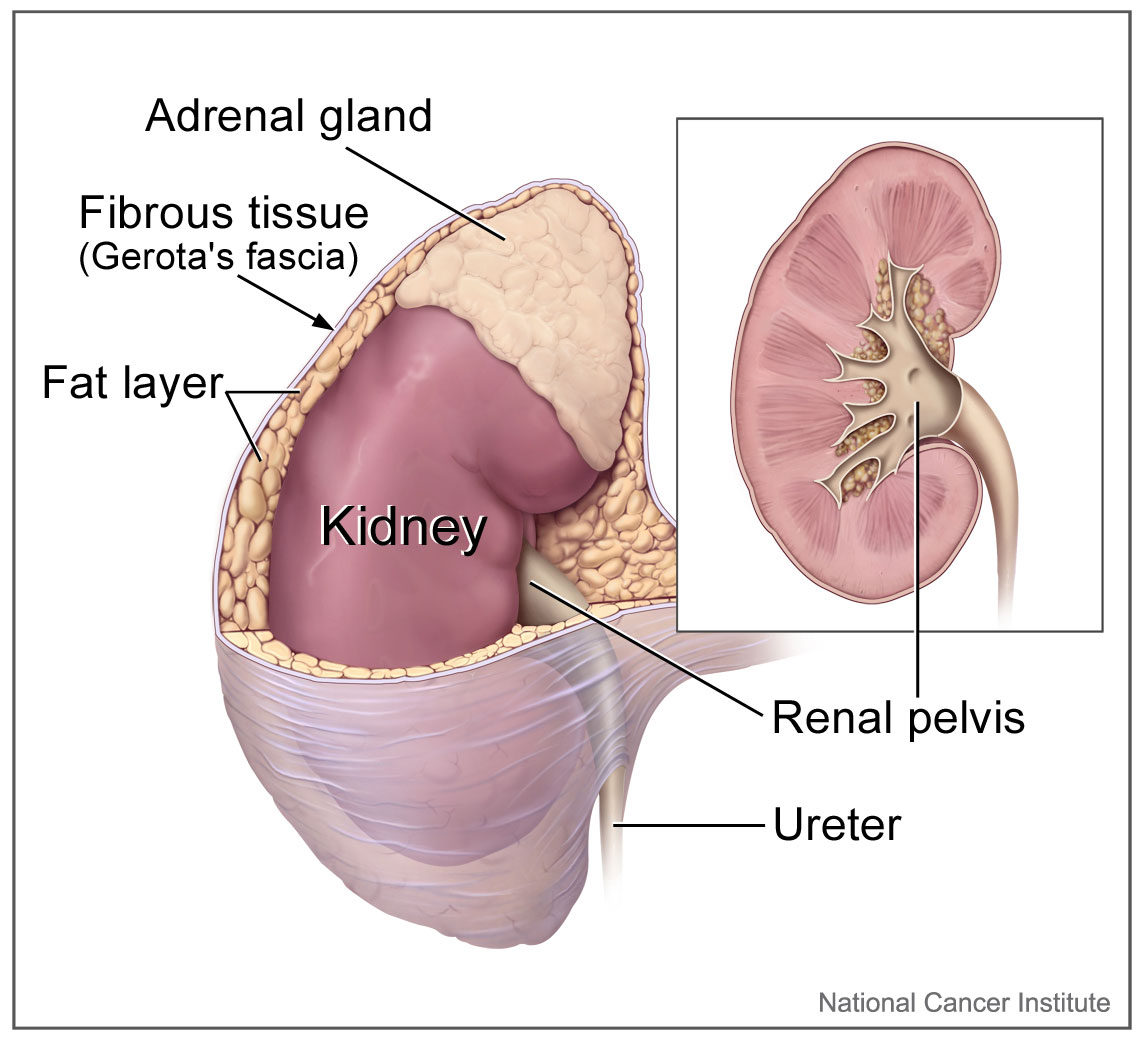
[1]
Feingold KR, Anawalt B, Blackman MR, Boyce A, Chrousos G, Corpas E, de Herder WW, Dhatariya K, Dungan K, Hofland J, Kalra S, Kaltsas G, Kapoor N, Koch C, Kopp P, Korbonits M, Kovacs CS, Kuohung W, Laferrère B, Levy M, McGee EA, McLachlan R, New M, Purnell J, Sahay R, Shah AS, Singer F, Sperling MA, Stratakis CA, Trence DL, Wilson DP, Kyritsi EM, Sertedaki A, Charmandari E, Chrousos GP. Familial or Sporadic Adrenal Hypoplasia Syndromes. Endotext. 2000:():
[PubMed PMID: 25905355]
[2]
Lotfi CFP, Kremer JL, Dos Santos Passaia B, Cavalcante IP. The human adrenal cortex: growth control and disorders. Clinics (Sao Paulo, Brazil). 2018 Sep 6:73(suppl 1):e473s. doi: 10.6061/clinics/2018/e473s. Epub 2018 Sep 6
[PubMed PMID: 30208164]
[3]
Aljabri KS, Bokhari SA, Assiri FY, Alshareef MA, Khan PM. The epidemiology of pituitary adenomas in a community-based hospital: a retrospective single center study in Saudi Arabia. Annals of Saudi medicine. 2016 Sep-Oct:36(5):341-345
[PubMed PMID: 27710986]
Level 2 (mid-level) evidence
[4]
Rojek A, Krawczynski MR, Jamsheer A, Sowinska-Seidler A, Iwaniszewska B, Malunowicz E, Niedziela M. X-Linked Adrenal Hypoplasia Congenita in a Boy due to a Novel Deletion of the Entire NR0B1 (DAX1) and MAGEB1-4 Genes. International journal of endocrinology. 2016:2016():5178953. doi: 10.1155/2016/5178953. Epub 2016 Aug 30
[PubMed PMID: 27656210]
[5]
Chung ST, Chi CH, Haymond MW, Jeha GS. Infantile Growth Hormone Deficiency and X- Linked Adrenal Hypoplasia Congenita. Jacobs journal of pediatrics. 2015 Nov:1(1):. pii: 003. Epub 2015 May 4
[PubMed PMID: 27110597]
[6]
Francke U, Harper JF, Darras BT, Cowan JM, McCabe ER, Kohlschütter A, Seltzer WK, Saito F, Goto J, Harpey JP. Congenital adrenal hypoplasia, myopathy, and glycerol kinase deficiency: molecular genetic evidence for deletions. American journal of human genetics. 1987 Mar:40(3):212-27
[PubMed PMID: 2883886]
[7]
Schwarz K, Thwaites R, Minford A, Day C, Butler G. Congenital adrenal hypoplasia presenting as a chronic respiratory condition. Archives of disease in childhood. 2003 Mar:88(3):261-2
[PubMed PMID: 12598398]
[8]
Feingold KR, Anawalt B, Blackman MR, Boyce A, Chrousos G, Corpas E, de Herder WW, Dhatariya K, Dungan K, Hofland J, Kalra S, Kaltsas G, Kapoor N, Koch C, Kopp P, Korbonits M, Kovacs CS, Kuohung W, Laferrère B, Levy M, McGee EA, McLachlan R, New M, Purnell J, Sahay R, Shah AS, Singer F, Sperling MA, Stratakis CA, Trence DL, Wilson DP, Asa SL. Pituitary Histopathology in Man: Normal and Abnormal. Endotext. 2000:():
[PubMed PMID: 25905234]
[9]
Kyriakakis N, Shonibare T, Kyaw-Tun J, Lynch J, Lagos CF, Achermann JC, Murray RD. Late-onset X-linked adrenal hypoplasia (DAX-1, NR0B1): two new adult-onset cases from a single center. Pituitary. 2017 Oct:20(5):585-593. doi: 10.1007/s11102-017-0822-x. Epub
[PubMed PMID: 28741070]
Level 3 (low-level) evidence
[10]
Shima H, Yatsuga S, Nakamura A, Sano S, Sasaki T, Katsumata N, Suzuki E, Hata K, Nakabayashi K, Momozawa Y, Kubo M, Okamura K, Kure S, Matsubara Y, Ogata T, Narumi S, Fukami M. NR0B1 Frameshift Mutation in a Boy with Idiopathic Central Precocious Puberty. Sexual development : genetics, molecular biology, evolution, endocrinology, embryology, and pathology of sex determination and differentiation. 2016:10(4):205-209
[PubMed PMID: 27648561]
[11]
Dineen R, Thompson CJ, Sherlock M. Adrenal crisis: prevention and management in adult patients. Therapeutic advances in endocrinology and metabolism. 2019:10():2042018819848218. doi: 10.1177/2042018819848218. Epub 2019 Jun 13
[PubMed PMID: 31223468]
Level 3 (low-level) evidence
[12]
Loureiro M, Reis F, Robalo B, Pereira C, Sampaio L. Adrenal Hypoplasia Congenita: A Rare Cause of Primary Adrenal Insufficiency and Hypogonadotropic Hypogonadism. Pediatric reports. 2015 Sep 28:7(3):5936. doi: 10.4081/pr.2015.5936. Epub 2015 Sep 28
[PubMed PMID: 26500747]