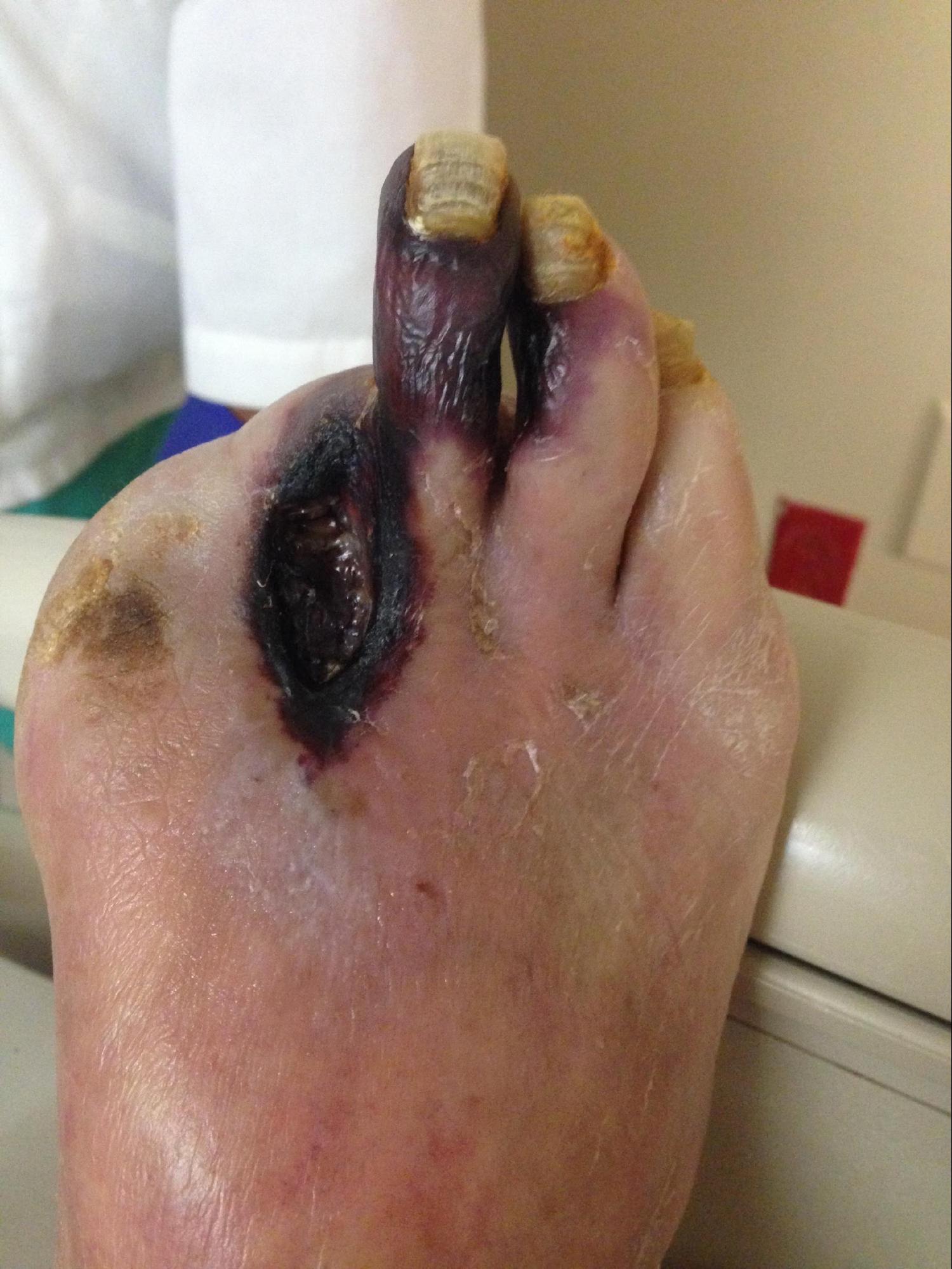
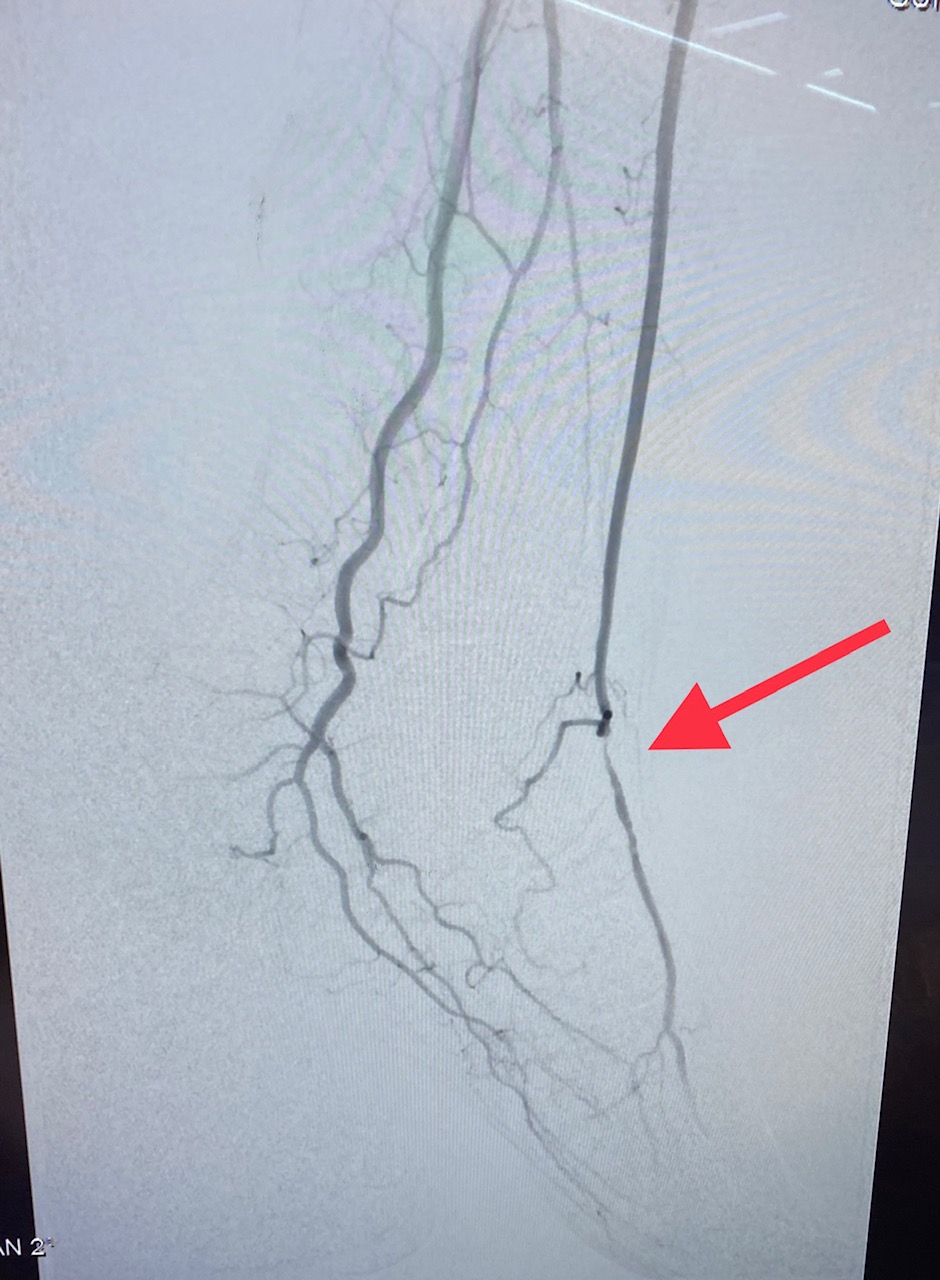
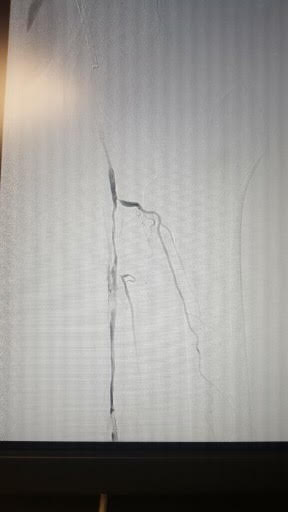
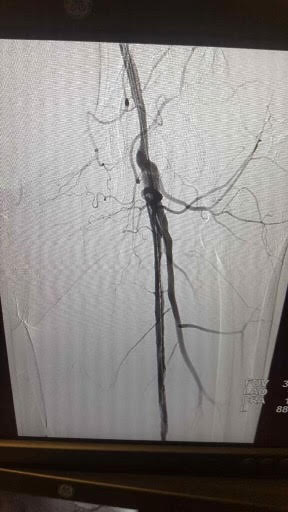
[1]
European Stroke Organisation, Tendera M, Aboyans V, Bartelink ML, Baumgartner I, Clément D, Collet JP, Cremonesi A, De Carlo M, Erbel R, Fowkes FG, Heras M, Kownator S, Minar E, Ostergren J, Poldermans D, Riambau V, Roffi M, Röther J, Sievert H, van Sambeek M, Zeller T, ESC Committee for Practice Guidelines. ESC Guidelines on the diagnosis and treatment of peripheral artery diseases: Document covering atherosclerotic disease of extracranial carotid and vertebral, mesenteric, renal, upper and lower extremity arteries: the Task Force on the Diagnosis and Treatment of Peripheral Artery Diseases of the European Society of Cardiology (ESC). European heart journal. 2011 Nov:32(22):2851-906. doi: 10.1093/eurheartj/ehr211. Epub 2011 Aug 26
[PubMed PMID: 21873417]
[2]
Meijer WT, Grobbee DE, Hunink MG, Hofman A, Hoes AW. Determinants of peripheral arterial disease in the elderly: the Rotterdam study. Archives of internal medicine. 2000 Oct 23:160(19):2934-8
[PubMed PMID: 11041900]
[3]
Fowkes FG, Rudan D, Rudan I, Aboyans V, Denenberg JO, McDermott MM, Norman PE, Sampson UK, Williams LJ, Mensah GA, Criqui MH. Comparison of global estimates of prevalence and risk factors for peripheral artery disease in 2000 and 2010: a systematic review and analysis. Lancet (London, England). 2013 Oct 19:382(9901):1329-40. doi: 10.1016/S0140-6736(13)61249-0. Epub 2013 Aug 1
[PubMed PMID: 23915883]
Level 1 (high-level) evidence
[4]
Krishna SM, Moxon JV, Golledge J. A review of the pathophysiology and potential biomarkers for peripheral artery disease. International journal of molecular sciences. 2015 May 18:16(5):11294-322. doi: 10.3390/ijms160511294. Epub 2015 May 18
[PubMed PMID: 25993296]
[5]
Subherwal S, Patel MR, Kober L, Peterson ED, Bhatt DL, Gislason GH, Olsen AM, Jones WS, Torp-Pedersen C, Fosbol EL. Peripheral artery disease is a coronary heart disease risk equivalent among both men and women: results from a nationwide study. European journal of preventive cardiology. 2015 Mar:22(3):317-25. doi: 10.1177/2047487313519344. Epub 2014 Jan 7
[PubMed PMID: 24398369]
[6]
Wakabayashi I, Sotoda Y. [Alcohol drinking and peripheral arterial disease of lower extremity]. Nihon Arukoru Yakubutsu Igakkai zasshi = Japanese journal of alcohol studies & drug dependence. 2014 Feb:49(1):13-27
[PubMed PMID: 24818355]
[7]
Periard D, Cavassini M, Taffé P, Chevalley M, Senn L, Chapuis-Taillard C, de Vallière S, Hayoz D, Tarr PE, Swiss HIV Cohort Study. High prevalence of peripheral arterial disease in HIV-infected persons. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2008 Mar 1:46(5):761-7. doi: 10.1086/527564. Epub
[PubMed PMID: 18230043]
[8]
Khandanpour N, Loke YK, Meyer FJ, Jennings B, Armon MP. Homocysteine and peripheral arterial disease: systematic review and meta-analysis. European journal of vascular and endovascular surgery : the official journal of the European Society for Vascular Surgery. 2009 Sep:38(3):316-22. doi: 10.1016/j.ejvs.2009.05.007. Epub 2009 Jun 27
[PubMed PMID: 19560951]
Level 1 (high-level) evidence
[9]
Prabhakaran D, Anand S, Gaziano TA, Mbanya JC, Wu Y, Nugent R, Sampson UKA, Fowkes FGR, Naidoo NG, Criqui MH. Peripheral Artery Disease. Cardiovascular, Respiratory, and Related Disorders. 2017 Nov 17:():
[PubMed PMID: 30212080]
[10]
Benjamin EJ, Muntner P, Alonso A, Bittencourt MS, Callaway CW, Carson AP, Chamberlain AM, Chang AR, Cheng S, Das SR, Delling FN, Djousse L, Elkind MSV, Ferguson JF, Fornage M, Jordan LC, Khan SS, Kissela BM, Knutson KL, Kwan TW, Lackland DT, Lewis TT, Lichtman JH, Longenecker CT, Loop MS, Lutsey PL, Martin SS, Matsushita K, Moran AE, Mussolino ME, O'Flaherty M, Pandey A, Perak AM, Rosamond WD, Roth GA, Sampson UKA, Satou GM, Schroeder EB, Shah SH, Spartano NL, Stokes A, Tirschwell DL, Tsao CW, Turakhia MP, VanWagner LB, Wilkins JT, Wong SS, Virani SS, American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee. Heart Disease and Stroke Statistics-2019 Update: A Report From the American Heart Association. Circulation. 2019 Mar 5:139(10):e56-e528. doi: 10.1161/CIR.0000000000000659. Epub
[PubMed PMID: 30700139]
[11]
Shu J, Santulli G. Update on peripheral artery disease: Epidemiology and evidence-based facts. Atherosclerosis. 2018 Aug:275():379-381. doi: 10.1016/j.atherosclerosis.2018.05.033. Epub 2018 May 22
[PubMed PMID: 29843915]
[12]
Kannel WB, McGee DL. Update on some epidemiologic features of intermittent claudication: the Framingham Study. Journal of the American Geriatrics Society. 1985 Jan:33(1):13-8
[PubMed PMID: 3965550]
[13]
Murabito JM, Evans JC, Nieto K, Larson MG, Levy D, Wilson PW. Prevalence and clinical correlates of peripheral arterial disease in the Framingham Offspring Study. American heart journal. 2002 Jun:143(6):961-5
[PubMed PMID: 12075249]
[14]
Aboyans V, Criqui MH, McClelland RL, Allison MA, McDermott MM, Goff DC Jr, Manolio TA. Intrinsic contribution of gender and ethnicity to normal ankle-brachial index values: the Multi-Ethnic Study of Atherosclerosis (MESA). Journal of vascular surgery. 2007 Feb:45(2):319-27
[PubMed PMID: 17264011]
[15]
Newman AB, Siscovick DS, Manolio TA, Polak J, Fried LP, Borhani NO, Wolfson SK. Ankle-arm index as a marker of atherosclerosis in the Cardiovascular Health Study. Cardiovascular Heart Study (CHS) Collaborative Research Group. Circulation. 1993 Sep:88(3):837-45
[PubMed PMID: 8353913]
[16]
Criqui MH, Aboyans V. Epidemiology of peripheral artery disease. Circulation research. 2015 Apr 24:116(9):1509-26. doi: 10.1161/CIRCRESAHA.116.303849. Epub
[PubMed PMID: 25908725]
[17]
Pande RL, Creager MA. Socioeconomic inequality and peripheral artery disease prevalence in US adults. Circulation. Cardiovascular quality and outcomes. 2014 Jul:7(4):532-9
[PubMed PMID: 24987053]
Level 2 (mid-level) evidence
[18]
Rafieian-Kopaei M, Setorki M, Doudi M, Baradaran A, Nasri H. Atherosclerosis: process, indicators, risk factors and new hopes. International journal of preventive medicine. 2014 Aug:5(8):927-46
[PubMed PMID: 25489440]
[20]
Olin JW, Sealove BA. Peripheral artery disease: current insight into the disease and its diagnosis and management. Mayo Clinic proceedings. 2010 Jul:85(7):678-92. doi: 10.4065/mcp.2010.0133. Epub
[PubMed PMID: 20592174]
[21]
Varu VN, Hogg ME, Kibbe MR. Critical limb ischemia. Journal of vascular surgery. 2010 Jan:51(1):230-41. doi: 10.1016/j.jvs.2009.08.073. Epub
[PubMed PMID: 20117502]
[22]
Kim HO, Kim W. Elucidation of the Diagnosis and Treatment of Peripheral Arterial Disease. Korean circulation journal. 2018 Sep:48(9):826-827. doi: 10.4070/kcj.2018.0155. Epub 2018 Jun 11
[PubMed PMID: 30088357]
[23]
Olinic DM, Stanek A, Tătaru DA, Homorodean C, Olinic M. Acute Limb Ischemia: An Update on Diagnosis and Management. Journal of clinical medicine. 2019 Aug 14:8(8):. doi: 10.3390/jcm8081215. Epub 2019 Aug 14
[PubMed PMID: 31416204]
[24]
Dhaliwal G, Mukherjee D. Peripheral arterial disease: Epidemiology, natural history, diagnosis and treatment. The International journal of angiology : official publication of the International College of Angiology, Inc. 2007 Summer:16(2):36-44
[PubMed PMID: 22477268]
[25]
McDermott MM, Greenland P, Liu K, Guralnik JM, Criqui MH, Dolan NC, Chan C, Celic L, Pearce WH, Schneider JR, Sharma L, Clark E, Gibson D, Martin GJ. Leg symptoms in peripheral arterial disease: associated clinical characteristics and functional impairment. JAMA. 2001 Oct 3:286(13):1599-606
[PubMed PMID: 11585483]
[26]
Høyer C, Sandermann J, Petersen LJ. The toe-brachial index in the diagnosis of peripheral arterial disease. Journal of vascular surgery. 2013 Jul:58(1):231-8. doi: 10.1016/j.jvs.2013.03.044. Epub 2013 May 18
[PubMed PMID: 23688630]
[27]
Dormandy JA, Rutherford RB. Management of peripheral arterial disease (PAD). TASC Working Group. TransAtlantic Inter-Society Consensus (TASC). Journal of vascular surgery. 2000 Jan:31(1 Pt 2):S1-S296
[PubMed PMID: 10666287]
Level 3 (low-level) evidence
[28]
Bailey MA, Griffin KJ, Scott DJ. Clinical assessment of patients with peripheral arterial disease. Seminars in interventional radiology. 2014 Dec:31(4):292-9. doi: 10.1055/s-0034-1393964. Epub
[PubMed PMID: 25435653]
[29]
Hennion DR, Siano KA. Diagnosis and treatment of peripheral arterial disease. American family physician. 2013 Sep 1:88(5):306-10
[PubMed PMID: 24010393]
[30]
Resnick HE, Lindsay RS, McDermott MM, Devereux RB, Jones KL, Fabsitz RR, Howard BV. Relationship of high and low ankle brachial index to all-cause and cardiovascular disease mortality: the Strong Heart Study. Circulation. 2004 Feb 17:109(6):733-9
[PubMed PMID: 14970108]
[31]
Olin JW, Kaufman JA, Bluemke DA, Bonow RO, Gerhard MD, Jaff MR, Rubin GD, Hall W, American Heart Association. Atherosclerotic Vascular Disease Conference: Writing Group IV: imaging. Circulation. 2004 Jun 1:109(21):2626-33
[PubMed PMID: 15173045]
[32]
Criqui MH, Langer RD, Fronek A, Feigelson HS, Klauber MR, McCann TJ, Browner D. Mortality over a period of 10 years in patients with peripheral arterial disease. The New England journal of medicine. 1992 Feb 6:326(6):381-6
[PubMed PMID: 1729621]
[33]
Jonason T, Bergström R. Cessation of smoking in patients with intermittent claudication. Effects on the risk of peripheral vascular complications, myocardial infarction and mortality. Acta medica Scandinavica. 1987:221(3):253-60
[PubMed PMID: 3591463]
[34]
Hirsch AT, Haskal ZJ, Hertzer NR, Bakal CW, Creager MA, Halperin JL, Hiratzka LF, Murphy WR, Olin JW, Puschett JB, Rosenfield KA, Sacks D, Stanley JC, Taylor LM Jr, White CJ, White J, White RA, Antman EM, Smith SC Jr, Adams CD, Anderson JL, Faxon DP, Fuster V, Gibbons RJ, Hunt SA, Jacobs AK, Nishimura R, Ornato JP, Page RL, Riegel B, American Association for Vascular Surgery, Society for Vascular Surgery, Society for Cardiovascular Angiography and Interventions, Society for Vascular Medicine and Biology, Society of Interventional Radiology, ACC/AHA Task Force on Practice Guidelines Writing Committee to Develop Guidelines for the Management of Patients With Peripheral Arterial Disease, American Association of Cardiovascular and Pulmonary Rehabilitation, National Heart, Lung, and Blood Institute, Society for Vascular Nursing, TransAtlantic Inter-Society Consensus, Vascular Disease Foundation. ACC/AHA 2005 Practice Guidelines for the management of patients with peripheral arterial disease (lower extremity, renal, mesenteric, and abdominal aortic): a collaborative report from the American Association for Vascular Surgery/Society for Vascular Surgery, Society for Cardiovascular Angiography and Interventions, Society for Vascular Medicine and Biology, Society of Interventional Radiology, and the ACC/AHA Task Force on Practice Guidelines (Writing Committee to Develop Guidelines for the Management of Patients With Peripheral Arterial Disease): endorsed by the American Association of Cardiovascular and Pulmonary Rehabilitation; National Heart, Lung, and Blood Institute; Society for Vascular Nursing; TransAtlantic Inter-Society Consensus; and Vascular Disease Foundation. Circulation. 2006 Mar 21:113(11):e463-654
[PubMed PMID: 16549646]
Level 3 (low-level) evidence
[35]
American Diabetes Association. 2. Classification and Diagnosis of Diabetes. Diabetes care. 2016 Jan:39 Suppl 1():S13-22. doi: 10.2337/dc16-S005. Epub
[PubMed PMID: 26696675]
[36]
Lane R, Harwood A, Watson L, Leng GC. Exercise for intermittent claudication. The Cochrane database of systematic reviews. 2017 Dec 26:12(12):CD000990. doi: 10.1002/14651858.CD000990.pub4. Epub 2017 Dec 26
[PubMed PMID: 29278423]
Level 1 (high-level) evidence
[37]
Society for Vascular Surgery Lower Extremity Guidelines Writing Group, Conte MS, Pomposelli FB, Clair DG, Geraghty PJ, McKinsey JF, Mills JL, Moneta GL, Murad MH, Powell RJ, Reed AB, Schanzer A, Sidawy AN, Society for Vascular Surgery. Society for Vascular Surgery practice guidelines for atherosclerotic occlusive disease of the lower extremities: management of asymptomatic disease and claudication. Journal of vascular surgery. 2015 Mar:61(3 Suppl):2S-41S. doi: 10.1016/j.jvs.2014.12.009. Epub 2015 Jan 28
[PubMed PMID: 25638515]
Level 1 (high-level) evidence
[38]
Thompson PD, Zimet R, Forbes WP, Zhang P. Meta-analysis of results from eight randomized, placebo-controlled trials on the effect of cilostazol on patients with intermittent claudication. The American journal of cardiology. 2002 Dec 15:90(12):1314-9
[PubMed PMID: 12480040]
Level 1 (high-level) evidence
[39]
Norgren L, Hiatt WR, Dormandy JA, Nehler MR, Harris KA, Fowkes FG, TASC II Working Group. Inter-Society Consensus for the Management of Peripheral Arterial Disease (TASC II). Journal of vascular surgery. 2007 Jan:45 Suppl S():S5-67
[PubMed PMID: 17223489]
Level 3 (low-level) evidence
[40]
Comerota AJ. Endovascular and surgical revascularization for patients with intermittent claudication. The American journal of cardiology. 2001 Jun 28:87(12A):34D-43D
[PubMed PMID: 11434898]