Continuing Education Activity
The fibrous pericardium is the outer layer and holds the heart in place and protect it from surrounding infections. It is composed of thick connective tissue. The serous pericardium has two layers, the visceral and parietal layers. The visceral pericardium helps to minimize friction, and the parietal pericardium provides additional defense. This activity reviews the cause, presentation and pathophysiology of fibrinous pericarditis and highlights the role of the interprofessional team in its management.
Objectives:
- Review the causes of fibrinous pericarditis.
- Describe the physical signs of fibrinous pericarditis.
- Summarize the treatment of fibrinous pericarditis.
- Outline modalities to improve care coordination among interprofessional team members in order to improve outcomes for patients affected by fibrinous pericarditis.
Introduction
The pericardium is a fluid-filled doubled-walled membrane sac that surrounds the heart. The fluid is separated by two layers, the fibrous and serous pericardium.[1] The fibrous pericardium is the outer layer and holds the heart in place and protect it from surrounding infections.[1] It is composed of thick connective tissue. The serous pericardium has two layers, the visceral and parietal layers. The visceral pericardium helps to minimize friction, and the parietal pericardium provides additional defense.[1]
The pericardium can develop an inflammatory process for a variety of reasons and is morphologically classified as follows:
- Fibrinous
- Purulent
- Serous
- Caseous
- Hemorrhagic
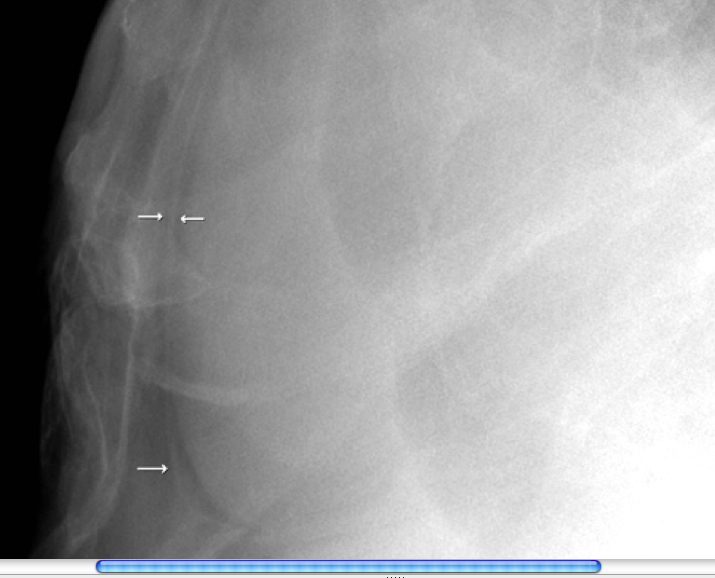
The normal pericardium is shiny, glistening, and smooth. In fibrinous pericarditis, the pericardial texture is rough, granular, and has many fibrous adhesions. Fibrinous pericarditis is usually caused by trauma, surgery, acute myocardial infarction, uremia, collagen vascular disorders, and malignancies.
Etiology
Injury to the pericardium leads to the release of inflammatory cells, fibrin, and fluid. Most commonly, acute pericarditis is idiopathic, and in 90% of the cases, an etiology remains undetermined. The remaining 10% are secondary to bacterial and viral infections, autoimmune disease, uremia, myocardial infarction, cardiac surgery, malignancy, trauma, and radiation.[2]
Malignancies associated with fibrinous pericarditis include lung and breast cancer, lymphomas, leukemia, and metastatic disease. Tuberculosis is one of the common causes of pericarditis in countries where tuberculosis is endemic. Several autoimmune and inflammatory conditions like lupus, scleroderma, rheumatoid arthritis, and mixed connective tissue disorder are known to cause pericarditis.
Epidemiology
The incidence and prevalence of acute pericarditis are unknown, and many cases are considered to go undiagnosed. One study showed that acute pericarditis might account for up to 5% of presentation to the emergency department and account for 0.1% of admissions for chest pain.[1] Typically, the demographic affected are men aged between 20 to 50 years old.[2]
Pathophysiology
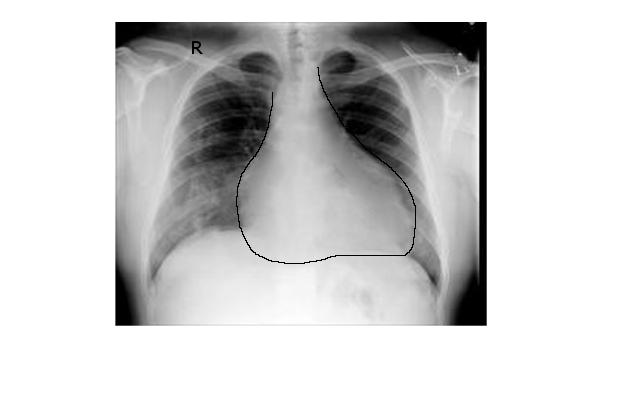
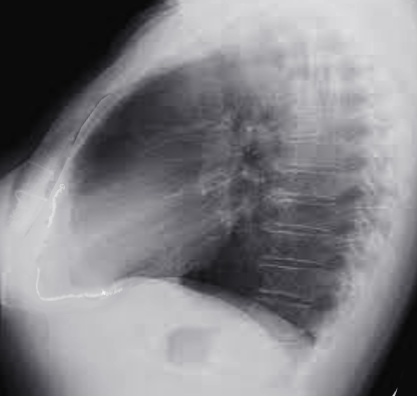
The normal pericardium sac contains anywhere from 20-50 ml of plasma. A sudden increase in the pericardial fluid can increase pressure on the right atria and right ventricles, which can compromise right atrial filling, reduce stroke volume, and diminish cardiac output. It is not the volume of pericardial fluid that is important but the speed of accumulation. With gradual accumulation of fluid, the pericardium has time to stretch and accommodate the increase in fluid.
Pericardial effusion may impede diastolic filling of the right heart if it accumulates too quickly, and it may also result in constriction of the heart if the accumulation is persistent.[3] This fluid accumulation may result in pericardial tamponade.
Inflammation of the pericardium produces a serous or purulent discharge. For example, in viral pericarditis, the fluid is serous. Purulent discharge can present in neoplastic or tuberculous associated episodes of pericarditis.[4][5] In most cases of acute pericarditis, the inflammatory exudate and influx of neutrophils result in a fibrinous reaction with adhesions and fluid accumulation.
Histopathology
The pericardium is a fibro-serous membrane that covers the heart. The fibrous pericardium is composed of connective tissue cells, microvasculature, collagen fibers and, small elastic fibers. The serosal layer is composed of a surface layer of flattened mesothelial cells. The luminal surface of the serosal layer is lined with surface microvilli and few cilia to help increase the area for fluid transport.[6]
History and Physical
Acute retrosternal chest pain that is sharp and pleuritic presents in over 95% of cases of acute pericarditis.[7] The chest pain is classically pleuritic and worsened by coughing, breathing, and sitting up-right.[7] Chest pain is often relieved by leaning forward. [7]
The most important physical sign in pericarditis is the pericardial friction rub.[8] The friction rub is best heard while the patient is upright and leaning forward. It is a high pitched, scratching sound best heard at the left lower sternal border during expiration.[8] The pericardial friction rub has three distinct components that correspond to the cardiac cycle. These components are heard during atrial systole, ventricular systole, and rapid ventricular filling during early diastole. It can be best heard with the patient sitting or leaning forward. [1]
Other critical clinical signs to be aware of are signs of tamponade such as raised jugular venous pressure (JVP), muffled heart sounds, and decreased blood pressure. If pericardial tamponade is concerned, checking for a pulsus paradoxus is recommended. It is defined as a drop of systolic blood pressure by more than 10 mm hg during inspiration.
Evaluation
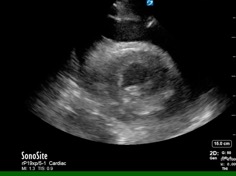
Diagnosis requires two of the following criteria.[9] Pericardial friction rub; the characteristic sharp and pleuritic chest pain; electrocardiography changes; and a new or worsening pericardial effusion.[9] Additionally, electrocardiography, chest radiography, and laboratory studies should support the diagnosis. The white blood cell count, erythrocyte sedimentation rate, and serum CRP level usually are elevated.[10] Electrocardiographic changes occur in stages. Stage I shows diffuse, concave ST-segment elevation. Stage II shows ST-segment normalization, J point return to baseline, T wave amplitude begins to decrease, PR-segment depression begins to appear. Stage III is symmetric, diffuse T-wave inversions. Stage IV occurs when the T-wave inversions normalize or become permanent.[11] Echocardiography should be performed to assess for pericardial effusion and tamponade physiology in pericarditis patients with hypotension or signs of congestive heart failure in a timely manner.
Treatment / Management
The therapy for acute pericarditis should be targeted as much as possible to the underlying etiology. It is not necessary to hospitalize patients with acute pericarditis unless they have high-risk features such as leukocytosis, fever, acute trauma, large pericardial effusion, tamponade physiology, or an immunocompromised state.[1] There are no randomized controlled trials, but pericarditis is usually treated empirically. One study showed nonsteroidal anti-inflammatory drugs (NSAIDs), and colchicine worked well for patients with presumed viral or idiopathic pericarditis. It was recommended the therapy be continued for two weeks or until symptoms resolve.[12] Commonly used NSAIDs are ibuprofen, indomethacin, and aspirin. NSAIDs are known to cause gastrointestinal toxicity, particularly in high-risk patients. Concomitant treatment with proton pump inhibitors should be considered in patients with high risk for gastrointestinal toxicity.
Glucocorticoids should be used for the initial treatment of acute pericarditis only in patients with contraindications to NSAIDs or for specific indications such as systemic inflammatory diseases. Systemic glucocorticoids should be slowly tapered to reduce the risk of recurrence.
Pericardiocentesis is the management of choice for cardiac tamponade. If it re-occurs, a pericardial window can be performed. In constrictive pericarditis, the treatment of choice is pericardiectomy.[13]
Treatment of recurrent pericarditis is similar to acute pericarditis. First-line treatment is concomitant use of NSAIDs and colchicine. Systemic glucocorticoids and colchicine can be used in patients who are unable to tolerate NSAIDs. Alternative treatment options including Azathioprine, Biological agents, and Intravenous immunoglobulins should be considered in cases of refractory pericarditis. [14]
Differential Diagnosis
Pericarditis may be confused with myocardial infarction, esophageal disorders, or other non-cardiac chest pain. It is therefore essential to use the diagnostic criteria and rule out other life-threatening causes of chest pain. Respiratory conditions including pneumonia, pulmonary embolism, and pneumothorax can cause pleuritic chest pain and should be considered in the differential diagnosis of pericarditis.
Prognosis
Prognosis of acute pericarditis is very good in idiopathic or viral pericarditis. Most cases resolve with no reoccurrence. In purulent pericarditis, the underlying etiology and treatment will determine the prognosis.[7][15]
Complications
Three potential complications may occur from pericarditis. These complications arise from the compression of the heart. Cardiac tamponade occurs when the pericardial pressure is high enough to impede filling of the right heart.[16] It is one of the life-threatening complications which requires urgent intervention. Constrictive pericarditis occurs when scarring and loss of the elasticity lead to impairment of diastolic filling and effusive-constrictive pericarditis occurs when there is constrictive physiology with a coexisting pericardial effusion resulting in a mixed hemodynamic picture.[17][18]
Deterrence and Patient Education
Patients with chest pain that is suggestive of pericarditis should seek immediate medical care. Pericarditis can become a life-threatening condition and may lead to compression of the heart. Acute pericarditis usually can not be prevented, but it is treatable. Symptoms of fainting and rapid breathing are signs that pericarditis may be progressing.
Enhancing Healthcare Team Outcomes
Expert opinion is primarily used to treat pericarditis. NSAIDs and colchicine are considered effective treatment options. Currently recommended doses are aspirin 650 mg every 4 to 6 hours or ibuprofen 400 mg every 4 to 6 hours. One study named the Colchicine for Acute Pericarditis Trial (COPE), showed colchicine to be the first-line adjuvant drug for the treatment and prevention of pericarditis (Level of Evidence A).[19] Colchicine is thus recommended for patients with their index event of pericarditis. Patients with renal insufficiency, motility disorders, or bleeding disorders should avoid or be cautious with the use of colchicine. Currently, recommended doses for colchicine are 2 to 3 mg per day for at least 6 months to 1 year.[1]