contributing factors of the pathogenesis of cutaneous LE. Lupus. 1997:6(2):181-92
[PubMed PMID: 9061667]
[8]
Robinson ES, Werth VP. The role of cytokines in the pathogenesis of cutaneous lupus erythematosus. Cytokine. 2015 Jun:73(2):326-34. doi: 10.1016/j.cyto.2015.01.031. Epub 2015 Mar 9
[PubMed PMID: 25767072]
[9]
Norris DA, Lee LA. Pathogenesis of cutaneous lupus erythematosus. Clinics in dermatology. 1985 Jul-Sep:3(3):20-35
[PubMed PMID: 2463862]
[10]
Norris DA, Lee LA. Antibody-dependent cellular cytotoxicity and skin disease. The Journal of investigative dermatology. 1985 Jul:85(1 Suppl):165s-175s
[PubMed PMID: 3874244]
[11]
Furukawa F, Kashihara-Sawami M, Lyons MB, Norris DA. Binding of antibodies to the extractable nuclear antigens SS-A/Ro and SS-B/La is induced on the surface of human keratinocytes by ultraviolet light (UVL): implications for the pathogenesis of photosensitive cutaneous lupus. The Journal of investigative dermatology. 1990 Jan:94(1):77-85
[PubMed PMID: 2132545]
[12]
Bangert JL, Freeman RG, Sontheimer RD, Gilliam JN. Subacute cutaneous lupus erythematosus and discoid lupus erythematosus. Comparative histopathologic findings. Archives of dermatology. 1984 Mar:120(3):332-7
[PubMed PMID: 6703733]
Level 2 (mid-level) evidence
[13]
Lee LA, Roberts CM, Frank MB, McCubbin VR, Reichlin M. The autoantibody response to Ro/SSA in cutaneous lupus erythematosus. Archives of dermatology. 1994 Oct:130(10):1262-8
[PubMed PMID: 7944507]
[14]
Nutan F, Ortega-Loayza AG. Cutaneous Lupus: A Brief Review of Old and New Medical Therapeutic Options. The journal of investigative dermatology. Symposium proceedings. 2017 Oct:18(2):S64-S68. doi: 10.1016/j.jisp.2017.02.001. Epub
[PubMed PMID: 28941497]
[15]
Ortiz A, Grando SA. Smoking and the skin. International journal of dermatology. 2012 Mar:51(3):250-62. doi: 10.1111/j.1365-4632.2011.05205.x. Epub
[PubMed PMID: 22348557]
[16]
Miyawaki S, Nishiyama S, Aita T, Yoshinaga Y. The effect of methotrexate on improving serological abnormalities of patients with systemic lupus erythematosus. Modern rheumatology. 2013 Jul:23(4):659-66. doi: 10.1007/s10165-012-0707-9. Epub 2012 Jul 19
[PubMed PMID: 23011357]
[17]
Manzi S, Sánchez-Guerrero J, Merrill JT, Furie R, Gladman D, Navarra SV, Ginzler EM, D'Cruz DP, Doria A, Cooper S, Zhong ZJ, Hough D, Freimuth W, Petri MA, BLISS-52 and BLISS-76 Study Groups. Effects of belimumab, a B lymphocyte stimulator-specific inhibitor, on disease activity across multiple organ domains in patients with systemic lupus erythematosus: combined results from two phase III trials. Annals of the rheumatic diseases. 2012 Nov:71(11):1833-8. doi: 10.1136/annrheumdis-2011-200831. Epub 2012 May 1
[PubMed PMID: 22550315]
[18]
Kieu V, O'Brien T, Yap LM, Baker C, Foley P, Mason G, Prince HM, McCormack C. Refractory subacute cutaneous lupus erythematosus successfully treated with rituximab. The Australasian journal of dermatology. 2009 Aug:50(3):202-6. doi: 10.1111/j.1440-0960.2009.00539.x. Epub
[PubMed PMID: 19659984]
[19]
Goodfield M, Davison K, Bowden K. Intravenous immunoglobulin (IVIg) for therapy-resistant cutaneous lupus erythematosus (LE). The Journal of dermatological treatment. 2004 Jan:15(1):46-50
[PubMed PMID: 14754650]
[20]
Ziemer M, Milkova L, Kunz M. Lupus erythematosus. Part II: clinical picture, diagnosis and treatment. Journal der Deutschen Dermatologischen Gesellschaft = Journal of the German Society of Dermatology : JDDG. 2014 Apr:12(4):285-301; quiz 302. doi: 10.1111/ddg.12254. Epub 2014 Jan 15
[PubMed PMID: 24423191]
[21]
Cohen MR, Crosby D. Systemic disease in subacute cutaneous lupus erythematosus: a controlled comparison with systemic lupus erythematosus. The Journal of rheumatology. 1994 Sep:21(9):1665-9
[PubMed PMID: 7799346]
[22]
Tiao J, Feng R, Carr K, Okawa J, Werth VP. Using the American College of Rheumatology (ACR) and Systemic Lupus International Collaborating Clinics (SLICC) criteria to determine the diagnosis of systemic lupus erythematosus (SLE) in patients with subacute cutaneous lupus erythematosus (SCLE). Journal of the American Academy of Dermatology. 2016 May:74(5):862-9. doi: 10.1016/j.jaad.2015.12.029. Epub 2016 Feb 18
[PubMed PMID: 26897388]
[23]
Sontheimer RD. Subacute cutaneous lupus erythematosus. Clinics in dermatology. 1985 Jul-Sep:3(3):58-68
[PubMed PMID: 3880024]
[24]
Callen JP, Kulick KB, Stelzer G, Fowler JF. Subacute cutaneous lupus erythematosus. Clinical, serologic, and immunogenetic studies of forty-nine patients seen in a nonreferral setting. Journal of the American Academy of Dermatology. 1986 Dec:15(6):1227-37
[PubMed PMID: 3543071]