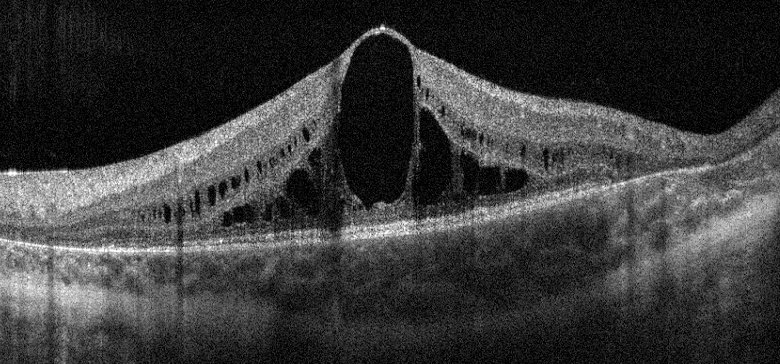
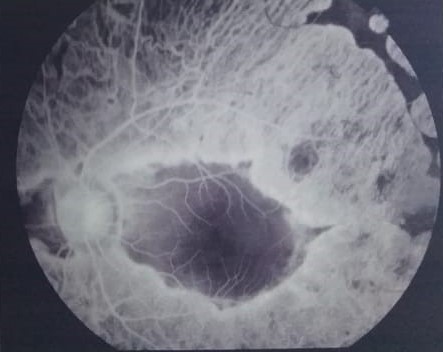
[1]
Takki K. Gyrate atrophy of the choroid and retina associated with hyperornithinaemia. The British journal of ophthalmology. 1974 Jan:58(1):3-23
[PubMed PMID: 4841281]
[2]
Simell O, Takki K. Raised plasma-ornithine and gyrate atrophy of the choroid and retina. Lancet (London, England). 1973 May 12:1(7811):1031-3
[PubMed PMID: 4122112]
[3]
Kaiser-Kupfer MI, Valle D, Del Valle LA. A specific enzyme defect in gyrate atrophy. American journal of ophthalmology. 1978 Feb:85(2):200-4
[PubMed PMID: 623190]
[4]
Michaud J, Brody LC, Steel G, Fontaine G, Martin LS, Valle D, Mitchell G. Strand-separating conformational polymorphism analysis: efficacy of detection of point mutations in the human ornithine delta-aminotransferase gene. Genomics. 1992 Jun:13(2):389-94
[PubMed PMID: 1612597]
[5]
Takki KK, Milton RC. The natural history of gyrate atrophy of the choroid and retina. Ophthalmology. 1981 Apr:88(4):292-301
[PubMed PMID: 7254775]
[6]
Elnahry AG, Hassan FK, Abdel-Kader AA. Bevacizumab for the treatment of intraretinal cystic spaces in a patient with gyrate atrophy of the choroid and retina. Ophthalmic genetics. 2018 Dec:39(6):759-762. doi: 10.1080/13816810.2018.1536220. Epub 2018 Oct 18
[PubMed PMID: 30335551]
[7]
Hayasaka S, Shiono T, Mizuno K, Sasayama C, Akiya S, Tanaka Y, Hayakawa M, Miyake Y, Ohba N. Gyrate atrophy of the choroid and retina: 15 Japanese patients. The British journal of ophthalmology. 1986 Aug:70(8):612-4
[PubMed PMID: 3741829]
[8]
Bargum R. Differential diagnosis of normoornithinaemic gyrate atrophy of the choroid and retina. Acta ophthalmologica. 1986 Aug:64(4):369-73
[PubMed PMID: 3776498]
[9]
Kaiser-Kupfer MI, Caruso RC, Valle D. Gyrate atrophy of the choroid and retina. Long-term reduction of ornithine slows retinal degeneration. Archives of ophthalmology (Chicago, Ill. : 1960). 1991 Nov:109(11):1539-48
[PubMed PMID: 1755734]
[10]
Weleber RG, Kennaway NG. Clinical trial of vitamin B6 for gyrate atrophy of the choroid and retina. Ophthalmology. 1981 Apr:88(4):316-24
[PubMed PMID: 6789268]
[11]
Kaiser-Kupfer MI, de Monasterio F, Valle D, Walser M, Brusilow S. Visual results of a long-term trial of a low-arginine diet in gyrate atrophy of choroid and retina. Ophthalmology. 1981 Apr:88(4):307-10
[PubMed PMID: 6973117]
[12]
Valle D, Kaiser-Kupfer MI, Del Valle LA. Gyrate atrophy of the choroid and retina: deficiency of ornithine aminotransferase in transformed lymphocytes. Proceedings of the National Academy of Sciences of the United States of America. 1977 Nov:74(11):5159-61
[PubMed PMID: 270753]
[13]
Berson EL. Nutrition and retinal degenerations. International ophthalmology clinics. 2000 Fall:40(4):93-111
[PubMed PMID: 11064860]
[14]
Brody LC, Mitchell GA, Obie C, Michaud J, Steel G, Fontaine G, Robert MF, Sipila I, Kaiser-Kupfer M, Valle D. Ornithine delta-aminotransferase mutations in gyrate atrophy. Allelic heterogeneity and functional consequences. The Journal of biological chemistry. 1992 Feb 15:267(5):3302-7
[PubMed PMID: 1737786]
[15]
Wang T, Steel G, Milam AH, Valle D. Correction of ornithine accumulation prevents retinal degeneration in a mouse model of gyrate atrophy of the choroid and retina. Proceedings of the National Academy of Sciences of the United States of America. 2000 Feb 1:97(3):1224-9
[PubMed PMID: 10655512]
[16]
Khan MY, Ibraheim AS, Firoozmand S. Gyrate atrophy of the choroid and retina with hyperornithinaemia, cystinuria and lysinuria. Eye (London, England). 1994:8 ( Pt 3)():284-7
[PubMed PMID: 7958031]
[17]
Valle D, Walser M, Brusilow S, Kaiser-Kupfer MI, Takki K. Gyrate atrophy of the choroid and retina. Biochemical considerations and experience with an arginine-restricted diet. Ophthalmology. 1981 Apr:88(4):325-30
[PubMed PMID: 7254778]
[18]
De Jonge WJ, Dingemanse MA, de Boer PA, Lamers WH, Moorman AF. Arginine-metabolizing enzymes in the developing rat small intestine. Pediatric research. 1998 Apr:43(4 Pt 1):442-51
[PubMed PMID: 9544996]
[19]
Whitcup SM, Iwata F, Podgor MJ, Valle D, Sran PK, Kaiser-Kupfer MI. Association of thyroid disease with retinitis pigmentosa and gyrate atrophy. American journal of ophthalmology. 1996 Dec:122(6):903-5
[PubMed PMID: 8956655]
[20]
Renner AB, Walter A, Fiebig BS, Jägle H. Gyrate atrophy: clinical and genetic findings in a female without arginine-restricted diet during her first 39 years of life and report of a new OAT gene mutation. Documenta ophthalmologica. Advances in ophthalmology. 2012 Aug:125(1):81-9. doi: 10.1007/s10633-012-9335-0. Epub 2012 Jun 7
[PubMed PMID: 22674428]
Level 3 (low-level) evidence
[21]
Jasani KM, Parry NRA, Black G, Kelly SP. Unique case of gyrate atrophy with a well-preserved electroretinogram (ERG). BMJ case reports. 2018 Feb 5:2018():. pii: bcr-2016-217556. doi: 10.1136/bcr-2016-217556. Epub 2018 Feb 5
[PubMed PMID: 29437727]
Level 3 (low-level) evidence
[22]
Tripathy K, Chawla R, Sharma YR, Gogia V. Ultrawide field fluorescein angiogram in a family with gyrate atrophy and foveoschisis. Oman journal of ophthalmology. 2016 May-Aug:9(2):104-6. doi: 10.4103/0974-620X.184529. Epub
[PubMed PMID: 27433038]
[23]
Huang J, Fu J, Fu S, Yang L, Nie K, Duan C, Cheng J, Li Y, Lv H, Chen R, Liu L, Fu J. Diagnostic value of a combination of next-generation sequencing, chorioretinal imaging and metabolic analysis: lessons from a consanguineous Chinese family with gyrate atrophy of the choroid and retina stemming from a novel OAT variant. The British journal of ophthalmology. 2019 Mar:103(3):428-435. doi: 10.1136/bjophthalmol-2018-312347. Epub 2018 Oct 26
[PubMed PMID: 30366948]
[24]
Moloney TP, O'Hagan S, Lee L. Ultrawide-field fundus photography of the first reported case of gyrate atrophy from Australia. Clinical ophthalmology (Auckland, N.Z.). 2014:8():1561-3. doi: 10.2147/OPTH.S64248. Epub 2014 Aug 20
[PubMed PMID: 25187693]
Level 3 (low-level) evidence
[25]
Angioï-Duprez K, Maalouf T, George JL. [Gyrate atrophy and craniopharyngioma: a case report]. Journal francais d'ophtalmologie. 2001 May:24(5):513-6
[PubMed PMID: 11397989]
Level 3 (low-level) evidence
[26]
Zhioua Braham I, Ammous I, Maalej R, Boukari M, Mili Boussen I, Errais K, Zhioua R. Multimodal imaging of foveoschisis and macular pseudohole associated with gyrate atrophy: a family report. BMC ophthalmology. 2018 Apr 12:18(1):89. doi: 10.1186/s12886-018-0755-9. Epub 2018 Apr 12
[PubMed PMID: 29649987]
[27]
Kim SJ, Lim DH, Kim JH, Kang SW. Gyrate atrophy of the choroid and retina diagnosed by ornithine-δ-aminotransferase gene analysis: a case report. Korean journal of ophthalmology : KJO. 2013 Oct:27(5):388-91. doi: 10.3341/kjo.2013.27.5.388. Epub 2013 Sep 10
[PubMed PMID: 24082780]
Level 3 (low-level) evidence
[28]
Vasconcelos-Santos DV, Magalhães EP, Nehemy MB. Macular edema associated with gyrate atrophy managed with intravitreal triamcinolone: a case report. Arquivos brasileiros de oftalmologia. 2007 Sep-Oct:70(5):858-61
[PubMed PMID: 18157315]
Level 3 (low-level) evidence
[29]
Shrestha SP, Arora R, Pradhan R, Bhatt S. First reported cases of gyrate atrophy of the choroid from Nepal. BMJ case reports. 2010 Nov 29:2010():. doi: 10.1136/bcr.04.2010.2951. Epub 2010 Nov 29
[PubMed PMID: 22798087]
Level 3 (low-level) evidence
[30]
Alparslan Ş, Fatih MT, Muhammed Ş, Adnan Y. Cystoid macular edema secondary to gyrate atrophy in a child treated with sub-tenon injection of triamcinolone acetonide. Romanian journal of ophthalmology. 2018 Jul-Sep:62(3):246-249
[PubMed PMID: 30505995]
[31]
Mäntyjärvi M, Tuppurainen K. Colour vision in gyrate atrophy. Vision research. 1998 Nov:38(21):3409-12
[PubMed PMID: 9893857]
[32]
Kaiser-Kupfer MI, Valle D, Bron AJ. Clinical and biochemical heterogeneity in gyrate atrophy. American journal of ophthalmology. 1980 Feb:89(2):219-22
[PubMed PMID: 7355975]
[33]
Wang T, Milam AH, Steel G, Valle D. A mouse model of gyrate atrophy of the choroid and retina. Early retinal pigment epithelium damage and progressive retinal degeneration. The Journal of clinical investigation. 1996 Jun 15:97(12):2753-62
[PubMed PMID: 8675686]
[34]
Ratzlaff K, Baich A. Comparison of ornithine aminotransferase activities in the pigment epithelium and retina of vertebrates. Comparative biochemistry and physiology. B, Comparative biochemistry. 1987:88(1):35-7
[PubMed PMID: 3677612]
Level 2 (mid-level) evidence
[35]
Rao GN, Cotlier E. Ornithine delta-aminotransferase activity in retina and other tissues. Neurochemical research. 1984 Apr:9(4):555-62
[PubMed PMID: 6462326]
[36]
Korte GE, Reppucci V, Henkind P. RPE destruction causes choriocapillary atrophy. Investigative ophthalmology & visual science. 1984 Oct:25(10):1135-45
[PubMed PMID: 6480292]
[37]
Valle D, Walser M, Brusilow SW, Kaiser-Kupfer M. Gyrate atrophy of the choroid and retina: amino acid metabolism and correction of hyperornithinemia with an arginine-deficient diet. The Journal of clinical investigation. 1980 Feb:65(2):371-8
[PubMed PMID: 7356686]
[38]
Valtonen M, Näntö-Salonen K, Jääskeläinen S, Heinänen K, Alanen A, Heinonen OJ, Lundbom N, Erkintalo M, Simell O. Central nervous system involvement in gyrate atrophy of the choroid and retina with hyperornithinaemia. Journal of inherited metabolic disease. 1999 Dec:22(8):855-66
[PubMed PMID: 10604138]
[39]
Sipilä I, Simell O, Rapola J, Sainio K, Tuuteri L. Gyrate atrophy of the choroid and retina with hyperornithinemia: tubular aggregates and type 2 fiber atrophy in muscle. Neurology. 1979 Jul:29(7):996-1005
[PubMed PMID: 572946]
[40]
Wilson DJ, Weleber RG, Green WR. Ocular clinicopathologic study of gyrate atrophy. American journal of ophthalmology. 1991 Jan 15:111(1):24-33
[PubMed PMID: 1985486]
[41]
Valle DL, Boison AP, Jezyk P, Aguirre G. Gyrate atrophy of the choroid and retina in a cat. Investigative ophthalmology & visual science. 1981 Feb:20(2):251-5
[PubMed PMID: 7461927]
[42]
Valtonen M, Näntö-Salonen K, Heinänen K, Alanen A, Kalimo H, Simell O. Skeletal muscle of patients with gyrate atrophy of the choroid and retina and hyperornithinaemia in ultralow-field magnetic resonance imaging and computed tomography. Journal of inherited metabolic disease. 1996:19(6):729-34
[PubMed PMID: 8982944]
[43]
Oliveira TL, Andrade RE, Muccioli C, Sallum J, Belfort R Jr. Cystoid macular edema in gyrate atrophy of the choroid and retina: a fluorescein angiography and optical coherence tomography evaluation. American journal of ophthalmology. 2005 Jul:140(1):147-9
[PubMed PMID: 16038665]
[44]
Parameswarappa DC, Agarwal K. Bilateral macular hole in gyrate atrophy: A rare association. Indian journal of ophthalmology. 2020 Apr:68(4):652. doi: 10.4103/ijo.IJO_984_19. Epub
[PubMed PMID: 32174595]
[45]
Inanc M, Tekin K, Teke MY. Bilateral choroidal neovascularization associated with gyrate atrophy managed with intravitreal bevacizumab. International ophthalmology. 2018 Jun:38(3):1351-1355. doi: 10.1007/s10792-017-0579-2. Epub 2017 May 30
[PubMed PMID: 28560651]
[46]
Peltola KE, Jääskeläinen S, Heinonen OJ, Falck B, Näntö-Salonen K, Heinänen K, Simell O. Peripheral nervous system in gyrate atrophy of the choroid and retina with hyperornithinemia. Neurology. 2002 Sep 10:59(5):735-40
[PubMed PMID: 12221166]
[47]
Kennaway NG, Stankova L, Wirtz MK, Weleber RG. Gyrate atrophy of the choroid and retina: characterization of mutant ornithine aminotransferase and mechanism of response to vitamin B6. American journal of human genetics. 1989 Mar:44(3):344-52
[PubMed PMID: 2916580]
[48]
Valayannopoulos V, Boddaert N, Mention K, Touati G, Barbier V, Chabli A, Sedel F, Kaplan J, Dufier JL, Seidenwurm D, Rabier D, Saudubray JM, de Lonlay P. Secondary creatine deficiency in ornithine delta-aminotransferase deficiency. Molecular genetics and metabolism. 2009 Jun:97(2):109-13. doi: 10.1016/j.ymgme.2008.12.010. Epub 2009 Mar 31
[PubMed PMID: 19345633]
[49]
Vannas-Sulonen K. Progression of gyrate atrophy of the choroid and retina. A long-term follow-up by fluorescein angiography. Acta ophthalmologica. 1987 Feb:65(1):101-9
[PubMed PMID: 3577698]
[50]
Chatziralli I, Theodossiadis G, Emfietzoglou I, Theodossiadis P. Intravitreal ranibizumab for choroidal neovascularization secondary to gyrate atrophy in a young patient: a multimodal imaging analysis. European journal of ophthalmology. 2015 Oct 21:25(6):e119-22. doi: 10.5301/ejo.5000660. Epub 2015 Oct 21
[PubMed PMID: 26419008]
[51]
Marano F, Deutman AF, Pinckers AJ, Aandekerk AL. Gyrate atrophy and choroidal neovascularization. Archives of ophthalmology (Chicago, Ill. : 1960). 1996 Oct:114(10):1295
[PubMed PMID: 8859105]
[52]
Salcedo-Villanueva G, Paciuc-Beja M, Villanueva-Mendoza C, Harasawa M, Smith JM, Velez-Montoya R, Olson JL, Oliver SC, Mandava N, Quiroz-Mercado H. Progression of gyrate atrophy measured with ultra-wide-field imaging. International ophthalmology. 2016 Feb:36(1):111-120. doi: 10.1007/s10792-015-0085-3. Epub 2015 May 26
[PubMed PMID: 26003990]
[53]
Raitta C, Carlson S, Vannas-Sulonen K. Gyrate atrophy of the choroid and retina: ERG of the neural retina and the pigment epithelium. The British journal of ophthalmology. 1990 Jun:74(6):363-7
[PubMed PMID: 2378845]
[54]
Weleber RG, Kennaway NG, Buist NR. Gyrate atrophy of the choroid and retina. Approaches to therapy. International ophthalmology. 1981 Aug:4(1-2):23-32
[PubMed PMID: 7028650]
[55]
Kaiser-Kupfer MI, Caruso RC, Valle D. Gyrate atrophy of the choroid and retina: further experience with long-term reduction of ornithine levels in children. Archives of ophthalmology (Chicago, Ill. : 1960). 2002 Feb:120(2):146-53
[PubMed PMID: 11831916]
[56]
Sergouniotis PI, Davidson AE, Lenassi E, Devery SR, Moore AT, Webster AR. Retinal structure, function, and molecular pathologic features in gyrate atrophy. Ophthalmology. 2012 Mar:119(3):596-605. doi: 10.1016/j.ophtha.2011.09.017. Epub 2011 Dec 17
[PubMed PMID: 22182799]
[57]
Mansour AM, Elnahry AG, Tripathy K, Foster RE, Mehanna CJ, Vishal R, Çavdarlı C, Arrigo A, Parodi MB. Analysis of optical coherence angiography in cystoid macular oedema associated with gyrate atrophy. Eye (London, England). 2021 Jun:35(6):1766-1774. doi: 10.1038/s41433-020-01166-6. Epub 2020 Sep 1
[PubMed PMID: 32873946]
[58]
Raval V, Kapoor A, Nayak S, Rao S, Das T. Optical Coherence Tomography Angiography and Macular Vessel Density Analysis of Cystoid Macular Edema in Gyrate Atrophy. Ophthalmic surgery, lasers & imaging retina. 2019 Jul 1:50(7):423-427. doi: 10.3928/23258160-20190703-03. Epub
[PubMed PMID: 31344241]
[59]
McInnes RR, Arshinoff SA, Bell L, McCulloch C. Treatment of gyrate atrophy of the choroid and retina with low arginine diet. Transactions of the American Ophthalmological Society. 1980:78():226-42
[PubMed PMID: 7257057]
[60]
Mashima YG, Weleber RG, Kennaway NG, Inana G. Genotype-phenotype correlation of a pyridoxine-responsive form of gyrate atrophy. Ophthalmic genetics. 1999 Dec:20(4):219-24
[PubMed PMID: 10617919]
[61]
Santinelli R, Costagliola C, Tolone C, D'Aloia A, D'Avanzo A, Prisco F, Perrone L, del Giudice EM. Low-protein diet and progression of retinal degeneration in gyrate atrophy of the choroid and retina: a twenty-six-year follow-up. Journal of inherited metabolic disease. 2004:27(2):187-96
[PubMed PMID: 15159649]
[62]
Vannas-Sulonen K, Simell O, Sipilä I. Gyrate atrophy of the choroid and retina. The ocular disease progresses in juvenile patients despite normal or near normal plasma ornithine concentration. Ophthalmology. 1987 Nov:94(11):1428-33
[PubMed PMID: 3684217]
[63]
Ros E. Nuts and CVD. The British journal of nutrition. 2015 Apr:113 Suppl 2():S111-20. doi: 10.1017/S0007114514003924. Epub
[PubMed PMID: 26148914]
[64]
Michaud J, Thompson GN, Brody LC, Steel G, Obie C, Fontaine G, Schappert K, Keith CG, Valle D, Mitchell GA. Pyridoxine-responsive gyrate atrophy of the choroid and retina: clinical and biochemical correlates of the mutation A226V. American journal of human genetics. 1995 Mar:56(3):616-22
[PubMed PMID: 7887415]
[65]
Hayasaka S, Saito T, Nakajima H, Takahashi O, Mizuno K, Tada K. Clinical trials of vitamin B6 and proline supplementation for gyrate atrophy of the choroid and retina. The British journal of ophthalmology. 1985 Apr:69(4):283-90
[PubMed PMID: 3922397]
[66]
Javadzadeh A, Gharabaghi D. Gyrate atrophy of the choroid and retina with hyper-ornithinemia responsive to vitamin B6: a case report. Journal of medical case reports. 2007 Jun 12:1():27
[PubMed PMID: 17565677]
Level 3 (low-level) evidence
[67]
Sipilä I, Rapola J, Simell O, Vannas A. Supplementary creatine as a treatment for gyrate atrophy of the choroid and retina. The New England journal of medicine. 1981 Apr 9:304(15):867-70
[PubMed PMID: 7207523]
[68]
Vannas-Sulonen K, Sipilä I, Vannas A, Simell O, Rapola J. Gyrate atrophy of the choroid and retina. A five-year follow-up of creatine supplementation. Ophthalmology. 1985 Dec:92(12):1719-27
[PubMed PMID: 4088625]
[69]
Casalino G, Pierro L, Manitto MP, Michaelides M, Bandello F. Resolution of cystoid macular edema following arginine-restricted diet and vitamin B6 supplementation in a case of gyrate atrophy. Journal of AAPOS : the official publication of the American Association for Pediatric Ophthalmology and Strabismus. 2018 Aug:22(4):321-323. doi: 10.1016/j.jaapos.2017.12.016. Epub 2018 Apr 12
[PubMed PMID: 29654911]
Level 3 (low-level) evidence
[70]
Çavdarlı C, Şahlı E, Çavdarlı B, Alp MN. Regression of macular edema with topical brinzolamide and nepafenac alone and identification of a novel gyrate atrophy mutation. Arquivos brasileiros de oftalmologia. 2020 Mar-Apr:83(2):149-152. doi: 10.5935/0004-2749.20200028. Epub
[PubMed PMID: 32159596]
[71]
Piozzi E, Alessi S, Santambrogio S, Cillino G, Mazza M, Iggui A, Cillino S. Carbonic Anhydrase Inhibitor with Topical NSAID Therapy to Manage Cystoid Macular Edema in a Case of Gyrate Atrophy. European journal of ophthalmology. 2017 Nov 8:27(6):e179-e183. doi: 10.5301/ejo.5001010. Epub
[PubMed PMID: 28708224]
Level 3 (low-level) evidence
[72]
Elnahry AG. Letter to the editor regarding: "cystoid macular edema secondary to gyrate atrophy in a child treated with sub-tenon injection of triamcinolone acetonide". Romanian journal of ophthalmology. 2018 Oct-Dec:62(4):317-318
[PubMed PMID: 30891531]
Level 3 (low-level) evidence
[73]
Abdelmassih Y, El-Khoury S, Cherfan CG. Dexamethasone implant for the treatment of gyrate atrophy associated macular edema. Journal francais d'ophtalmologie. 2019 Jan:42(1):e1-e4. doi: 10.1016/j.jfo.2018.03.029. Epub 2018 Dec 14
[PubMed PMID: 30559013]
[74]
Elnahry AG, Aboulfotouh MR, Nassar GA. Treatment of Intraretinal Cystic Spaces Associated With Gyrate Atrophy of the Choroid and Retina With Intravitreal Bevacizumab. Journal of pediatric ophthalmology and strabismus. 2020 Nov 1:57(6):400-406. doi: 10.3928/01913913-20200813-01. Epub
[PubMed PMID: 33211898]
[75]
Tsilou E, Rubin BI, Abraham FA, Kaiser-Kupfer M. Bilateral late posterior chamber intraocular lens dislocation with the capsular bag in a patient with gyrate atrophy. Journal of cataract and refractive surgery. 2004 Jul:30(7):1593-4
[PubMed PMID: 15210246]
[76]
Berbel RF, Rauen PI, Pallone RF, Casella AM. Retinal detachment and gyrate atrophy of the choroid and retina: case report. Arquivos brasileiros de oftalmologia. 2012 Jan-Feb:75(1):59-60
[PubMed PMID: 22552420]
Level 3 (low-level) evidence
[77]
Takahashi O, Hayasaka S, Kiyosawa M, Mizuno K, Saito T, Tada K, Igarashi Y. Gyrate atrophy of choroid and retina complicated by vitreous hemorrhage. Japanese journal of ophthalmology. 1985:29(2):170-6
[PubMed PMID: 4046225]
[78]
Dimopoulos IS, Radziwon A, St Laurent CD, MacDonald IM. Choroideremia. Current opinion in ophthalmology. 2017 Sep:28(5):410-415. doi: 10.1097/ICU.0000000000000392. Epub
[PubMed PMID: 28520608]
Level 3 (low-level) evidence
[79]
Verbakel SK, van Huet RAC, Boon CJF, den Hollander AI, Collin RWJ, Klaver CCW, Hoyng CB, Roepman R, Klevering BJ. Non-syndromic retinitis pigmentosa. Progress in retinal and eye research. 2018 Sep:66():157-186. doi: 10.1016/j.preteyeres.2018.03.005. Epub 2018 Mar 27
[PubMed PMID: 29597005]
[80]
Zeitz C, Robson AG, Audo I. Congenital stationary night blindness: an analysis and update of genotype-phenotype correlations and pathogenic mechanisms. Progress in retinal and eye research. 2015 Mar:45():58-110. doi: 10.1016/j.preteyeres.2014.09.001. Epub 2014 Oct 13
[PubMed PMID: 25307992]
[81]
Molday RS, Kellner U, Weber BH. X-linked juvenile retinoschisis: clinical diagnosis, genetic analysis, and molecular mechanisms. Progress in retinal and eye research. 2012 May:31(3):195-212. doi: 10.1016/j.preteyeres.2011.12.002. Epub 2012 Jan 3
[PubMed PMID: 22245536]
[82]
García-García GP, Martínez-Rubio M, Moya-Moya MA, Pérez-Santonja JJ, Escribano J. Current perspectives in Bietti crystalline dystrophy. Clinical ophthalmology (Auckland, N.Z.). 2019:13():1379-1399. doi: 10.2147/OPTH.S185744. Epub 2019 Jul 30
[PubMed PMID: 31440027]
Level 3 (low-level) evidence
[83]
Tsang SH, Sharma T. Pigmented Paravenous Chorioretinal Atrophy (PPCRA). Advances in experimental medicine and biology. 2018:1085():111-113. doi: 10.1007/978-3-319-95046-4_22. Epub
[PubMed PMID: 30578495]
Level 3 (low-level) evidence
[84]
Godley BF, Tiffin PA, Evans K, Kelsell RE, Hunt DM, Bird AC. Clinical features of progressive bifocal chorioretinal atrophy: a retinal dystrophy linked to chromosome 6q. Ophthalmology. 1996 Jun:103(6):893-8
[PubMed PMID: 8643244]
[85]
Elnahry AG, Khafagy MM, Esmat SM, Mortada HA. Prevalence and Associations of Posterior Segment Manifestations in a Cohort of Egyptian Patients with Pathological Myopia. Current eye research. 2019 Sep:44(9):955-962. doi: 10.1080/02713683.2019.1606252. Epub 2019 Apr 30
[PubMed PMID: 30964360]
[86]
Hwang JC, Kim DY, Chou CL, Tsang SH. Fundus autofluorescence, optical coherence tomography, and electroretinogram findings in choroidal sclerosis. Retina (Philadelphia, Pa.). 2010 Jul-Aug:30(7):1095-103. doi: 10.1097/IAE.0b013e3181cd48f9. Epub
[PubMed PMID: 20224472]
[87]
Kellner U, Weleber RG, Kennaway NG, Fishman GA, Foerster MH. Gyrate atrophy-like phenotype with normal plasma ornithine. Retina (Philadelphia, Pa.). 1997:17(5):403-13
[PubMed PMID: 9355188]
[88]
Labiano AT, Arroyo MH. Gyrate atrophy-like phenotype with normal plasma ornithine and low plasma taurine. GMS ophthalmology cases. 2020:10():Doc04. doi: 10.3205/oc000131. Epub 2020 Feb 27
[PubMed PMID: 32269902]
Level 3 (low-level) evidence
[89]
Tripathy K, Sharma YR, Chawla R, Jain S, Behera A. Ultra-wide Field Imaging of an Operated Macular Hole in Gyrate Atrophy. Journal of ophthalmic & vision research. 2016 Jul-Sep:11(3):336-7. doi: 10.4103/2008-322X.188404. Epub
[PubMed PMID: 27621797]