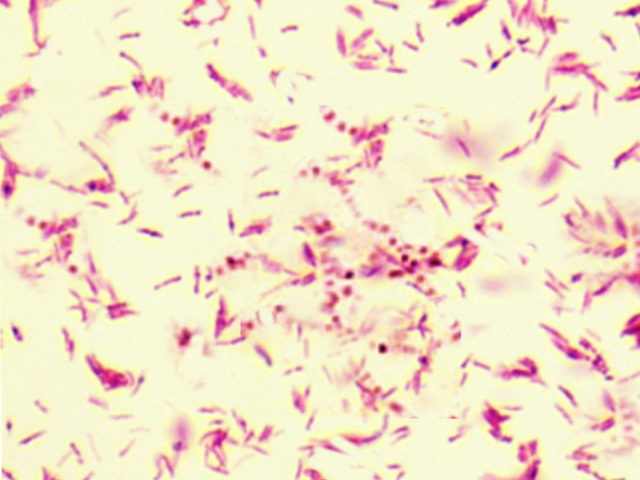
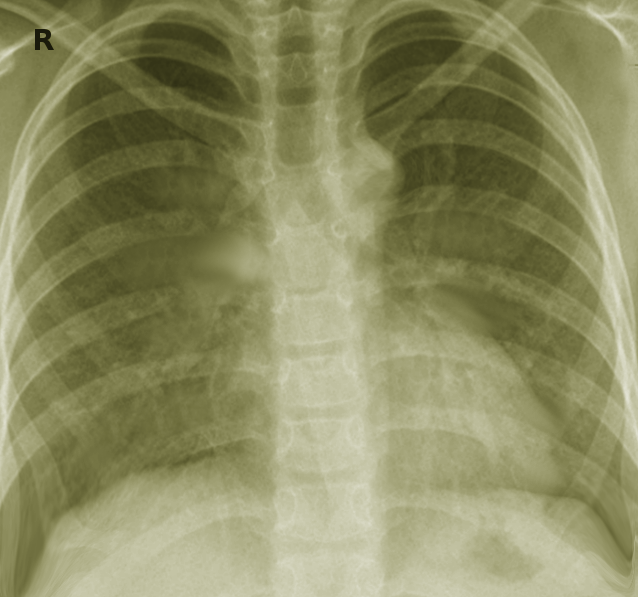
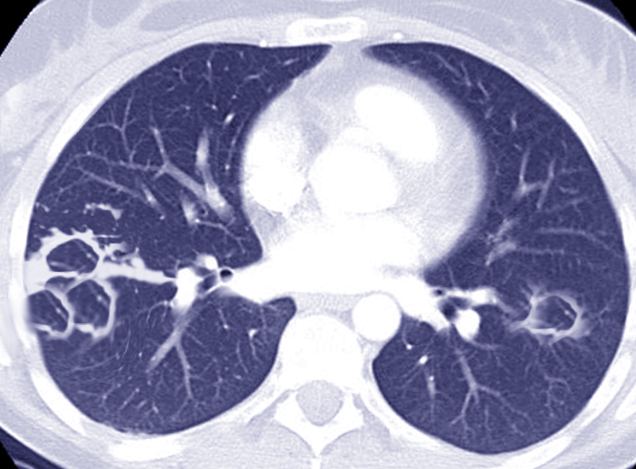
[1]
Pinner M. IV. Atypical Acid Fast Organisms: II. Some Observations on Filtration Experiments. Journal of bacteriology. 1933 Jun:25(6):576-9
[PubMed PMID: 16559638]
[2]
Kendall BA, Winthrop KL. Update on the epidemiology of pulmonary nontuberculous mycobacterial infections. Seminars in respiratory and critical care medicine. 2013 Feb:34(1):87-94. doi: 10.1055/s-0033-1333567. Epub 2013 Mar 4
[PubMed PMID: 23460008]
[3]
Prevots DR, Marras TK. Epidemiology of human pulmonary infection with nontuberculous mycobacteria: a review. Clinics in chest medicine. 2015 Mar:36(1):13-34. doi: 10.1016/j.ccm.2014.10.002. Epub 2014 Nov 6
[PubMed PMID: 25676516]
[4]
Kee SJ,Suh SP, Increasing Burden of Nontuberculous Mycobacteria in Korea. Journal of Korean medical science. 2017 Aug
[PubMed PMID: 28665053]
[5]
Griffith DE, Aksamit T, Brown-Elliott BA, Catanzaro A, Daley C, Gordin F, Holland SM, Horsburgh R, Huitt G, Iademarco MF, Iseman M, Olivier K, Ruoss S, von Reyn CF, Wallace RJ Jr, Winthrop K, ATS Mycobacterial Diseases Subcommittee, American Thoracic Society, Infectious Disease Society of America. An official ATS/IDSA statement: diagnosis, treatment, and prevention of nontuberculous mycobacterial diseases. American journal of respiratory and critical care medicine. 2007 Feb 15:175(4):367-416
[PubMed PMID: 17277290]
[6]
Haworth CS, Banks J, Capstick T, Fisher AJ, Gorsuch T, Laurenson IF, Leitch A, Loebinger MR, Milburn HJ, Nightingale M, Ormerod P, Shingadia D, Smith D, Whitehead N, Wilson R, Floto RA. British Thoracic Society guidelines for the management of non-tuberculous mycobacterial pulmonary disease (NTM-PD). Thorax. 2017 Nov:72(Suppl 2):ii1-ii64. doi: 10.1136/thoraxjnl-2017-210927. Epub
[PubMed PMID: 29054853]
[7]
Daley CL, Iaccarino JM, Lange C, Cambau E, Wallace RJ Jr, Andrejak C, Böttger EC, Brozek J, Griffith DE, Guglielmetti L, Huitt GA, Knight SL, Leitman P, Marras TK, Olivier KN, Santin M, Stout JE, Tortoli E, van Ingen J, Wagner D, Winthrop KL. Treatment of nontuberculous mycobacterial pulmonary disease: an official ATS/ERS/ESCMID/IDSA clinical practice guideline. The European respiratory journal. 2020 Jul:56(1):. doi: 10.1183/13993003.00535-2020. Epub 2020 Jul 7
[PubMed PMID: 32636299]
Level 1 (high-level) evidence
[8]
Cambau E, Drancourt M. Steps towards the discovery of Mycobacterium tuberculosis by Robert Koch, 1882. Clinical microbiology and infection : the official publication of the European Society of Clinical Microbiology and Infectious Diseases. 2014 Mar:20(3):196-201. doi: 10.1111/1469-0691.12555. Epub
[PubMed PMID: 24450600]
[9]
RUNYON EH. Anonymous mycobacteria in pulmonary disease. The Medical clinics of North America. 1959 Jan:43(1):273-90
[PubMed PMID: 13612432]
[10]
Grange JM, Yates MD, Boughton E. The avian tubercle bacillus and its relatives. The Journal of applied bacteriology. 1990 May:68(5):411-31
[PubMed PMID: 2196253]
[11]
Wolinsky E. Nontuberculous mycobacteria and associated diseases. The American review of respiratory disease. 1979 Jan:119(1):107-59
[PubMed PMID: 369415]
[12]
Runyon EH. Micobacterium intracellulare. The American review of respiratory disease. 1967 May:95(5):861-5
[PubMed PMID: 6023519]
[13]
Tortoli E. Microbiological features and clinical relevance of new species of the genus Mycobacterium. Clinical microbiology reviews. 2014 Oct:27(4):727-52. doi: 10.1128/CMR.00035-14. Epub
[PubMed PMID: 25278573]
[14]
Cook GM, Berney M, Gebhard S, Heinemann M, Cox RA, Danilchanka O, Niederweis M. Physiology of mycobacteria. Advances in microbial physiology. 2009:55():81-182, 318-9. doi: 10.1016/S0065-2911(09)05502-7. Epub
[PubMed PMID: 19573696]
Level 3 (low-level) evidence
[15]
Gangadharam PR. Microbiology of nontuberculosis mycobacteria. Seminars in respiratory infections. 1996 Dec:11(4):231-43
[PubMed PMID: 8976577]
[16]
Bittner MJ, Preheim LC. Other Slow-Growing Nontuberculous Mycobacteria. Microbiology spectrum. 2016 Nov:4(6):. doi: 10.1128/microbiolspec.TNMI7-0012-2016. Epub
[PubMed PMID: 27837745]
[17]
Kim CJ, Kim NH, Song KH, Choe PG, Kim ES, Park SW, Kim HB, Kim NJ, Kim EC, Park WB, Oh MD. Differentiating rapid- and slow-growing mycobacteria by difference in time to growth detection in liquid media. Diagnostic microbiology and infectious disease. 2013 Jan:75(1):73-6. doi: 10.1016/j.diagmicrobio.2012.09.019. Epub 2012 Oct 29
[PubMed PMID: 23114094]
[18]
Springer B, Stockman L, Teschner K, Roberts GD, Böttger EC. Two-laboratory collaborative study on identification of mycobacteria: molecular versus phenotypic methods. Journal of clinical microbiology. 1996 Feb:34(2):296-303
[PubMed PMID: 8789004]
[19]
Asaoka M, Hagiwara E, Etori S, Higa K, Ikeda S, Sekine A, Kitamura H, Baba T, Komatsu S, Ogura T. Identification and Characteristics of Co-isolation of Multiple Nontuberculous Mycobacteria. Internal medicine (Tokyo, Japan). 2021 Oct 15:60(20):3213-3219. doi: 10.2169/internalmedicine.5300-20. Epub 2021 Apr 26
[PubMed PMID: 33896860]
[20]
Jun HJ, Jeon K, Um SW, Kwon OJ, Lee NY, Koh WJ. Nontuberculous mycobacteria isolated during the treatment of pulmonary tuberculosis. Respiratory medicine. 2009 Dec:103(12):1936-40. doi: 10.1016/j.rmed.2009.05.025. Epub 2009 Jul 2
[PubMed PMID: 19576745]
[21]
Chaptal M, Andrejak C, Bonifay T, Beillard E, Guillot G, Guyomard-Rabenirina S, Demar M, Trombert-Paolantoni S, Jacomo V, Mosnier E, Veziris N, Djossou F, Epelboin L, French Guiana PNTM working group. Epidemiology of infection by pulmonary non-tuberculous mycobacteria in French Guiana 2008-2018. PLoS neglected tropical diseases. 2022 Sep:16(9):e0010693. doi: 10.1371/journal.pntd.0010693. Epub 2022 Sep 9
[PubMed PMID: 36084148]
[22]
Chindam A, Vengaldas S, Srigiri VR, Syed U, Kilaru H, Chenimilla NP, Kilaru SC, Patil E. Challenges of diagnosing and treating non-tuberculous mycobacterial pulmonary disease [NTM-PD]: A case series. Journal of clinical tuberculosis and other mycobacterial diseases. 2021 Dec:25():100271. doi: 10.1016/j.jctube.2021.100271. Epub 2021 Aug 30
[PubMed PMID: 34541338]
Level 2 (mid-level) evidence
[23]
Pravosud V, Mannino DM, Prieto D, Zhang Q, Choate R, Malanga E, Aksamit TR. Symptom Burden and Medication Use Among Patients with Nontuberculous Mycobacterial Lung Disease. Chronic obstructive pulmonary diseases (Miami, Fla.). 2021 Apr 27:8(2):243-254. doi: 10.15326/jcopdf.2020.0184. Epub
[PubMed PMID: 33610137]
[24]
Daley CL, Iaccarino JM, Lange C, Cambau E, Wallace RJ Jr, Andrejak C, Böttger EC, Brozek J, Griffith DE, Guglielmetti L, Huitt GA, Knight SL, Leitman P, Marras TK, Olivier KN, Santin M, Stout JE, Tortoli E, van Ingen J, Wagner D, Winthrop KL. Treatment of Nontuberculous Mycobacterial Pulmonary Disease: An Official ATS/ERS/ESCMID/IDSA Clinical Practice Guideline. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2020 Aug 14:71(4):e1-e36. doi: 10.1093/cid/ciaa241. Epub
[PubMed PMID: 32628747]
Level 1 (high-level) evidence
[25]
Schildkraut JA, Zweijpfenning SMH, Nap M, He K, Dacheva E, Overbeek J, Tostmann A, Wertheim HFL, Hoefsloot W, van Ingen J. The epidemiology of nontuberculous mycobacterial pulmonary disease in the Netherlands. ERJ open research. 2021 Jul:7(3):. pii: 00207-2021. doi: 10.1183/23120541.00207-2021. Epub 2021 Jul 12
[PubMed PMID: 34262970]
[26]
Thomson R, Donnan E, Konstantinos A. Notification of Nontuberculous Mycobacteria: An Australian Perspective. Annals of the American Thoracic Society. 2017 Mar:14(3):318-323. doi: 10.1513/AnnalsATS.201612-994OI. Epub
[PubMed PMID: 28118021]
Level 3 (low-level) evidence
[27]
Adjemian J, Frankland TB, Daida YG, Honda JR, Olivier KN, Zelazny A, Honda S, Prevots DR. Epidemiology of Nontuberculous Mycobacterial Lung Disease and Tuberculosis, Hawaii, USA. Emerging infectious diseases. 2017 Mar:23(3):439-447. doi: 10.3201/eid2303.161827. Epub
[PubMed PMID: 28221128]
[28]
Deutsch-Feldman M, Springer YP, Felix D, Tsang CA, Brostrom R, Haddad M. Tuberculosis Among Native Hawaiian and Other Pacific Islander Persons: United States and U.S.-Affiliated Pacific Islands, 2010-2019. Health equity. 2022:6(1):476-484. doi: 10.1089/heq.2022.0065. Epub 2022 Jun 27
[PubMed PMID: 35801148]
[29]
Lin C, Russell C, Soll B, Chow D, Bamrah S, Brostrom R, Kim W, Scott J, Bankowski MJ. Increasing Prevalence of Nontuberculous Mycobacteria in Respiratory Specimens from US-Affiliated Pacific Island Jurisdictions(1). Emerging infectious diseases. 2018 Mar:24(3):485-491. doi: 10.3201/eid2403.171301. Epub
[PubMed PMID: 29460734]
[30]
Hojo M, Iikura M, Hirano S, Sugiyama H, Kobayashi N, Kudo K. Increased risk of nontuberculous mycobacterial infection in asthmatic patients using long-term inhaled corticosteroid therapy. Respirology (Carlton, Vic.). 2012 Jan:17(1):185-90. doi: 10.1111/j.1440-1843.2011.02076.x. Epub
[PubMed PMID: 21995339]
[31]
O'Brien RJ, Geiter LJ, Snider DE Jr. The epidemiology of nontuberculous mycobacterial diseases in the United States. Results from a national survey. The American review of respiratory disease. 1987 May:135(5):1007-14
[PubMed PMID: 3579001]
Level 3 (low-level) evidence
[32]
Adjemian J, Olivier KN, Seitz AE, Holland SM, Prevots DR. Prevalence of nontuberculous mycobacterial lung disease in U.S. Medicare beneficiaries. American journal of respiratory and critical care medicine. 2012 Apr 15:185(8):881-6. doi: 10.1164/rccm.201111-2016OC. Epub 2012 Feb 3
[PubMed PMID: 22312016]
[33]
Shah NM, Davidson JA, Anderson LF, Lalor MK, Kim J, Thomas HL, Lipman M, Abubakar I. Pulmonary Mycobacterium avium-intracellulare is the main driver of the rise in non-tuberculous mycobacteria incidence in England, Wales and Northern Ireland, 2007-2012. BMC infectious diseases. 2016 May 6:16():195. doi: 10.1186/s12879-016-1521-3. Epub 2016 May 6
[PubMed PMID: 27154015]
[34]
Dohál M, Porvazník I, Krivošová M, Solovič I, Mokrý J. Epidemiology of non-tuberculous mycobacterial diseases in Slovakia during the years 2016-2021. Respiratory physiology & neurobiology. 2023 Aug:314():104090. doi: 10.1016/j.resp.2023.104090. Epub 2023 Jun 12
[PubMed PMID: 37315773]
[35]
Brode SK, Daley CL, Marras TK. The epidemiologic relationship between tuberculosis and non-tuberculous mycobacterial disease: a systematic review. The international journal of tuberculosis and lung disease : the official journal of the International Union against Tuberculosis and Lung Disease. 2014 Nov:18(11):1370-7. doi: 10.5588/ijtld.14.0120. Epub
[PubMed PMID: 25299873]
Level 1 (high-level) evidence
[36]
. Rapid increase of the incidence of lung disease due to Mycobacterium kansasii in Japan. Chest. 1983 Jun:83(6):890-2
[PubMed PMID: 6851691]
[37]
Thomson RM, Furuya-Kanamori L, Coffey C, Bell SC, Knibbs LD, Lau CL. Influence of climate variables on the rising incidence of nontuberculous mycobacterial (NTM) infections in Queensland, Australia 2001-2016. The Science of the total environment. 2020 Oct 20:740():139796. doi: 10.1016/j.scitotenv.2020.139796. Epub 2020 Jun 13
[PubMed PMID: 32563864]
[38]
Lee SJ, Ju S, You JW, Jeong YY, Lee JD, Kim HC, Choi H, Lee H, Oh YM, Ra SW. Trends in the Prevalence of Non-TB Mycobacterial Infection in Patients With Non-Cystic Fibrosis Bronchiectasis in South Korea, 2012-2016. Chest. 2021 Mar:159(3):959-962. doi: 10.1016/j.chest.2020.10.093. Epub 2020 Dec 7
[PubMed PMID: 33301746]
[39]
Thomson RM, NTM working group at Queensland TB Control Centre and Queensland Mycobacterial Reference Laboratory. Changing epidemiology of pulmonary nontuberculous mycobacteria infections. Emerging infectious diseases. 2010 Oct:16(10):1576-83. doi: 10.3201/eid1610.091201. Epub
[PubMed PMID: 20875283]
[40]
Park SC, Kang MJ, Han CH, Lee SM, Kim CJ, Lee JM, Kang YA. Prevalence, incidence, and mortality of nontuberculous mycobacterial infection in Korea: a nationwide population-based study. BMC pulmonary medicine. 2019 Aug 1:19(1):140. doi: 10.1186/s12890-019-0901-z. Epub 2019 Aug 1
[PubMed PMID: 31370826]
[41]
Falkinham JO 3rd. Nontuberculous mycobacteria from household plumbing of patients with nontuberculous mycobacteria disease. Emerging infectious diseases. 2011 Mar:17(3):419-24. doi: 10.3201/eid1703.101510. Epub
[PubMed PMID: 21392432]
[42]
Shen Y, Haig SJ, Prussin AJ 2nd, LiPuma JJ, Marr LC, Raskin L. Shower water contributes viable nontuberculous mycobacteria to indoor air. PNAS nexus. 2022 Nov:1(5):pgac145. doi: 10.1093/pnasnexus/pgac145. Epub 2022 Nov 10
[PubMed PMID: 36712351]
[43]
Dirac MA, Horan KL, Doody DR, Meschke JS, Park DR, Jackson LA, Weiss NS, Winthrop KL, Cangelosi GA. Environment or host?: A case-control study of risk factors for Mycobacterium avium complex lung disease. American journal of respiratory and critical care medicine. 2012 Oct 1:186(7):684-91. doi: 10.1164/rccm.201205-0825OC. Epub 2012 Aug 2
[PubMed PMID: 22859521]
Level 2 (mid-level) evidence
[44]
Prevots DR, Adjemian J, Fernandez AG, Knowles MR, Olivier KN. Environmental risks for nontuberculous mycobacteria. Individual exposures and climatic factors in the cystic fibrosis population. Annals of the American Thoracic Society. 2014 Sep:11(7):1032-8. doi: 10.1513/AnnalsATS.201404-184OC. Epub
[PubMed PMID: 25068620]
[45]
Kahana LM, Kay JM, Yakrus MA, Waserman S. Mycobacterium avium complex infection in an immunocompetent young adult related to hot tub exposure. Chest. 1997 Jan:111(1):242-5
[PubMed PMID: 8996025]
[46]
Cowman S, van Ingen J, Griffith DE, Loebinger MR. Non-tuberculous mycobacterial pulmonary disease. The European respiratory journal. 2019 Jul:54(1):. pii: 1900250. doi: 10.1183/13993003.00250-2019. Epub 2019 Jul 11
[PubMed PMID: 31221809]
[47]
Garcia B, Wilmskoetter J, Grady A, Mingora C, Dorman S, Flume P. Chest Computed Tomography Features of Nontuberculous Mycobacterial Pulmonary Disease Versus Asymptomatic Colonization: A Cross-sectional Cohort Study. Journal of thoracic imaging. 2022 May 1:37(3):140-145. doi: 10.1097/RTI.0000000000000610. Epub 2021 Jul 21
[PubMed PMID: 34292274]
Level 2 (mid-level) evidence
[48]
Fowler SJ, French J, Screaton NJ, Foweraker J, Condliffe A, Haworth CS, Exley AR, Bilton D. Nontuberculous mycobacteria in bronchiectasis: Prevalence and patient characteristics. The European respiratory journal. 2006 Dec:28(6):1204-10
[PubMed PMID: 16807259]
[49]
Bai X, Bai A, Honda JR, Eichstaedt C, Musheyev A, Feng Z, Huitt G, Harbeck R, Kosmider B, Sandhaus RA, Chan ED. Alpha-1-Antitrypsin Enhances Primary Human Macrophage Immunity Against Non-tuberculous Mycobacteria. Frontiers in immunology. 2019:10():1417. doi: 10.3389/fimmu.2019.01417. Epub 2019 Jun 26
[PubMed PMID: 31293581]
[50]
Wijers CD, Chmiel JF, Gaston BM. Bacterial infections in patients with primary ciliary dyskinesia: Comparison with cystic fibrosis. Chronic respiratory disease. 2017 Nov:14(4):392-406. doi: 10.1177/1479972317694621. Epub 2017 Mar 6
[PubMed PMID: 29081265]
[51]
Benmerzoug S, Marinho FV, Rose S, Mackowiak C, Gosset D, Sedda D, Poisson E, Uyttenhove C, Van Snick J, Jacobs M, Garcia I, Ryffel B, Quesniaux VFJ. GM-CSF targeted immunomodulation affects host response to M. tuberculosis infection. Scientific reports. 2018 Jun 5:8(1):8652. doi: 10.1038/s41598-018-26984-3. Epub 2018 Jun 5
[PubMed PMID: 29872095]
[52]
Lombardi A, Villa S, Castelli V, Bandera A, Gori A. T-Cell Exhaustion in Mycobacterium tuberculosis and Nontuberculous Mycobacteria Infection: Pathophysiology and Therapeutic Perspectives. Microorganisms. 2021 Nov 28:9(12):. doi: 10.3390/microorganisms9122460. Epub 2021 Nov 28
[PubMed PMID: 34946062]
Level 3 (low-level) evidence
[53]
McCarthy KD, Cain KP, Winthrop KL, Udomsantisuk N, Lan NT, Sar B, Kimerling ME, Kanara N, Lynen L, Monkongdee P, Tasaneeyapan T, Varma JK. Nontuberculous mycobacterial disease in patients with HIV in Southeast Asia. American journal of respiratory and critical care medicine. 2012 May 1:185(9):981-8. doi: 10.1164/rccm.201107-1327OC. Epub 2012 Feb 16
[PubMed PMID: 22345581]
[54]
Horsburgh CR Jr. Mycobacterium avium complex infection in the acquired immunodeficiency syndrome. The New England journal of medicine. 1991 May 9:324(19):1332-8
[PubMed PMID: 2017230]
[55]
Maglione PJ, Xu J, Chan J. B cells moderate inflammatory progression and enhance bacterial containment upon pulmonary challenge with Mycobacterium tuberculosis. Journal of immunology (Baltimore, Md. : 1950). 2007 Jun 1:178(11):7222-34
[PubMed PMID: 17513771]
[56]
Lutt JR, Pisculli ML, Weinblatt ME, Deodhar A, Winthrop KL. Severe nontuberculous mycobacterial infection in 2 patients receiving rituximab for refractory myositis. The Journal of rheumatology. 2008 Aug:35(8):1683-5
[PubMed PMID: 18671331]
[57]
Okumura M, Iwai K, Ogata H, Ueyama M, Kubota M, Aoki M, Kokuto H, Tadokoro E, Uchiyama T, Saotome M, Yoshiyama T, Yoshimori K, Yoshida N, Azuma A, Kudoh S. Clinical factors on cavitary and nodular bronchiectatic types in pulmonary Mycobacterium avium complex disease. Internal medicine (Tokyo, Japan). 2008:47(16):1465-72
[PubMed PMID: 18703856]
[58]
Chan ED, Iseman MD. Slender, older women appear to be more susceptible to nontuberculous mycobacterial lung disease. Gender medicine. 2010 Feb:7(1):5-18. doi: 10.1016/j.genm.2010.01.005. Epub
[PubMed PMID: 20189150]
[59]
Iseman MD, Buschman DL, Ackerson LM. Pectus excavatum and scoliosis. Thoracic anomalies associated with pulmonary disease caused by Mycobacterium avium complex. The American review of respiratory disease. 1991 Oct:144(4):914-6
[PubMed PMID: 1928970]
[60]
Dawrs SN, Kautz M, Chan ED, Honda JR. Mycobacterium abscessus and Gastroesophageal Reflux: An In Vitro Study. American journal of respiratory and critical care medicine. 2020 Aug 1:202(3):466-469. doi: 10.1164/rccm.202001-0011LE. Epub
[PubMed PMID: 32298605]
[61]
Koh WJ, Kwon OJ, Lee KS. Diagnosis and treatment of nontuberculous mycobacterial pulmonary diseases: a Korean perspective. Journal of Korean medical science. 2005 Dec:20(6):913-25
[PubMed PMID: 16361797]
Level 3 (low-level) evidence
[62]
Thomson R, Tolson C, Carter R, Coulter C, Huygens F, Hargreaves M. Isolation of nontuberculous mycobacteria (NTM) from household water and shower aerosols in patients with pulmonary disease caused by NTM. Journal of clinical microbiology. 2013 Sep:51(9):3006-11. doi: 10.1128/JCM.00899-13. Epub 2013 Jul 10
[PubMed PMID: 23843489]
[63]
Thomson RM, Carter R, Tolson C, Coulter C, Huygens F, Hargreaves M. Factors associated with the isolation of Nontuberculous mycobacteria (NTM) from a large municipal water system in Brisbane, Australia. BMC microbiology. 2013 Apr 22:13():89. doi: 10.1186/1471-2180-13-89. Epub 2013 Apr 22
[PubMed PMID: 23601969]
[64]
Pennington KM, Vu A, Challener D, Rivera CG, Shweta FNU, Zeuli JD, Temesgen Z. Approach to the diagnosis and treatment of non-tuberculous mycobacterial disease. Journal of clinical tuberculosis and other mycobacterial diseases. 2021 Aug:24():100244. doi: 10.1016/j.jctube.2021.100244. Epub 2021 May 8
[PubMed PMID: 34036184]
[65]
Pathak K, Hart S, Lande L. Nontuberculous Mycobacteria Lung Disease (NTM-LD): Current Recommendations on Diagnosis, Treatment, and Patient Management. International journal of general medicine. 2022:15():7619-7629. doi: 10.2147/IJGM.S272690. Epub 2022 Oct 1
[PubMed PMID: 36213301]
[66]
Kendall BA, Varley CD, Choi D, Cassidy PM, Hedberg K, Ware MA, Winthrop KL. Distinguishing tuberculosis from nontuberculous mycobacteria lung disease, Oregon, USA. Emerging infectious diseases. 2011 Mar:17(3):506-9. doi: 10.3201/eid1703.101164. Epub
[PubMed PMID: 21392445]
[67]
Gopalaswamy R, Shanmugam S, Mondal R, Subbian S. Of tuberculosis and non-tuberculous mycobacterial infections - a comparative analysis of epidemiology, diagnosis and treatment. Journal of biomedical science. 2020 Jun 17:27(1):74. doi: 10.1186/s12929-020-00667-6. Epub 2020 Jun 17
[PubMed PMID: 32552732]
Level 2 (mid-level) evidence
[68]
Musaddaq B, Cleverley JR. Diagnosis of non-tuberculous mycobacterial pulmonary disease (NTM-PD): modern challenges. The British journal of radiology. 2020 Feb 1:93(1106):20190768. doi: 10.1259/bjr.20190768. Epub 2019 Dec 11
[PubMed PMID: 31794241]
[69]
Parkar AP, Kandiah P. Differential Diagnosis of Cavitary Lung Lesions. Journal of the Belgian Society of Radiology. 2016 Nov 19:100(1):100. doi: 10.5334/jbr-btr.1202. Epub 2016 Nov 19
[PubMed PMID: 30151493]
[70]
Kartalija M, Ovrutsky AR, Bryan CL, Pott GB, Fantuzzi G, Thomas J, Strand MJ, Bai X, Ramamoorthy P, Rothman MS, Nagabhushanam V, McDermott M, Levin AR, Frazer-Abel A, Giclas PC, Korner J, Iseman MD, Shapiro L, Chan ED. Patients with nontuberculous mycobacterial lung disease exhibit unique body and immune phenotypes. American journal of respiratory and critical care medicine. 2013 Jan 15:187(2):197-205. doi: 10.1164/rccm.201206-1035OC. Epub 2012 Nov 9
[PubMed PMID: 23144328]
[71]
Chu H, Zhao L, Xiao H, Zhang Z, Zhang J, Gui T, Gong S, Xu L, Sun X. Prevalence of nontuberculous mycobacteria in patients with bronchiectasis: a meta-analysis. Archives of medical science : AMS. 2014 Aug 29:10(4):661-8. doi: 10.5114/aoms.2014.44857. Epub
[PubMed PMID: 25276148]
Level 1 (high-level) evidence
[72]
Pursner M, Haller JO, Berdon WE. Imaging features of Mycobacterium avium-intracellulare complex (MAC) in children with AIDS. Pediatric radiology. 2000 Jun:30(6):426-9
[PubMed PMID: 10876832]
[73]
Soetaert K, Subissi L, Ceyssens PJ, Vanfleteren B, Chantrenne M, Asikainen T, Duysburgh E, Mathys V. Strong increase of true and false positive mycobacterial cultures sent to the National Reference Centre in Belgium, 2007 to 2016. Euro surveillance : bulletin Europeen sur les maladies transmissibles = European communicable disease bulletin. 2019 Mar:24(11):. doi: 10.2807/1560-7917.ES.2019.24.11.1800205. Epub
[PubMed PMID: 30892180]
[74]
Kim N, Yi J, Chang CL. Recovery Rates of Non-Tuberculous Mycobacteria from Clinical Specimens Are Increasing in Korean Tertiary-Care Hospitals. Journal of Korean medical science. 2017 Aug:32(8):1263-1267. doi: 10.3346/jkms.2017.32.8.1263. Epub
[PubMed PMID: 28665061]
[75]
Somoskövi A, Hotaling JE, Fitzgerald M, O'Donnell D, Parsons LM, Salfinger M. Lessons from a proficiency testing event for acid-fast microscopy. Chest. 2001 Jul:120(1):250-7
[PubMed PMID: 11451846]
[76]
Sawatpanich A, Petsong S, Tumwasorn S, Rotcheewaphan S. Diagnostic performance of the Anyplex MTB/NTM real-time PCR in detection of Mycobacterium tuberculosis complex and nontuberculous mycobacteria from pulmonary and extrapulmonary specimens. Heliyon. 2022 Dec:8(12):e11935. doi: 10.1016/j.heliyon.2022.e11935. Epub 2022 Nov 26
[PubMed PMID: 36471833]
[77]
Alonso H, Samper S, Martín C, Otal I. Mapping IS6110 in high-copy number Mycobacterium tuberculosis strains shows specific insertion points in the Beijing genotype. BMC genomics. 2013 Jun 25:14():422. doi: 10.1186/1471-2164-14-422. Epub 2013 Jun 25
[PubMed PMID: 23800083]
[78]
Harboe M, Nagai S, Patarroyo ME, Torres ML, Ramirez C, Cruz N. Properties of proteins MPB64, MPB70, and MPB80 of Mycobacterium bovis BCG. Infection and immunity. 1986 Apr:52(1):293-302
[PubMed PMID: 3514457]
[79]
Alexander KJ, Furlong JL, Baron JL, Rihs JD, Stephenson D, Perry JD, Stout JE. Evaluation of a new culture medium for isolation of nontuberculous mycobacteria from environmental water samples. PloS one. 2021:16(3):e0247166. doi: 10.1371/journal.pone.0247166. Epub 2021 Mar 3
[PubMed PMID: 33657154]
[80]
Ryu YJ, Koh WJ, Daley CL. Diagnosis and Treatment of Nontuberculous Mycobacterial Lung Disease: Clinicians' Perspectives. Tuberculosis and respiratory diseases. 2016 Apr:79(2):74-84. doi: 10.4046/trd.2016.79.2.74. Epub 2016 Mar 31
[PubMed PMID: 27066084]
Level 3 (low-level) evidence
[81]
Caverly LJ, Carmody LA, Haig SJ, Kotlarz N, Kalikin LM, Raskin L, LiPuma JJ. Culture-Independent Identification of Nontuberculous Mycobacteria in Cystic Fibrosis Respiratory Samples. PloS one. 2016:11(4):e0153876. doi: 10.1371/journal.pone.0153876. Epub 2016 Apr 19
[PubMed PMID: 27093603]
[82]
Im Y, Hwang NY, Kim K, Kim H, Kwon OJ, Jhun BW. Impact of Time Between Diagnosis and Treatment for Nontuberculous Mycobacterial Pulmonary Disease on Culture Conversion and All-Cause Mortality. Chest. 2022 May:161(5):1192-1200. doi: 10.1016/j.chest.2021.10.048. Epub 2021 Nov 16
[PubMed PMID: 34793759]
[83]
Dohál M, Porvazník I, Solovič I, Mokrý J. Whole Genome Sequencing in the Management of Non-Tuberculous Mycobacterial Infections. Microorganisms. 2021 Oct 27:9(11):. doi: 10.3390/microorganisms9112237. Epub 2021 Oct 27
[PubMed PMID: 34835363]
[84]
Donnan EJ, Marais BJ, Coulter C, Waring J, Bastian I, Williamson DA, Sherry NL, Bond K, Sintchenko V, Meumann EM, Horan K, Cooley L, Denholm JT. The use of whole genome sequencing for tuberculosis public health activities in Australia: a joint statement of the National Tuberculosis Advisory Committee and Communicable Diseases Genomics Network. Communicable diseases intelligence (2018). 2023 Feb 28:47():. doi: 10.33321/cdi.2023.47.8. Epub 2023 Feb 28
[PubMed PMID: 36850064]
[85]
Rodriguez-Temporal D, Alcaide F, Mareković I, O'Connor JA, Gorton R, van Ingen J, Van den Bossche A, Héry-Arnaud G, Beauruelle C, Orth-Höller D, Palacios-Gutiérrez JJ, Tudó G, Bou G, Ceyssens PJ, Garrigó M, González-Martin J, Greub G, Hrabak J, Ingebretsen A, Mediavilla-Gradolph MC, Oviaño M, Palop B, Pranada AB, Quiroga L, Ruiz-Serrano MJ, Rodríguez-Sánchez B. Multicentre study on the reproducibility of MALDI-TOF MS for nontuberculous mycobacteria identification. Scientific reports. 2022 Jan 24:12(1):1237. doi: 10.1038/s41598-022-05315-7. Epub 2022 Jan 24
[PubMed PMID: 35075208]
[86]
Johnson TM, Byrd TF, Drummond WK, Childs-Kean LM, Mahoney MV, Pearson JC, Rivera CG. Contemporary Pharmacotherapies for Nontuberculosis Mycobacterial Infections: A Narrative Review. Infectious diseases and therapy. 2023 Feb:12(2):343-365. doi: 10.1007/s40121-022-00750-5. Epub 2023 Jan 7
[PubMed PMID: 36609820]
Level 3 (low-level) evidence
[87]
Kwon YS, Koh WJ. Diagnosis and Treatment of Nontuberculous Mycobacterial Lung Disease. Journal of Korean medical science. 2016 May:31(5):649-59. doi: 10.3346/jkms.2016.31.5.649. Epub 2016 Mar 22
[PubMed PMID: 27134484]
[88]
Richard M, Gutiérrez AV, Kremer L. Dissecting erm(41)-Mediated Macrolide-Inducible Resistance in Mycobacterium abscessus. Antimicrobial agents and chemotherapy. 2020 Jan 27:64(2):. doi: 10.1128/AAC.01879-19. Epub 2020 Jan 27
[PubMed PMID: 31791943]
[89]
Lyu J, Jang HJ, Song JW, Choi CM, Oh YM, Lee SD, Kim WS, Kim DS, Shim TS. Outcomes in patients with Mycobacterium abscessus pulmonary disease treated with long-term injectable drugs. Respiratory medicine. 2011 May:105(5):781-7. doi: 10.1016/j.rmed.2010.12.012. Epub 2011 Jan 5
[PubMed PMID: 21211956]
[90]
Gill LI, Dominic C, Tiberi S. Atypical mycobacterial infections - management and when to treat. Current opinion in pulmonary medicine. 2021 May 1:27(3):216-223. doi: 10.1097/MCP.0000000000000764. Epub
[PubMed PMID: 33560672]
Level 3 (low-level) evidence
[91]
Griffith DE, Eagle G, Thomson R, Aksamit TR, Hasegawa N, Morimoto K, Addrizzo-Harris DJ, O'Donnell AE, Marras TK, Flume PA, Loebinger MR, Morgan L, Codecasa LR, Hill AT, Ruoss SJ, Yim JJ, Ringshausen FC, Field SK, Philley JV, Wallace RJ Jr, van Ingen J, Coulter C, Nezamis J, Winthrop KL, CONVERT Study Group. Amikacin Liposome Inhalation Suspension for Treatment-Refractory Lung Disease Caused by Mycobacterium avium Complex (CONVERT). A Prospective, Open-Label, Randomized Study. American journal of respiratory and critical care medicine. 2018 Dec 15:198(12):1559-1569. doi: 10.1164/rccm.201807-1318OC. Epub
[PubMed PMID: 30216086]
Level 1 (high-level) evidence
[92]
Martiniano SL, Esther CR, Haworth CS, Kasperbauer SH, Zemanick ET, Caverly LJ. Challenging scenarios in nontuberculous mycobacterial infection in cystic fibrosis. Pediatric pulmonology. 2020 Feb:55(2):521-525. doi: 10.1002/ppul.24604. Epub 2019 Dec 10
[PubMed PMID: 31821718]
[93]
Ballarino GJ, Olivier KN, Claypool RJ, Holland SM, Prevots DR. Pulmonary nontuberculous mycobacterial infections: antibiotic treatment and associated costs. Respiratory medicine. 2009 Oct:103(10):1448-55. doi: 10.1016/j.rmed.2009.04.026. Epub 2009 May 21
[PubMed PMID: 19467851]
[94]
Jo KW, Kim S, Lee JY, Lee SD, Kim WS, Kim DS, Shim TS. Treatment outcomes of refractory MAC pulmonary disease treated with drugs with unclear efficacy. Journal of infection and chemotherapy : official journal of the Japan Society of Chemotherapy. 2014 Oct:20(10):602-6. doi: 10.1016/j.jiac.2014.05.010. Epub 2014 Jun 26
[PubMed PMID: 24981714]
[95]
Zhang Y, Hill AT. Amikacin liposome inhalation suspension as a treatment for patients with refractory mycobacterium avium complex lung infection. Expert review of respiratory medicine. 2021 Jun:15(6):737-744. doi: 10.1080/17476348.2021.1875821. Epub 2021 May 26
[PubMed PMID: 34039231]
[96]
Santin M, Dorca J, Alcaide F, Gonzalez L, Casas S, Lopez M, Guerra MR. Long-term relapses after 12-month treatment for Mycobacterium kansasii lung disease. The European respiratory journal. 2009 Jan:33(1):148-52. doi: 10.1183/09031936.00024008. Epub
[PubMed PMID: 19118226]
[97]
Lee MR,Sheng WH,Hung CC,Yu CJ,Lee LN,Hsueh PR, Mycobacterium abscessus Complex Infections in Humans. Emerging infectious diseases. 2015 Sep;
[PubMed PMID: 26295364]
[98]
Yang B, Jhun BW, Moon SM, Lee H, Park HY, Jeon K, Kim DH, Kim SY, Shin SJ, Daley CL, Koh WJ. Clofazimine-Containing Regimen for the Treatment of Mycobacterium abscessus Lung Disease. Antimicrobial agents and chemotherapy. 2017 Jun:61(6):. doi: 10.1128/AAC.02052-16. Epub 2017 May 24
[PubMed PMID: 28348153]
[99]
Winthrop KL, Ku JH, Marras TK, Griffith DE, Daley CL, Olivier KN, Aksamit TR, Varley CD, Mackey K, Prevots DR. The tolerability of linezolid in the treatment of nontuberculous mycobacterial disease. The European respiratory journal. 2015 Apr:45(4):1177-9. doi: 10.1183/09031936.00169114. Epub 2015 Jan 22
[PubMed PMID: 25614169]
[100]
Inoue T, Tsunoda A, Nishimoto E, Nishida K, Komatsubara Y, Onoe R, Saji J, Mineshita M. Successful use of linezolid for refractory Mycobacterium abcessus infection: A case report. Respiratory medicine case reports. 2018:23():43-45. doi: 10.1016/j.rmcr.2017.11.007. Epub 2017 Nov 28
[PubMed PMID: 29234594]
Level 3 (low-level) evidence
[101]
Siegel SAR, Griffith DE, Philley JV, Brown-Elliott BA, Brunton AE, Sullivan PE, Fuss C, Strnad L, Wallace RJ Jr, Winthrop KL. Open-Label Trial of Amikacin Liposome Inhalation Suspension in Mycobacterium abscessus Lung Disease. Chest. 2023 Oct:164(4):846-859. doi: 10.1016/j.chest.2023.05.036. Epub 2023 Jun 17
[PubMed PMID: 37419144]
[102]
Hansen MP, Scott AM, McCullough A, Thorning S, Aronson JK, Beller EM, Glasziou PP, Hoffmann TC, Clark J, Del Mar CB. Adverse events in people taking macrolide antibiotics versus placebo for any indication. The Cochrane database of systematic reviews. 2019 Jan 18:1(1):CD011825. doi: 10.1002/14651858.CD011825.pub2. Epub 2019 Jan 18
[PubMed PMID: 30656650]
Level 1 (high-level) evidence
[103]
Mosholder AD, Mathew J, Alexander JJ, Smith H, Nambiar S. Cardiovascular risks with azithromycin and other antibacterial drugs. The New England journal of medicine. 2013 May 2:368(18):1665-8. doi: 10.1056/NEJMp1302726. Epub
[PubMed PMID: 23635046]
[104]
Vasquez EM, Maddux MS, Sanchez J, Pollak R. Clinically significant hearing loss in renal allograft recipients treated with intravenous erythromycin. Archives of internal medicine. 1993 Apr 12:153(7):879-82
[PubMed PMID: 8466379]
[105]
Barbieri MA, Cicala G, Cutroneo PM, Mocciaro E, Sottosanti L, Freni F, Galletti F, Arcoraci V, Spina E. Ototoxic Adverse Drug Reactions: A Disproportionality Analysis Using the Italian Spontaneous Reporting Database. Frontiers in pharmacology. 2019:10():1161. doi: 10.3389/fphar.2019.01161. Epub 2019 Oct 8
[PubMed PMID: 31649536]
[106]
Nakajima A, Fukami T, Kobayashi Y, Watanabe A, Nakajima M, Yokoi T. Human arylacetamide deacetylase is responsible for deacetylation of rifamycins: rifampicin, rifabutin, and rifapentine. Biochemical pharmacology. 2011 Dec 1:82(11):1747-56. doi: 10.1016/j.bcp.2011.08.003. Epub 2011 Aug 12
[PubMed PMID: 21856291]
[107]
Huang YS, Chern HD, Su WJ, Wu JC, Chang SC, Chiang CH, Chang FY, Lee SD. Cytochrome P450 2E1 genotype and the susceptibility to antituberculosis drug-induced hepatitis. Hepatology (Baltimore, Md.). 2003 Apr:37(4):924-30
[PubMed PMID: 12668988]
[109]
Holdiness MR. A review of the Redman syndrome and rifampicin overdosage. Medical toxicology and adverse drug experience. 1989 Nov-Dec:4(6):444-51
[PubMed PMID: 2689837]
[110]
Kim JH, Moon JI, Kim JE, Choi GS, Park HS, Ye YM, Yim H. Cutaneous leukocytoclastic vasculitis due to anti-tuberculosis medications, rifampin and pyrazinamide. Allergy, asthma & immunology research. 2010 Jan:2(1):55-8. doi: 10.4168/aair.2010.2.1.55. Epub 2009 Dec 30
[PubMed PMID: 20224679]
[111]
Mehta YS, Jijina FF, Badakere SS, Pathare AV, Mohanty D. Rifampicin-induced immune thrombocytopenia. Tubercle and lung disease : the official journal of the International Union against Tuberculosis and Lung Disease. 1996 Dec:77(6):558-62
[PubMed PMID: 9039451]
[112]
Venkatesan K. Pharmacokinetic drug interactions with rifampicin. Clinical pharmacokinetics. 1992 Jan:22(1):47-65
[PubMed PMID: 1559307]
[113]
Tan S, Kasperbauer S. Nontuberculous Mycobacteria. Seminars in respiratory and critical care medicine. 2021 Aug:42(4):567-586. doi: 10.1055/s-0041-1730997. Epub 2021 Jul 14
[PubMed PMID: 34261181]
[114]
Yu JA, Pomerantz M, Bishop A, Weyant MJ, Mitchell JD. Lady Windermere revisited: treatment with thoracoscopic lobectomy/segmentectomy for right middle lobe and lingular bronchiectasis associated with non-tuberculous mycobacterial disease. European journal of cardio-thoracic surgery : official journal of the European Association for Cardio-thoracic Surgery. 2011 Sep:40(3):671-5. doi: 10.1016/j.ejcts.2010.12.028. Epub 2011 Feb 15
[PubMed PMID: 21324708]
[115]
Shiraishi Y, Katsuragi N, Kita H, Hyogotani A, Saito MH, Shimoda K. Adjuvant surgical treatment of nontuberculous mycobacterial lung disease. The Annals of thoracic surgery. 2013 Jul:96(1):287-91. doi: 10.1016/j.athoracsur.2013.03.008. Epub 2013 Apr 22
[PubMed PMID: 23618520]
[116]
Brode SK, Marchand-Austin A, Jamieson FB, Marras TK. Pulmonary versus Nonpulmonary Nontuberculous Mycobacteria, Ontario, Canada. Emerging infectious diseases. 2017 Nov:23(11):1898-1901. doi: 10.3201/eid2311.170959. Epub
[PubMed PMID: 29048292]
[117]
Carpenter JL. Klebsiella pulmonary infections: occurrence at one medical center and review. Reviews of infectious diseases. 1990 Jul-Aug:12(4):672-82
[PubMed PMID: 2201068]
[118]
Macfarlane J. An overview of community acquired pneumonia with lessons learned from the British Thoracic Society Study. Seminars in respiratory infections. 1994 Sep:9(3):153-65
[PubMed PMID: 7831537]
Level 3 (low-level) evidence
[119]
Thambidurai L, Prabhuradhan R, Singhvi P, Ilanchezhian S, Ramachandran R, Shankar H. Cryptococcal pneumonia: the great mimicker. BJR case reports. 2017:3(2):20150358. doi: 10.1259/bjrcr.20150358. Epub 2017 Jan 5
[PubMed PMID: 30363287]
Level 3 (low-level) evidence
[120]
Sobonya RE, Yanes J, Klotz SA. Cavitary pulmonary coccidioidomycosis: pathologic and clinical correlates of disease. Human pathology. 2014 Jan:45(1):153-9. doi: 10.1016/j.humpath.2013.08.014. Epub
[PubMed PMID: 24321524]
[121]
Goodwin RA Jr, Owens FT, Snell JD, Hubbard WW, Buchanan RD, Terry RT, Des Prez RM. Chronic pulmonary histoplasmosis. Medicine. 1976 Nov:55(6):413-52
[PubMed PMID: 792626]
[122]
Domej W, Hermann J, Krause R, Wehrschütz M, Maier A, Flögel E. [Lung cavities, mycetomas and hemoptysis]. Wiener medizinische Wochenschrift (1946). 2007:157(19-20):466-72
[PubMed PMID: 18030549]
[123]
Gadkowski LB, Stout JE. Cavitary pulmonary disease. Clinical microbiology reviews. 2008 Apr:21(2):305-33, table of contents. doi: 10.1128/CMR.00060-07. Epub
[PubMed PMID: 18400799]
[124]
Lee H, Myung W, Lee EM, Kim H, Jhun BW. Mortality and Prognostic Factors of Nontuberculous Mycobacterial Infection in Korea: A Population-based Comparative Study. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2021 May 18:72(10):e610-e619. doi: 10.1093/cid/ciaa1381. Epub
[PubMed PMID: 32926135]
Level 2 (mid-level) evidence
[125]
Mourad A, Baker AW, Stout JE. Reduction in Expected Survival Associated With Nontuberculous Mycobacterial Pulmonary Disease. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2021 May 18:72(10):e552-e557. doi: 10.1093/cid/ciaa1267. Epub
[PubMed PMID: 32856690]
[126]
Sin S, Han S, Lee YJ, Cho YJ, Park JS, Yoon HI, Lee CT, Lee JH. Prognosis of nontuberculous mycobacterial pulmonary disease according to the method of microbiologic diagnosis. Scientific reports. 2021 Apr 13:11(1):8036. doi: 10.1038/s41598-021-87197-9. Epub 2021 Apr 13
[PubMed PMID: 33850204]
[127]
Kotilainen H, Valtonen V, Tukiainen P, Poussa T, Eskola J, Järvinen A. Clinical findings in relation to mortality in non-tuberculous mycobacterial infections: patients with Mycobacterium avium complex have better survival than patients with other mycobacteria. European journal of clinical microbiology & infectious diseases : official publication of the European Society of Clinical Microbiology. 2015 Sep:34(9):1909-18. doi: 10.1007/s10096-015-2432-8. Epub 2015 Jul 9
[PubMed PMID: 26155783]
[128]
Graybill JR, Silva J Jr, Fraser DW, Lordon R, Rogers E. Disseminated mycobacteriosis due to Mycobacterium abcessus in two recipients of renal homografts. The American review of respiratory disease. 1974 Jan:109(1):4-10
[PubMed PMID: 4588091]
[129]
Jhun BW, Moon SM, Jeon K, Kwon OJ, Yoo H, Carriere KC, Huh HJ, Lee NY, Shin SJ, Daley CL, Koh WJ. Prognostic factors associated with long-term mortality in 1445 patients with nontuberculous mycobacterial pulmonary disease: a 15-year follow-up study. The European respiratory journal. 2020 Jan:55(1):. pii: 1900798. doi: 10.1183/13993003.00798-2019. Epub 2020 Jan 2
[PubMed PMID: 31619468]
[130]
Knoll BM, Kappagoda S, Gill RR, Goldberg HJ, Boyle K, Baden LR, Fuhlbrigge AL, Marty FM. Non-tuberculous mycobacterial infection among lung transplant recipients: a 15-year cohort study. Transplant infectious disease : an official journal of the Transplantation Society. 2012 Oct:14(5):452-60. doi: 10.1111/j.1399-3062.2012.00753.x. Epub 2012 Jun 8
[PubMed PMID: 22676720]
[131]
Bachar K, Shulimzon T, Segel MJ. Nontuberculous mycobacteria infections of the pleura: A systematic review. Respiratory medicine. 2022 Dec:205():107036. doi: 10.1016/j.rmed.2022.107036. Epub 2022 Nov 1
[PubMed PMID: 36335889]
Level 1 (high-level) evidence
[132]
Khoor A, Leslie KO, Tazelaar HD, Helmers RA, Colby TV. Diffuse pulmonary disease caused by nontuberculous mycobacteria in immunocompetent people (hot tub lung). American journal of clinical pathology. 2001 May:115(5):755-62
[PubMed PMID: 11345841]
[133]
Kuroda F, Tanabe N, Igari H, Sakurai T, Sakao S, Tada Y, Kasahara Y, Tatsumi K. Nontuberculous mycobacterium diseases and chronic thromboembolic pulmonary hypertension. Internal medicine (Tokyo, Japan). 2014:53(20):2273-9
[PubMed PMID: 25318788]
[134]
Shah SK, McAnally KJ, Seoane L, Lombard GA, LaPlace SG, Lick S, Dhillon GS, Valentine VG. Analysis of pulmonary non-tuberculous mycobacterial infections after lung transplantation. Transplant infectious disease : an official journal of the Transplantation Society. 2016 Aug:18(4):585-91. doi: 10.1111/tid.12546. Epub 2016 Jul 2
[PubMed PMID: 27368989]
[135]
Olivier KN, Yankaskas JR, Knowles MR. Nontuberculous mycobacterial pulmonary disease in cystic fibrosis. Seminars in respiratory infections. 1996 Dec:11(4):272-84
[PubMed PMID: 8976581]
[136]
Piersimoni C, Scarparo C. Extrapulmonary infections associated with nontuberculous mycobacteria in immunocompetent persons. Emerging infectious diseases. 2009 Sep:15(9):1351-8; quiz 1544. doi: 10.3201/eid1509.081259. Epub
[PubMed PMID: 19788801]
[137]
Zenone T, Boibieux A, Tigaud S, Fredenucci JF, Vincent V, Chidiac C, Peyramond D. Non-tuberculous mycobacterial tenosynovitis: a review. Scandinavian journal of infectious diseases. 1999:31(3):221-8
[PubMed PMID: 10482048]
[138]
Gascón P, Sathe SS, Rameshwar P. Impaired erythropoiesis in the acquired immunodeficiency syndrome with disseminated Mycobacterium avium complex. The American journal of medicine. 1993 Jan:94(1):41-8
[PubMed PMID: 8093587]
[139]
Havlik JA Jr, Horsburgh CR Jr, Metchock B, Williams PP, Fann SA, Thompson SE 3rd. Disseminated Mycobacterium avium complex infection: clinical identification and epidemiologic trends. The Journal of infectious diseases. 1992 Mar:165(3):577-80
[PubMed PMID: 1347060]
[140]
Davies PD. The role of DOTS in tuberculosis treatment and control. American journal of respiratory medicine : drugs, devices, and other interventions. 2003:2(3):203-9
[PubMed PMID: 14720002]
[141]
Courtney R, Ballard E, Fauver S, Gariota M, Holland L. The partnership model: working with individuals, families, and communities toward a new vision of health. Public health nursing (Boston, Mass.). 1996 Jun:13(3):177-86
[PubMed PMID: 8677233]