Introduction
The physiological concept of muscle contraction is based on two variables: length and tension. In physiology, muscle shortening and muscle contraction are not synonymous. Tension within the muscle can be produced without changes in the length of the muscle, as when holding a dumbbell in the same position or holding a sleeping child in your arms. Upon termination of muscle contraction, muscle relaxation occurs, which is the return of muscle fibers to a low-tension state.
Mammals have three types of muscles: skeletal, cardiac, and smooth. Skeletal muscles are attached to bones and give the body structure and strength. Cardiac muscle comprises the walls of the heart, allowing blood to be pumped through the vasculature. Smooth muscle is found throughout the blood vessels, gastrointestinal (GI) tract, bronchioles, uterus, and bladder.[1]
Muscle contraction throughout the human body can be broken down based on muscle subtype specialization.[2] In general, muscle fibers are classified into two large categories: striated muscle fibers and smooth muscle fibers.
Striated muscle fibers contain actin and myosin filaments that power contraction and are organized into repeating arrays, called sarcomeres, with a striated microscopic appearance.[1] Cardiac muscle tissue is a striated muscle fiber under involuntary control by the body's autonomic nervous system (ANS).[3] Skeletal muscle tissue is a striated muscle fiber under voluntary control.
Smooth muscle fibers do not contain sarcomeres but use actin and myosin contraction to constrict blood vessels and move the contents of hollow organs in the body. These fibers are under involuntary control by reflexes and the body's ANS.[4]
Cellular Level
Striated Muscle
To understand the mechanism of striated muscle contraction, it is first essential to understand its structure. The striated muscles in our body comprise many individual muscle fibers. Inside these muscle fibers are smaller units called myofibrils made of parallel thin and thick filaments. These filaments are arranged longitudinally in small units known as sarcomeres, which give the muscle a striated appearance under microscopy.[5]
The thick filaments are made from the protein myosin, which has one pair of heavy chains and two pairs of light chains; these heavy and light chains differ from the thin and thick filaments of myofibrils. At the tail of the thick filament, the two heavy chains are intertwined in a helical formation. At the other end of the thick filament, each heavy chain is paired with two light chains, giving rise to two heads. The myosin heads have an actin-binding site that helps them attach to the thin filaments.[6]
The thin filaments are composed of actin, tropomyosin, and troponin. Actin is a globular protein that combines with other actin globules to form two intertwined strands with positive and negative ends. The double-stranded actin filaments are covered by tropomyosin, which blocks the interaction between myosin and actin when the muscle is inactive. The troponin group comprises troponins I, T, and C and is located along the actin filaments next to tropomyosin.[7]
The complex process leading to muscle contraction, called excitation-contraction coupling, begins when an action potential causes depolarization in the myocyte membrane. The depolarization is spread via the transverse (T) tubules, invaginations of the muscle cell membrane, that help spread depolarization signals to the entire muscle fiber. Depolarization of the T tubules causes a conformational change in the dihydropyridine receptors, which causes the opening of nearby ryanodine receptors on the sarcoplasmic reticulum (SR), the storage site for calcium within muscle cells. When calcium is released from the SR, it binds to troponin C. This causes a conformation change, which shifts tropomyosin, allowing the myosin heads to attach to the actin filaments, creating what is known as a cross-bridge.
Cross-bridge cycling begins when ATP binds to an ATP-binding domain on the myosin head. Myosin dissociates from the actin, breaking the cross-bridge. ATP is then hydrolyzed into ADP and P, which causes the myosin heads to change conformation and move toward the positive end of the actin, cocking the myosin head. The phosphate is released, and the ADP-bound myosin binds to a new location on the actin filament. ADP is then released, which causes the myosin to return to its original position, pulling on the actin filament and causing the sarcomere (and, therefore, the muscle fiber) to contract. These cycles continue until calcium levels in the myocyte fall, causing tropomyosin to cover the actin filaments' myosin-binding sites.[8]
Smooth Muscle
The same thin and thick filaments discussed in striated muscles are present in smooth muscles. However, in smooth muscle tissue, these filaments are not organized into sarcomeres. As a result, smooth muscle does not contain the troponin complex required for skeletal muscle contraction and, thus, has a different mechanism for controlling contraction. This difference is characterized by how calcium (Ca) enters the cell, with three mechanisms increasing intracellular concentration:
- Voltage-gated Ca channels are activated by membrane depolarization, allowing Ca to enter the cell.
- Hormones or neurotransmitters can open ligand-gated channels on the cell membrane.
- Hormones and neurotransmitters such as norepinephrine and angiotensin II can, via the phospholipase-C (PLC) pathway, cause an increase in intracellular inositol triphosphate (IP3).
IP3 can bind to receptors on the SR and cause Ca to be released. Once Ca is released, it binds to a calmodulin protein instead of troponin C, as it does in striated muscle. Calmodulin then activates myosin light chain kinase (MLCK), which, as the name suggests, phosphorylates the myosin light chain. The phosphorylated myosin light chain has ATPase activity, which hydrolyzes ATP, increasing its affinity to actin. The myosin can then readily bind to actin. From this point, cross-bridge cycling is the same as in striated muscle. The smooth muscle will remain contracted as long as Ca is bound to calmodulin and the MLCK is phosphorylated. This allows for prolonged periods of vasoconstriction in blood vessels.[4]
A step-by-step outline of each type of muscle contraction is discussed in a later section.
Development
Skeletal, cardiac, and smooth muscles are derived from the mesoderm. More specifically, skeletal muscle is derived from the paraxial mesoderm, cardiac muscle is derived from the lateral splanchnic mesoderm, and smooth muscle fibers differentiate from the splanchnic mesoderm.
Smooth muscle exceptions not derived from the mesoderm include the muscles of the iris (sphincter and dilator pupillae) and the myoepithelial cells in mammary and sweat glands, which are derived from mesenchymal cells originating from the ectoderm.
Organ Systems Involved
Organ Systems With Striated Muscle
- Skeletal system: muscle attached to bone
- Gastrointestinal system: upper esophagus, external anal sphincter
- Urinary system: external urethral sphincter
- Visual system: extraocular muscles
Organ Systems With Smooth Muscle
- Integumentary system: arrector pili
- Gastrointestinal system: walls of hollow visceral organs (e.g., intestines, pancreas, liver, gallbladder)
- Respiratory system: bronchi and bronchioles
- Urinary system: ureters, bladder, internal urethral sphincter
- Reproductive system: uterus
- Visual system: ciliary muscle
- Vascular system: blood vessels
- Lymphatic system: lymphatic vessels
Function
The primary function of skeletal muscle contraction is to allow for the performance of specific movements. Skeletal muscle also provides structural support, maintains the body's posture, stores amino acids, and maintains core body temperature via shivering.[9]
There are two types of cardiac muscle cells: autorhythmic and contractile. Autorhythmic cardiac cells do not contract; they set the pace of contraction for other cardiac muscle cells, which the ANS can modulate. Contractile cardiac cells (cardiomyocytes) constitute the majority of the heart muscle and can contract.[10]
There are two types of smooth muscle cells: single-unit and multiunit. Single-unit smooth muscle cells are found in the gut and blood vessels. They are linked together via gap junctions, allowing for contraction as a functional syncytium. They also contract myogenically and can be modulated by the ANS.[11] Multiunit smooth muscle cells are found in the muscles of the eye and at the base of the hair follicles. They contract by being separately stimulated by nerves of the ANS and allow for fine control and gradual response.[11]
Mechanism
Types of Striated Muscle Contraction
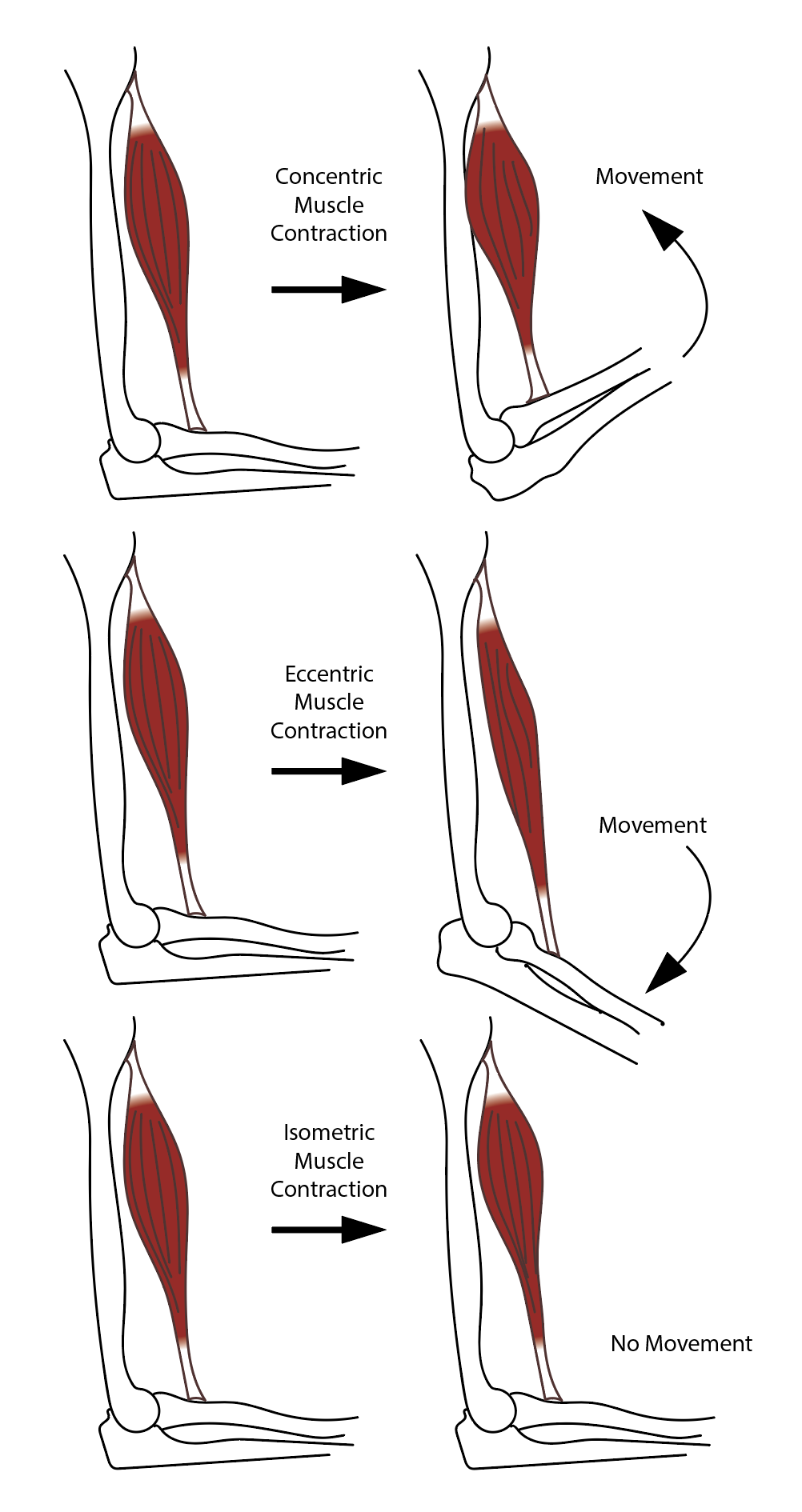
There are four types of striated muscle contractions: isometric, isotonic, concentric, and eccentric.
Isometric striated muscle contraction is characterized by a change in muscle tension without a change in muscle length. Isometric contractions are seen when pushing against an immovable object or trying to lift a weight that is too heavy.[12]
Isotonic striated muscle contraction is characterized by constant muscle tension with a change in muscle length. This type of contraction occurs when the contraction force matches the total load on a muscle. Isotonic contractions are seen during activities such as walking, running, or squatting.
Concentric striated muscle contraction occurs when there is sufficient muscle tension to overcome the load, and the muscle contracts and shortens.[13] During this type of contraction, a muscle is stimulated to contract according to the sliding filament theory. Concentric contractions are seen during activities such as a biceps curl or standing from a squatting position.
Eccentric striated muscle contraction occurs when the muscle works to decelerate a joint at the end of a movement as opposed to pulling a joining in the direction of the contraction. This type of contraction can occur involuntarily (eg, while attempting to move a weight too heavy for the muscle to lift) or voluntarily (e.g., when the muscle is 'smoothing out' a movement or resisting gravity, such as during downhill walking). Eccentric contractions act as a braking force in opposition to a concentric contraction to protect joints from damage.
General Mechanism of Skeletal Muscle Contraction
Skeletal muscle contraction initiation and execution occur in the following steps.
- An action potential (AP) travels along a motor nerve to its endings on muscle fibers.
- At each motor nerve ending, the nerve secretes acetylcholine (ACh).
- ACh acts locally on the muscle fiber membrane to open ACh-gated cation channels.
- The opening of ACh-gated channels allows large quantities of sodium (Na) ions to diffuse to the interior of the muscle fiber membrane.
- This action causes a local depolarization, leading to the opening of voltage-gated sodium (Na) channels, which initiates an AP at the membrane.
- The AP depolarizes the muscle membrane, causing the sarcoplasmic reticulum (SR) to release large quantities of Ca ions stored within the reticulum.
- The Ca ions produce attractive forces to act between actin and myosin filaments, causing them to slide alongside each other, leading to the contractile process.
- After a fraction of a second, the Ca ions are pumped back into the SR by a Ca-membrane pump and remain stored in the SR until a new muscle AP occurs.
- The removal of Ca ions from the myofibrils causes muscle contraction to cease.
General Mechanism of Cardiac Muscle Contraction
Cardiac muscle contraction occurs via excitation-contraction coupling (ECC), utilizing a mechanism called calcium-induced calcium release (CICR). ECC is the process of converting an electrical stimulus (AP) into a mechanism response (muscle contraction). CICR involves the conduction of Ca ions into the cardiomyocyte, leading to the further release of ions into the cytoplasm. Ca prolongs the period of cardiac muscle cell depolarization before repolarization begins. Contraction of cardiac muscle occurs due to the binding of the myosin head to ATP, which pulls actin filaments to the center of the sarcomere, the mechanical force of contraction.
Cardiac muscle contraction initiation and execution occur in the following steps.
- An AP, induced by the pacemaker cells in the sinoatrial (SA) and atrioventricular (AV) nodes, is conducted to contractile cardiomyocytes through gap junctions.
- As the AP travels between sarcomeres, it activates the Ca channels in the T tubules, leading to an influx of Ca ions into the cardiomyocyte.
- Ca in the cytoplasm then binds to cardiac troponin C, which moves the troponin complex away from the actin-binding site. Removal of the troponin complex frees actin, which becomes bound by myosin and initiates contraction.
- Intracellular Ca is then removed by the SR, dropping the concentration of intracellular Ca. This decrease in intracellular Ca concentration returns the troponin complex to its inhibiting position on the active site of actin, ending contraction as the actin filaments return to their initial position, relaxing the muscle.
General Mechanism of Smooth Muscle Contraction
The contraction of smooth muscle is not regulated by the binding of Ca to the troponin complex, as is seen in cardiac and skeletal muscle contraction. Smooth muscle instead utilizes calmodulin, an intracellular second messenger that binds calcium.
Smooth muscle contraction initiation and execution occur in the following steps.
- Intracellular Ca concentration increases when calcium enters the cell and is released from the SR.
- Calcium binds to calmodulin.
- Ca-calmodulin activates myosin light chain kinase (MLCK).
- MLCK phosphorylates myosin head light chains and increases myosin ATPase activity.
- Active myosin cross-bridges slide along actin and create muscle tension.
- Smooth muscle relaxation occurs as free Ca in the cytosol decreases when Ca is pumped out of the cell or back into the SR.
- Ca unbinds from calmodulin.
- Myosin phosphatase removes phosphate from myosin, decreasing myosin ATPase activity and muscle tension.
Related Testing
Manual Muscle Testing (MMT)
MMT is used in the physical examination of patients to assess muscle strength. It may be used to evaluate weakness and effectively differentiate true weakness from imbalance or poor endurance. The Oxford scale is the most widely and commonly accepted method of assessing muscle strength, as it does not require special equipment and demonstrates reliability among independent evaluators. The Oxford scale involves testing key muscles from the upper and lower extremities against an examiner's resistance and grading the patient's strength on a scale of 0 to 5, documented as x/5 (e.g., 1/5, 2/5 muscle strength).[14][15]
- 0: no contraction
- 1: visible/palpable muscle contraction but no movement
- 2: movement with gravity eliminated
- 3: movement against gravity only
- 4: movement against gravity with some resistance
- 5: movement against gravity with full resistance
Electromyography (EMG)
EMG is the recording of electrical activity occurring within a muscle, with its primary use being that distinguish myopathy from neurogenic muscle wasting and weakness. EMGs can detect fasciculations or chronic denervation in muscles that appear clinically normal and differentiate focal nerve, plexus, or radicular pathology by determining the distribution of neurogenic abnormalities.
EMGs can provide supportive evidence of the pathophysiology of peripheral myopathy and is an obligatory investigation in motor neuron disease to demonstrate the widespread denervation and fasciculation required to secure a diagnosis. EMG recordings are made by inserting a needle electrode into the muscle. Typical EMG needles record a radius of approximately 1 mm where, within this volume, there can be 100 muscle fibers. Analyzing the waveforms and firing rates of single-motor or multiple-motor units can give the diagnostic information needed.[16]
Pathophysiology
As muscles are found throughout the entire body, the pathologies associated with abnormal muscle contraction are extensive. Therefore, while this list delineates high-yield pathophysiological conditions related to muscle contraction, it is not exhaustive.
Skeletal Muscle Contraction Pathology
- Duchenne muscular dystrophy
- Becker muscular dystrophy
- Myotonic dystrophy
- Lambert-Eaton syndrome
- Myasthenia gravis
- Myositis
- Bell palsy
Smooth Muscle Contraction Pathology
- Asthma
- Gastroparesis
- Nephrolithiasis
- Chronic kidney disease
- Neurogenic bladder
- Infertility
- Pulmonary hypertension
Cardiac Muscle Contraction Pathology
- Congestive heart failure
- Cardiomyopathy (eg, dilated, hypertrophic, restrictive)[17]
Clinical Significance
Troponin Assays
Troponin I and Troponin T subtypes can serve as markers of cardiac muscle damage. Levels of troponin found in the blood are used to differentiate between unstable angina and myocardial infarction (MI), as they may present with similar symptoms; the elevated troponin levels in myocardial infarction would not be seen in unstable angina. Troponin levels peak a few hours following injury and can remain elevated for up to two weeks after the event.
Troponin release is not specific to MI, as troponin release merely indicates cardiac muscle damage has occurred. As such, troponin levels may also be elevated in conditions such as heart failure, pericarditis, and amyloidosis. In addition, non-cardiac diseases such as sepsis or renal failure can also cause elevated troponin levels.[18]
Muscle Spasm
Muscle spasms, also known as muscle cramps, are involuntary, painful, and localized contractions of an entire muscle group, individual muscle, or select muscle fibers. Muscle spasms can last anywhere from a few seconds to a few weeks, depending on the muscle affected and the mechanism of the spasm.[19]
In sports, exercise-associated muscle spasms are the most frequent condition requiring therapeutic intervention.[20] The specific etiology is poorly understood, and possible causes depend on the physiological or pathological situation in which the spasms occur. It is important to note that a painful contraction limited to a specific area does not mean that the cause of the onset of the cramp is necessarily local.
In certain clinical scenarios, the underlying etiology may be related to persistent spastic muscle contractions that can significantly impact body function. A common example of this condition is manifested in the sternocleidomastoid muscle. Clinically, this is recognized as congenital torticollis or spasmodic torticollis.[21] Other relevant conditions in this realm include but are not limited to exercise-induced and heat-related muscle cramping and piriformis syndrome.
Manual Muscle Testing (MMT)
As previously discussed, MMT is used as a clinical tool to assess muscle strength and is graded on a scale from 0 to 5. MMT is also used in rehabilitation and recovery practices to assess impairments and deficits in muscle performance, including strength, power, and endurance. Identifying impairment in specific muscles or groups of muscles is vital in determining the course of rehabilitation, which may include particular therapy exercises, bracing, or functional movement training.[14]
Pharmacology
Clinically, it is essential to recognize that many medications today will affect muscle contraction at some level. Therefore, whether it be the main effect of the drug or a side effect, it is crucial to understand the uses and consequences of administering medications to patients. Pharmacological agents that directly affect muscle contraction include, but are not limited to:
- Skeletal muscle relaxants (e.g., botulinum toxin, cyclobenzaprine, methocarbamol)
- Vasodilating medications (e.g., hydralazine, nitrates, milrinone)
- Volatile anesthetics or succinylcholine can lead to malignant hyperthermia