Introduction
Functioning as vital cells in the bone matrix that maintains adequate bone mineral density through stimulating bone turnover and maintaining plasma calcium levels, osteoclasts are multinucleate cells that arise from hematopoietic stem cells.[1][2][3][4] As a result, they are identifiable with the presence of CD13, CD14, and CD68 antigens and the lack of CD56, GrB, and Ki67 antigens.[5] When they become activated through their receptor activator nuclear factor-kB (RANK), they function primarily to resorb bone through various acids and collagenases. They have a complex interplay with the immune system, which can stimulate them and result in various pathologies.[1][2][3][6][7] Many bone diseases are associated with dysfunctional osteoclasts, such as osteoarthritis, osteoporosis, and osteopetrosis.[4][8][9][10]
Issues of Concern
What happens to osteoclasts in space? (e.g., the bones of astronauts)
In the presence of microgravity (μG), there is a monthly bone loss of about 1 to 2%.
The genomic activity of osteoclasts is high, involving maturation, differentiation, and metabolic stimulation processes. In particular, according to more recent literature, we can find increases in:
- c-fos
- DNA damage-inducible transcript 4 (DDIT4)
- jun B proto-oncogene
- TRAIL
- DCSTAMP
- Syncytin-A
Pre-osteoclastic cells with μG show increases in other molecules, such as:
- Metalloproteinase type 9
- Cathepsin K
- TRAP
- RANK
- LC3
- Atg5
Structure
Osteoclasts are large dome-shaped multinucleated cells comprised of fused mononuclear osteoclast progenitor cells. Osteoclast progenitors fuse together on the bone surfaces but are primarily found in bone marrow, where they derive from macrophage/dendritic cell precursors, which arise from myeloid progenitor cells, which ultimately originate from hematopoietic stem cells. Therefore, osteoclasts have a hematopoietic origin, which may explain their diverse and dynamic function.[1][2][3][4] On average, osteoclasts have eight nuclei and are 300 micrometers large, which researchers theorize gives them a strong resorptive property.[2] Average osteoclasts are 15 times larger than macrophages, which are 20 micrometers large. In some pathological conditions, they have been reported to have as many as 100 nuclei.[2] Osteoclasts are well-known for their characteristic ruffled border, which forms on their inferior surface due to the high concentration of vesicles in that region of their cell membrane.[1] Osteoclasts contain many mitochondria, which provides them with abundant energy for bone resorption. Additionally, other common cell components such as Golgi bodies and lysosomes are also present in osteoclasts.[1]
A lifecycle has been proposed for osteoclasts, with the order being the following[1]:
- Pre-Osteoclasts (Precursors/Progenitors)
- This was the subject of prior discussion.
- Active Osteoclasts
- Multinucleated cells attached to the bone surface, with a ruffled border
- Inactive Osteoclasts
- Multinucleated cells attached to the bone surface, with no ruffled border
- Resting Osteoclasts
- Multinucleated cells not attached to the bone surface, with no ruffled border
- Post-Osteoclasts
- Mononuclear cells with a high capacity for phagocytosis
Osteoclasts and osteoclast progenitor cells have many receptors present on their cell surface, which play important roles that will be discussed later. The most important ones include receptor activator nuclear factor-kB (RANK) and toll-like receptors (TLRs).
Function
Osteoclasts serve a vital role in bone metabolism and turnover, being the cells responsible for bone resorption. As a result, they have two essential functions: maintaining strong and healthy bone architecture and maintaining normal plasma calcium levels.[2] Bone resorption is defined as the dissolution and degradation of the bone matrix, both mineral and organic components. It occurs through five osteoclast-mediated steps:[1][2][3][6][7]
- Osteoclast Formation and Osteoclast Migration
- Mononuclear osteoclast progenitors express receptor activator nuclear factor-kB (RANK), which interacts with receptor activator nuclear factor-kB ligand (RANKL) on osteoblasts and stromal cell stems, to cause the differentiation and formation of osteoclasts (known as osteoclastogenesis), with the help of the expression of macrophage-colony stimulating factor (m-CSF) by osteoblasts and stromal cell smells. Additionally, mononuclear osteoclast precursors have dendritic cell-specific transmembrane protein (DC-STAMP), which helps them fuse to form osteoclasts. Osteoclast stimulatory transmembrane protein (OC-STAMP) also plays a role in precursor fusion, but its impact on fusion is not as much as DC-STAMP.
- Osteoclasts are theorized to migrate to the bone matrix via chemoattractants such as osteocalcin, type I collagen peptides (such as alpha-2-HS glycoprotein), and stromal cell-derived factor-1 (SDF-1), which are all produced by osteoblasts. Additionally, it has been hypothesized that living osteocytes in the bone matrix inhibit bone resorption.
- Bone Mineral Attachment and Formation of the Actin-Ring and Sealing Zone
- For resorption to occur in a precise and efficient manner, osteoclasts create a sealed microenvironment over the area which needs to be resorbed, preventing the leakage of any osteolytic proteins or ions. Osteoclasts attach to the bone matrix via integrin alpha-V-beta-3, which activates the non-receptor tyrosine kinase (NRTK) c-Src. Activation of the c-Src leads to a cascade that results in the polymerization of F-actin in a circular fashion, which forms the actin-ring that has small actin processes called podosomes. The podosomes directly attach to the bone matrix and create the sealing zone, within which bone resorption takes place.
- Ruffled Border Formation
- The ruffled border of an osteoclast is the folded membrane facing the side of the sealed zone. Since it faces the sealed zone, it plays a vital role in bone resorption. The ruffled border has a high concentration of vesicles, which help acidify and secrete enzymes into the microenvironment formed by the sealed zone. The NRTK c-Src helps form the ruffled border through mediating trafficking of those vesicles.
- Release of Collagenolytic Enzymes and Degradation of the Bone Matrix
- Once all of the structures that form the microenvironment are assembled, then a proton pump-ATPase, coupled with a chloride ion channel, pumps protons and chloride ions into the microenvironment. They combine to form HCl, which dissolves the solid mineral hydroxyapatite [Ca(PO)]Ca(OH) into its constituent ions, degrading the mineral component of the bone matrix. The organic component of the bone matrix, especially type I collagen (the most abundant protein in the extracellular matrix (ECM)), is mainly degraded by cathepsin K. Additionally, cathepsin B, D, and L are also proteinases that degrade the organic bone matrix, but their effect is not as prominent as cathepsin K. Matrix metalloproteinases (MMPs), especially MMP-9, are enzymes that degrade the ECM proteins of bone as well.
- Disposal of Resorptive Byproducts
- The byproducts of resorption are endocytosed from the resorption microenvironment into the osteoclast. The endocytic vesicles fuse to the functional secretory domain (FSD), which is a part of the plasma membrane that is separate from the ruffled border, and the materials are released into the extracellular space. This process is called transcytosis. This results in the increase of plasma calcium levels (from the breakdown of hydroxyapatite), which is essential for many bodily functions.
Upon completion of those five steps, the osteoclast detaches from the bone matrix and finds another spot to perform bone resorption, or it undergoes apoptosis.
Tissue Preparation
There have been many different preparations utilized for the analysis of bone tissue containing osteoclasts. One preparation that can be utilized requires obtaining bone tissue, fixed in formalin for 24 hours, then decalcified using a strong acid (such as 5% nitric acid) for 48 hours. The bone tissue can then be placed into paraffin wax, and a routine hematoxylin & eosin (H&E) preparation can be done. H&E preparation is a common choice for the visualization of bone tissue. Additionally, if using immunohistochemistry, an indirect immunoperoxidase technique can be utilized for bone tissue.[5]
Histochemistry and Cytochemistry
Osteoclasts arise from macrophage precursors, so they share many of the same antigens found on macrophages and giant cells. Therefore, identifying osteoclasts relies on the presence and lack of certain antigens. Antigens associated with osteoclasts are CD13, CD14, and CD68, which have a particularly strong association with osteoclasts. The lack of CD56, GrB, and Ki67 expression help distinguish osteoclasts from macrophages and giant cells.[5]
Microscopy, Light
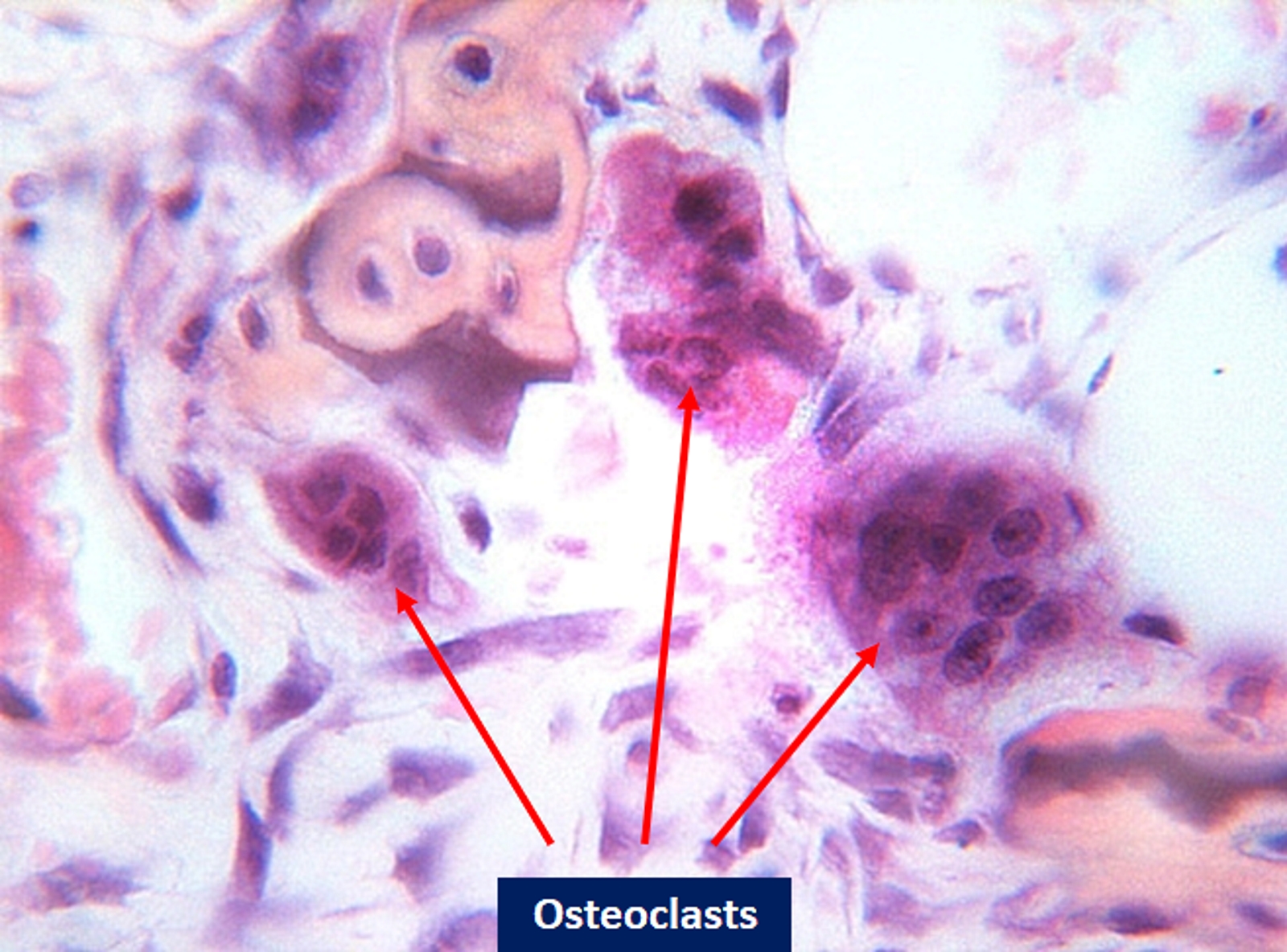
On light microscopy, osteoclasts are identifiable as containing red in their cytoplasm if stained with tartrate-resistant acid phosphatase (TRAP). The multinucleated appearance of osteoclasts can typically be visualized on light microscopy, and large vacuoles can also be seen. Light microscopy allows visualization of the osteoclasts being surrounded by osteocytes and other components of the bone matrix, allowing viewers to gain an understanding of the complex interactions between different cells within the bone matrix.[11] Additionally, since osteoclasts contain a large number of mitochondria, observing the increased cytochrome C oxidase activity of osteoclasts in bone marrow can help differentiate them from other cells.[1]
Microscopy, Electron
On electron microscopy, which has much higher magnifying power than light microscopy, the extensive multinucleated appearance of osteoclasts can be visualized, along with a closer view of the ruffled border adjacent to the bone undergoing resorption. The lacunae in which osteoclasts are contained, resorbing bone, are known as Howship's lacunae. Lying under the ruffled membrane are numerous visible vacuoles. Some of the vacuoles contain cells visibly undergoing apoptosis.[11]
Pathophysiology
The RANK-RANKL interaction is essential for osteoclastogenesis, and that interaction is under the regulation of osteoprotegerin (OPG). Osteoblasts and stromal cells secrete OPG, and it binds to RANKL on osteoblasts and stromal stem cells and prevents the interaction between RANKL and RANK, inhibiting osteoclastogenesis. This serves as a control mechanism to prevent excess bone resorption from taking place.[12] OPG opposes the effects of 1, 25-dihydroxy-vitamin D, parathyroid hormone (PTH), parathyroid hormone-related peptide (PTH-rP), and prostaglandin E2 (PGE), which all increase bone resorption.[6] Therefore, the interplay between RANK-RANKL-OPG is crucial in regulating osteoclastogenesis and the maintenance of bone mineral density and plasma calcium levels. Dysregulation of the RANK-RANKL-OPG system plays a vital role in the pathogenesis of many conditions, such as osteoporosis, osteopetrosis, and bone tumors.
A complex interplay exists between the musculoskeletal and immune systems. Bone metabolism and immune activity are directly and indirectly connected, and immune system dysregulation can lead to osteoclast-mediated pathology. Two mechanisms can cause immune system-mediated osteoclast generation: T-cell mediated generation of osteoclasts and innate immune system generation of osteoclasts[2]:
T-Cell Mediated Generation of Osteoclasts
Similar to osteoblast expression of RANKL, arguably the most important osteoclast determining factor, activated T-cells also express RANKL. When there is a persistence of elevated active T-cells, it can lead to increased osteoclast differentiation, and ultimately, excess bone resorption.
Additionally, active T-cells produce pro-inflammatory cytokines, which can stimulate the expression of RANKL on osteoblasts. Inflammatory cytokines that can stimulation osteoclast-mediated bone resorption are IL-1, IL-6, IL-11, IL-15, IL-17, and TNF-alpha. Additionally, IL-1 and TNF-alpha stimulate osteoblasts, which can cause RANKL-induced osteoclasts differentiation by the osteoblasts. Inflammatory cytokines that inhibit osteoclast-mediated bone resorption are IL-4, IL-10, IL-12, IL-13, IL-18, IFN-alpha, and GM-CSF.
Innate Immune System Regulation of Osteoclasts
Immune cells in the body have toll-like receptors (TLRs), which recognize pathogen-associated molecular patterns (PAMPs) on invading organisms, allowing the body to mount an immune response to the foreign organisms. Upon activation of the TLRs and immune cells, pro-inflammatory cytokines are produced, which can indirectly affect bone metabolism through activation of osteoclast differentiation, as previously mentioned. This is an indirect mechanism through which the innate immune system can cause osteoclast-mediated resorption of bone. Additionally, osteoclasts themselves have TLRs, which can be directly activated by foreign organisms. When TLRs on late osteoclast progenitors and osteoclasts are active, that increases the generation of osteoclasts. However, when TLRs on early osteoclast progenitors are activated, it decreases the generation of osteoclasts. This has been theorized to be the body’s way of preventing excess osteoclast generation.
Aside from the interaction between the musculoskeletal and immune systems, the formation of many of the osteoclast structures previously described is important to prevent the malfunctioning of osteoclasts. For example, failure of the ruffled border to form may result in the failure of bone resorption.[3]
Clinical Significance
Osteoarthritis
- Osteoarthritis (OA), a degenerative disorder of the hyaline cartilage and subchondral bone at the ends of bones, is the most common joint condition in the United States. It affects many individuals throughout the world.[13] Osteoarthritis is associated with an altered ratio of osteoblasts to osteoclasts, with an increased number of osteoclasts causing bone resorption of subchondral bone, leading to thinning of the subchondral bone plate. Not only is there an increased number of osteoclasts, but their activity is also increased, with a study finding that their surface area was increased in OA, leading to increased bone resorption.[8] An interesting finding of the study was that loading a joint with osteoarthritis decreased the expression of RANKL, Cathepsin K, and TNF-alpha, which may provide a link between mechanical loading of a joint and reduced activity of osteoclasts.[8] Therefore, osteoclasts play a significant role in the pathogenesis of osteoarthritis.
Osteopetrosis
- Known as “marble bone disease,” osteopetrosis refers to several genetic diseases where patients have an increase in bone mass, but the bone is brittle and prone to pathological fractures.[9][10] There are many presentations of osteopetrosis, but they have a common link in that osteoclast function is decreased. The number of osteoclasts is typically normal or even increased. However, they are not functional and typically have problems with adhesion to the bone and activation, leading to impaired secretion of acids that cause bone resorption.[9]
Osteoporosis
- Osteoporosis is an age-related microarchitectural deterioration of bone, hypothesized to occur due to an imbalance between osteoblasts and osteoclasts. While osteoporosis is common in postmenopausal females, it can occur in males as well. Postmenopausal females have lower estrogen levels, and estrogen is known to be a potent inhibitor of osteoclasts. Therefore, postmenopausal females lose that osteoclast inhibition, which then leads to increased osteoclast bone resorption. The hypothesis is that decreased testosterone levels in men also increase osteoclast activity, but it is not clear through what mechanism.[4] A significant cause of morbidity and mortality in elderly individuals, osteoporotic-related fractures (especially of the vertebrae and spine), should be prevented through effective patient counseling and treatment.[14]
Paget Disease
- Paget’s disease is a systemic bone disorder that starts occurring in middle-aged individuals and older. It is the second most common bone disorder in that age group in the United States and the United Kingdom. While it can manifest with bony abnormalities and disfigurations of the skull, pelvis, and spine, it can also include deafness and nerve compression. In the majority of patients with the disease, it is asymptomatic. Paget disease has correlations with increased osteoclastic activity, but the exact cause is still unknown. Some studies have linked it to paramyxoviruses, but there has not been concrete evidence regarding its pathogenesis. Paget disease typically occurs in four stages. The first stage primarily shows increased osteoclastic activity. The second stage is a mix of osteoclastic and osteoblastic activity, the third stage being primarily osteoblastic activity, and the fourth stage is the degeneration of the bone. This progression ultimately leads to softening and enlargement of bone, which makes patients prone to pathological fractures. One rare complication of Paget disease is osteosarcoma, which can be deadly in patients. Histologically, there is typically a hypercellularity of osteoblasts and osteoclasts, with the osteoclasts normally having a very large number of nuclei (can reach up to 100) and intracytoplasmic inclusions.[9][4][15]
Juvenile Paget Disease (JPD)
- JPD is an autosomal recessive condition where there is a loss of function mutation of OPG, which causes a loss of regulation of the RANK/RANKL interaction, leading to increased osteoclast differentiation and activity, and increased osteoblast activity. Patients have a high turnover in their bones, potentially leading to skeletal abnormalities such as skull thickening, axial spine vertebral compression fractures, kyphosis, widening of the diaphysis of long bones, and pelvic deformity. The key feature of JPD is its early onset, with newborn infants potentially showing symptoms of the disorder. JPD has a highly variable clinical presentation. The histology of patients with JPD is also variable, with some patients not having any woven bone and some highly disorganized bone matrix.[4][9]
FEO (Familiar Expanile Osteolysis), Expansile Skeletal Hyperphosphatasia (ESH), and Early Onset Paget’s Disease of Bone (Early Onset PDB)
- FEO, ESH, and early onset PDB are all characterized by focal skeletal abnormalities located in many regions throughout the body. Their onset of symptoms are during the second decade of life, and they range from progressive hearing loss found in all of the conditions (due to degeneration of resorption of the bones in the inner ear), to mainly the involvement of the appendicular skeleton in FEO, with the axial and appendicular skeleton involvement in early-onset PDB. ESH and early onset PDB are differentiated from FEO by episodic hypercalcemia. Skeletal abnormalities can lead to pathological fractures in patients. In all of these conditions, osteoclasts are hypothesized to have activating mutations of the RANK (not requiring the RANKL signal), which can cause excess resorption and lead to widespread bony involvement. Osteoclasts become enlarged in this condition, but the number of osteoclasts does not typically elevate in these conditions.[9] With histological examination, there is a high amount of woven bone present in patients with these conditions, which ultimately can progress to lytic lesions of the bone and replacement with adipocytes and fibrovascular tissue.[9]
Pycnodysostosis
- Pycnodysostosis is an extremely rare genetic disease that presents as short stature with dysmorphic features of the skull, clavicles, and digits. It results from a genetic mutation of Cathepsin K, a proteinase secreted by osteoclasts that destroys Type I collagen. As a result, osteoclasts can resorb the mineralized portion of the bone matrix, but they are unable to resorb the organic portion of the bone matrix. This impairment of type I collagen degradation leads to osteoclasts phagocytosing the organic components of the bone matrix, which can be visualized in osteoclasts as intracellular vacuoles containing fibrillary collagen.[4][9] Patients with Pycnodysostosis typically have an increased number of osteoclasts.
Adult T-Cell Leukemia (ATL)
- As previously mentioned, T-cells can have RANKL ligands that bind to RANK on osteoclast precursors, causing osteoclastogenesis. In patients with ATL, a common side-effect is hypercalcemia, caused by increased osteoclast bone resorption activity. This directly results from increased RANK/RANKL interaction and indirectly by producing inflammatory cytokines such as PTHrP, IL-1, and TGF-beta. Other forms of cancer have correlations with increased expression of RANKL, such as breast cancer and multiple myeloma, which can present as hypercalcemia via osteoclastogenesis.[16][17][16]
Periodontitis
- The pathophysiology of periodontitis is that as bacterial cells increase in number, they ultimately attract immune cells such as macrophages. Those immune cells release inflammatory cytokines such as IL-6, which increases the migration of osteoclasts and results in increased bone resorption of the alveolar bone of teeth, destroying the insertion sites of teeth.[11] Additionally, T-lymphocytes can be called on to help destroy the bacterial infection, and they have RANKL, which can directly stimulate osteoclastogenesis and cause bone resorption of alveolar bone.[18]