Introduction
Neurotransmitters are endogenous chemicals that allow neurons to communicate with each other throughout the body. They enable the brain to provide a variety of functions, through the process of chemical synaptic transmission. These endogenous chemicals are integral in shaping everyday life and functions.[1]
Chemical synaptic transmission primarily through the release of neurotransmitters from presynaptic neural cells to postsynaptic receptors. Alterations in the levels of specific neurotransmitters have been observed in various neurological disorders, including Parkinson disease, schizophrenia, depression, and Alzheimer disease.
Development
Neurotransmitters are involved in the processes of early human development, including neurotransmission, differentiation, the growth of neurons, and the development of neural circuitry. Certain neurotransmitters may appear at different points of development. For example, monoamines are present before the neurons are differentiated. Norepinephrine levels are high in the notochord, even in the very early stages of the embryo. Serotonin has a role in morphogenesis. Excitatory amino acids tend to appear later in ontogenesis. The levels of neurotransmitters and neuromodulators tend to increase as new synapses form. Others will appear in the perinatal period, like glutamate, and plateau afterward. Hypoxia and drug-exposure can disturb the formation of neuronal circuity, leading to long-term deleterious effects in the body.[2][3]
Function
There are a number of neurotransmitters used by the body for different functions, including acetylcholine, glutamate, GABA, glycine, dopamine, norepinephrine, and serotonin. Glutamate is the principal excitatory neurotransmitter used in the brain. It is also the primary mediator of nervous system plasticity.[4] Glutamate has been implicated in modifiable synapses, which researchers suspect are the memory-storage elements of the brain.[5] Gamma-aminobutyric acid (GABA) and glycine, conversely, serve as the major inhibitory neurotransmitters. GABA, for example, can account for approximately 40% of the inhibitory processing in the brain. Glycine is found primarily in the spinal cord.[6] Dopamine, another major neurotransmitter, plays an essential role in several brain functions, including learning, motor control, reward, emotion, and executive functions. Dopamine has also been implicated in psychiatric and neurological disorders.[7] Serotonin is a neurotransmitter that modulates multiple neuropsychological processes and neural activity — many drugs used in psychiatry and neurology target serotonin. Serotonin also has implications that affect gastrointestinal processes like bowel motility, bladder control, and cardiovascular function.[8] Norepinephrine is a monoamine that is synthesized in the central nervous system and sympathetic nerves. The locus coeruleus of the brain plays a vital role in the signaling of norepinephrine. The release of norepinephrine in the brain exerts effects on a variety of processes, including stress, sleep, attention, focus, and inflammation. It also plays a role in modulating the responses of the autonomic nervous system.[9] Histamine is another neurotransmitter that mediates homeostatic functions in the body, promotes wakefulness, modulates feeding behavior, and controls motivational behavior.[10]
Mechanism
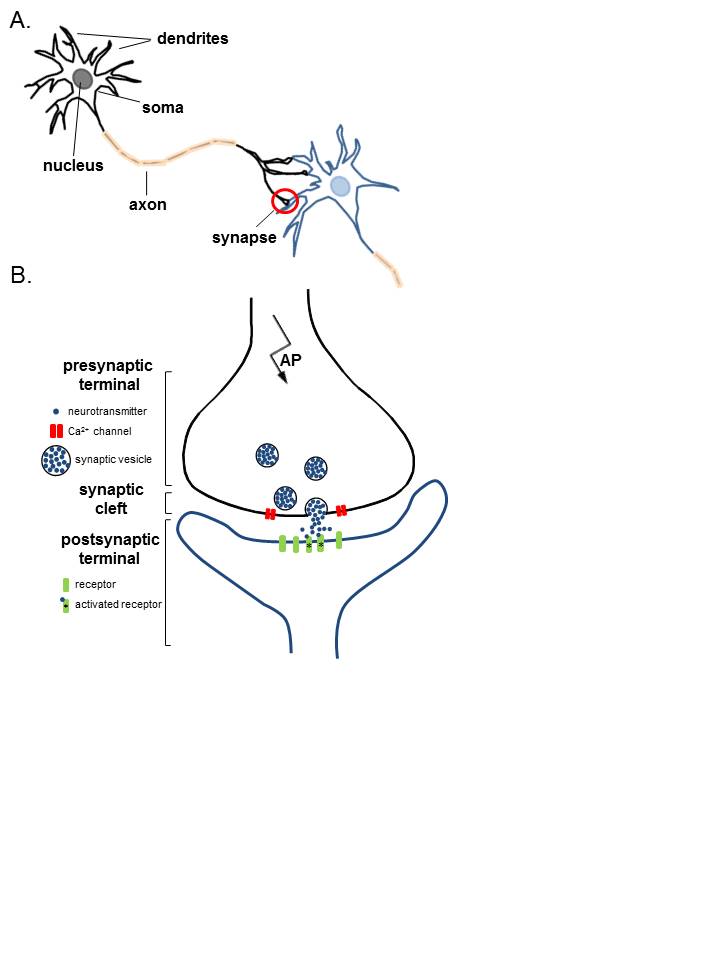
Neurotransmission medication occurs via the vesicular release of neurotransmitters at presynaptic nerve terminals. Specifically, calcium-evoked exocytosis of the presynaptic vesicles is what enables the release of neurotransmitters into the synapse. Active zones, specialized areas on the presynaptic plasma membranes, tether the neurotransmitter-containing vesicles to the plasma membrane. Once an action potential triggers calcium influx into the presynaptic cleft, active zones undergo fusion with the vesicles, allowing neurotransmitter release.[1] There are multiple proteins involved in the fusion of neurotransmitter-containing vesicles and the active zone. The soluble N-ethyl maleimide sensitive factor attachment protein receptors (SNAREs) syntaxin-1, SNAP-25, and synaptobrevin-2 together form a SNARE complex, a key component in membrane fusion and ultimately exocytosis. A number of the proteins involved in this process may act as inhibitors and activators of the exocytosis of neurotransmitters from the presynapse.[11]
Pathophysiology
The neurotransmitter glutamate has been implicated in multiple neurodegenerative studies. Researchers agree that glutamate excitotoxicity undoubtedly has a role in the pathogenesis of Alzheimer disease, the most common neurodegenerative pathology affecting the elderly population. Research suggests glutamate excitotoxicity accelerates the progression of Alzheimer disease.[12] Glutamate is also implicated in the pathogenesis of Parkinson disease. Mutations in genes encoding the parkin and DJ1 proteins are present in Parkinson disease, which are involved in the regulation of excitatory glutamate synapses. These proteins may also protect neurons against glutamate excitotoxicity.[13][14]
Clinical Significance
Gamma-aminobutyric acid (GABA), the major inhibitory neurotransmitter in the central nervous system, is targeted in the treatment of anxiety disorder, insomnia, epilepsy, and other conditions. In particular, these drugs alter GABAergic function by targeting the GABA-A and GABA-B receptors.[15]
Not only does dopamine play an important role in multiple physiological processes, but it also has a role in the pathology of psychiatric and neurodegenerative diseases. Disturbances in the neurotransmission of dopamine are implicated in schizophrenia, psychosis, depression, Tourette syndrome, and attention deficit hyperactivity disorder. Regarding neurodegenerative diseases, dopamine is related to Parkinson disease, multiple sclerosis, and Huntington disease. There has been ample research on the role of dopaminergic neurons in Parkinson disease. Currently, research suggests that the degeneration of dopaminergic neurons in the substantia nigra pars compacta is involved in the pathogenesis of Parkinson disease.[16]
Serotonin, a neurotransmitter that controls several neuropsychiatric processes, has been implicated in the pathogenesis of depression. Research has shown that patients with endogenous depression have low plasma levels of tryptophan, a precursor of serotonin. Furthermore, postmortem studies found an association between decreased serotonin levels in the brain and suicide, among depressed patients. In light of this, quite a few drugs have been developed that target serotonin in the treatment of depression. For example, tricyclic antidepressants work by increasing serotonin levels in the synapse.[17]
Norepinephrine is involved in the pathogenesis of neuropsychiatric disorders. Changes in locus coeruleus firing, dysregulation of norepinephrine function, synaptic receptor regulation, and norepinephrine availability are what result in pathogenesis. Conditions related to norepinephrine dysfunction include anxiety disorders, mood disorders, attention-deficit hyperactivity disorder, Alzheimer’s disease, and posttraumatic stress disorder. Furthermore, many symptoms in these disorders are directly attributable to norepinephrine dysfunction in the neural circuitry.[18]
A prominent contributor to the pathogenesis of IgE-mediated diseases is the neurotransmitter histamine. Produced in mast cells, histamine exerts its effects in the body by binding to certain histamine receptors. Two of the cardinal features of asthma, bronchospasm, and mucosal edema, are directly related to histamine receptor stimulation.[19] Histamine is also implicated in the pathogenesis of multiple sclerosis, which is characterized by inflammatory demyelination in the central nervous system. In animal models, histamine has been shown to change the blood-brain barrier permeability. This change in permeability led to an increase of cells infiltrating the central nervous system, subsequently increasing neuroinflammation.[20]